Abstract
A novel high-performance electrode material based on fluoro-functionalized graphene oxide (GOF)/polyaniline (PANI) composites was synthesized via in situ polymerization of monomer in the presence of GOF. The composite was characterized by Fourier transform infrared spectrometry, X-ray diffraction (XRD), and the field emission scanning electron micrographs. The electrochemical properties of the composites were measured based on cyclic voltammetry, galvanostatic charge/discharge, and AC impedance spectroscopy. When the obtained composites with a mass ratio is 3% GOF/PANI, the results showed that GOF as a support material provides additional electron transfer paths, as well as active sites, for the electrochemical redox reaction of PANI, which helps to increase its pseudocapacitance. A specific capacitance of 502 F·g−1 was obtained at a current density of 1.0 A·g−1 in 1 mol·L−1 H2SO4, as well as excellent cycling stability with 95% of its initial capacitance after 600 cycles. These encouraging results illustrate the exciting potential for high-performance, electrical energy storage devices based on this new class of carbon material.
Introduction
The increasing popularity of various portable electronic devices and motor vehicles has increased the demand of energy storage devices. In this regard, different batteries and high-performance capacitors are the focus of the scientific community. Supercapacitors, also called electrochemical capacitors or ultracapacitors, based on electrochemical double layer capacitance are electrical energy storage devices that store and release energy by nanoscopic charge separation at the electrochemical interface between an electrode and an electrolyte. 1 In addition, supercapacitors have excellent advantages over conventional batteries, such as high power density, long cycle life, and low maintenance. 2,3 However, the supercapacitor with high performance highly depends on the properties of electrode materials. 4 In recent years, people have devoted many efforts in graphene-based materials due to their outstanding physical and chemical properties (high theoretical surface area, good electrical conductivity, and open porous structure). 5 However, graphene nanosheets derived from chemical reduction of graphene oxide (GO) often show the aggregation or restacking due to the intersheet van der Waals attractions, resulting in the loss of their unique property. 6,7 GO, one of the most important derivatives of graphene, is characterized by a layered structure with oxygen functional groups bearing on the basal planes and edges, which promotes pseudocapacitance. At present, some work has been carried out on the application of GO 8 -11 and carboxyl-functionalized graphene oxide in supercapacitors. 12
However, the pure graphene-based supercapacitor materials actually exhibit unsatisfactory capacitance because of the inevitable aggregation of the graphene sheets. 5,13 Polyaniline (PANI), owing to its good electrochemical activity, high reversible pseudocapacitance, thermal stability, and environmentally friendly properties, has shown great potential for supercapacitor applications. 14 Composite materials based on the combination of graphene (or GO) and PANI are expected not only to improve the PANI conductivity, but also relieve graphene aggregation via a synergistic effect. To date, a great number of papers have reported graphene (or GO)/PANI composites for supercapacitors. 15
Herein, we present an approach of using fluoro-functionalized graphene oxide (GOF)/PANI composites as electrode material for supercapacitors by in situ polymerization. GOF was prepared in our previous work. 16 As a functionalized GO, GOF have increased average interlayer spacing, excellent dispersion in water and wetting performance, which is the prerequisite of dispersion of functionalized GO in PANI. In addition, the fluorine and other oxygen-containing functional groups improve pseudocapacitance for supercapacitors.
Experiment
Preparation of carboxylated graphene oxide (GOCOOH)
GO was prepared by a modified Hummers’ method. GO aqueous suspension (1 mg mL−1, 250 mL) was dispersed into ultrasonic treatment for 1 h. After the GO was entirely dispersed, sodium hydroxide (10 g) and sodium chloroacetate (ClCH2COONa, 14 g) were added to the GO suspension and a magnetic stirring at room temperature for 12 h, and the pH of the solution was adjusted to 5–6 by dilute hydrochloric acid. Then the mixture was filtered and washed with anhydrous ethanol and a large amount of distilled water in sequence. Then the samples were collected and dialyzed against distilled water for 2 days to remove any impurities. To prepare freeze-dried GOCOOH, the obtained GO colloid was directly subjected to freeze-drying for 24 h on a vacuum freeze dryer.
Preparation of GOF
To a stirred solution of GOCOOH (20 mg) in distilled water (50 mL) were added Selectfluor (100 mg) and silver nitrate (10 mg), and the reaction mixture was stirred at 90°C for 10 h under an atmosphere of nitrogen. Then the mixture was filtered under vacuum, and the filtrate was extracted with ethyl acetate (3 × 15 mL) to remove organic impurities. Then the water layer repeated centrifugation for 15 min at 8000 r min−1 until the silver ion has not been detected in the process. Then the samples were collected and dialyzed against distilled water for 2 days to remove any impurities. The obtained GOF colloid was directly subjected to freeze-drying for 24 h on a vacuum freeze dryer.
