Abstract
The present work was designed to study several functions of Silk coated with polyaniline (PANI), graphene oxide (GO), and a composite of PANI@GO. The samples were characterized by XRD, SEM, FTIR, and thermal analysis
Introduction
Textiles with multi functionalities are increasingly demanded as a part of advanced and future marketing strategies. Global trends are shifting towards environmentally friendly fabrics. Therefore, natural fiber applications are gaining traction globally that have expanded the use of silk fiber beyond the conventional textile industry due to their unique properties and functionality over synthetic fibers and their sustainability. Silk is a natural protein polymer with reactive functional groups, allowing them to be cross-linked with other polymers to be used in several applications. Coating and lamination are two operational processes that are used to make a proper finishing to the textile material. Textile coatings usually provide material layers that adhere to the textile structures and extend the range of their functional performance properties. Properties of coated textile materials primarily depend on their application. Modern technologies, optimization of the production process conditions, and the use of specific agents and recipes enable making a target product that will meet all the requirements. Since it is impossible to use the classic textile for many technical purposes, excellent properties are obtained by combining it with other substances coated in the form of paste or laminated to the material. Using value-added material, nano products, and modern technology can enhance the use of textile materials several times.1–3 Several routes have been used to fabricate conductive textiles, such as creating metal fibers, coating the textile with metallic salts, and introducing intrinsically conductive polymers.4,5
Amongst these approaches, textiles coated with electrically conducting polymers are the most encouraging and simplistic methods for fabricating textile supercapacitors. Electric-heat conversion is a crucial way to be used in numerous applications such as heaters and thermoelectric power generators. Many thermal materials, such as conducting polymers, have been used for these applications. Polyaniline (PANI) is the most significant polymer due to its environmental stability, moderate conductivity, and diverse oxidation states. The in-situ chemical polymerization method is one of the various methods used for synthesizing polyaniline. 6 This method has some benefits over the other reported methods due to its eco-friendly, simplicity, and lower cost. The aniline monomer can be quickly deposited on the fabric surface. 6
Due to graphene oxide’s excellent dispersion and solubility, which contains many hydrophilic groups on its surface, it is easy to join with PANI.7–9 Thus, PANI@GO composite material turns out to be an appropriate applicant for the manufacture of functional fabrics by deposition and chemical polymerization methods. The considerable benefit of the presence of PANI in the composite is to increase the electrical conductance in the coated fabric.
Silk fabric is a highly water-absorbent material. Moreover, it is porous and lightweight. Therefore, the production of compressible silk supercapacitors integrated into clothes is worth investigating. The electrodes used for making supercapacitors should have a large area to provide appropriate energy. 10
Little research has been registered about conducting fabrics' electrical properties, and there is a shortage in their electronic fields. Therefore, the present work was done to prepare conductive silk based on low-price abundant raw materials and simple preparation methods but tried to improve the performances of its electronic and thermal properties significantly. We have used PANI-Silk, GO-Silk, and PANI@GO-Silk fabrics via in-situ chemical oxidation polymerization and vacuum filtration. Several techniques characterized the control silk and the coated silk samples. Furthermore, we studied the treated silk’s conductivity and thermal and electrochemical properties to test the possibility of using them as electrodes in supercapacitor devices. We also investigated the possibility of using the treated fabrics in UV protection and antimicrobial clothes.
Experimental
Materials
All chemicals are reagents used without further purification. Aniline, H2SO4 (98%), HNO3 (32.5%), potassium permanganate (KMnO4) (97%), sodium nitrate (NaNO3) (95%), and sodium hydroxide (NaOH) (98.5%) delivered from ADWIC, HNO3 (55%), and HCl (30%) supplied from DOP ORGANIK KIMYA SAN. VE TIC.LTD.STI, ammonium persulphate delivered from oxford (98%), graphite (99.995%) supplied from Fluka AG, and H2O2 (50%) provided from El Salam for chemical industries. The silk fabrics were purchased from the Egyptian market with a specification of warp and weft count: 23.3 dtex/2; warp density: 50 threads/cm; weft density: 45 threads/cm; and weight per unit area: 35 g/m2.
Graphene oxide preparation
Modified Hummers-Offeman’s method synthesized the graphene oxide (GO).11,12 5 g of graphite powder was placed in a flask. Then, 120 mL of H2SO4 was added by stirring in an ice-water bath, and 2.5 g NaNO3 followed by 22.5 g of KMnO4 was slowly added over about 2 h. The temperature of the reaction mixture should not exceed 20°C. After that, the flask was placed in an oil bath at 35°C for 2 h, and 250 mL of distilled water was added into the flask, which caused the temperature to increase to 95°C; after 1 h stirring, 50 mL of H2O2 was added, and the mixture was stirred for 2 h at room temperature. Finally, the mixture was centrifuged and washed with boiling distilled water until it became neutral. The resultant powder was dried at 60°C for 48 h to get the GO sample.
