Abstract
Cotton fabric with enhanced ultraviolet shielding behavior was successfully developed by coating functionalized activated carbon and grafting polyaniline hybrid material through in situ polymerization. Initially, the activated carbon was prepared from bio-mass rice husk and then surface functionalized using suitable silane coupling agent. The optimized sample delivers strong ultraviolet shielding ability with a Ultraviolet Protection Factor (UPF) value of 63.9, which is nearly 20 times higher than that of pristine cotton (UPF = 3.2). Moreover, the same sample shows enhanced tensile strength behavior. The structure and morphological studies are also discussed using Fourier transform infrared spectroscopy and scanning electron microscopy for both pristine cotton and hybrid coated fabrics. The results show that the activated carbon derived from rice husk could be a suitable alternate carbon source for ultraviolet shielding when compared with high-cost carbon derivatives. Thus, the developed material can find good high-performance application with better ultraviolet shielding property.
Keywords
Introduction
Flexible fibrous textile materials have received great attention due to their enhancement in functional properties [1–5] through adopting advancing engineering approach [6–11]. Cotton fabric is commercially available and has high specific surface, porous structure, flexibility and less cost. Due to these properties, cotton has received industrial interest in addition to wearable application [11–13]. Properties like hydrophilic, impotent against antimicrobial, low strength and poor sensitivity to the ultraviolet (UV) light, restrict the cotton fabrics in wide industrial applications and make them to fail as functional fabrics [14–16]. Consequently, considerable industrial attentions have been paid on this issue which has paved way to promote the industrial value of cotton due to its properties as well as scope for the advanced materials for many industrial and technical applications [17–20].
Among various available techniques, coating of organic compounds (dyes), inorganic particles (nanoparticles) and organic-inorganic hybrids (polymer composites) is the foremost technique to improve the functional behaviors of fabrics [21–24]. The coating of organic–inorganic hybrids affords superior and enormous engineering quality than their combinational building blocks. In textile fabric processing, hybrid materials perform their important role in enhancing the functional behaviors. UV protection, EMI shielding, flame retardance and super hydrophobicity are such properties that have been enhanced by coating of hybrid materials [25–28]. Cotton fabrics coated with various organic inorganic hybrids have been studied for their UV shielding behavior [29–32]. Since, UV radiations cause serious damages to the cotton fabric and confine their wide applications, especially in some high-end areas [33–36], recently various carbon derivatives such as carbon nanotubes (CNT), graphene and graphene oxide have been hybridized with polypyrrole, polyaniline (PANI) and polyurethane, respectively and used as a UV-blocking coating for cotton fabric [37–41]. However, the high cost and tedious reaction condition of graphene, CNT, etc., restrict their commercial utility. So, still there is a need for low cost, photo stable material obtained from relatively easier synthetic method without harsh reaction conditions [42,43].
Rice husk is a well-known biomass for its rich cellulose and lignin content. It possesses various constituents such as 38% cellulose, 22% lignin, 18% hemicelluloses, 20% ash and 2% extracts, which differ according to growing conditions [44,45]. Carbonization of rice husk yields bio-based activated carbon (AC) that possesses sp2 hybridized carbon skeletons and graphitic structures. Moreover, they exhibit excellent physicochemical properties such as high surface area, porosity and conductivity, which make them suitable for wide range of engineering applications. However, there are no significant reports of coating either rice husk AC or its hybrid with cotton fabric towards industrial application. In view of this, it was expected that AC derived from rice husk may result in better UV-shielding property for cotton fabrics.
Among the various intrinsically conductive polymers, polyaniline is a promising one from an industrial perspective due to its good conductivity, easy synthesis, environmental stability and relative low cost [46,47]. Recently, inorganic nanoparticles-incorporated polyaniline nanocomposite materials have attracted much interest worldwide because of the improved stability, conductivity and unique optical properties, etc. [48–50]. Hence, in the present work, polyaniline and rice husk-derived ACs are used together. The resulting hybrid coated fabrics were studied for their UV shielding and thermal properties.
