Abstract
In the present work, we unveil a facile and effective method to directly grow Ni–Mo–Co oxy-hydroxide–based 3-dimensional hierarchical nanostructures on carbon microfibers (nano-on-micro) by using a facile hydrothermal synthesis route. Further, the electrochemical activity for directly grown fiber electrode as well as electrode formed by slurry coating of active material formed after hydrothermal reaction has been investigated. In this study, the metal ratios (nickel and cobalt) were selected to cover the wide spectrum of the concentration in order to obtain the optimum concentration for the best electrochemical performance. Electrochemical analysis of these ternary metal oxy-hydroxide–based active materials on the carbon microfiber shows significantly high electrochemical activity with a specific capacitance of 519 Fg−1 in hydrothermally activated sample and 890 Fg−1 in a slurry coated sample (at 1 Ag−1). This simple technique provides a novel method to fabricate high energy-storage devices with the advantage of being lighter and flexible and can be easily integrated for various flexible electronic applications potential applications including e-textiles, personal electronics, military apparel devices, and antimicrobial and biomedical textiles.
Introduction
The rapid development of wearable and portable electronics has increased the need for flexible, lightweight but high-performance energy-storage devices.1,2 They have notable application potential in the field of micro-robotics, e-textiles, energy harvesting, and medical devices.2–5
Fiber-based supercapacitors have been broadly studied for numerous wearable applications such as military garment devices, antimicrobial textiles, personal electronics and biomedical textiles, flexible optical and electronic devices, and chemical and biological sensors. Devices prepared from fibers exhibit greater electrochemical performance and good mobility. 6
Batteries and supercapacitors are the most extensively used energy-storage devices, among which batteries possess greater energy density and lesser power density, though they have a long charging and discharging time. 7 Supercapacitors have gained greater interest as energy-storage devices, due to their prompt charging, power delivering capability, and great power density. 8 The efficiency of the supercapacitor usually depends upon the active electrode materials, and they can typically be classified into three main types: carbon materials, oxides/hydroxides of transition metals, and conducting polymers. 9 Due to large flexibility and smaller volume, yarn/fiber-shaped supercapacitors can be easily twisted, stretched, or woven, which facilitates easy integration with the device. 10 Hence, it is easy to integrate an energy-storage system in a textile configuration like with ordinary clothes; thus, wearable electronics have attracted significant attention. Various textile yarns such as cotton fiber, carbon material–based fiber, Kevlar fiber; polymer nanofibers, and metal wires are used by various researchers for preparing fiber-shaped electrodes.11–16 The rapid developments of the fiber-based supercapacitor have been extensively summarized in several recently reviewed papers.10,17,18
Carbon materials, including carbon nanotube, graphene, carbon nanofiber, activated carbon, and carbon fiber, are commonly used electrode materials (without functional groups on carbon material surfaces), demonstrating a higher power density, better rate capability, and longer operation life.19–21 However, carbon-based supercapacitors still show restricted capacitances as carbon-based materials totally depend upon electrochemical double layer capacitance. 22
Pseudocapacitive oxides of transition metals (Ni, Fe, Co, Cu, Mn, etc.) are broadly explored owing to their low cost and high theoretical capacitances, but their conductivity is usually low, causing a noticeable decrease in rate capability. Various transition metal oxides, hydroxides, and sulfides have been extensively used as the electrode material such as RuO2, 23 CuO, 24 NiO, 25 ZnO/NiO, 26 FeO2, 16 and MnO2,27,28 NiCoO4, 29 NiCo2O4,29–31NiMoO4, 32 Co3O4, 33 CuCo2O4, 34 MnS, 35 NiS, 19 CoS2, 36 and CoS 37 due to their abundance, low price, and their accessible and facile preparation process.
Furthermore, the broad range of morphologies are found, including nanofilms, 38 nanosheets, 39 nanoneedles, 40 nanowires, 41 nanotubes, 42 urchin-like structures, 43 and nanoflakes. 41 These nanostructured morphologies can greatly affect electrochemical performance. From the literature, it is clear that carbon-based materials are most explored because of their remarkable mechanical and electronic properties.44–49 These well-aligned nanostructures are thought to be useful to facilitate high electrochemical activity and quick charge transport on the carbon fiber substrate.
