Abstract
This research aims to develop chemical protective clothing via a functionalised surface coating on cotton fabric for simultaneous barrier performance and thermal comfort. An omniphobic protective layer on the cotton fabric was developed through a polyurethane – superabsorbent polymer – fluoropolymer coating to protect the penetration of water, oils and liquid chemicals. It was found that the padding of coated specimens with fluoropolymer provided a substantial improvement of hydrophobicity, and therefore, exhibited a high-water contact angle (114.06°) and a maximum water repellency rating of 100. The highest aqueous liquid repellency (8.0) and satisfactory oil repellency (7.5) with resistance against liquid chemical penetration were also achieved after the treatment with fluoropolymer. Moreover, the coated fabrics integrated with superabsorbent polymer showed an improved dry heat resistance and moisture vapour transmittance, consequently a high evaporative cooling index to create a favourable thermal comfort between the skin and the apparel in hot and humid conditions. Other comfort parameters, including air permeability and moisture management performance were compromised to a certain level due to a continuous and hydrophobic barrier layer on the fabric surface that hindered the transferring of air and liquid sweat through the coating.
Keywords
Introduction
Chemical protective clothing (CPC) refers to apparel and other textile-based products that are designed to safeguard the wearer from harmful chemicals. 1 These clothing materials are expected to provide reliable protection from the detrimental effects of poisonous chemicals, and therefore, they are required to be completely impervious to water and liquid chemical penetration. 2 The fabrication of CPC with an impermeable chemical barrier layer on the cloth surface includes the technique called laminating or coating. 3 Applying polymer materials to the fabric as a thin and smooth surface layer, coating brings functionality and incorporates versatile protection capability to clothing materials. 4 Hence, this method is widely used to develop functional textile fabrics for diverse applications.
Impermeable coated layers on textiles are commonly developed by using several polymeric materials, including polyurethane (PU), polytetrafluoroethylene (PTFE), poly(vinyl chloride) (PVC) and so on.5–8 These coated fabrics are generally hydrophobic and can be employed to protect against water and aqueous liquid chemical. 9 However, due to the extreme threats of various hazardous chemicals and the complexity of diverse environmental conditions, it has become obligatory to develop CPC with versatile protection. 10 Hence, the design of performance fabric with an omniphobic surface coating is a key interest of current research to protect the body from water, oils, toxic industrial chemicals, petroleum and lubricants. 11
The development of an omniphobic surface on textiles requires a combined action of several techniques or a combination of different materials. 12 A water-oil repellent surface layer can be developed on PU-coated fabrics after treatment with materials like fluoropolymers with low surface tension. Fluorocarbon-based materials are traditionally used in oil-repellent coating because of their low surface energy. 11 Such substances are comprised of close-packed CF3-groups at the end of the polymer chain. 13 With high electronegativity and low polarisability of C–F bonds, they have low intermolecular attractive forces; therefore, these materials are oleophobic in addition to being hydrophobic/nonpolar. 14 Due to its low surface energy, the omniphobic surface exhibits self-cleaning properties as well as resistance to water, oil and liquid chemicals penetration. 15 However, the coating adds an extra layer to fabric materials that are usually impermeable in both directions. 16 Hence, the thermal comfort of clothing is severely affected because the transmission of air and perspiration vapour through the impervious coating is hindered.
Designing chemical protective textiles with maximum protection and minimum heat stress entails two critical attributes that are difficult to accomplish in a clothing system. 17 CPC is expected to be completely impermeable to oil, water and other liquid chemical penetration. At the same time, it also needs to have a mechanism for the transferring of metabolic heat and sweat from the body. The resistance against penetration of liquid should be absolutely the major concern, and any porosity in the protective layer for metabolic heat transmission may have a catastrophic effect on the protection performance of clothing. The balance between protection and thermal comfort is, therefore, the crucial issue in fabricating any chemical barrier material. Considering this complex aspect of CPC, research efforts are being directed widely to the development of novel CPC having maximum protection performance and an acceptable level of thermal comfort.18–20
Moiz et al. 12 developed a superomniphobic surface on cotton fabrics via coating of silicone rubber and fluoropolymer. Shen et al. 21 designed a chemical repellent robust coating based on aramid for protection against organic and inorganic liquid chemicals. Besides, polyurethane coating with the combination of polydimethylsiloxane and trimethylated silica on cotton 11 and poly-cotton fabrics 22 was developed for versatile chemical protection. All these research works demonstrated the potential application of coating on the fabric surface for reliable protection against liquid chemicals. However, the air and moisture vapour permeability of coating for thermal comfort remains a challenging matter and requires further attention. In this regard, PU-coated protective clothing via micro-cracking has been developed for chemical protection and improved breathability for thermal comfort in a contemporary research study. 23 Although any porosity in the protective layer can provide permeability to air and perspiration vapour it has a considerable negative impact on the barrier performance of the clothing, especially for protection against chemical penetration. 2 Thus, the modification of an impervious surface coating by integrating hydrophilic materials with PU can be considered as a potential alternative approach for developing CPC with permeability to moisture vapour but without compromising chemical barrier performance.
