Abstract
Damage to the skin makes the body vulnerable to microorganisms; wound dressings with desirable properties such as antibacterial activity is used in order to accelerate the healing of the injury. An available natural substance investigated for its antibacterial property is honey; also, zinc-oxide nanoparticles (ZnO-NPs) have shown great antibacterial activities. This study investigated some properties of PU-Gel nanofibrous membranes, loaded with honey and ZnO-NPs, including antibacterial activity against Staphylococcus aureus, Escherichia coli, and Bacillus subtilis, mechanical properties and cell proliferation. The results indicate that PU/Gel/ZnO and PU/Gel/ZnO/H are suitable in inhibiting bacterial growth compared with PU/Gel/H and control membranes. Moreover, PU/Gel/H membranes had considerable antibacterial effect on E-coli. The addition of ZnO-NPs improved the mechanical properties. Cell culture studies (MTT test) proved the biocompatibility of the developed nanofibrous membranes. The obtained nanofibrous membrane PU/Gel/ZnO/H is a promising candidate for the development of improved bandage materials.
Introduction
The skin has an important function in protecting the body, and once its structure is disturbed, the body is vulnerable to invasion of microorganisms and wound infection, which slows the healing of wounds and can even lead to death. 1 To accelerate wound healing, some supportive agents such as wound dressings have been used for many years. A suitable wound dressing should have some properties such as high porosity for gas transferring and being a barrier to prevent dehydration and infection. According to these requirements, the electrospinning method can be a practical approach for fabricating the membranes.2,3 Because it is an efficient, relatively easy working and adjustable method for fabricating nanofibers 4 and the porous structure of electrospun membrane lets the fluids exit from the wound, it has great oxygen permeability and adjustable water evaporation, but still it prevents invasion of the microorganism. 3
Among polymers, we chose one synthetic polymer, polyurethane (PU), and one natural polymer (gelatin). PU is a suitable barrier and has good permeability; also, some studies report its wound healing capability.3,5,6 Some wound dressings containing PU which have been commercialized are TegadermTM and Op-Site®, both of which reduce water loss and are antibacterial. 2
Gelatin also has appropriate properties such as biocompatibility, biodegradability, and hydrophilic surface, but it is very quickly degradated.2,7 Therefore, to increase the biodegradability and biocompatibility of PU, we can modify it with gelatin. 8
For a wound dressing, another important property is being antibacterial. An available natural substance that has been investigated for its antibacterial activity is honey. 9 It also has anti-inflammatory and antiviral properties,10,11 as well as antioxidant and wound healing activities. 12 The reasons for its antibacterial activity include low pH, its osmolarity which makes it anti-moisture, and hydrogen peroxide activity of its enzymes. 9 The other natural substance which has anti-inflammatory and antioxidant properties is curcumin which is used by Merrell et al. for diabetic wound dressing. 13
Wessam et al. developed a high concentration of honey in Honey-Polyvinyl-chitosan nanofibers and they observed enhanced antibacterial activity against E.coli. 14 Aysu Arslan et al. investigated different concentrations of honey in the electrospinning process, and their result showed that addition of honey had good effects on the spinning of chitosan, good water-absorbing, and no toxic effects on cells. 15
Also, ZnO particles have shown great antibacterial properties in both microscale and nanoscale formulations with different morphologies. 16 According to previous studies, it is considered as a nontoxic agent to mammalian cells17,18; besides nontoxicity, it has low cost and raises keratinocytes by releasing Zn ion on the wound surface and accelerates the wound healing process.19,20 Several studies have documented that the antibacterial mechanism for ZnO is cell lysis through the cell membrane disruption. 21 Release of antibacterial ions, mainly Zn2+22 and Reactive oxygen species (ROS) generation, was reported as the main factor for damaging cell wall and enhancing the internalization of toxic zinc ions which lead to cell death. 18 ZnO-NPs can be synthesized in different methods. Some parameters such as pH, temperature, and solvent type have effects on particle size and morphology. 18 Wahab et al. have produced ZnO nanostructure with the shape of a flower by using zinc acetate solution which showed high antibacterial activity. 23 For assessing the antibacterial activity, one of the most frequent ways is the use of agar diffusion method. 18 There are researches which report incorporation of various nanoparticles in electrospun membranes.16,24,25 In a study, for preventing the detachment of ZnO-NPs loaded in electrospun mats, the sequential electrospinning method was used that made a sandwich structure. 26
The other property which should be considered in the manufacturing of wound healing is mechanical properties. The mechanical properties of materials that are used in the field of regenerative medicine are very important because tissue-engineered scaffolds should mimic human tissues. 27 Skin is an anisotropic, viscoelastic tissue and usually uniaxial tensile test is done to investigate its mechanical properties. 28
The objective of this study was to fabricate a PU/gelatin nanofibrous membrane treated with honey and zinc-oxide nanoparticles. These structures were characterized by (1) testing their antibacterial activity, (2) assessing their tensile properties, (3) morphology by scanning electron microscopy (SEM) and transition electron microscopy (TEM), and (4) toxicity test by MTT.