Preparation of GO-Silk fabric
First, the silk fabric was washed with 60°C deionized water to remove impurities before use. 10 mL of an ultrasonic dispersing aqueous solution of GO with a concentration of 5 mg mL−1 was poured onto the silk fabric fitted in the Buchner funnel of a suction flask (5 cm diameter) to fabricate the GO/silk material. After filtration, the composites were removed from the funnel and air-dried. The produced fabric showed black color and symbolized GO-SL.
Preparation of PANI-Silk coated fabrics
Silk fabrics were cut into a circle diameter of 2.5 cm and cleaned before the coating using ethyl alcohol for 30 min in an ultrasonic bath; after that, washed them with distilled water before drying in an oven at 45°C. The silk fabrics were coated with PANI via the precipitation method. This was done by oxidation of aniline dissolved in 1M nitric acid solution (0.2 M) in the presence of fabrics by 0.2 M solution of (NH4)2S2O8 under vigorously stirring under ultrasonication for 1 h. Finally, the fabrics were left overnight in the reaction mixture, washed with distilled water, and dried in an oven for half an hour at 60oC. After coating with polyaniline, the produced fabrics had dark green and symbolized PANI-SL.
Preparation of PANI@GO-Silk fabric
The prepared GO-Silk fabric sample was immersed in a beaker containing 80 mL anhydrous ethyl alcohol, nitric acid, and 20 mL aniline monomer for 90 min. Ammonium persulfate in a molar ratio with aniline and HNO3 of 1: 1: 0.5 was added gradually to the above solution. The polymerization procedure was carried out at 20°C for another 120 min to get silk fabric coated with dark green nanocomposites, symbolized as PANI@GO-SL.
Characterization
The untreated and treated silk fabrics were dried at 40°C for 24 h and kept in a desiccator before making any measurements. XRD analysis was carried out by a Diano (U.S.A.). The patterns were measured with CuKα radiation (λ ═ 1.5418 Å). The diffractograms were recorded at 2θ angles between 5 and 70°. The SEM images were taken using the scanning electron microscope (Philips SEM 515), where the accelerating voltage was kept at 3 kV. Prior to imaging, all the samples were coated with Leica EM ACE600 gold coater (Leica Microsystems Pty Ltd., Macquarie Park, NSW, Australia). An infrared spectrophotometer (Perkin-Elmer Model 883) was employed to record the ATR FT-IR spectra in the range of 4000-400 cm−1. The thermal stability of the silk and the PANI@GO-silk fabrics was measured by the TG/DTA instrument of Shimadzu model TGA-50H. In the experiment, argon was used as the carrier gas with a flow rate of 50 mL min−1, and the heating rate was controlled at 10°C min−1. Fabric tensile strength test was conducted according to ASTM method 1682 (1994) using an Electronic Universal Testing Machine (UTM), Instron 3369, at 30°C with a tensile speed of 0.009 mm/s. The fabric width and length were 50 mm and 200 mm respectively. Each sample was repeated five times. The UV- protection efficiency of the coated fabrics is analyzed at a wavelength between 280-400 nm. The mean UPF values were evaluated based on the Australian/New Zealand Standard (AS/NZS 4399:1996 Sun protective clothing–evaluation and classification, Standards Australia, Sydney). The heat generation capacity of the examined conducting fabric was measured using a 9 V battery as a power source connected to a square ship of the fabric (6 cm × 6 cm). The obtained current was measured using an electrometer model 6517, Keithley, and the temperature was determined using an Omega infrared thermometer sited in the middle of the sample. All the determinations of the heat generation were done in triplicate, and the results present mean values.