The binary metallic system has been considered as a promising electrode material for high-performance energy-storage devices since it comprises greater electrochemical performance and higher capacity than the monometallic system.30,50
In particular, the ternary transition metal system, for instance, MCo2S4 (M = Ni,51–53 Co, and Mo), ZnNiCo, 54 and NiCoMo, 55 offers higher electrochemical performance and capacity than the monometallic system due to its various oxidation states and richer redox reactions. 52 By taking benefit of the synergetic effect to enhance electrochemical activity and stability, it is important to find mixed metal oxide–based systems for the electrode material to achieve high performance. 55
Recently, few studies have examined the mixed metal oxides/hydroxide nanostructured materials for SC applications that show synergetic effect and exceptional energy-storage behavior in comparison to the corresponding binary and monometal oxides. Among various bimetallic oxide electrode materials, NiMoO4 electrode material is extensively used because of environmental friendliness, low cost, and excellent electrochemical performance due to occurrence of various reversible redox reactions because of its variable valence state. 32 The hydrothermal method is commonly used to synthesize NiMoO4 nanorods and fabricate NiMoO4//activated carbon asymmetric supercapacitor devices delivering exceptionally high specific capacitance. Recently, an optimization study was conducted on the synthesis of NiMoO4 nanorods using the hydrothermal process for high electrochemical performance. 56
Co-based oxide has been considered as another promising transition metal–based electrode material for its high specific and exceptional pseudocapacitive properties. 50 Chen et al. reported Co3O4 microspheres by using cobalt nitrate hexahydrate (Co(NO3)2·6H2O) and urea (NH2CONH2) as reactants in a simple hydrothermal reaction for SC applications, showing a high capacitance of 372.7 F g−1. 57
It is observed that most published works have focused on the different materials, synthesis parameters like temperature during reaction, deposition current, and reaction time, which affects the material morphology, fabrication of devices, and their potential applications; however, the effect of the different metal ratio is rarely reported in the literature. 6
Variation in the metal ratio significantly affects the morphology of synthesized nanostructures and in turn the electrochemical performance of the electrodes. 58 So, combining salts of different metals to make a distinctive nanostructure is an effective approach to improve the electrochemical performance further.
Herein, we study the synergistic effects of mixed metal oxy-hydroxides and investigate their electrochemical performance in the preview of a flexible fiber-based supercapacitor. We have utilized the nano-on-micro approach with nanostructures grown over carbon microfibers wherein providing ease of handling nanomaterials as well as a conducting addressable electrode for ease of fabrication apart from being flexible and hence potential for futuristic flexible functional textile. For fabricating supercapacitor electrodes, we have used different proportions of salts of the ternary metal (Ni–Mo–Co) synthesized via the hydrothermal route. The reaction was carried out for 5 h at 150°C, followed by two different approaches to prepare carbon microfiber–based electrodes: one by taking out hydrothermally synthesized carbon microfibers directly after reaction and other was by using the synthesized material which was left over after the reaction, by making its slurry and coating it over neat carbon fiber. Further, both types of fiber-based electrodes were examined for electrochemical performance; that is, electrochemical impedance spectroscopy (EIS), cyclic voltammetry (CV), and galvanostatic charge–discharge (GCD) were carried out.
Materials
Carbon microfibers were taken from the carbon fabric of 200 gsm. Carbon microfiber bundle with diameter ≈ 60 µm was taken as the base material for fabricating electrodes. Nickel nitrate hexahydrate (Ni (NO3)2.6H2O) (99%< purity) and polyvinylidene fluoride (PVDF) were procured from Sigma Aldrich, USA. Cobalt Nitrate hexahydrate (Co (NO3)2.6H2O) (98% purity), KOH (85% purity), N-methyl 2-pyrrolidone (99.5% purity), and ethanol were obtained from Merck, India. Other chemicals which were used like urea (NH2CONH2), hydrochloric acid (HCL), sodium molybdate di hydrate (Na2MoO4.2H2O), NMP (N-methyl pyrrolidone), and carbon babric were obtained from Samir Tech. Pvt. Limited, India. Conducting carbon black (Grade: EQ-Lib-Superp, density: 160 kgm−3, BET surface area: 62 m2g−1) was bought from MTI Corporation, USA.
Methods
Pretreatment of carbon fiber
Different lengths of fiber were taken and then cleaned with 10% dilute solution of HCl in order to convert their surface more hydrophilic, followed by washing with deionized water, in a sonication bath for 15 min. After cleaning, drying of fibers was carried out at 60o C for 5 h, followed by measuring their weight.