Materials and methods
Nanoparticle synthesis
For zinc-oxide nanoparticle synthesis, first, 2g zinc acetate dehydrate (CH3COO)2Zn*2H2O, M.W.=219 g/mol, Titrachem. ir 6108800) was added to 15 mL distilled water and stirred for 10 min. Then, 10 mL of 0.2 M NaOH was added to the solution and stirred for 10 min. As soon as the solution was added, the white precipitate containing the zinc-oxide nanoparticles began to form. The resulting solution is titrated with 100 mL of ethanol to keep the oxide nanoparticles stable; it was washed several times with water-ethanol solution to purify the resulting precipitate. Finally, it was dried at 80°C for 2 h.
Nanofiber fabrication
concentrations of electrospinning solutions in different groups.

Cross-linking of nanofiber membranes
The cross-linking phase of the electrospun membranes was performed at ambient temperature for 12 h through exposure to glutaraldehyde vapor (50%/H2O). Subsequently, they were exposed to the residual glutaraldehyde vapor for 6 h in the laminar hood.
Characterization of electrospun fibers
Tensile test
To measure tensile properties, we cut the samples into rectangle shape in 10 mm in width and 40 mm in length, (based on: ASTM D882) and their thickness was measured by micrometer, showing a range from 0.2 to 0.3 mm. Then, these samples were fixed on the homemade cardboard frames. The tests were done using SANTAM, STM 20, Iran, at a speed of 5 mm/min by a cell load of 50-Kgf. Each experiment was repeated three times; after that, the average values for Ultimate Tensile Strength (MPa), Elastic Modulus (MPa), Elongation at peak (%), and Elongation at break (%) were determined.
Morphology
The morphology of electrospun fibers was investigated by SEM (VEGA3 TESCAN). The samples were sputtered with gold. A voltage of 20 kV was applied. Image J software was used to calculate the diameters of the fibers. Approximately, 50 random fibers were measured for each sample and the average diameter was determined. The result is shown in Figure 1. Also, transition electron microscopy (TEM, 906E, Philips, Germany) was employed for morphological study. (Figure 2) Scanning electron microscopy images of (a) PU/Gel, (b) PU/Gel/ZnO, (c) PU/Gel/H, and (d) PU/Gel/H/ZnO, (d’) PU/Gel/H/ZnO with different magnifications. This shows changes in the morphology and nanofiber diameter of the PU/Gel membranes due to enrichment with honey and ZnO nanoparticles and their combination. Transition electron microscopy images derived from two regions of the electrospun PU/Gel membranes with 1 wt.% of ZnO nanoparticles. Size of nanoparticles derived from Image J software in (a): 82 nm and (b): 63 nm.

Evaluation of antibacterial activity
To assess the antibacterial effect of synthesized nanofibers, we used the Kirby–Bauer method.9,25,29,30. The analysis was against Staphylococcus aureus (ATCC 6538P), Escherichia coli (ATCC 8739), and Bacillus subtilis (ATCC 6633). The membranes were cut in circular shape with a size of 8 mm in diameter and left on Agar plates which were seeded with bacterial cells. This test was done triplicate for each membrane by incubation at 37°C overnight.
To assess the cell viability, we seeded them in 1 mL of LB medium at 37°C for 24 h. Then, the cells with a concentration of 1:100 v/v were cultured onto an agar plate and assessed overnight. The antibacterial effect of the samples was analyzed by measuring the created distance between the samples and colonies.