Electrical properties
The electrical properties were measured at a constant voltage (1 V), in a frequency range between103 and 106 Hz at 25°C, using a programmable automatic LCR bridge (HIOKI: 3532–50). The a.c. Conductivity (σac) is obtained from the data of dielectric constant (ε/) and loss tan δ using the relation σac = ε/εοω tan δ, where εο is the permittivity of vacuum and ω is the angular frequency. The ε/−values are calculated using the relation ε/ = (C/εο)x(t/A), where C is the equivalent capacitance, t is the thickness of the sample, A is the area of the specimen, tan δ is the dissipative factor. The bulk dc-conductivity σdc of the material was measured using an electrical circuit consisting of a voltameter (Keithley, 2182), an electrometer (model 6517, Keithley), and a 5 V dc power supply. The bulk dc-conductivity σdc of the material was estimated utilizing the equation: σdc = (t/A) × (1/Rdc); where Rdc is the resistance of the sample. The Surface resistance was also determined on the fabric sample (6 cm2) using the four-probe method, which indicates the ratio of the voltage drop between one pair of electrodes with current flow between the other pair of electrodes placed opposite each other in the case of square-shaped samples. The measurements were made using a combination of a low current DC source Keithley 6220 precision current source and a high resistance electrometer – Keithley 6517 electrometer. To measure the surface resistance, a small current ranging from 1 μA to 1 mA was passed through the sample, and the voltage drop was measured with the voltameter (Keithley, 2182). The surface electrical conductivity is evaluated using the relation: σs = (I/ ΔV) × (t/A); where ΔV is the measured voltage drop, I is the current passed through the sample. Each of the conductivity results is the observation of five repetitions, and the measurement error is estimated using the standard deviation formula. Electrochemical measurements were performed with an SP 300, BioLogic potentiostat by performing galvanostatic charge/discharge (GCD), cyclic voltammetry (CV), and electrochemical impedance spectroscopy (EIS) analyses.
Electrochemical Properties
Aqueous Supercapacitor
The silk electrode’s electrical capacitance was investigated by employing a three-electrode cell containing the saturated calomel electrode (SCE) as a reference electrode and the platinum sheet with a surface area of 2 cm2 as a counter electrode. The working electrodes were made up of pieces of fabrics where their upper edges were placed between two titanium plates to hang down with high stability in the electrolyte solution. The titanium plates were put to be not contacted with the electrolyte. The electrolyte used was a 1 M Na2SO4 solution. The experiments were done under a nitrogen atmosphere to remove oxygen from the solution.
Flexible solid-state supercapacitors fabrication
A solid-state symmetric supercapacitor was fabricated using a Swagelok cell with two PANI@GO-SL electrodes of the same mass and PVA-H3PO4 gel electrolytes. The two identical PANI@GO-SL electrodes were immersed in the H3PO4/PVA gel electrolyte for 10 min, then cured at room temperature over 4 h. The two electrodes were then tightly pressed for 10 min to allow the polymer gel electrolyte on each electrode to combine into one thin separating layer to form an integrated device. One cm-diameter disk was punched out of the fabric device and placed inside the Swagelok type two-electrode cells with stainless steel 316 current collectors. The PVA-H3PO4 gel electrolyte was prepared: 1 g of PVA was dispersed in 10 mL of double distilled water with stirring at 90°C to form a clear homogeneous solution. Next, 0.02 mol of H3PO4 was added to the solution with continuous stirring at room temperature for 24 h.
Antimicrobial Activity test
The silk fabric’s antimicrobial activity n reducing microbe growth was determined according to AATCC Test Method 100–2004. The tested micro-organisms are Escherichia coli (E. coli), S. aureus, and Candida Albicans. The initial micro-organism concentration used was about 105 CFU ml−1 for all experiments. The percentage reduction in the colony (R%) between the untreated and treated silks was determined after incubation for 24 h at 37°C by using the equation: R% = (B – A)/ B × 100, where B and A are the numbers of CFU ml−1 of micro-organisms determined for the untreated and the treated fabrics respectively.
Statistical analysis
All the quantitative data were expressed as a mean ± standard deviation (SD). The statistical analysis was carried out using one-way ANOVA, and a value of p < 0.05 was considered to be statistically significant.
Results and Discussion
XRD
XRD patterns of the coating materials (PANI, GO), raw silk (SL), and coated fabrics (PANI-SL, GO-SL, and PANI@GO-SL) are represented in Figure 1. The diffraction pattern of GO reveals a sharp peak at 2θ = 12.01°, attributed to the inter-planar spacing of GO sheets, and another peak at 42.3°, attributed to incomplete oxidation of graphite.
13
The X-ray diffraction of PANI shows mainly two peaks at 2θ = 20.3° and 24.9°, corresponding to (020) and (200) crystal planes of PANI related to the periodicity parallel and perpendicular of PANI chains, respectively.
14
The diffraction patterns of raw silk fabric show a prominent peak at 2θ = 20.5°, which is attributed to the exceptionally ordered β-structure of silk.
15
However, the XRD patterns of the treated silk fabrics with PANI, GO, and PANI@GO (composites), given in Figure (1), show the same characteristic peaks of the coating material with a slight shift in their positions, besides a significant difference in the diffraction peak of the silk observed at 20.5°. It was also observed that the intensity of the peak appearing at 24.48o in PANI was decreased in the PANI@GO-SL. This may be attributed to decreasing the fabric’s crystallinity as a result of some interactions of GO and PANI with the peptide linkages in the structure of silk fabrics.
16
XRD patterns of the investigated fabrics.