Materials and methods
Materials
Cotton fabric comprising 44s Ne warp yarn count and 40s Ne weft yarn count with weight per unit area of 96 g/m2 obtained from a textile industry in Erode, Tamil Nadu was used for the experiment. Rice husk was collected from a local rice processing mill, Salem, Tamil Nadu, India. Aniline, ammonium persulphate (APS), ethanol and sodium hydroxide were obtained from Merck, India. 3-Aminopropyltriethoxysilane (3-APS), KBr and acetic acid were purchased from Sigma Aldrich, India.
Synthesis of AC from rice husk
Rice husk-derived AC was prepared in accordance with reported literature [46,50,51]. Initially, after complete purification by washing with water, the dried rice husk was carbonized at 400℃ under a nitrogen atmosphere for 90 min. Later on, the carbonized sample was digested with strong NaOH with a weight ratio of 1:3 and dried at 120℃ for 12 h. Consequently, carbonization was carried out at 400℃ for 30 min and then at 800℃ for 60 min to achieve complete graphitization. The carbonized product was neutralized by 0.1 M HCl solution and washed several times with hot distilled water to attain a constant pH (6.6–7.0). The resulted AC sample was dried under vacuum at 120℃ for 24 h and then taken for surface functionalization.
Surface functionalization of AC
The prepared AC was surface functionalized with an amine group in order to have a compatibility with aniline as per the reported procedure [46]. The surfaces of the AC were initiated by forming functional group such as acid, hydroxyl, etc., via thermal treatment at 300℃ for 3 h. Then, the sample was dispersed in ethanol by way of ultrasonic agitation for 30 min, and a calculated amount of 3-APS was added. The pH of this mixture was adjusted to 4–5 using acetic acid and subjected to reflux for 24 h. Finally, the functionalized activated carbon (FAC) was collected through centrifugation and washed three times using ethanol followed by hexane to remove the unreacted silane and dried in a vacuum oven at 60℃ for 24 h.
Preparation of FAC-coated and polyaniline-grafted cotton fabric (CA1–50Ca1–4) samples
Coded samples with respect to percentage of PANI and FAC.