Cytotoxicity assessment (MTT assay)
MTT assay was used to evaluate the cell viability and determine the toxicity of the scaffolds. First, the HEK cells were seeded in 96-well plates in DMEM, containing 10% FBS and 1% penicillin-streptomycin at 37°C. The supernatant of the cultured cells (20000 cells in each well) was removed; then, the size of 5x5 mm of each scaffold, which was enclosed to UV radiation for 2 h, was placed into the wells and incubated for 48 h. Then, the MTT solution was added and incubated for 4 h. For dissolving formazan, DMSO was added. The cell proliferation was analyzed by testing the absorbance at 570 nm. The result is shown in Figure 3. The nanofiber membrane without ZnO exhibited the lowest cell viability of 96.2 ± 3% survival, while the cells cultured in the leachate of the honey-loaded membranes showed higher viability. The nanofibrous membrane with H/ZnO led to the best cell viability of 155.1 ± 5%.
Statistical analysis
Presented results were shown as mean values ±SD. The data obtained were statistically analyzed using SPSS (version 22). One‐way analysis of variance followed by the LSD post-hoc test was performed to compare the means of samples; A p value of ≤.05 was considered as statistically significant.
Results
EDX analysis
EDX analysis: Energy dispersive X-ray diffractive (the IDFix report) was done to analyze the synthesized ZnO-NPs loaded on PU/Gel nanofibrous mats (Figure 4). Analysis conditions were accelerating the voltage of 20 kV, beam current of 1.000 nA, and live time of 30 s. EDX corroborated the presence of elements oxygen (96.51%) and zinc (3.49%). IDFix plot of the synthesized ZnO-NPs loaded on nanofibrous membranes.
Transition electron microscopy photographs revealed the distribution and size of ZnO-NPs in PU/Gel nanofibrous membranes, as shown in Figure 2. Nanoparticles are totally isolated and agglomeration is not observed; altogether, a moderated acceleration of NPs release could be obtained based on the TEM images.
Tensile test
Antibacterial activity
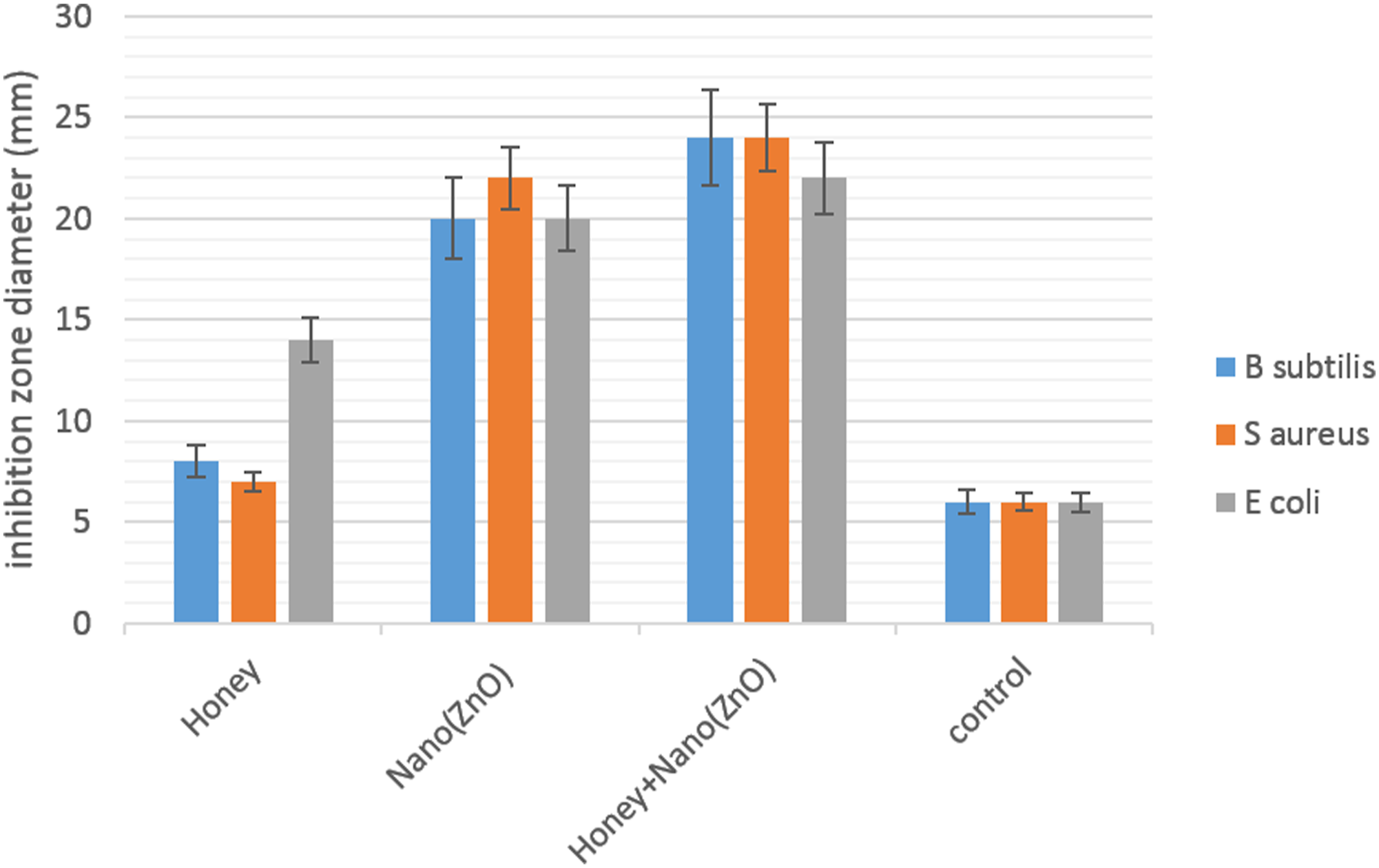