SEM
SEM analysis was done to confirm the presence of the coating materials on the Silk’s surface. The results obtained are shown in Figure 2. The SEM image of the raw silk fabric shows a smooth and uniform appearance (Figure 2(a)), whereas the photograph of the GO-SL sample (Figure 2(b)) shows some aggregation of GO particles on the silk’s surface. The micrograph of the silk fabrics coated with PANI (Figure 2(c)) shows an almost uniform surface covered with PANI sheets. On the other hand, the SEM image of PANI@GO-silk (Figure 2(d)) shows silk fibers coated with aggregated composite material. These results agree with XRD and FTIR results and elucidate the introduction of the coating materials in Silk fabric. The SEM image of PANI@GO-silk (Figure 2(d)) shows regular and uniform morphology with some aggregation. These results agree with XRD and FTIR results and elucidate the introduction of the coating materials in Silk fabric.
17
SEM image of (a) SL (b) PANI –SL (c) GO-SL (d) PANI@GO-S1.
FTIR spectra analysis
The FTIR spectra of the studied fabrics are recorded in 4000–400 cm−1 and illustrated in Figure 3. The spectrum of control silk fabric reveals the prominent peak’s vibrational modes: C=O at 1726 cm−1, N—H stretching at 1611 cm−1, C-N stretching at 1251 cm−1, and -CH3 stretching at 1446 cm−1. The FTIR spectra of coating materials (PANI and GO) powders were performed to assign the discrete impacts of these materials, which might be seen in the spectra of coated fabrics. The existence of various categories of oxygen functionalities in GO was approved by the presence of a band at 3300 cm−1 that can be ascribed to the O-H stretching vibrations of the C-OH groups and H2O,18,19 a band at 1600 cm−1 (skeletal vibrations from unoxidized graphitic domains),
20
and band at 1050 cm−1 (C-O stretching vibrations).
21
However, the FT-IR spectrum of the polyaniline sample demonstrates its most essential characteristic bands at 1488 cm−1 and 1570 cm−1, referring to C-N vibration of the benzenoid N–B–N and quinoid N=Q=N rings
22
(where B denotes the benzoic-type rings and Q represents to the quinolinic-type rings). These peaks approve of the formation of PANI. The peak detected at 1140 cm−1 is associated with the doped PANI chain with H+ .
23
The peak observed at 1100 cm−1 is a distinguishing for PANI conductance and the extent of the electron delocalization and is assigned to the vibration of C- H.
22
The band at 1295 cm−1 is agreed to the displacement of π electrons stimulated polymer by doping it by acid.
24
The spectrum of GO-silk shows bands at 1465 cm−1 due to C-OH stretching mode and C-C stretching mode at 1598 cm−1 and peaks at 1723 cm−1, 1690 cm−1 corresponding to C=O (RCOOH). Moreover, absorption peaks observed for PANI-Silk at 817 cm−1, 1170 cm−1, 1300 cm−1, and 1490 cm−1 are credited to the vibration of the C-H plane, C-N in PANI, and the aromatic C=C stretching vibration of the benzenoid ring, respectively. The change in the position and intensity of the absorption bands of untreated silk, shown in Figure 3, ensure that the silk fabric is coated by GO nanosheet and PANI. The coating materials seem to mask the fibroin band at 965 cm−1 characterized of −gly−gly− and −gly−ala− peptide backbone motifs.