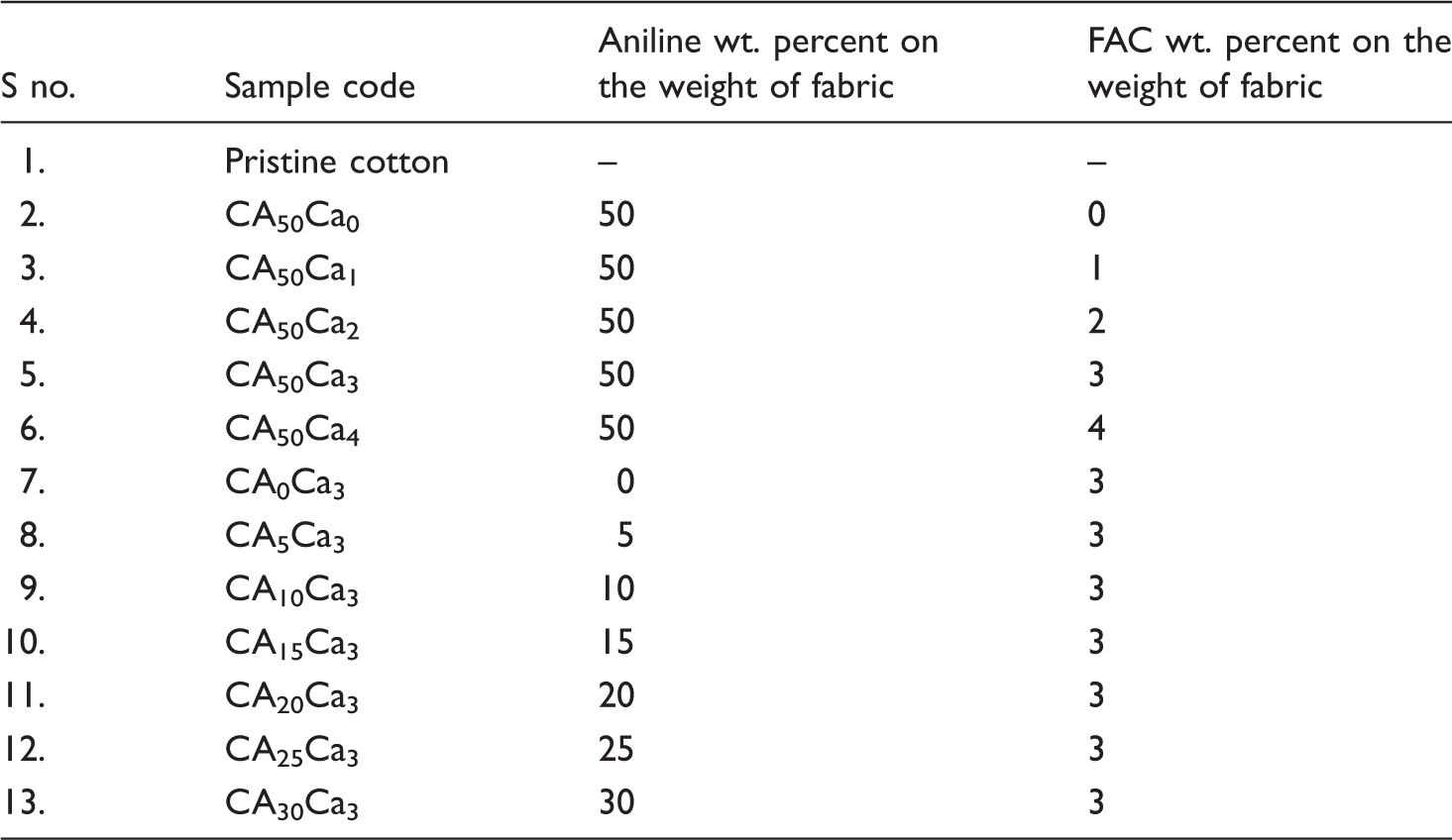
FAC: functionalized activated carbon.
Characterization
Fourier transform infrared spectroscopy (FTIR)
FT-IR spectra data were obtained from FT-IR 7600 Spectrophotometer of Shimadzu Model. KBr was used as a reference. It was ground with samples (2%) and used to record the spectrum in the form of pellets.
UV-Visible spectroscopy
The UV-Visible transmittance spectra were recorded using a UV-Visible spectrophotometer—UV 2450 Shimadzu Model. The fabric samples of dimension 1 cm breadth and 4.5 cm length were used to record the spectrum. Further, to evaluate the UV shielding tendency of the pristine cotton and all other samples, their respective Ultraviolet Protection Factor has been calculated on the basis of the recorded data in accordance with Australia/New Zealand standard AC/NZS 439:1996 [37,38]. The formula used to calculate the UPF is given below
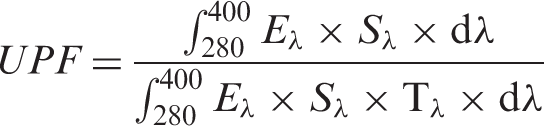
Scanning electron microscopy (HR-SEM)
A Hitachi S-4800 high resolution scanning electron microscope attached with Horiba equipment was used to record the surface morphology and elemental composition of cotton fabrics samples at 15 kV.
Thermal gravimetric analysis
Thermal gravimetric analysis (TGA) was performed on a PerkinElmer TGA 4000 thermogravimetric analyzer. The thermograms were recorded at a heating rate of 10℃ min−1 under an air atmosphere.
Tensile strength
The tensile properties of the pristine cotton and treated fabric were measured using a universal testing machine (Tensile Tester Z10 of Zwick/Roell, Germany), with a constant rate of extension. The test was done according to ASTM standard D5035-11 (Reapproved 2015). The clamp speed was kept as 300 mm/min, and the gauge length (grip to grip separation at the start position) was 75 mm.
Air permeability
Air permeability of the pristine cotton and the treated fabric was tested using air permeability tester of P.S.I Sales Pvt Ltd, New Delhi, India as per the standard BS 3424. The test was done on the fabric area of 5.07 cm2. The rate of flow of air was measured when a steady pressure drop of 1 cm head of water was achieved.