Growth of Ni–Mo–Co ternary metal oxy-hydroxides
Two different routes were followed for the deposition of ternary metal oxy-hydroxides over carbon microfibers. In the first route, the metal oxy-hydroxides were directly grown or in situ deposited over the carbon microfibers by simply dipping the fibers in the reaction solution during the reaction. In second route, slurry coating of the synthesized product which was formed during the hydrothermal reaction was done over carbon fibers. In both cases, the concentration of the Ni–Mo–Co metals was individually varied by varying the concentrations of reactants Ni(NO3)2.6H2O, Na2MoO4.2H2O, and Co(NO3)2.6H2O. The ratios were selected such as to cover the wide spectrum of the concentration and enable us to find an optimum concentration for the best electrochemical performance.
In a typical hydrothermal synthesis,
59
Ni (NO3)2.6H2O and Na2MoO4.2H2O were dissolved in 50 mL de-ionized (DI) water, and Co(NO3)2.6H2O and urea were dissolved in 50 mL DI water for 10 min under stirring, and then both the solutions were mixed slowly. After stirring for an additional 10 min, the solution was transferred into a 100 mL Teflon-lined stainless steel autoclave containing samples of carbon fibers. The reaction was carried out for 6 h by keeping the temperature at 150°C. After the reaction, the reactor was cooled down at room temperature. After that, fibers were taken out of the reactor and washed with DI water several times, and then dried at 60°C overnight. The resulting product formed after the reaction was taken out in a beaker, cleaned with water and ethanol for few times, and dried at 60°C in the oven until it dried, as illustrated in Figure 1(a). Schematic illustration of preparatory process of (a) hydrothermally deposited electrodes and (b) slurry coated electrodes.
In order to study the synergistic effect of different metals, Ni:Mo:Co (NMC) ratios were varied. Initially, it was 2:1:2 denoted as Ni2xMoxCo2x (Ni(NO3)2.6H2O: 3 Mmol, Na2MoO4.2H2O: 1.5 Mmol, Co(NO3)2.6H2O: 2 Mmol and NH2CONH2: 7.5 Mmol). We further investigated the Ni2xMoxCo4x (2:1:4) and Ni2xMoxCox (2:1:1) ratios. The ratio of Ni to Mo was kept constant in all the reactions. Apart from this, the Ni–Mo (NM) concentration was doubled and halved with respect to Co to observe the effect on electrochemical performance. For comparison, all the materials with varying concentrations ratios were synthesized by using a similar hydrothermal process. Dried fibers were directly weighed, and the weight of the deposited material after the reaction was calculated.
Slurry preparation
The carbon microfiber–based working electrodes were also prepared by the slurry coating method apart from the above-described
Materials characterization
Surface morphology of the nanomaterial deposited fibers after hydrothermal reaction analyzed by using Zeiss Supra-40VP Field-Emission scanning electron microscopy (FE-SEM). To analyze the structure of the crystalline phase and chemical composition, X-ray spectroscopic analysis of prepared samples was carried out on a Bruker D8 ADVANCE diffractometer in the scanning range of 20° < 20 < 80°.
Electrochemical measurements
All the electrochemical experimentations were carried out by using a Multi Autolab M204, Metrohm, Netherlands, electrochemical workstation. The electrochemical performance of single electrodes was studied by using CV, EIS, and galvanostatic charge/discharge (GCD) measurements in a three-electrode system with Ag/AgCl reference and platinum as counter electrode in 2M KOH aqueous electrolyte. CV scans were recorded from 0 to 0.5 V (vs Ag/AgCl) at scan rates of 20mV s–1. GCD cycle tests were carried out in the range of 0–5.0 V. Specific capacitance C (Fg−1) was calculated from the GCD curves by (I Δt)/(m ΔV), in which I signifies discharge current, Δt signifies discharge time, ΔV is the potential window, and m denotes the mass of active electrode materials. 60
Electrochemical impedance spectroscopy measurements of the supercapacitor electrodes was performed at open circuit potential with an AC amplitude of 5 mV and frequency range of 0.1 Hz–110 kHz.
The specific energy density E (W h kg−1) and power density P (W kg−1) as E = (1/2) C (ΔV) 2 and P = E/Δt, where C is the specific capacitances of the device and is calculated by using mass of the active material from galvanostatic discharging curves.
