Abstract
Contaminants deposited on the firefighter protective clothing increase the risk of carcinogenesis by transdermal absorption. The effects of the inclination angle, waterproof grade, and fabric structure of the flame retardant outer layer of firefighting protective clothing on polycyclic aromatic hydrocarbon (PAH) contamination and laundering were studied by simulating smoking exposure. The permeation and transmission process of PAHs in multilayered assemblies were investigated. Results showed that the adsorption capacity of soot and PAH contamination on fabric with a water repellent finish relatively increased, and outer layer absorbed more PAHs at 180° compared to other inclination angles. The fabric structure with single layer had a strong adsorption capacity and low removal capacity for PAHs. The highest amount of PAH contaminant was found in the moisture barrier layer, and the removal rate (44.7%) was much lower than that of the outer layer cleaned through conventional laundering (91.3%). The research can better provide scientific support for pollution control and clothing cleaning in the actual firefight.
Introduction
Firefighters are becoming increasingly aware of smoke exposure risks of personal protective equipment (PPE) on firegrounds and health risks associated with smoke exposure.1–3 The composition and properties of soot vary according to the types of materials burned and the state of combustion. 4 In general, soot consists of carbon material, inorganic compounds, and organic matter. 5 The exposure rates of urban firefighters to toxic combustion products during their operational duties have been investigated,6,7 and research on dermal exposure to contaminants has focused on PAHs. 8 Polycyclic aromatic hydrocarbons are derived naturally from coal and tar deposits and produced by the incomplete combustion of organic matter and have studied in relation to the occupational health of firefighters because some PAHs have mutagenic and carcinogenic properties. 9 Some cancer types are related to PAHs, including lung, bladder, and skin cancer.10,11
High contamination levels are found after inside attack or inside search. 12 Firefighters are most commonly exposed to PAHs, which are inhaled by firefighters not wearing airway protection or enter their bodies through dermal absorption during and after a firefight even when they are wearing PPE.13,14 The surface loads of PAHs on the outer surface of firefighter PPE and tools used by firefighters have been examined after training or simulation in a closed environment.15,16 However, data for assessing the relationship between outer layer conditions (fabric structure and waterproof level) and exposure to airborne ultrafine smoke particles are currently limited.
Some of the contaminants may directly enter the body through inhalation, but others are deposited on the exterior surfaces and inner layers of firefighter protective clothing, which increasing the risk of exposure between clothing and skin. 17 Furthermore, dermal absorption occurs during firefighting through permeation or through the penetration of contaminants during doffing of hoods, jackets, and trousers, into the interface regions of this ensemble or through the cross-transfer of contaminants on a gear to skin.9,12 However, whether toxic substances accumulated on a gear during a fire response are transfer to the skin through multilayer firefighter PPE needs further confirmation. In addition, the pollution mechanism, especially the mechanism by which PAHs penetrates into multiple layers, is not clear. Understanding the level of exposure and exposure pathways from firefighting uniforms to firefighters is of great importance. 14 In addition, rarely study has investigated the effectiveness of laundering in removing contaminants from multilayer firefighting PPE.
In this research, the PAH contamination of firefighter PPE in a fire exposure simulator was characterized. Further, real smoke particle deposition and PAH concentration under three angles (45°, 90°, and 180°) were obtained. The effects of water repellent and outer layer fabric structure on PAH deposition were investigated in the smoke simulator, and the contamination mechanisms in multilayer fabrics before and after decontamination measures were investigated.
Methods
Experimental materials
As shown in Figure 1, three pieces of fire service outer fabric based on meta- and para-aramid were prepared as representative outer fabric in the marketplace. The three outer layers were named WRF1, WRF2 and WRF3. The specific fabric parameters of three outer layers are shown in the Table 1. The grams per square meter of the outer layer was 250 g/m2 (WRF1), 200 g/m2 (WRF2), and 210 g/m2 (WRF3). The filtration resistance with 0.3 μm diameter aerosol was tested under the flow rate of 32 L/min. An aged version of Fabric 1 (WRAF1) was washed 20 times and used to emulate aging fabric subjected to extensive use. In addition, the version of Fabric 1 (NWRF1) was a preproduction sample of the fabric without water repellent finish. Therefore, the fabric of NWRF1 was completely wet, generating a version of Fabric 1 (NWRSF1). In addition, WRF1 was used as an outer shell layer constituting a typical multilayer structure of fire cloth. The moisture barrier (MB) layer and comfortable layer (CL) were purchased from DuPont. Plastic cup, as standard burning material for fire tests, was selected to carry out the smoke exposure test. The main component of the plastic cup was polystyrene and has a mass of 30.0 ± 2.0 g, density of 1.05 g/cm3, and volume of 450 ± 10 mL. The outer fabric of three different structures. Fabric parameters of the three outer layers.