Preparation of GOF/PANI composite materials
To a stirred solution of GOCOOH (20 mg) in distilled water (50 mL) were added Selectfluor (100 mg) and silver nitrate (10 mg), and the reaction mixture was stirred at 90°C for 10 h under an atmosphere of nitrogen. Then the mixture was filtered under vacuum, and the filtrate was extracted with ethyl acetate (3 × 15 mL) to remove organic impurities. Then the water layer repeated centrifugation for 15 min at 8000 r min−1 until the silver ion has not been detected in the process. Then the samples were collected and dialyzed against distilled water for 2 days to remove any impurities. The obtained GOF colloid was directly subjected to freeze-drying for 24 h on a vacuum freeze dryer. Then it was dissolved into deionized water to prepare GOF by ultrasonic treatment. PANI was synthesized by the oxidation polymerization. 17 The weight ratio of GOF to aniline is 1:100–5:100 (labeled as 1% GOF/PANI to 5% GOF/PANI). The GOF was dispersed in aqueous solution (2.0 mg mL−1, 30.0 mL) by ultrasonication. Aniline (2.76 mL) in 1.0 mol·L−1 HCl was introduced. The dispersed solution was ultrasonicated for another 1 h. Then 1.71 mg of ammonium persulfate dissolved in 1.0 mol L−1 HCl was dropwise added into the reaction system and stirred continuously for 24 h. The precipitates were collected by filtration and repeatedly washed with deionized water, ethanol, and hexane in sequence until the filtrates were colorless, and dried at 50°C in vacuum for 24 h.
Characterization
X-ray diffractometry (XRD: PANalytical X’Pert Powder) was performed using Cu Kα radiation of 40 kV. Fourier transform infrared (FTIR) spectrum (BRUKER, IF660V) analysis was carried out in a FTIR-N3896. The field emission scanning electron micrographs (FESEM) of the samples were obtained on a SU8020 (Hitachi). Electrochemical workstation (PARSTAT 2273, AMETEK) was used to evaluate the electrochemical performance of samples; the samples were tested in a standard three-electrode test system. The counter electrode was a piece of platinum foil, the reference electrode was saturation mercury electrode, and the working electrode was assembled from the GOF/PANI composites as follows. Electrode material was made of the composites (75 wt%), conductive agent (Super P Li:KS6 = 1:5, 20 wt%), and polytetrafluoroethylene (5 wt%). The mixture was painted on stainless steel sheet current collectors and pressed under 15 MPa to make the electrode.
Results and discussion
Figure 1 shows the FT-IR spectra of GOF, PANI, and 3% GOF/PANI composites. For GOF, the absorption bands at 1646 cm−1 and 1070 cm−1 are ascribed to the C=O stretching of the –COOH and the C–O stretching of the C–OH/C–O–C groups, respectively. The sample GOF has a weaker C–F stretching absorption at 1269 cm−1. FT-IR spectrum of 3% GOF/PANI composite paper (Figure 1(a)) showed a group of typical bands of PANI; C=C stretchings of the quinonoid ring and benzenoid ring at 1565 and 1484 cm−1, respectively; C–N stretching of secondary aromatic amines at 1290 cm−1; and C–H bendings of the benzenoid ring and the quinonoid ring at 1229 and 1126 cm−1, respectively. XRD patterns GOF and GOF/PANI composites are shown in Figure 1(b). The as-prepared PANI exhibits a wider reflection peak at 2θ = 9.88°, 14.46°, 20.18°, and 25.55°, with the characteristic peak of PANI. The GOF/PANI has the same crystalline phase as pure PANI. The GOF/PANI composites have no characteristics peak of GOF in FT-IR and XRD, which may be caused by the low content of GOF.