Results and discussion
Morphological properties
Field-emission scanning electron microscopy has been performed to examine the morphological structure. Figure 2(a) shows images of pristine carbon fiber with a smoother surface. Figure 2(b) shows hydrothermally grown 3D nanostructure at initial concentrations. Ni2xMoxCo2x consists of many nanotube- and nanoflake-like structures; similarly, Figure 2(c) represents double concentration. Ni2xMoxCo4x of cobalt nitrite shows the formation of nanoflakes. In Figure 2(d), magnification reveals unique nanosheet-like structures along with nanotubes Ni2xMoxCox at a half concentration of Co. Several sharp tips and circled pores appear in Figure 2(e) at a double concentration of NM (Ni2xMoxCox); Figure 2(f) SEM images reveal nanostructure different from previous (Ni2xMoxCo4x NM halved) with nanoflakes. These well-grown nanostructures show well-defined interior which favors the diffusion process of the electrolyte solution. This helps in improving the electrochemical performance. The peripheral of the NMC over the surface of carbon fiber is continuous and relatively thin with a porous architecture, which could be attributed to the successive discharge and loss of the produced gas during various thermal reactions.61–63 In the case of slurry deposited samples, Figure 2(g)–(k); Figure 2(g) consist of numerous nanotubes and nanosheet-like structures at initial concentrations (Ni2xMoxCo2x) like in situ samples. Figure 2(h) represents double concentration (Ni2xMoxCo4x) of cobalt nitrite unique alignment of nanosheet-like structures which can increase specific capacitance by delivering large electrochemically active surface and can help in shortening paths for electron transport; the average thickness of these nanosheets is around 20nm. Figure 2(i) shows the nanosheets and nanotubes of NMC homogeneously deposited over carbon fiber revealed at a half concentration of cobalt nitrite Ni2xMoxCox. The highly dense nanostructure is distributed at double concentration of NM (Ni2xMoxCox) Figure 2(i); Figure 2(k) (Ni2xMoxCo4x NM halved) shows well-aligned nanostructure composed of nanoflakes and nanotubes. These nanostructures help to enhance electrochemical activity. SEM images of (a) untreated fiber; (b–f) hydrothermal reactions of NMC with the NMC ratio of (b) Ni2xMoxCo2x initial concentration, (c) Ni2xMoxCo4x (Co double), (d) Ni2xMoxCox (Co halved), (e) Ni2xMoxCox (NM double), and (f) Ni2xMoxCo4x (NM halved); (g–k) SEM of slurry deposited samples with the NMC ratio of (g) Ni2xMoxCo2x initial concentration, (h) Ni2xMoxCo4x (Co double), (i) Ni2xMoxCox (Co halved), (j) Ni2xMoxCox (NM double), and (k) Ni2xMoxCo4x (NM halved).
X-ray spectroscopic analysis
The phase composition of the synthesized carbon fiber samples was characterized by using XRD, and the XRD pattern is illustrated in Figure 3. Although these peaks are all weak, they still can be recognized. The detected diffraction peaks at the degrees of 33.9 and 59.7 could be indexed to Ni(OH)2 (JCPDF card No. 73-1520), and the other diffraction peaks can be indexed to Co(OH)2 (JCPDF card No. 74-1057) and MoO2 (JCPDF card No. 78-1071). It indicates that the product comprises three compounds: Ni–Co double hydroxide and some MoO2.
59
Moreover, the peak intensities are moderately wide and weak, which shows the reduced crystallization of the NMC nanostructures. Poor crystallization is advantageous for better electrochemical performance
55
because more defect transportations are present in materials with weak crystallinity.