Stiffness testing
Shirley Stiffness tester was used to test the bending length of fabric as a measure of stiffness. The test was done as per the standard B.S 3356:1961 keeping the specimen size 6″ × 1″.
Results and discussion
Spectral analysis
FTIR
In order to have better compatibility, the surfaces of the AC were modified using amine terminated silane. Figure 1 illustrates the FTIR of AC and surface FAC. The appearance of peak at 1080 cm−1 confirms the presence of Si-O-Si network of silane coupling agent. In addition, the peaks at 2922 and 2832 cm−1 correspond to the asymmetric and symmetric stretching modes of the –CH2-aliphatic group of 3-aminopropyltriethoxysilane [15]. It was reported that the amino group present on the surface could involve in the growth of polyaniline through interfacial properties, which can control the deposition and adhesion of the polymer chain [52–54]. Initially, during polymerization, the amino (NH2) groups present in the surface of FAC were converted to radicals ( FTIR spectra of AC and FAC. Synthesis of FAC coated polyaniline grafted cotton fabrics.

Figure 2 illustrates the FTIR spectra of pristine cotton and FAC-coated polyaniline-grafted cotton fabrics. The pristine cotton fabric shown in the inset of Figure 2 exhibits a number of peaks, from 1200 to 1700 cm−1, which were attributed to the stretching vibration of both C–O and C=O functional groups. Minor peak at 2832 cm−1 shows the presence of carboxylic group (–COOH) on the surface of the cotton fabric. After the coating process, the cotton fabric shows dark green color, which confirms the grafting of polyaniline on the cotton surface [56]. The same has been supported in Figure 2. The FAC-coated polyaniline grafted fabrics show distinguished FTIR spectra. The peaks at 1570 and 1476 cm−1 correspond to the typical C=N stretching vibration of the quininoid and benzenoid rings of polyaniline respectively, whereas the peak at 1302 cm−1 is attributed to C–N stretching vibration [38].
FTIR spectra of pristine cotton (inset) and FAC coated polyaniline grafted cotton fabric.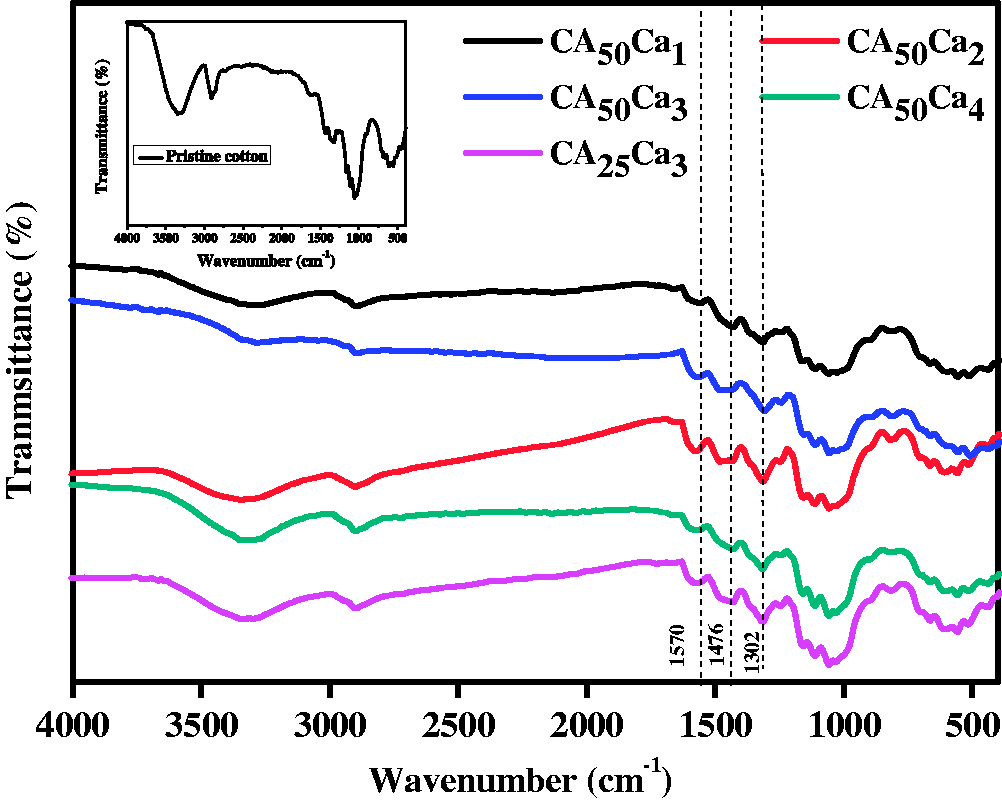
UV shielding analysis
Figure 3 illustrates the UV-visible transmittance spectra of pristine cotton, and FAC-coated polyaniline grafted cotton fabrics (CA0–50Ca0–4). The UPF values of the samples were tabulated in Table 2. From Figure 3 and UPF values presented in Table 2, it is clear that the pristine cotton retains poor UV shielding tendency (shows 98% of transmittance with UPF 3.2). In order to promote UV shielding property, the bio-mass derived AC along with polyaniline polymer was used as hybrid coating materials in different ratios. Initially, the UV shielding behavior was studied with respect to coating percentage of AC, (1, 2, 3 and 4 wt.%) in which aniline concentration was kept constant (50 wt.%). In this approach, the sample CA50Ca3 demonstrates highest UV shielding behavior and possesses UPF value as 58.7. Further, the carbon-coated sample CA50Ca4 shows reverse trend, whose UPF was found to be only 24.9. The decrease in UPF is because of aggregation and improper distribution of hybrids over the cotton fabric. From the obtained result, we came to know that the optimum AC coating could be 3 wt.%.