The antibacterial activity of the nanofibrous membranes was studied by disc diffusion assay. Photographs of the inhibition zone of different PU/Gel nanofibrous membranes against E. coli, S. aureus, and B. subtilis in disc diffusion assay are shown in Figure 5. With the honey and ZnO nanoparticle in the nanofibers, the inhibition zone became increasingly clearer and larger. Figure 6 shows the histograms of the inhibition zones diameters. For E. coli, the diameter of the inhibition zone was enlarged for PU/Gel/H to 8 ± 0.2 mm, PU/Gel/ZnO to 14 ± 0.2 mm, and PU/Gel/H/ZnO to 16 ± 0.2 mm, while for S. aureus, the diameter rose for Pu/Gel/H to 1 ± 0.2 mm, PU/Gel/ZnO to 16 ± 0.2 mm, and PU/Gel/H/ZnO to 18 ± 0.2 mm. Also, for B. subtilis, the diameter of the inhibition zone was enlarged for PU/Gel/H to 2 ± 0.2 mm, PU/Gel/ZnO to 14 ± 0.2 mm, and PU/Gel/H/ZnO to 18 ± 0.2 mm. Antibacterial activity of treated PU-Gel membranes, by appearance of growth inhibition zones on solid media in Petri dishes after 24 h incubation. (a, d) Bacillus subtilis, (b, e) Staphylococcus aureus, (c, f) Escherichia coli. (0) Control, (1) Honey, (2) Nano ZnO, (3) Honey + Nano ZnO. Antibacterial activity of PU/Gel/H/ZnO nanofibrous membranes against E. coli, S. aureus and B. subtilis evaluated by disc diffusion assay after 24 h. Results are mean ± SD (n = 3).
Cytotoxicity
To evaluate the cytotoxicity of the PU/Gel/H/ZnO nanofibers, we measured the viability of the cells cultured in the leachate medium of nanofibers and PU/Gel/common medium (control group) by MTT assay. As shown in Figure 3, the cells cultured in the leachate medium of all groups of nanofibers with honey exhibited high viability, which demonstrates the good biocompatibility of the PU/Gel/H nanofibers. Among these, the nanofiber membrane without ZnO exhibited the lowest cell viability of 96.2 ± 3% survival, while the cells cultured in the leachate of the honey-loaded membranes showed higher viability. The nanofibrous membrane with H/ZnO led to the best cell viability of 155.1 ± 5%. The results indicate that PU/Gel/H/ZnO nanofibers facilitate cell proliferation with honey content, and all PU/Gel/H/ZnO nanofibrous membranes have good biocompatibility.