64
Effects of concentration changes are not clearly visible through diffraction peaks; only minor shift in 20 values was there. X-ray diffraction (XRD) peaks of the nickel–cobalt–molybdenum metal (NMC) nanostructured samples at different metal ratios.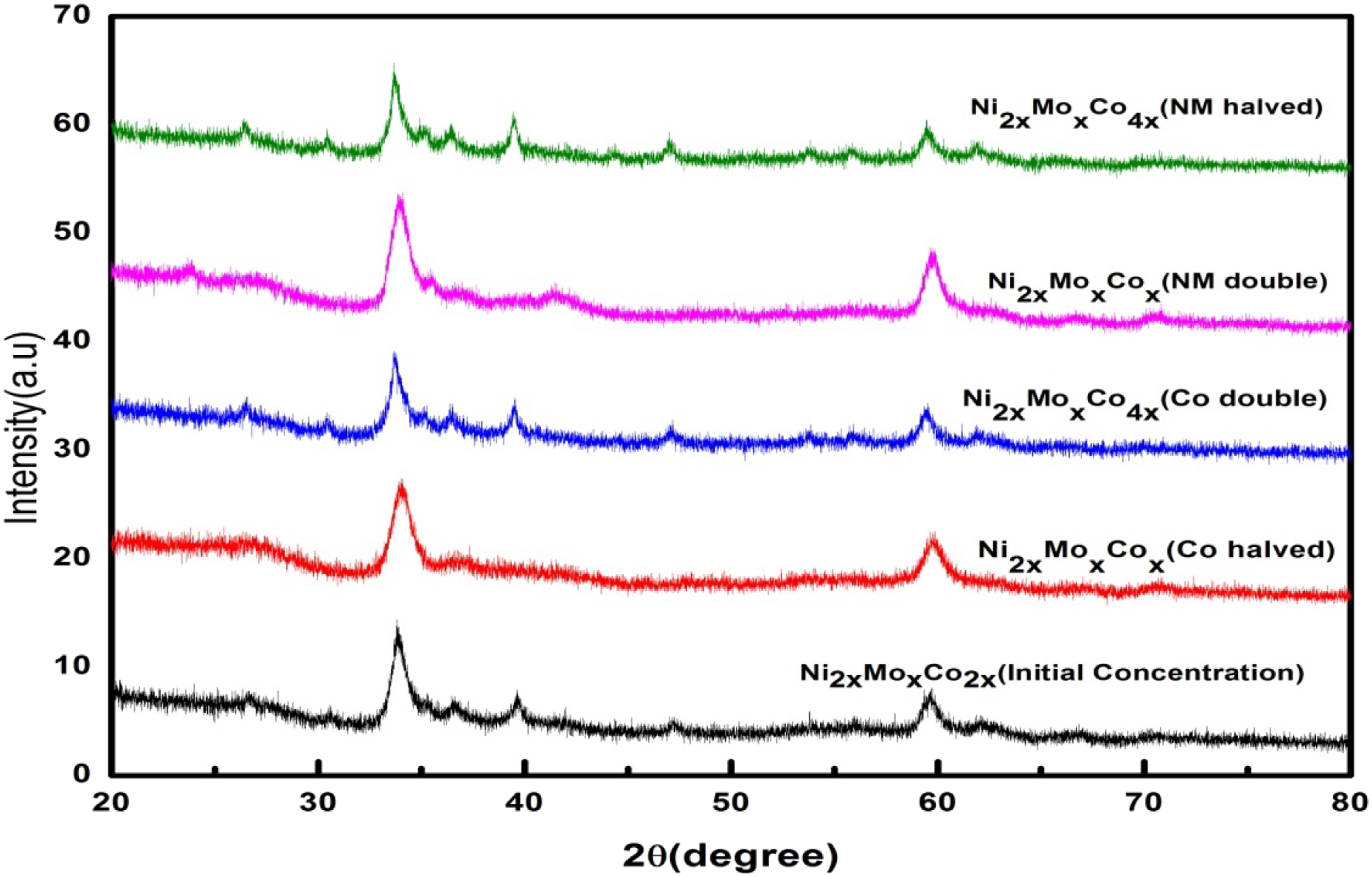
Electrochemical performance
To examine the electrochemical properties of NMC nanoarrays, electrode prepared with hydrothermal deposition and slurry coating were separately tested; a series of electrochemical investigations were carried out at various concentrations, which includes CV, GCD, and EIS in a three-electrode arrangement by using an aqueous solution of 2 molL−1 KOH as an electrolyte. Figure 4 shows carbon fiber at different positions, which clearly shows it is flexible enough to be used for wearable electronics applications. Previous studies show that carbon fiber is a promising material for the fabrication of flexible supercapacitors.
10
Photographs of flexible carbon fiber: (a) normal state, (b) twisted-state, and (c) bent-state.
Figure 5 represents values of capacitance at different concentrations. At optimized concentration, ratios show better electron transportation from active materials to the current collector and less electrode resistance which can be confirmed by the Nyquist plots in Figure 6(a) and (b). Capacitance at different concentrations of (a) hydrothermally activated samples and (b) slurry deposited samples. (a), (c), (e) EIS, CV, and GCD curves, respectively, for the hydrothermally deposited carbon fiber sample. (b), (d), (f) EIS, CV, and GCD curves, respectively, for the slurry deposited carbon fiber sample. (g) Cycling after 200 cycles. (h) CF retention after 200 cycles.