Hydrogels are cross-linked hydrophilic materials that have three-dimensional (3D) polymeric networks with numerous polar groups (–NH2, –OH, –COOH and –SO3H) in the polymer chain and can absorb a huge proportion of water in their interstitial structures.24,25 Commercial synthetic hydrogels are cross-linked sodium polyacrylate or sodium salt of poly(acrylic acid), 26 commonly known as superabsorbent polymers (SAPs). Having many carboxylate anions (–COO-) and sodium cations (Na+) in the backbone of the polymer chain, SAPs are exceptionally hydrophilic. 27 Because of there hygroscopic nature, SAPs have garnered much attention for extensive applications in healthcare and hygiene materials and personal safety products.28–30 So far, a minimal study has been conducted on the application of SAP in protective textiles, including in chemical barrier clothing systems. Our earlier study has revealed the successful application of SAP in the chemical protective coating as a hygroscopic material for thermophysiological wear comfort of clothing. 9 However, high thickness and surface roughness because of integrating SAP particles in the coating created several undesirable effects on the physical and functional properties of the clothing.
In this regard, we have extended our previous work by mixing the SAP solution with a PU binder for the development of a thin hybrid surface coating on cotton fabric. The treatment of the coated surface with fluoropolymer is anticipated to develop an omniphobic surface that can repel water, oils and other liquid chemicals. The development of a PU–SAP–fluoropolymer coating membrane on the fabric surface can be considered as an innovative approach that has versatile protection performance and improved transmission of perspiration vapour. Accordingly, this new technique of coating can be employed for the fabrication of high-performance textiles accomplishing with chemical barrier performance and adequate thermal comfort.
Experimental
Materials
Cotton woven fabric from Bruck Textiles, Australia, was used for coating. The woven fabric in the current study was selected for its dimensional stability, which is crucial during the coating of the fabric. The detailed specification of the fabric is listed in supplemental Table S1. Superabsorbent polymer powder was procured from Dongying Naxing Trading Company Ltd., China. Waterborne polyurethane coating paste (Tubicoat WP 1665) was purchased from CHT Australia Pty Ltd., Australia. The physical characteristics of both the SAP and PU binder are provided in supplemental Table S2. Oleophobol CP-C (fluorocarbon-based oil-water repellent chemical) was obtained from Huntsman, Germany. Liquid chemicals methanol, acetone, N, N-dimethylformamide (DMF), toluene, sulphuric acid and sodium hydroxide were obtained from Chem-Supply Pty Ltd., Australia. Hexadecane, n-tetradecane, n-dodecane, n-decane, n-octane and n-heptane were procured from Sigma Aldrich Pty Ltd., Australia.
Preparation
Coating of cotton fabric
The coating paste for the development of coated cotton fabric was prepared by using a superabsorbent polymer solution and polyurethane binder. The SAP solution (0.75% w/v) was prepared by dissolving the SAP particles in water. The SAP solution was then added with the PU binder paste at different ratios (PU: SAP = 75: 25 and PU: SAP = 50: 50) and stirred continuously until a homogeneous solution was achieved. The maximum concentration of the SAP up to the ratio of 50: 50 could be maintained in the binder paste, with the required viscosity suitable for coating.
A knife (blade) coating machine was used to conduct the coating on the fabric surface. At first, fabric samples were coated with PU and the mixture of PU and SAP solution. The coated fabrics were dried at 60°C for 40 min to solidify the coating by removing any low boiling organic solvents from liquid binder paste and then cured at 140°C for 5 min. To impart the water-oil repellent properties, the fabrics were treated in the padding machine (Ernst Benz Pad m/c) using the omniphobic finishing agent Oleophobol CP-C. During the padding, the fabrics were immersed in the solution (10% v/v) of Oleophobol CP-C and then passed through between the rollers of the machine. After padding, fabric specimens were dried at 60°C for 30 min and cured at 120°C for 3 min. The design of experiment and the test fabric coding are shown schematically in Figure 1. Schematics of the experimental design and coding of fabric material.