A washing machine (SXT-250FDQ) with a capacity of 25 kg was purchased from Jinan Oasis Washing Equipment Co., LTD., and the drying equipment (DC6-15) was purchased from Electrolux.
Experimental procedure
In the experiment, the contaminated fabric containing PAHs was prepared by simulating the smoking pollution. In order to repeatedly generate PAHs of city fire, a smoke exposure simulator was developed from a steel laboratory chamber. The simulator was divided into two compartments, as shown in Figure 2. The left section was served as the combustion area and the right section was as exposure area. The fabric of 15 cm × 40 cm fixed by a holder was exposed through burning a plastic cup with an electric furnace. The whole smoking process lasted for 25 min, in which plastic cup started from melting to carbonization. The fabric holder could adjust the angle to change the pollution degree of fabrics. The inclination angle refers to the angle between the fabric and the top of steel laboratory chamber. For instance, 180° is completely parallel to the top of chamber. The change in temperature around the fabric was patterned through two thermocouples under the smoking test. The chamber allowed the testing of multiple pieces of fabric in a single trial, and many different conditions with excellent repeatability were used within a group of samples and the influences of ambient conditions were prevented. The steel laboratory chamber.
Contaminated fabric was sewn onto the protective clothing for laundering. The clothing was washed according to the relevant washing guidelines of NFPA 1851. The volume of water was set to 60 L and the amount of detergent was 40 g. The rotation speed of washing was 45 r/min and clothes were washed for 20 min at room temperature (24°C).
The clothing was suspended in the drying equipment after being fully dehydrated. The hot air of 60°C generated by drying equipment was blown to the inside and outside of the garment for 2 hours.
The presence of smoke particles in fabric was confirmed through scanning electron microscopy (SEM, Hitachi TM3030). The PAHs of contaminated fabrics before and after laundering were analyzed in accordance with Afps GS 2019:01 by Intertek Testing Services. Analysis was performed through gas chromatography mass spectrometry (GC-MS), and PAHs were detected and quantified in the collected samples after the preparation procedure (extraction, clean up, and concentration).
Results and discussion
Effects of angles on PAH contamination
The smoke during burning plastic cup could be transported swiftly up until met an top obstacle of steel chamber. Subsequently, spread direction was changed and finally turned down under the influence of heat and gravity. Photographs were used for observing pollution level on the fabric surface. As can be seen from Figure 3, deposition levels during smoking test at different inclination angles varied. From Figure 3(e)–(g), the soot on the fabric surface was scattered when the inclination angle was 90°, and the soot adsorbed on the fabric surface increased at an inclination angle of 45°. As expected, the fabric surface was full of soot when the inclination angle was 180°. Furthermore, the back of this fabric exhibited the lowest deposition level, as shown in Figure 3(j). The deposition level depends on whether the fabric was completely perpendicular to the spreading direction of smoke. The fabric under 180°, which was completely perpendicular to the spreading direction of smoke, was exposed to more smoke from horizontal direction. When inclination angle changed, the horizontal smoke possessed by fabric was reduced. In addition, the soot dropped partially along the fabric surface under the gravity and the influence of heat. The deposition level was the lowest under 90° due to the inclination angle was consistent with the spreading direction. However, the deposition level of smoke particle in the upper of 90° was higher than that in the bottom. This result was consistent with the results of Horn,
16
who reported that pollution level at the upper part of clothing was higher than that at the bottom part. Photographs of fabric contaminated at different inclination angles: (a) blank fabric, (b) smoking test under 45°, (c) smoking test under 90°, (d) smoking test under 180°, (e) front side of contaminated fabric at 45°, (f) front side of contaminated fabric at 90°, (g) front side of contaminated fabric at 180°, (h) back side of contaminated fabric at 45°, (i) back side of contaminated fabric at 90°, (j) back side of contaminated fabric at 180°.