UV-visible transmittance spectra of pristine cotton and CA0–50Ca1–4 from 200 to 800 nm (inset shows the spectra from 280 to 400 nm). UPF value of pristine cotton and CA0–50Ca1–4. UPF: Ultraviolet Protection Factor; FAC: functionalized activated carbon.

Further, optimization studies were performed with respect to aniline (5, 10, 15, 20, 25 and 30%) by keeping the AC content as constant with 3 wt.%. Here, CA25Ca3 sample with 3 wt.% FAC and 25 wt.% aniline shows the highest UPF value 63.9. Comparably, the obtained result was nearly 20 times higher than that of pristine cotton, 8 times higher than that of cotton fabric with FAC (CA0Ca3) alone and 4.8 times higher than coated cotton fabric with polyaniline alone (CA50Ca0).
From the above results, the enhanced UV shielding tendency may be attributed to the synergistic effect contributed by both FAC and polyaniline hybrid materials. It was well known that the AC possesses high surface area due to its nano form. Further, the ability of the carbon nano material to absorb the photons from the light provides a strong protection against the UV rays for the host cotton fabric. It is reported that the energy of C=C bonds is about 335 kJ which is approximately equal to the energy of UV photons [57,58]. The large enough number of C=C bonds in the AC distributed with graphitic configuration thereby probably protect the fabric cellulose structure from UV radiations. In addition, the quininoid and benzenoid rings of polyaniline also possess strong UV absorption behavior due to their C=C skeleton and thereby provide the UV shielding behavior. Thus, synergistic effect of both functionalized activated carbon (FAC) and polyaniline promotes the UV shielding behavior, when they are applied over the surface of cotton fabric.
Previously, various semiconductor metal oxide nanoparticles (TiO2, ZnO and CeO2) and carbon (graphene and graphene oxide) have been coated over cotton fabric and higher UV-shielding functional behavior was achieved. However, the photocatalytic activity of semiconductor nanoparticles would also induce photo-degradation of fabric which is an organic substance. High cost and hard reaction condition in synthesis of graphene materials led to an undesirable and complicated approach. As a consequence, there is a demand for novel UV-shielding materials with higher stability and UV light absorbing ability. In this endeavor, FAC derived from rice husk hybridized with polyaniline provides equivalent UV shielding behavior as that of the existing carbon derivatives [5,37,38,57].