Characterisation and measurement
Morphological analysis
The morphological study of the test specimens was conducted using scanning electron microscopy (FEI Quanta 200 ESEM). The observation was performed in low vacuum mode at the accelerating voltage of 15 kV. The specimens were coated with Au for 60 s using a sputter coater (SPI Sputter Coating Unit, model 11430, USA). An Fourier transform infrared (FTIR) spectrometer (PerkinElmer Spectrum 400 FTIR, USA) was used for the chemical characterisation of the coated fabrics.
Fabric physical properties
The weight, that is, mass per unit area of fabric was determined as per the standard ASTM D3776-96. The measurement of fabric thickness was carried out using a fabric thickness tester followed by the method ASTM D1777–15. Fabric stiffness was determined as per the standard ASTM D1388-08 by using Shirley stiffness tester, from Shirley Development Ltd., England.
Water resistance of fabrics
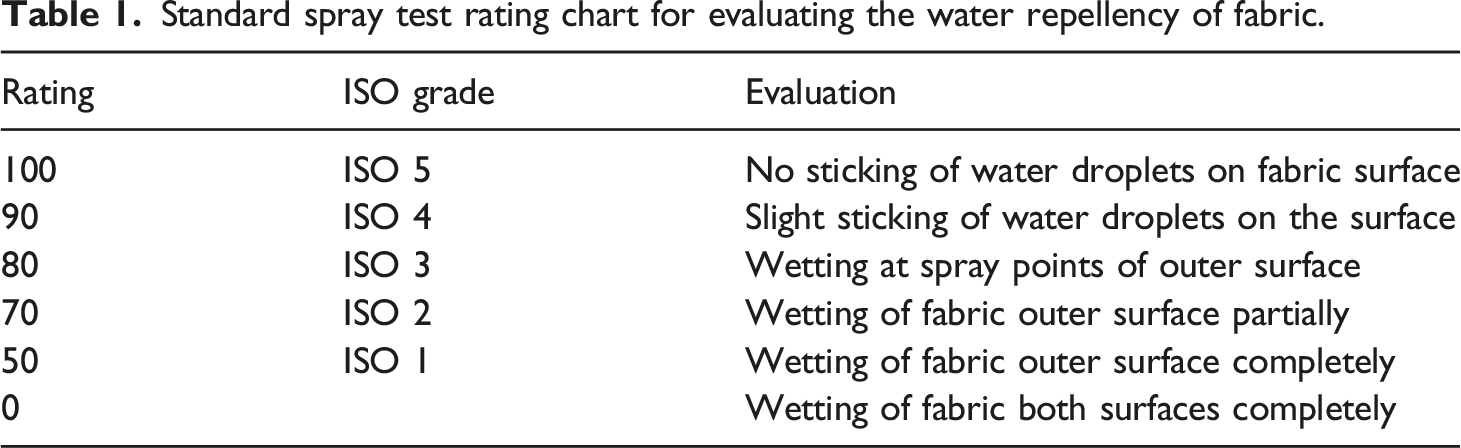
Standard spray test rating chart for evaluating the water repellency of fabric.
Oil and aqueous liquid repellency
The repellency of fabric against oil and the aqueous liquids (water alcohol solution) was carried out as per the test method AATCC 118-2013 (oil repellency: hydrocarbon resistance) and AATCC 193-2017 (liquid repellency: water/alcohol solution resistance), respectively. The test was conducted using eight different oil and the aqueous liquid solutions of different surface tensions, as shown in supplemental Table S3. For both experiments, the fabric specimen was placed on a flat and horizontal smooth surface, and a small volume (50 μL) of liquid was dropped on the fabric through the dropper tip, held at the height of 0.60 cm from the specimen. Then the wetting and wicking of the liquid at the liquid-fabric interface were observed at an angle of 45° (approximately) for 30 ± 2 s and 10 ± 2 s for oil and aqueous liquids, respectively.
Resistance to chemical penetration
The chemical resistance of fabric was determined following the method BS ISO 22608:2004 (Protective clothing—Protection against liquid chemicals—Measurement of repellency, retention, and penetration of liquid pesticide formulations through protective clothing materials). The liquid resistance was assessed in terms of fraction repellency, retention, and penetration of chemicals through the fabric. The test was performed using different organic and inorganic liquid chemicals specified in the standard ASTM F1001-12 (Standard Guide For Selection of Chemicals to Evaluate Protective Clothing Materials). The detailed procedure of the experiment was stated in our previous study. 9
Clothing thermal comfort
The thermal comfort of clothing was evaluated in terms of thermal and evaporative resistance, permeability to air and the moisture management performance of the fabric. The dry and evaporative heat resistance of fabrics was determined under steady-state conditions. The test was conducted by using an Integrated Sweating Hotplate (Model 431-213) according to ISO 11092:2014 (Textiles—Physiological effects—Measurement of thermal and water-vapour resistance under steady-state conditions (sweating guarded-hotplate test)) method. The detailed test procedure and standard atmospheric conditions of the test chamber for both investigations are mentioned in our previous report.