The variation in temperature under different inclination angle also contributes to the difference in deposition levels. The thermocouple on both sides of fabric holder could capture the specific changes in temperature during smoking test. Figure 4 depicts variability in temperatures on both sides of fabric holder. As demonstrated in Figure 3, change in inclination angle had a great influence on temperature at the left side of the fabric holder. The temperature on the left side of fabric holder at 45° was higher than that at 90°, and reached a maximum temperature in the 13th minute and plateaued over time. However, the temperature at the right side of the fabric holder varied with different angles. The maximum temperatures of smoke around the fabrics reached to 100°C within 11 min and then decreased rapidly over time. The main reason for this phenomenon is that plastic cup was decomposed in the early experiment, and began to burn with the temperature increasing. The burning continued for several minutes, then the flame was gone and the plastic cup was carbonized into ash. Therefore, it is obvious that the temperature near the fabric gradually decreased after 13 min. The temperature of smoke around fabrics was basically consistent with the environment at a height of 1.5 m in the fire exposure simulator study of Horn.
16
The temperature curve of smoke around fabrics during smoking process.
The total deposition of Polycyclic aromatic hydrocarbons before and after laundering at three inclination angles.

ND stands for the non-detected component.
Moreover, change in angle greatly influenced PAH deposition. All the PAHs, except acenaphthene were detected at the three angles. Notably, phenanthrene had the highest level on all exposed fabric samples, indicating that it was a key contaminant generated by the burning of the plastic cup. Furthermore, in all three cases, phenanthrene level was unchanged, indicating that the number of soot particles did not affect phenanthrene adsorption.
Polycyclic aromatic hydrocarbons had a concentration of 48.1 mg/kg at an inclination angle of 90°. The adsorption capacity of PAHs was 70.3 mg/kg at 180°. The main reason for this phenomenon was the weakened smoke sedimentation at 90°. Smoke diffused from high temperature to low temperature under the action of thermal convection and radiation in an accumulation mode. 19 By contrast, the sedimentation of smoke pollution was more obvious when the inclination angle was 180°. In addition, the combination rate of smoke was high because of the high temperature around the fabric. Moreover, physical sedimentation of smoke was observed, along with thermal motion at high temperature at 45°, both of which accelerated the contamination of the fabric.
The temperature design in the chamber referred to the relevant research and smoking temperature in the actual rescue. In practice, the air was filled with high-temperature smoke in the rescue process of interior fire, and the firefighters were directly exposed to the dark space. Different rescue actions of firefighters, such as standing or possession, would affect the pollution level on clothing and skin. The contamination varies with angle as a result of the exposure and deposition level varying from 45° to 180°. Under the premise in spreading direction of high-temperature dust, the change of angle reduced the accumulative exposure between fabric and smoke in the vertical direction. As a result, the contamination of different positions on clothing were also different. The results can better provide scientific support for pollution controlling in the actual firefight.
Polycyclic aromatic hydrocarbon concentration in fabric at different angles after laundering remarkably varied after the same laundering process. The residue contaminant at 180° had the highest level, while the lowest residue level was obtained at 90°. Routine laundering reduces PAH contamination in fabric by an average of 76%–81%.
20
As shown in Figure 5, the contaminated fabric samples at different angles before and after laundering were examined, and the reduction rates were 79.4% and 83.2% at 45° and 180°, respectively. Interestingly, 96.5% was the highest achievable contamination reduction rate at 90°. These findings indicated that the effectiveness of PAH removal by laundering may depend on the initial contamination levels in a load of laundry. Total Polycyclic aromatic hydrocarbon (PAH) and removal rate of contaminated fabric at different angles.
Effects of fabric structure on PAH contamination
There are many kinds of outer fabrics of firefighting protective clothing, which often provided by more than one supplier in the fire brigade. Studying the relationship between fabric structure and contamination is of great significance for the cleaning and maintenance of clothing. Figure 6 illustrates PAHs adsorption in different contaminated fabric structures. A significant difference in the adsorption rates of PAHs was observed, especially between single- and double-layer fabric. The amounts of 10 kinds of PAHs were higher in the single-layer fabric (WRF2) than those in the other two fabrics, as shown in Figure 6(a). Figure 6(b) shows that the sum of PAH contamination in the two double-layer fabric were basically the same, while total PAH contamination in single-layer fabric was 77% higher than those double-layer fabric. Furthermore, the contamination level and cleaning efficiency varied greatly according to fabric structure.