25
FTIR spectra of the investigated fabrics.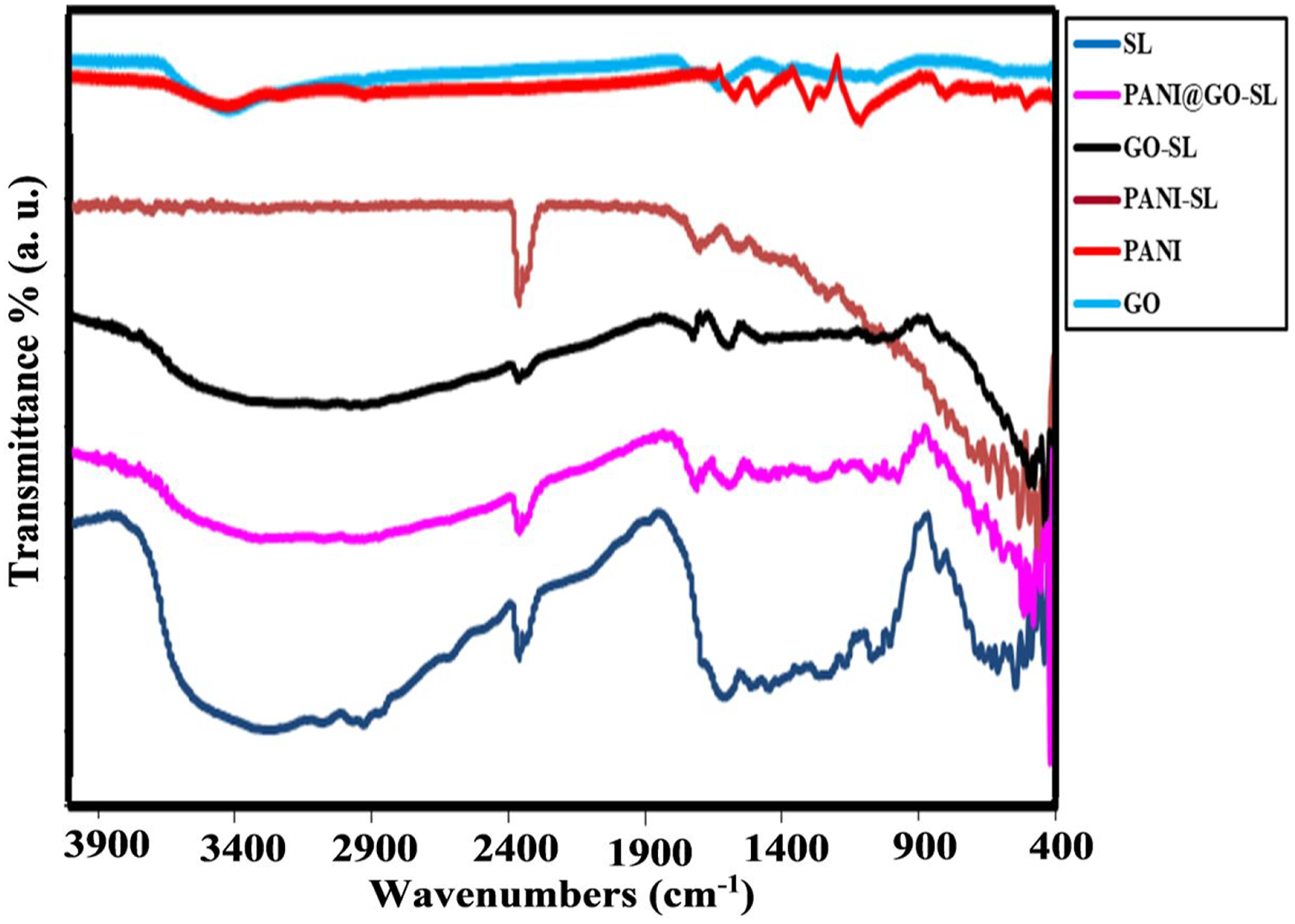
Thermal analysis
Thermal gravimetric analyses (TGA) were used to study the thermal stability of the silk (SL) and treated silk. The thermograms obtained are given in Figure 4. It shows that the thermal degradations for each sample occur mainly in several stages. The lower steps (at temperatures less than 250°C) are attributed to the loss of adsorption water and dopants and found to be 8.2, 8.4, 8.7, 9.1, and 9.5% for SL, SL-GO, SL-PANI, and PANI@GO-SL samples, respectively. The TGA steps observed at higher temperatures (more than ∼250 o) are ascribed to the degradation of the fabrics. The second stage appeared at a temperature range of 300–380°C showing a weight loss of 44.5.47.1, 47.9, and 49% are ascribed to starting the degradation of the silk proteins (sericin and fibroin) skeletal, PANI, and GO chains for SL, GO-SL PANI-SL, and PANI@GO-SL samples, respectively. The third step is attributed to the complete degradation of the samples. The TGA - results show that the thermal stability decreases in the order: SL > GO-SL > PAN-SL, and PANI@GO-SL. The decrease in the thermal stability of the silk fabric after its coating with GO and PANI refers to some interactions occurring between the coating materials and the silk causing weakness in the bonds of the fabric.
26
The results also show that the weight loss of coated fabrics is faster than that of untreated ones. From the remaining char, it is found that the percent of the undegradable part of the samples is 5.4, 5.45, 5.55, and 5.6% for SL, PANI-SL, GO-PANI, and PANI@GO-SL, respectively. TGA thermograms of the investigated uncoated and coated silk.
Mechanical properties
Tensile strength and antimicrobial data of the investigated fabrics.

Wrap value ( ).
Laundering durability
Weight changes of the investigated fabrics before and after washing.