2
Besides, the moisture permeability index or evaporative cooling index (imt) of fabric was calculated using equation (1).
The air permeability of fabric was determined following the standard ISO 9237:1995 (Textiles—Determination of the permeability of fabrics to air), using an air permeability tester. The mean air permeability of each fabric was calculated at least seven air flow readings and presented in mL·cm−2·s−1. The moisture management profile of fabrics was assessed by employing a moisture management tester (MMT) (M 290, SDL Atlas Ltd.), followed by the method AATCC-TM-195-2011 (Liquid Moisture Management Properties of Textile Fabrics).
Results and discussion
Protective surface coating on cotton fabric with resistance to water, oils and liquid penetration was investigated for simultaneous liquid barrier performance and clothing thermal comfort. The development of chemical protective clothing involved an initial surface coating of fabric using PU and PU–SAP and then padding with a fluorocarbon polymer solution. The subsequent drying and curing process established a durable coated layer on the fabric surface. As a result, the surface morphology, thickness and handle of the coated cotton fabrics have been altered. Meanwhile, improved chemical protective performance with evaporative heat transmittance were observed.
Morphological analysis
The morphological investigation of fabrics was conducted to determine the changes in surface properties upon coating. Optical and scanning electron microscopy (SEM) was employed to analyse the cloth surface topography and composition. The optical image in Figure 2(a1) shows a clear weave structure with distinct porous surface of uncoated cotton fabric. The typical morphology of the cotton, including a clear fibrous structure with a well-defined ribbon-like convolution,
9
can be observed from the SEM micrographs of the uncoated fabric (Figure 2(a2)). Surface morphology of test fabric specimens. Optical images and corresponding scanning electron microscopy images of (a1, a2) uncoated fabric, (b1, b2) polyurethane-coated fabric before and (c1, c2) after padding, (d1, d2) PU–SAP (50:50) coated fabric before and (e1, e2) after padding. Scanning electron microscopy. Insets: cross-sectional views of uncoated and coated fabrics.
The optical images of the coated specimens display a continuous PU and PU–SAP layer with a glossy appearance on the surface of the materials (Figures 2(b1) and (c1)). The lustre of the coating is slightly decreased after padding the fabrics with the fluoropolymer (Figures 2(d1) and (e1)). The continuous coating layer on the fabric is confirmed by the SEM micrographs (Figure 2(b2) and (c2)). This surface morphology indicates that the fabrics were covered successfully by filling the gaps between the fibres with a thin coating of PU and PU–SAP. A similar surface property with no considerable variation of coating can be observed for the fabrics after padding (Figures 2(d2) and (e2)). The continuous covering of fibrous materials after coating suggests the development of a consistent protective layer on the surface of the fabric that can be employed as an effective barrier against the penetration of water, oil and liquid chemicals through the coating.
FTIR analysis
The FTIR spectrum of uncoated cotton fabric (UC) in Figure 3 shows a characteristic broad peak located at 3337 cm−1, which belongs to OH–stretching vibration, specifying the hydrogen bonding of cellulose in the cotton fibre.
31
The C–O–C stretching of β(1→4) glycosidic linkages in cellulose polymer was detected from the absorption band at 1023 cm−1.
32
Other prominent peaks, including –CH stretching band at 2919 cm−1 and –CH wagging at 1314 cm−1, were also observed in the chemical structure of cotton fibre.
33
Fourier transform infrared (FTIR) spectra of uncoated and coated fabrics: (a) before padding and (b) after padding.