Morphological analysis
The morphological behavior of the samples with significant contribution towards UV shielding was recorded with HR-SEM. Thus, the morphological features of FAC-coated fabric (CA0C3), polyaniline-grafted fabric (CA50C0) and both FAC-coated and polyaniline-grafted fabrics (CA50Ca3, CA50Ca4 and CA25Ca3) are presented in Figure 4. The features were compared with pristine cotton and discussed. In Figure 4(a), the smooth fiber structure of the pristine cotton can be visualized clearly. However, Figure 4(b) shows the rough surfaces with coated particles which confirms the presence of FAC particles on the cellulose structure. The absence of a smooth original surface of fabric indicates that there exists adequate interfacial bonding between FAC and fabric. In addition, intercalation of the carbon particles in between the warp and weft of the fibers documents for their promoted UV shielding behavior. Figure 4(c) shows a contradict morphology with fine nanorods-like structures over the surface (highlighted through a circle), that indicates the presence of polyaniline network. The network found to be one-dimensional flexible and linear. Figures 4(d) to (f) show the morphologies of CA50Ca3, CA50Ca4 and CA25Ca3, respectively. It is to note that the nucleation and growth of polyaniline along with FAC increases the mass of the fabrics. Thus, the mass of the CA25Ca3 was found as 139 GSM, whereas the control fabric has 96 GSM only. As discussed earlier in FTIR, the polymerization of the amino group of FAC with polyaniline through interfacial mechanism contributes for the same behavior. Moreover, with an increase of concentration of FAC and aniline monomer beyond a limit, molecules of polyaniline and FAC were intercalated and distributed irregularly on the fabric surface and form agglomerates of polyaniline (Figure 4(f)). On the other hand, a more uniform distribution of FAC and polyaniline was observed (Figure 4(d)) in CA25Ca3 surface. This result supports their enhanced UV shielding behavior due to optimum concentration of aniline and FAC. Further, it was also suggested to be due to the strong interaction that occurs between the uniformly self-assembled 3APS-coated FAC and polyaniline layers achieved through silanization [53,59]. Thus, these homogenous coating of polyaniline and FAC over the cotton fabric have the significant impact in functional UV shielding property. Figures 5(a) and (b) show the elemental profile of CA0Ca3 and CA25Ca3. It was found that CA0Ca3 contains 51.4% carbon atoms, whereas CA25Ca3 has 65.7% carbon atoms. The concentration of carbon content in the fabric increases because of the growth of polyaniline over the surface of the cotton fabric. Further, the CA25Ca3 also shows the presence of nitrogen (7.8%), which confirms the presence of polyaniline in the fabric structure.