Heat Generation from Fabrics
One of the possible applications of the made coated silk fabrics, especially those exhibit high conductivity, is their use in heating devices. A setup, shown in Figure 5 (insert), was operated to measure the heat-generating capacity of the studied conducting silk. A 9 V battery as a power supply was connected to a square shape fabric (7 cm × 7 cm). The increase in temperature was recorded employing an Omega infrared thermometer located at the midpoint of the fabric. The change in temperature with time is illustrated in Figure 5. The results showed that a conventional 9-V battery could be used to warm the silk fabric treated with the different coated materials, and the heat generated decreases in the order: PANI@GO-SL > PANI-SL > GO-SL > SL. The current drawn in this experiment is very low. Thus, the lifespan of a battery will be high, and an on/off switching unit within the person’s clothing can utilize the power carefully as per needs. Heat-generation behavior of the investigated fabrics under voltage of 9V. The inset figure is the setup of the heating circuit.
UV radiation protection
To our knowledge, there are no studies on UV radiation protection for silk fabric coated with GO and PANI. The ultraviolet protection factor (UPF) for all the investigated samples was determined at a wavelength between 280-400 nm and found to be 37.1 ± 0.4, 44.3 ± 0.5, and 48.5 ± 0.4 for GO-Sl, PANI-SL, and PANI@GO-SL, respectively. They are higher than that of silk fabric (UPF rating at 4.6). The results indicate that the composite layer of PANI@GO on the silk surface causes a more reduction in the transmittance of ultraviolet light than that of other individual coating materials.
Antimicrobial activity
The Gram-negative Escherichia coli (E. coli), Gram-positive Staphylococcus aureus (S. aureus), and Fungi Candida Albicans are used to test the antimicrobial activity of the investigated fabrics using AATCC Test Method 100–2004. The acquired results are given in Table 1, which shows that all coated silk samples have antimicrobial activity decreases in the order: PANI@GO-SL > PANI-SL > GO-SL > SL. It is thought that the electrostatic attraction between the negatively microbial cell and positively charged nanomaterials (such as PANI in our study) is an essential parameter for their antimicrobial activity. 27 The antimicrobial mechanism of GO is explained based on the fact that GO can destroy the microbial membranes, leading to the efflux of intracellular material and killing microbes.28,29 It can also be said that the hydroxyl groups in GO interfere with microbial metabolism by piling at the cell surface and attaching with DNA to hinder m-RNA synthesis .29, 30 The difference observed in the antimicrobial efficiency values can also be attributed to the dissimilarities in the structures of the microorganisms.
Electrical properties
dc Conductivity
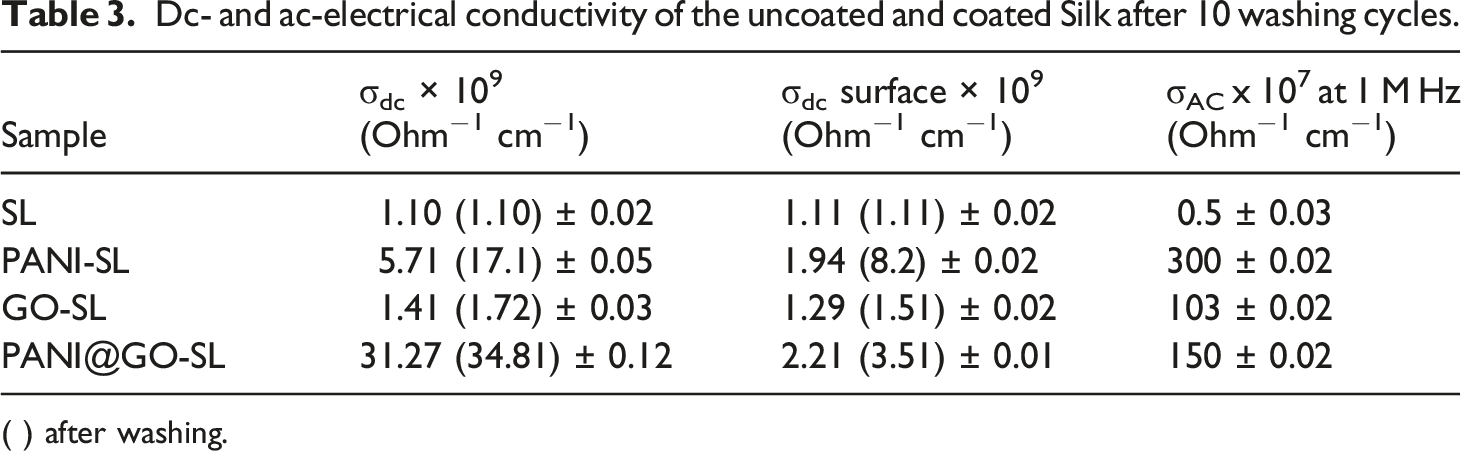
The electrical conductivity properties of the studied fabrics were also investigated. I–V plots of the uncoated and coated silk in a voltage ranging between −10 to +10 V show linear I–V relations for all the coated silk samples. The bulk (σdc) and surface dc-conductivity (σdc/Su) of all the investigated samples, before and after the sample. The obtained results are shown in Figure 6 and Table 3, which show that σdc values of the coated samples are higher than that of the uncoated sample. The initiation of conductivity within silk matrix fabric is attributed to the diffusion of PANI and GO inside the fabrics and their coating on the silk surface to form a conducting path in the fabric. It can be said that the observed enhancement in conductivity could be attributed to two supplies, namely microscopic and macroscopic conductivities which are participating in the improvement conduction process.