21
Compared with routinely laundered fabric, the single-layer fabric (WRF2) had a lower cleaning efficiency. Only 40.1% of the PAHs on the exposed fabric was removed. The removal rates in the two-layer structures, WRF1 and WRF3, were 79.4% and 86.1%, respectively. Moreover, WRF2 has the highest filtration resistance of 28.9 Pa, improving the ability to block smoke. All these changes may be responsible for effectiveness in PAH removal. (a) Polycyclic aromatic hydrocarbons in fabrics of different structure after smoking text; (b) Sum and removal rate of PAH contamination before and after laundering.
Effects of waterproof grade on PAH contamination
The waterproof capacity of protective clothing reduced due to the daily wearing and washing. However, the contamination mechanisms of waterproof effects on outer layer remain unclear. Figure 7 shows the microscopic photos of fabric with different spraying rating. Compared Figure 7(a) and (d) found that the hairiness and deformation of the aging fabric significantly increased after repeated laundering. (a) Microimage of WRF1 with 80×, (b) Microimage of WRF1 with 1000×, (c) experimental result of WRF1 after spraying test, (d) Microimage of WRAF1 with 80×, (e) Microimage of WRAF1 with 1000×, (f) experimental result of WRAF1 after spraying test, (g) Microimage of NWRF1 with 80×, (h) Microimage of NWRF1 with 1000×, (i) experimental result of NWRF1 after spraying test.
In addition, the fiber surface was obviously damaged, as shown in Figure 7(e). Figure 7(c) showed the appearance of sporadic water droplets on the surface of the waterproof fabric after the spraying test, and the spray rating was greater than level 3. And the waterproof fabric subjected to repeated laundering and drying was exhibited water stain on its surface, as shown in Figure 7(f), and the spray rating was level 2. However, the surface of the fabric without any water repellent finish was completely wet after water spraying, as shown in Figure 7(i).
Figure 8 shows that waterproof grade greatly influenced PAH contamination, and this influence was mainly reflected in the adsorption and removal rates of PAHs. As shown in Figure 8(a), phenanthrene had the highest content in all contaminated fabric samples, which was caused by combustion of a single plastic cup. The product type would change with the combustion materials.
6
In addition, as indicated in Figure 8(b), the PAH concentrations of NWRF1 without any water repellent finish were the lowest, reaching 32.5 mg/kg, whereas the PAH concentration of WRF1 was 70.3 mg/kg. However, the adsorption rate of PAHs in WRAF1 increased to 80.2 mg/kg. The cause of this phenomenon had two aspects. First, when combined with non-waterproof fabric, the waterproof coating accelerated the combination between fabric and soot particles under the action of high temperature. Second, the aging fabric exposed to serious hairiness possessed the higher roughness surface, which improving the soot holding capacity of fabric. Interestingly, when the fabric was exposed to 100% humidity, the PAH content was greatly reduced from 32.5 mg/kg to 18.1 mg/kg during smoking. The reason for this phenomenon may be that the ambient temperature gradually increased during fuming, the highest temperature exceeded 100°C, resulting in the gradual evaporation of moisture in the fabric. The upward evaporation process directly affected the physical process of soot deposition, leading to the final reduction of pollution level. (a) Polycyclic aromatic hydrocarbons in fabrics with different waterproof grade after smoking text; (b) Sum and removal rate of PAH contamination before and after laundering.
As indicated by the removal rates of PAHs in the Figure 8(b), the fabric without waterproof coating had the highest removal rate, reaching 87.7%, whereas the waterproof fabrics had the similar removal rates, reaching 79.4% and 79.9%. As a result, the difficulty in cleaning PAHs was increased because of the coating. The waterproof coating adsorbed a higher content of PAHs as mentioned above, and the PAHs had the higher combination fastness with coating. And this interaction was reinforced under the effect of high temperature. However, the removal rate of PAHs decreased from 87.7 to 79.9% when the contaminated fabric was humid, and the causes of this phenomenon may be the increased moisture content and enhanced hydrogen bonding interaction between soot particles and fibers. 22
The surface of fabric exposed in the smoke was covered with a lot of soot particles, as shown in Figure 9(a) and (b). The surface of different fibers was smooth after laundering. However, soot particles were still observed in the gaps among fibers of WRF1, as shown in Figure 9(c). Results indicated that particles on the surface of the fiber were loosely combined, which could be captured during laundering,
23
but particles in the gaps were difficult to be cleaned in the presence of extrusion force between fibers. In addition, by comparing Figure 9(c) and (e), there were more soot particles in the gap of fabric with waterproof coating. This main reason is that fastness between waterproof coating on fibers and soot particles was improved at high temperature. Figure 9(f) shows that the fiber surface of wet fabric is adhered by the individual particles, while the dry fabric surface is neat and smooth after laundering. Microstructure of WRF1 exposed in smoke: (a)400×, (b)4000×; Microstructure of fabrics after laundering: (c) WRF1, (d) WRAF1, (e) NWRF1, (f) NWRSF1.