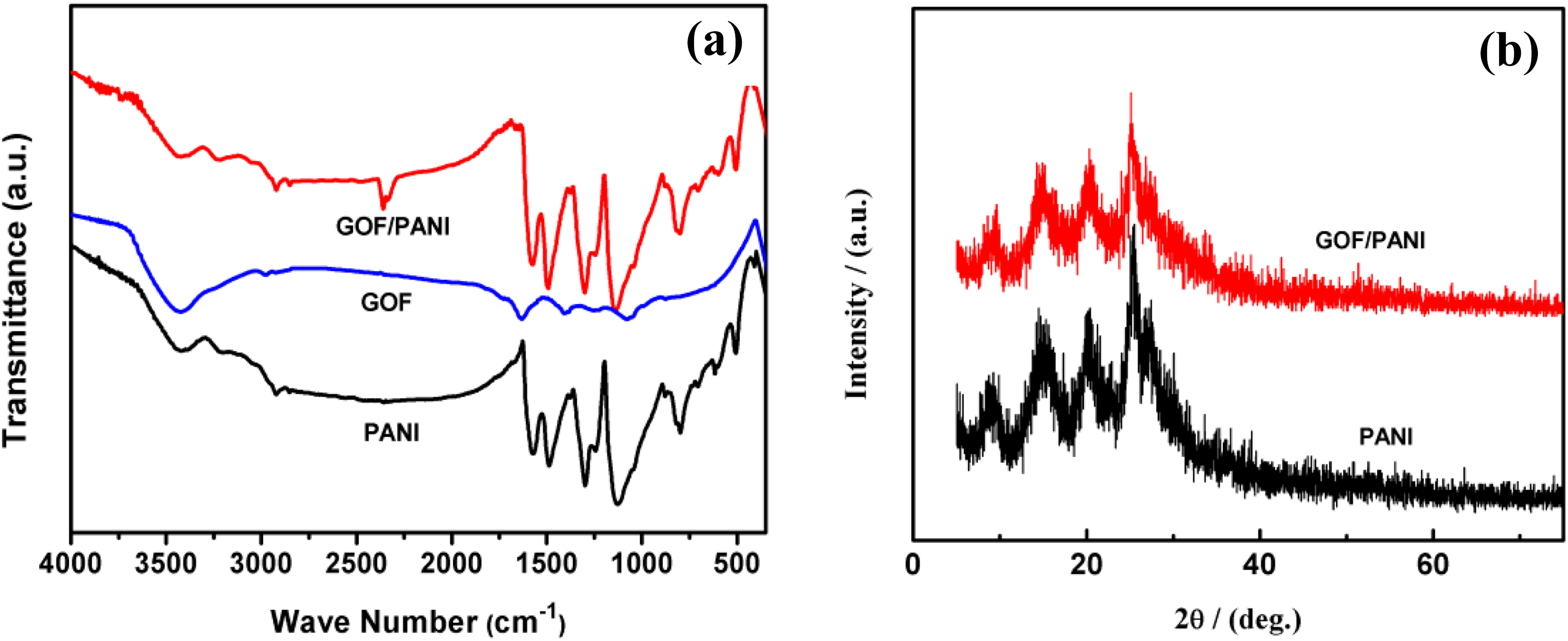
(a) Fourier transform infrared spectra of PANI, GOF, and 3% GOF/PANI composites, and (b) XRD patterns of PANI and 3% GOF/PANI composites.
The morphology of the prepared 3% GOF/PANI is imaged by FESEM in Figure 2. From the FESEM images of 3% GOF/PANI, it can be seen that graphene layers are irregularly curled or entangled (Figure 2(a)). In addition, the composites exhibited a homogeneous hierarchical morphology of a layered structure and granular PANI densely packed on the GOF plane (Figure 2(b)). Although there exists a portion of agglomerate morphology (Figure 2(c)), the composites have a good aqueous dispersion and have agglomeration under solvent-free condition. FESEM indicates that PANI is adsorbed on GOF sheets without disturbing graphene structure (Figure 2(d)).

FESEM images of 3% GOF/PANI composites.
To evaluate the electrochemical performances of the samples, cyclic voltammetry (CV) was first carried out in 1 mol·L−1 H2SO4 aqueous solution with a three-electrode system. The CV curves of composite samples with different GOF contents of GOF/PANI at a scanning rate of 1 mV·s−1 are shown in Figure 3(a); it can be observed that the CV curves of the composites exhibit capacitance contributions from both electric double layer capacitance and pseudocapacitance. 18 Curves of 1–5% GOF/PANI exhibit redox peaks, which are characteristic peaks of PANI. 19 Moreover, the areas of CV curves of the composites show 3% GOF/PANI has the largest CV area among the composites. The result suggests that the 3% GOF/PANI composite possesses larger capacity than other contents of GOF composites. In addition, at a current density of 1.0 A·g−1, the specific capacitance (Cs) of 1% GOF/PANI, 2% GOF/PANI, 3% GOF/PANI, 4% GOF/PANI, and 5% GOF/PANI is 284, 372, 502, 362, 335 F·g−1, respectively (Figure 3(b)); 3% GOF/PANI has the highest capacitance among the composites. The Cs values 20 were calculated from the charging and discharging curves (Figure 3(b)) according to Cs = I/m*(dV/dt), where I is the constant discharge current, m is the mass of the active materials within the electrode, and dV/dt can be obtained from the slope of the discharge curve. Whereas 3% GOF/PANI exhibits the highest capacitance than the other composites, and the results match well with the CV performances in above. The high capacitance of 3% GOF/PANI is attributed to the following reasons: (1) direct and close contact between GOF and PANI, resulting in high charge–transfer rate in the electrode material; (2) without the pseudocapacitance of PANI, high concentration of oxygen functional groups and fluorine atom on the graphene can provide high pseudocapacitance.