31
The microscopic conductivity hangs on several factors, such as conjugation and chain length. On the contrary, the macroscopic conductivity is dependent on external factors such as molecular orientation. The conjugated structure of PANI chains with high conductivity and high surface area is also considered the main reason for increasing the conductivity of the treated fabric containing PANI The surface and bulk conductivity of the investigated fabrics after washing (a) and before washing (b). Dc- and ac-electrical conductivity of the uncoated and coated Silk after 10 washing cycles. ( ) after washing.
ac Conductivity
The ac-electrical conductivity (σac), at a frequency range of 103–107 Hz, was investigated to get information on the polarization in the coated silk. The σac - value was evaluated using equation (1)
32

The frequency dependence of the ac-conductivity of SL, PANI-SL, GO-SL, and PANI@GO-SL is given in Figure S2. It shows that the electrical conductivity value (σac), at each measuring frequency decreases in the order: PANI-SL > PANI@GO-SL > GO-SL >SL
We can attribute the lower conductivity values of the samples containing GO compared with that of other coated pieces to the lower electrical conductivity value of GO than PANI; besides, it can make barrier ways to prevent the moving of electrons in the PANI@GO-SL sample. The lower conductivity values of the samples containing GO compared with that of other coated pieces may be attributed to GO’s lower electrical conductivity value than PANI; besides, it can make barrier ways prevent the moving of electrons in the PANI@GO-SL sample. Figure S2 also shows that as the frequency increases, the ac-conductivity increases. This is because the frequency acts as a pushing force for charges and facilitates the polarization processes.
Electrochemical Study
The capacitance behavior of the uncoated and coated silk electrodes was studied using cyclic voltammetry (CV) and galvanostatic charge-discharge (GCD). Figure 7 depicts the CV curves for the investigated electrode samples at a scan rate of 10 mVs−1. It is evident from the Figure that the silk electrode sample shows a rectangular shape that refers to a double-layer capacitance. In contrast, the coated silk electrodes containing the PANI, GO, and GO/PANI composites electrodes exhibit two pairs of redox peaks representing their pseudocapacitive characteristics. The redox peaks of the samples containing PANI are attributed to the redox transition of PANI from a semiconducting state (leucoemeraldine form) to a conducting state (polaronic emeraldine form) and faradaic transformation between the emeraldine and pernigraniline states .
33
The specific capacitance values Csp were calculated from CV plots using the following equation
34
Cyclic Voltammetry of the undoped and doped silk at 10 mV/s in 1 M Na2SO4.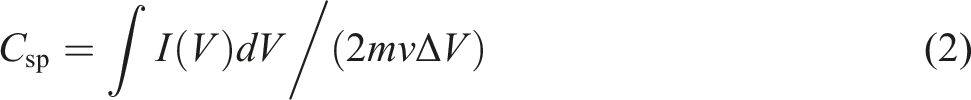

The highest capacitance of the binary composite electrode, PANI@GO-SL, is attributed to a synergetic effect of the individual component in the composite material and the contribution of double-layer capacitance with the pseudocapacitance. The composite electrode exhibits numerous distinctive characteristics causing it to be an encouraging applicant for high-performance electrode materials. These properties involve: (a) the porous structure of the textile permitted uniform coating of GO and the following loading of PANI. Accordingly, it simplified the connection of electrolyte ions to the electrode surface. (b) Across in-situ electrochemical polymerization, a good interface contact between PANI and GO can be produced to realize rapid electron transfer. (c) PANI guaranteed high pseudocapacitive capacitance and a small path ion diffusion throughout the charge-discharge procedure. Since the PANI@GO-SL electrode exhibits the highest capacitance of all studied coated electrodes; therefore, it was further characterized under different scan rates. Figure 8 shows the CV curves of PANI@GO-SL at the scan rates from 1 to 50 mV s−1. As the scan rate increases, the oxidation and reduction peaks shift to higher and lower potentials. Since the area within a CV curve is directly proportional to its specific capacitance, therefore, it can be seen that the specific capacitance decreases as the scan rate increases. This could be attached to the large ohmic resistance at the high scan rate. Besides, the electrolyte ions do not have enough time, at high scan rates, to fully diffuse through the electrode nanopores wherever the faradaic reactions occur. Electrochemical properties of symmetric cell PANI@GO-SL// PANI@GO-SL (a) Cyclic Voltamogram at different scan rates, (b) GCD at different current densities, (c) Stability of the symmetric cell during 5000 cycles, (d) Nyquist plots and (e) Ragone plot.