Effects of multilayered assemblies on PAH contamination
Most studies only focused on the cleaning of a single layer and ignored the relation with contamination and removal of multilayered assemblies. Three layers of typical fabric structures were sewn together to form fire protection clothing for contamination simulation. In almost every case, the levels of PAHs found on the middle layer were higher than those found on the outer layer, as shown in Figure 10(a). Although a range of PAHs was detected on the middle and outer layers, only one PAH was found on the inner layer. This finding was consistent with that of Alexander.
7
In addition, some PAHs found on the outer layer were not observed on the middle layer. Richard M. Kesler found that PAH level difference between the inner and outer layers for hood samples was significant[24]. PAHs with low molecular weights, such naphthalene, fluorene, phenanthrene, and anthracene, are considered semivolatile PAHs.
16
The flame retardant outer layer was the first physical barrier, and the content of phenanthrene reached 28.2 mg/kg, and 153.5 mg/kg phenanthrene was detected in the moisture barrier layer. This finding indicated that the penetration ability of phenanthrene in the multilayer fabric was the strongest and moisture barrier layer had a stronger adsorption capacity. (a) Polycyclic aromatic hydrocarbons in fabrics with different layers after smoking text; (b) Sum and removal rate of PAH contamination before and after laundering.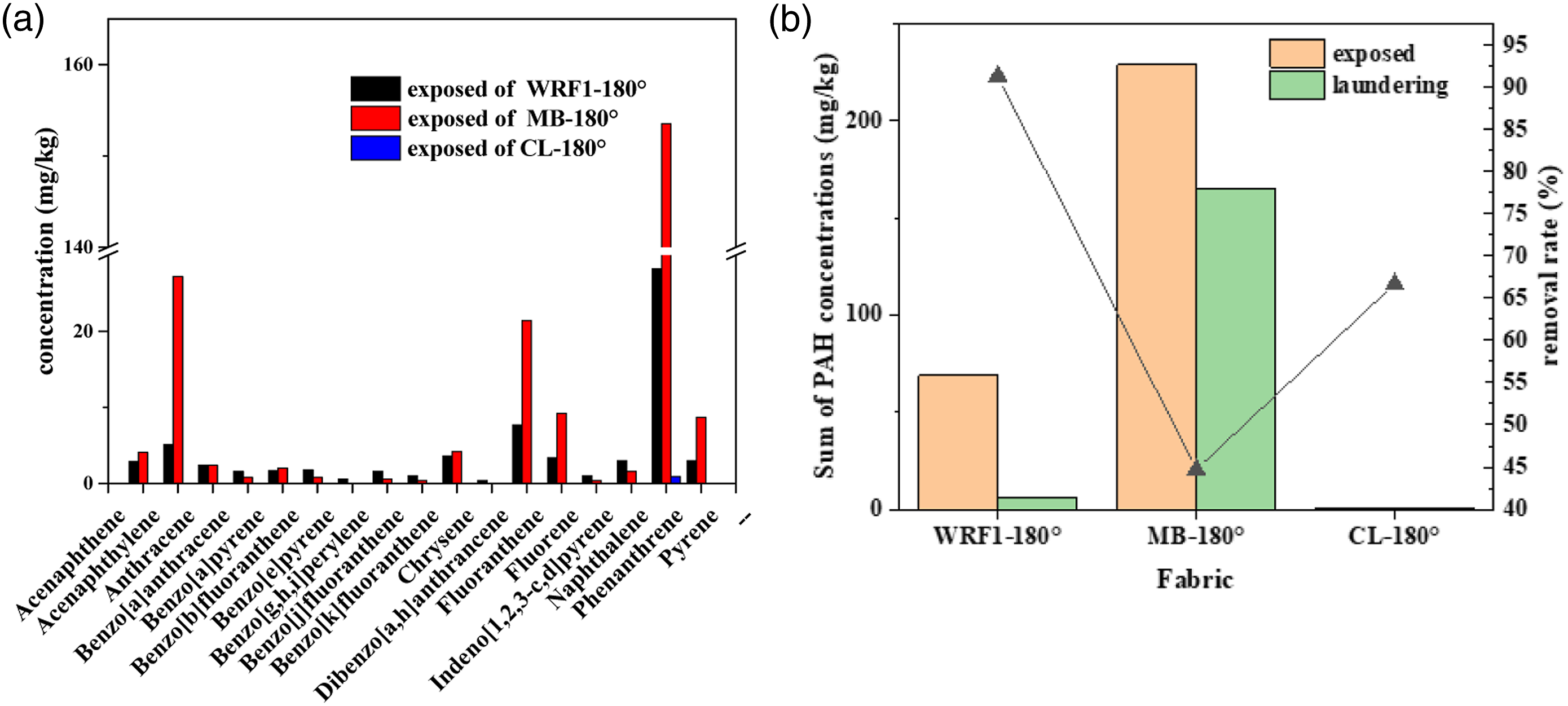
Polycyclic aromatic hydrocarbons in the middle layer was the highest, reaching 228.9 mg/kg. However, the PAH concentrations of the outer and comfort layers were 68.8 and 0.9 mg/kg, respectively. Although soot particles and part of PAHs were trapped in the outer layer, the most of PAHs moved towards the moisture barrier layer along the outer fabric pores under the action of high temperature. The moisture barrier layer has the larger specific surface area due to its microporous structure and therefore limited the movement of PAHs, which resulting in the highest contaminations in multilayered assemblies of firefighting protective clothing. FENT et al. 12 also found that a moisture barrier of gloves can stop PAHs from permeating between moisture and inner layers. The moisture barrier layer had the best insulation capacity, and PAHs could not continue to pollute the comfort layer and human body through the moisture barrier even at high-temperature. However, 0.9 mg/kg phenanthrene in the comfort layer should be caused by the direct pollution between smoking and this layer.