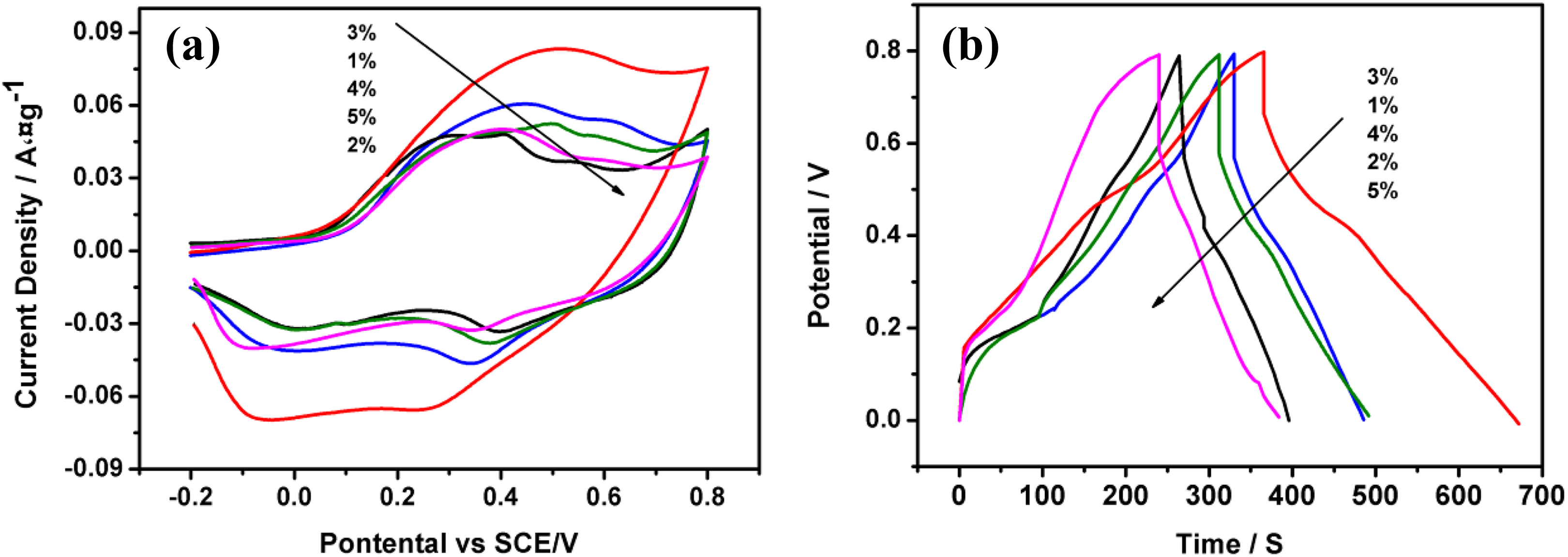
(a) Cyclic voltammetry curves of GOF/PANI composites with different contents of GOF at a scanning rate of 1.0 mV·s−1 and (b) galvanostatic charge–discharge curves of GOF/PANI composites with different contents of GOF obtained at a current density of 1.0 A·g−1.