Using PVA/H3PO4 gel as a solid electrolyte, two PANI@GO-SL (highest capacitance of all the electrodes) electrodes were collected into a solid-state symmetric supercapacitor. Figure 8(a) demonstrates the CV curves of the supercapacitor at different scan rates. The figure shows high capacitance rising from the high conductivity of GO and the large pseudocapacitance of PANI. The capacitance increases with increasing scan rates. The device supplies capacitance values of 74, 72, 70.5, 69.4, 68.1, and 66.9 Fg−1 at scan rates of 1, 2.5, 5, 10, 20, and 50 mVs−1.
The galvanostatic charge-discharge (GCD) behavior of the symmetric solid capacitors was also studied using different current densities. The obtained results are shown in Figure 8(b). The departure in the linearity of the discharge plots can be attributed to the pseudocapacitance mechanism resulting from the faradic process.
The cell capacitance (Ccell) values were calculated from the galvanostatic discharge curves using equation (3).
35

The cyclic stability of the symmetric cell was also tested by GCD measurement for 5000 cycles at a current density of one Ag−1 (Figure 8(c)). The capacitance remained 87.4% after 5000 cycles. The gradual decrease of capacitance was possibly attributed to drying the gel electrolyte.
Figure 8(d) demonstrates the Nyquist plot of the symmetric PANI@GO-SL supercapacitor. The figure shows a semicircle in the high-frequency region. The intersection point between the semicircle and the Z//- axis is about 0.38 Ω, representing a low equivalent series resistance (ESR). The semicircle diameter represents the charge-transfer resistance (Rct) produced from the Faradic reaction, and electrochemical double-layer capacitance is found to be 2.7 Ω. The fast rise at the nearly vertical line at the low-frequency region is due to the capacitive behavior. From the maximum frequency (f°, Hz) at Z// on the semicircle in the impedance spectrum, the time constant (τ, s) of the symmetric capacitor is determined using the equation: τ = 1/f°
35
and is found to be 7.2 ms. The small-time constant reveals the high power response of the device. The specific energy density and power density of the symmetric PANI@GO-SL supercapacitor device are calculated by equations (4) and (5),
34
respectively

A comparison between electrochemical data of some supercapacitors found in literature and the data of the present work.

Conclusions
The coating is a powerful tool for the advancement of textile technology. It provides the opportunities to produce unique fabrics for Medical applications, heat-sensitive fabrics, automotive fabrics, disposable hospital apparel, etc. The present study reports a superficial and eco-friendly methodology for preparing coated silk fabrics by GO, PANI, and PANI@GO nanocomposite. The silk surfaces of the raw and treated fabrics have been characterized using XRD, ATR-FTIR, SEM, and TGA The mechanical properties, dc-, ac-electrical conductivity, electrochemical properties, and antimicrobial tests were also studied. The FT-IR spectra approved the functional groups of the investigated samples, while XRD spectra reveal that the investigated coating material and fabrics possess amorphous structures. SEM studies showed that the coating materials were deposited on the treated fabric surface. Comparing the electrical conductivity values of the raw and treated fabrics refers to the penetration of coating materials inside the fabrics. The treated fabrics showed high UV protection, an improvement in the fabric’s tensile properties, and antimicrobial efficiency against Escherichia coli (E. coli), S. aureus, and Candida Albicans in the order: PANI@GO-SL > PANI-SL > GO-SL > SL. The electrical capacitance of the uncoated and coated silk in 1 M Na2SO4 showed an increase in the specific capacitance in the same order observed for antimicrobial efficiency. Symmetric solid supercapacitor using PANI@GO-SL electrodes and H3PO4/PVA gel showed a specific capacitance of 71.2 F/g at a current density of 1 A/g. The symmetric cell exhibits a maximum energy density of 25.31 kWh/kg and a maximum power density of 8018 W/kg. The obtained results are significant because they may allow using coated fabrics in industrial applications. Generally, the treated silk fabrics improved electrical conductivity, UV-protection ability, antibacterial activity, and other physical properties—including tensile properties. Therefore, it can be said that the investigated coated silk with multifunctional properties has high potential in the textile industry.
Supplemental Material
Supplemental Material - Preparation and physical properties of conductive silk fabrics used in wearable clothes and flexible supercapacitors
Supplemental Material for Preparation and physical properties of conductive silk fabrics used in wearable clothes and flexible supercapacitors by M Khairy, R Kamal and MA Mousa
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