Discussion
Morphology
The SEM images of treated and non-treated PU/Gel membranes are shown in Figure 1. The average fiber diameter of the treated membranes was 140–260 nm. The pictures show that adding honey to the electrospinning solution resulted in an increase in the average fiber diameter of the membranes, and this can be attributed to increased viscosity of the solution; some other studies reported a similar result.1,9 However, a study by Maleki et al. showed that the addition of honey decreased the fiber diameter and they attributed this result to increased conductivity. 31 However, some reported that the addition of honey would reduce the conductivity, and the reduced conductivity of the electrospinning solutions lead to less jet stretching, and finally a larger nanofiber diameter.1,32
In a study by Kim et al., it was shown that increasing the percentage of PU to an electrospun solution of gelatin/PU would lead to a decrease in the final fiber diameter size. 2
Mechanical properties
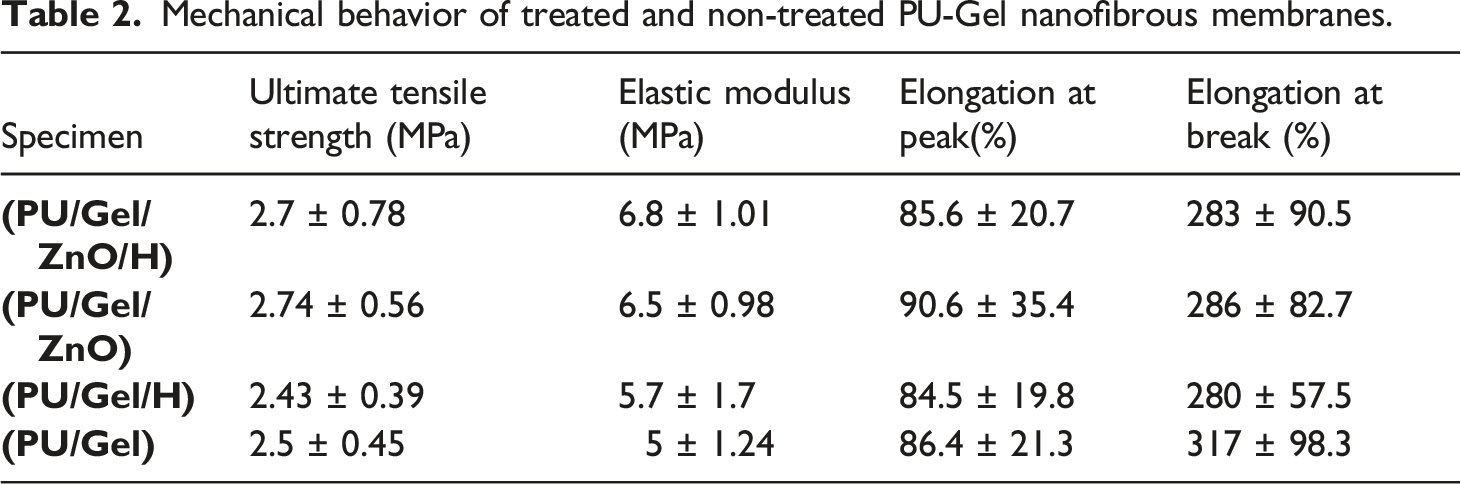
Previous studies showed that the scaffolds made of natural polymers do not provide adequate strength in applied tissue engineering; therefore, in this study, a synthetic polymer, PU, was added to improve the final mechanical properties. The results of the tensile test are summarized in Table 2 and Figure 7. As shown in the Table, they possess acceptable young modulus (5–6.8 MPa) for applying in soft tissue engineering.33,34 Mechanical behavior of treated and non-treated PU-Gel nanofibrous membranes.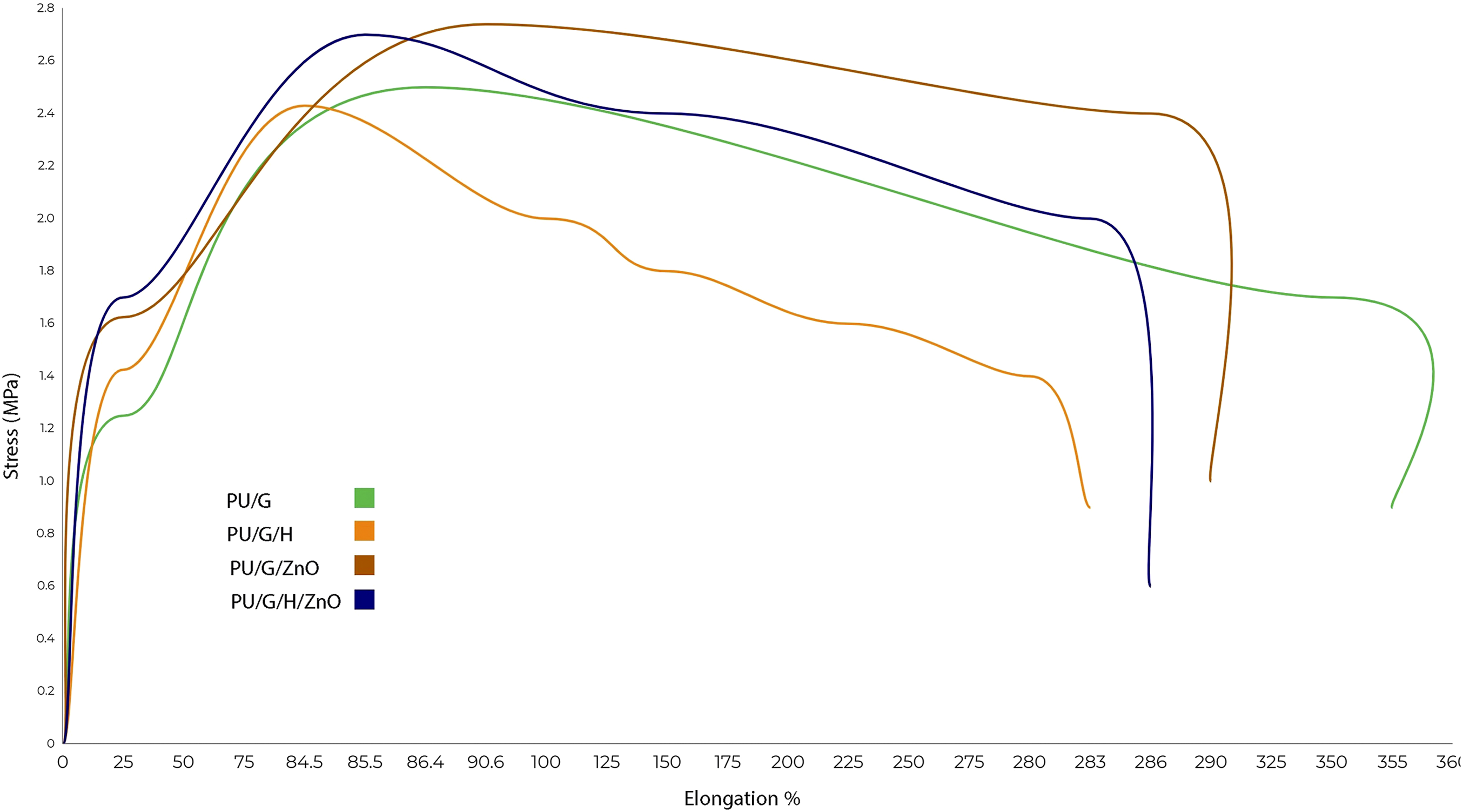
Based on the results, the addition of honey increased the elastic modulus and made the membranes stiffer, but it decreased elongation at peak and elongation at break, so it increased the rigidity of membranes.