Figure 4(a) shows galvanostatic charge–discharge curves of 3% GOF/PANI composites at different current densities (0.5–5 A·g−1). The discharge plateaus can be obviously observed at various current densities due to the redox reaction; 3% GOF/PANI was able to discharge Cs of 523, 502, 454, and 347 F·g−1 at current density of 0.5, 1, 2, and 5 A·g−1, respectively, with about 66% capacitance retention when current density increased by 10 times, indicating that 3% GOF/PANI composites exhibited good electron and ionic migration performance. To obtain a comprehensive understanding of the capacitive response of the 3% GOF/PANI composites, an electrochemical impedance test was conducted and the results are shown in Figure 4(b). The nearly vertical arm of the AC impedance in the low frequency region indicates an excellent capacitive behavior, representative of fast ion diffusion and adsorption in/on the electrode material. 21 The low resistance and fast ion diffusion may be attributed to the special properties of PANI and functionalized graphene in 3% GOF/PANI composites which may render this composite material very promising as a supercapacitor electrode. 22 At high frequencies, the diameter of high-frequency semicircle for 3% GOF/PANI composites is a little smaller than that for pure PANI, indicating that the 3% GOF/PANI electrode system has relatively higher electrical conductivity.
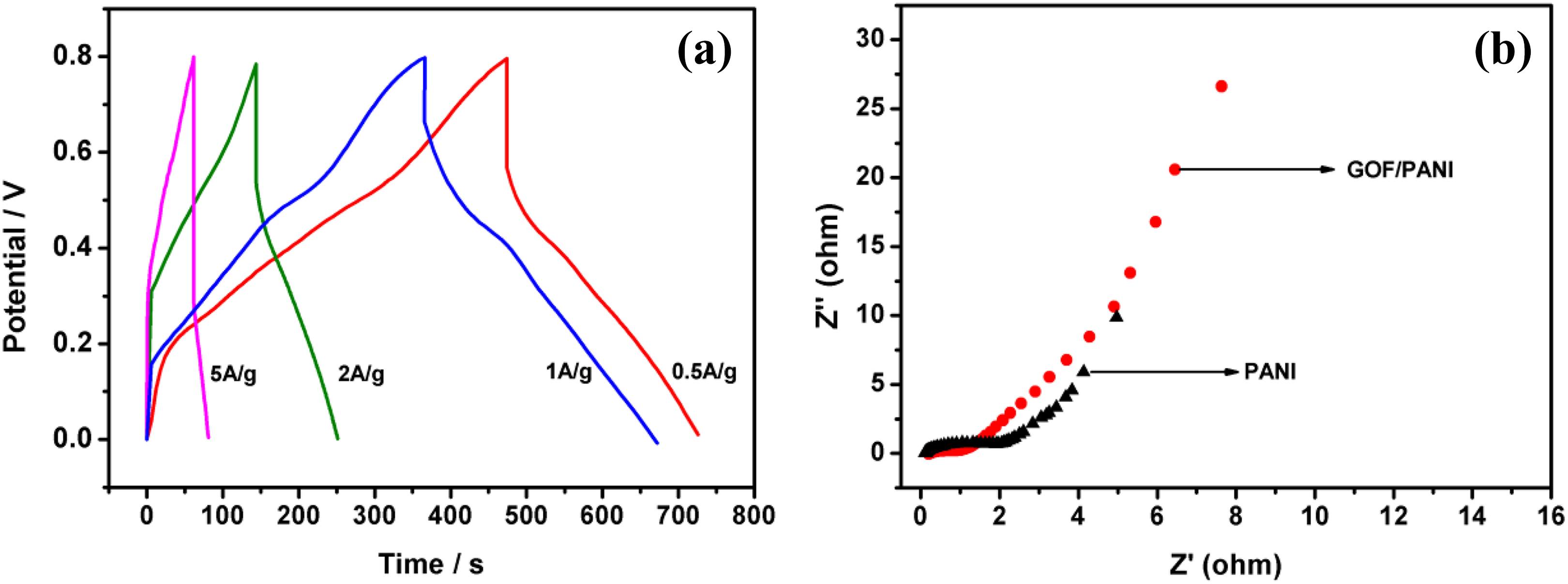
(a) Galvanostatic charge–discharge curves of 3% GOF/PANI composites under different current densities and (b) electrochemical impedance spectroscopies of 3% GOF/PANI composites and pure PANI in the frequency range of 10−2–105 Hz.
The long life cycle of supercapacitors is also a crucial parameter for their practical application. Figure 5 presents the Cs of 3% GOF/PANI composites as a function of charge–discharge cycle numbers at a specific current of 1 A·g− 1 in 1 mol·L− 1 H2SO4 aqueous solution. The Cs of 3% GOF/PANI decreases slightly with continuous cycles, and the initial value of the composite can maintain 95% after 600 cycles, which demonstrated good electrochemical stability of this electrode material.