Figure 6(c) and (d) show the typical CV curves of NMC nanoarrays electrode recorded in a potential window of 0–0.5 V at a scan rate of 20 mVs−1. It can be clearly noticed that all the CV curves comprised one strong redox peak, representing the redox reactions and pseudocapacitive characteristic of the NMC-based mixed material oxy-hydroxide.
To further evaluate the capacitive performance of NMC electrodes, GCD measurements were carried out at a current density of 1Ag−1 with 0 to 0.5 V potential range window. As shown in Figure 6(e) and (f), the GCD curves are clear nonlinear lines with distinct voltage plateaus around 0.30–0.20V, furthermore indicating the pseudocapacitive behavior of the prepared electrode materials due to the Faradaic reactions which are reliable with the CV results. Moreover, the discharge time decreases with the increasing concentration of nickel nitrate, which leads to the decrease of specific capacity. This could be accredited to the point that the active materials may be lacking to contribute in redox reactions. In other words, the electrochemical capacitance of NMC is recognized to the quasi-reversible electron transfer process that typically involves the redox couples Co2+/Co3+ and Ni2+/Ni3+ and is perhaps intervened by the OH− ions in the alkaline electrolyte medium. However, the Mo atoms do not contribute in the measured capacitance and any redox reaction cleared by XRD analysis also. Even though the Mo constituent did not contribute in the Faraday reaction, it affects the crystal structure. Additionally, molybdenum oxide through reversible intercalation/DE intercalation of K+ ions displayed intercalation pseudocapacitive performance into/out of the nanostructures.65,66
Furthermore, the direct growth of NMC on the surface of carbon microfibers shows good adhesion, and this approach leads to avoiding the use of polymer and conductive additives, which generally increases the series resistance and declines the capacitance in redox reactions. However, in case of slurry coated samples, significantly decreased equivalent series resistance is shown in comparison with hydrothermally deposited samples due to the addition of conducting carbon and activated material, which fills the gaps between micron-sized activated particles and acts as a conductive link.
The positive synergistic effect of nickel molybdenum and cobalt nitrate ions in the complex metal oxy-hydroxide can also considerably increase its capacitance characteristics.
According to electrochemical analyses, the specific capacitance of carbon microfibers and different nanostructures shows excellent synergistic effects. The enhanced electrochemical performance might be associated with the subsequent structural features.
Firstly, ion diffusion becomes easier due to the large surface area, which ensures utilization active materials. So undoubtedly, the use of NCM-based material contributes to the high capacitance. Secondly, to improve the electrochemical performances, carbon cloth in the hybrid materials provides fast electronic transfer channels. 40
These excellent electrochemical properties, combined with other advantages in the synthesis process, make this ternary complex compound a very competitive electrode material for supercapacitors. Results shows (Figure 6 (g) and (h)) even after 200 cycles, no obvious change in specific capacitance (CF) is observed. These unique nano structures provide effective contact surface area, provide more active sites for the electrolyte to effectively diffuse to the whole electrode, and improve the charge transfer kinetics of ions and electrons.
Conclusions
In summary, we have successfully fabricated the ternary compound–based electroactive 3D nanostructures over carbon microfibers by using two approaches: (1) hydrothermal deposition and (2) slurry coating for flexible high-performance pseudocapacitors. The study reveals that NMC performed as an outstanding pseudocapacitive material, and obtained unique nanostructures are also capable of fast electron transmission and ion diffusion for loading a large amount of additional electroactive materials. The effect of the varying metal ratio on NMC-based supercapacitor electrodes prepared by the hydrothermal synthesized method and slurry deposition method was analyzed by characterizing structural, morphological, and electrochemical properties. Specific capacitance was found to increase on doubling the concentration of cobalt nitrate in both hydrothermal deposition and slurry coating methods. In both routes, the maximum capacitance value was achieved when doubling the concentration of cobalt nitrite; in case of hydrothermal deposition, it reaches up to 519 Fg−1, while in the case of slurry coating, maximum specific capacitance of 890 Fg−1 was observed at 1 Ag−1 current density, which indicates that cobalt metal actively participated in the reaction and enhanced the electrochemical performance. Nickel also helps to increase the electrochemical activity, but the change is appreciable in comparison with cobalt. These NMC-based composite products have immense potential as a unique electrode material for supercapacitor, and we have shown that they are equally suitable for flexible carbon fiber–based supercapacitors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