SEM images of (a) pristine cotton, (b) CA0Ca3, (c) CA50Ca0, (d) CA25Ca3, (e) CA50Ca3 and (f) CA50Ca4. EDAX profiles of (a) CA0Ca3 and (b) CA25Ca3.

Thermal studies
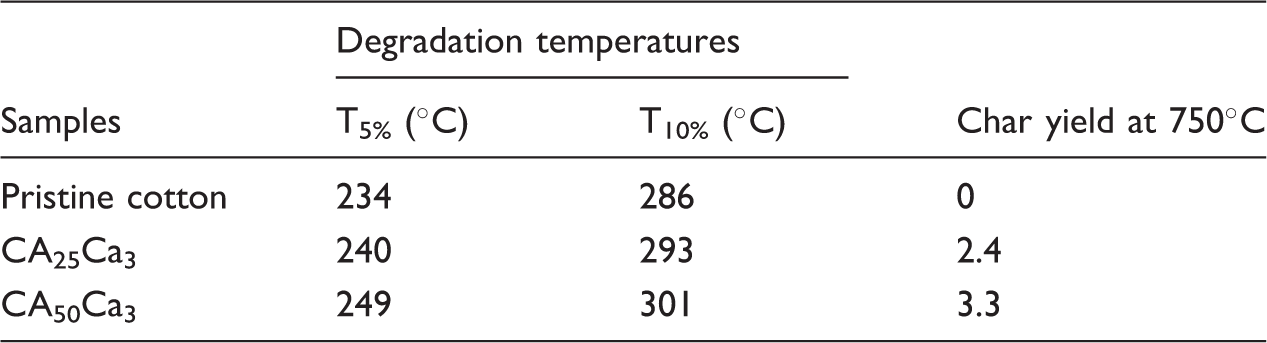
Figure 6 details about the thermal degradation behavior of pristine cotton in comparison with those of CA50Ca3 and CA25Ca3 in air atmosphere. Generally, degradation mechanism of cotton fabric happens in three stages; however, degradation near 100℃ is associated with removal of moisture. First-stage (300℃–400℃) degradation is associated with formation (aliphatic) char and volatilization. In the second stage (400℃–600℃), the significant degradation is observed, due to the formation of residue from aliphatic to aromatic char, due to the carbonization process. In other words, due to degradation of crystallization of cellulose fabric, major combustion gases are generated. Finally, the last or third stage (above 600℃), degradation is associated with the complete decomposition or oxidization of carbonaceous species (CO and CO2) [60,61]. The degradation of samples CA50Ca3 and CA25Ca3 also shows similar pattern. But, their onset temperatures increase, and the values are tabulated in Table 3, which infers that cotton coating and polyaniline grafting improve thermal stability marginally. This improvement in thermal stability is a desiring factor when the treated fabric is used in high-end industrial purpose for UV absorbency. High char yield was also obtained through the effective coating [62,63].
TGA profiles of pristine cotton, CA50Ca3 and CA25Ca3. Thermal properties of pristine cotton, CA50Ca3 and CA25Ca3.
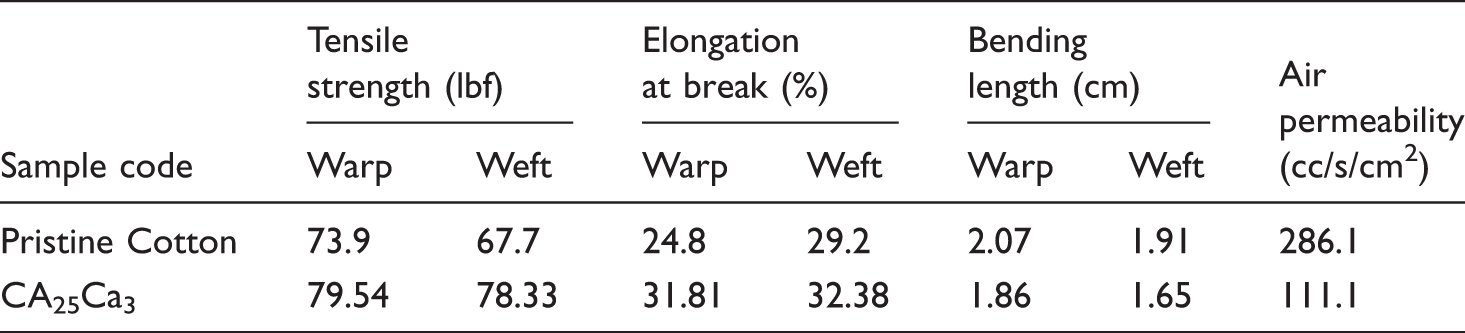
Basic properties of pristine cotton and CA25Ca3
Basic properties of the treated samples.
Conclusion
In the present work, a low-cost AC from the bio-mass rice husk was prepared and functionalized. Using in situ polymerization method, FAC-coated and polyaniline hybrid material-grafted cotton fabric was developed. The presence of both FAC and polyaniline was found to be essential for providing the better UV shielding behavior to the cotton fabric. The formation and morphologies of organic-hybrids coated over the fabric were discussed elaborately. The UPF value (63.9) indicated that strong UV absorbing ability of carbon and polyaniline provides protection to the core cotton fabric. Moreover, the coated cotton fabric samples show enhanced tensile strength when compared to the pristine cotton fabric. Therefore, it is understood that the process and coating to improve UV resistance does not adversely affect the fabric but improve the tensile property additionally. Thus, the low cost and environmental friendly organic–inorganic hybrids of FAC and polyaniline show great advantages in improving the functional behavior of textiles which will help technocrats to use these treated cotton fabrics to use for high-end applications as UV protecting textile materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