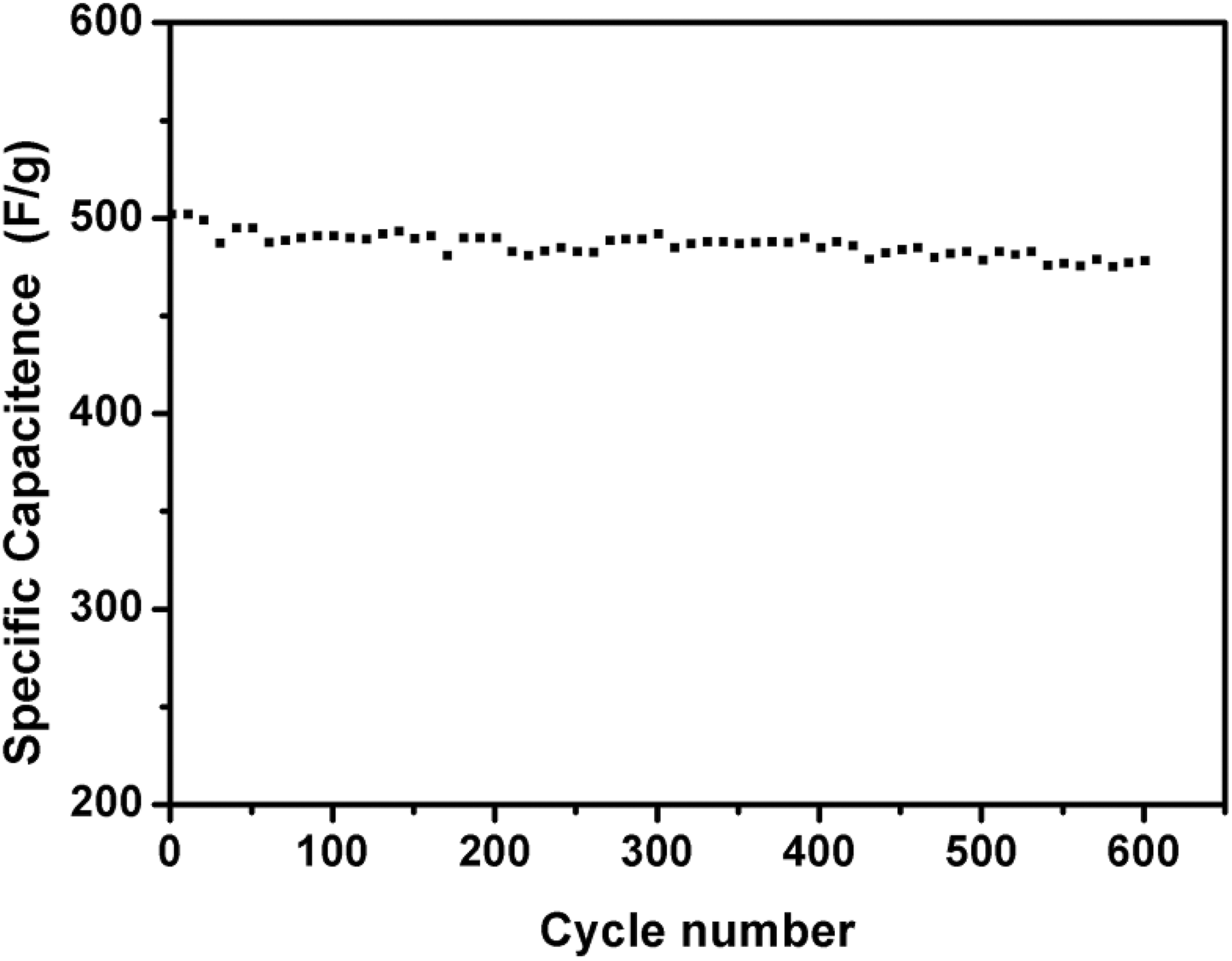
The cycle lifetime of 3% GOF/PANI composites at 1 A·g−1.
Conclusions
The GOF/PANI composites were prepared via a simple in-situ polymerization approach without the use of any templates or surfactants. GOF/PANI composite suitable for use as a supercapacitor electrode material has been constructed and characterized. The GOF/PANI composite with a mass ratio of aniline/GOF is 3%, possessed the highest Cs of 502 F·g−1, higher than that of the pure PANI. In addition, 3% GOF/PANI composites had a good cycling stability. The main origin is coming from PANI and the surface oxygen containing fluorine groups, leading to large pseudocapacitance. Therefore, the GOF/PANI composite is expected to be a very promising supercapacitor material.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Inner Mongolia Autonomous Region (No. 2018LH05020, 2018LH02003).