Kim et al. indicated that the gelatin/PU nanofiber scaffolds would mimic the mechanical properties of the soft tissues. 2 Also, Patil et al. reported the tensile strength and elongation percentage of the Silk Fibroin–Polyinylalcohol/ZnO composite film increased by adding ZnO-NPs. 20 Similarly, our result showed the tensile strength and Young modulus were increased when ZnO-NPs were loaded on nanofibers, but elongation at break was reduced. Increase in Young modulus16,35 and reduction in elongation at break are similar to previous studies after addition of ZnO nanoparticles to nanofibers.16,36 These changes could be caused by agglomeration of nanoparticles, 16 and some reported decrease in Young modulus 36 and attributed this to bead formation. 36 These amounts are appropriate for wound dressing applications because they were higher than those reported for the composite bandage. 37
Antibacterial activity
Figures 5 and 6 represent the results of the antibacterial assessment of the membranes. Control groups showed no zones of inhibition. In this regard, Turaga et al. observed no antibacterial activity after treatment of PVA nano-webs with honey. 9 Similarly, Puttamayutanon et al. reported some antibacterial activity of pure honey, but no activity was observed on PVA nano-webs treated with honey. 38
The results indicated that the antibacterial activity was improved with honey and ZnO nanoparticles content in the nanofibers. The antibacterial property of honey predominantly originates from its hydrogen peroxide production ability, high sugar content, acidity, and flavonoids. 39 Additionally, the nanofiber membranes with honey displayed higher antibacterial activity against E. coli in comparison with S. aureus and B. subtilis, which does not approve the previous study showing that honey exhibits better inhibition effect against gram-positive bacterium than gram-negative bacterium.1,40 It is obvious that the antibacterial activity of nanofibers comes mainly from the addition of ZnO nanoparticles. Especially, the antibacterial activity of the PU/Gel/H/ZnO nanofibers was dramatically improved after 24 h of incubation compared with 12 h, which could be attributed to the sustained release of honey and ZnO from the nanofibers over time. Besides, it was also observed that the antibacterial activity of the PU/Gel/H/ZnO nanofibers against S. aureus, E. coli, and B. subtilis was the same. The results demonstrate that PU/Gel/H/ZnO nanofibers possess effective and broad-spectrum antibacterial activity, as is favorable for the wound healing process.
In antibacterial tests against bacteria such as Escherichia coli, ZnO-NPs are claimed to increase the inhibition zones.19,41,42 A study showed the inhibition zone also increased as concentration of nanoparticles increased, and the highest inhibition zone was observed in S. aureus; this result is similar to what Schwartz et al. have reported. 43 Since S. aureus can cause serious infections, it is useful to use ZnO-NPs in wound care applications. Based on what Khorasani et al. stated, the better antibacterial effect of ZnO-NPs on S. aureus in comparison with E. coli and B. subtilis was because of the difference in the structure of the cell wall in these bacteria. 19 Studies stated that mechanism of its antibacterial activity could be attributed to increased specific surface area because of reduced particle size and consequently enhanced particle size activity, as well as generation of ROS and particle abrasive surface which lead to cell wall damage. 18 Previous researches showed the smaller the size of ZnO-NPs, the better the antibacterial activity.44,45
Some studies reported an increase in inhibition zone dimension by increasing the amount of honey.1,46 Also, Abou Zekry et al. in a study have used pomegranate/honey nanofibers for wound dressing and concluded that honey-based nanofibers had a good antibacterial activity against both gram-positive and gram-negative species. 47 Furthermore, a study on Manuka honey showed it inhibited the production of pathogens and biofilms found in wounds.48,49 Some studies attribute honey antibacterial activity to hydrogen peroxide content which activates the neutrophils,39,50 phytochemicals, or flavonoids content which are antioxidants, 39 low water, and high sugar content; the water amount is not sufficient for bacterial growth, and the last factor, its acidity (pH: 3.2–4.5). 39
Conclusion
In summary, PU/Gel/H/ZnO nanofibrous membranes were fabricated via electrospinning. Their morphology, antibacterial activity, mechanical properties, and cytotoxicity were investigated. It could be observed by SEM that by adding honey, the average diameter of the nanofibers increased. A moderated distribution of ZnO-NPs was obtained based on the TEM images which is suitable for the rate of release. The antibacterial assessment showed that honey affected gram-negative, but not on the gram-positive bacterium. Additionally, MTT assay suggests the appropriate biocompatibility of the PU/Gel/H/ZnO nanofibrous membranes. Therefore, PU/Gel/H/ZnO nanofibrous membranes could be a good candidate for efficient wound dressing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.