It was worth noting that the fabric samples were washed twice for increasing cleaning efficiency. Figure 10(b) shows that the residual content of PAHs in the washed samples in the outer layer was 6.0 mg/kg, and the residual content in the comfort layer was 0.3 mg/kg, indicating that PAH contamination in the outer and comfort layers was relatively easy to clean. However, the residual content of phenanthrene in the moisture barrier layer was 126.6 mg/kg, and the proportion was 59.8%. The removal rate of PAHs in the outer and moisture barrier layers were 91.3% and 44.7%, respectively. Polycyclic aromatic hydrocarbons in moisture barrier layer was more difficult to be removed by washing than outer layer finished by water repellent. Firstly, the moisture barrier layer is the middle layer and cannot be washed directly in the solution. Moreover, because of the small diameter and large specific surface area, the moisture barrier layer possesses strong binding power with PAHs. In addition, the removal rate of PAHs in moisture barrier layer was the lowest and phenanthrene was probably more difficult to remove with the current laundering procedure. This is mostly due to the lack of special detergents and decontamination equipment, even though the conventional detergent molecules can enter the moisture barrier layer for cleaning. In the future, the experimental research in developing detergents and washing process will be carry out.
Conclusion
The effects of the inclination angle, waterproof grade, and fabric structure of outer layer on PAH contamination and laundering were studied through the simulation of smoke pollution. In the simulation, a plastic cup was fired through an exposure simulator. The infiltration process and removal rate of PAHs in multilayered assemblies were studied. The results showed that the highest contamination level was observed at an inclination angle of 180°. Polycyclic aromatic hydrocarbons in fabric with outer and comfortable inner layers could be easily cleaned through conventional laundering regardless of PAH contamination level. The moisture barrier layer acted as the most powerful barrier that prevented the permeation and transmission of PAHs but adsorbed PAHs, resulting in inefficient cleaning through conventional laundering.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Application Innovation Project of Fire and Rescue Department Ministry of Emergency Management (2020XFCX24) and Key Research and Development Program of Tianjin (20YFZCSN00920).