In the case of fabrics after coating (Figure 3(a)), the >C = O bond of urethane groups of PU are confirmed from the strong absorption at 1724 cm−1. 34 The extra peaks at 2954 cm−1 and 2845 cm−1 correspond to the asymmetric and the symmetric stretching of methylene (–CH2–) groups. 35 The –NH and –CH2 vibrations were detected by the peaks at 1448 and 1260 cm−1, respectively. 36
The broadening in the band between 3200 cm−1 to 3600 cm−1 corresponding to OH-stretching of cotton fibre (Figures 3(a) and (b)) indicates the formation of H-bond between hydroxyl groups (–OH) of cellulose polymer and carbonyl groups (>C = O) of PU. 37 This chemical interaction suggests the interfacial bonding of polyurethane with cotton fibre after the coating. In the spectra of fabrics treated with fluoropolymer (Figure 3(b)), the strong peak at 1143 cm−1 represents the symmetric stretching vibration of the peripheral CF2 groups. 12 In addition, the existence of the fluoropolymer in the coating is confirmed from the C–F stretching at the region of 1235 cm−1 of the spectra, 38 as shown in Figure 3(b). Besides, a possible chemical interaction between the carbonyl group of PU and electronegative fluorine of fluoropolymer is predicted due to the change of intensity and shifting of carbonyl absorption peak to a higher frequency at 1730 cm−1. The presence of fluoropolymer on the surface of the coated layer and the strong interfacial adhesion between polyurethane and the constituent cotton fibres indicate the formation of a stable omniphobic barrier layer on the fabric that can provide consistent liquid chemical protection by repelling water, oils and aqueous liquids of various surface tensions.
Fabric physical properties
The physical parameters of the fabrics, including the thickness, weight and stiffness, were measured to determine the changes in material characteristics after coating. Both the thickness and weight (mass per unit area) have increased significantly after the coating of fabric (Figure 4(a)). The development of a thin layer on the fabric surface due to the coating has contributed to this increase in thickness and weight of the coated fabrics. However, the fabrics coated with PU–SAP are thinner and lighter compared with the PU-coated specimens, and the thickness and weight decreased as SAP concentration in the coating increased. Fabric physical parameters: (a) thickness and weight, and (b) flexural rigidity of uncoated and coated cotton fabrics.
The stiffness of fabric expressed in terms of flexural rigidity decreased considerably after the coating and all the fabrics showed a clear difference in flexural rigidity between lengthwise and widthwise directions. Among the coated specimens, a higher stiffness in both directions was found for the fabrics coated with PU–SAP. This is due to the change in flexibility of PU after the integration of SAP in the solution. The blending of SAP with PU created a negative impact on the flexibility of the coating, which increased with the SAP concentration. Therefore, the coated specimens that had the maximum SAP demonstrated the highest flexural rigidity, as revealed in Figure 4(b).
Protective performance
Water resistance of fabric
Test results showed that cotton fabric without coating exhibited a zero (0°) contact angle because of its excellent hydrophilicity. The existence of countless polar hydroxyl groups (−OH) in the cellulose polymer
24
was responsible for such hydrophilicity of the cotton fibre. Hence, water was absorbed rapidly without forming an angle when dropped onto the cloth surface. However, the WCA has increased abruptly after coating the fabric with a PU membrane. The low PU surface energy contributed to this significant improvement of hydrophobicity on the coated fabric. Therefore, PU-cotton fabric exhibits a high contact angle with an average value of 84.6°, as shown in Figure 5(a). Protective performance concerning water-resistance of fabric: (a) water contact angle, and (b) water repellency of fabrics before and after washing.
A slight decrease in WCA was noticed after integrating the SAP in the coating. The blending of hydrophilic SAP with PU binder has altered the surface energy of the coated fabric. This high surface energy reduced the contact angle by employing strong solid–liquid interaction between the coated surface and water droplets. 9 However, the treatment of coated cotton fabrics with a fluoropolymer solution results in a further improvement of the hydrophobicity of the coating as demonstrated by a maximum WCA with minimum surface energy (Figure 5(a)). This high WCA suggested the development of a superhydrophobic surface on the fabric, where liquids will form into a spherical droplet and roll off the surface of the clothing. 39 The hydrophobicity of fabric was slightly reduced after laundering, and the fabric PF, SF1 and SF2 showed the average WCA of 112.8°, 109.5° and 105.7°, respectively. The minimal change of WCA indicates the development of durable polyurethane–fluoropolymer coating on the surface of cotton fabric. 12 Besides, no significant change of WCA was also observed for the fabrics coated with PU and PU–SAP, suggesting the formation of a stable layer on the fabric surface.
The change of fabric water repellency due to coating and padding is presented in Figure 5(b). The uncoated cotton fabric possesses high wettability because of its excellent hydrophilicity, as discussed earlier. Thus, the fabric demonstrated the lowest repellency rating of 0, which means wetting on both the inner and outer surfaces after showering (Table 1). A considerable improvement of repellency, with a rating of 90 (ISO 4), was observed after coating the fabric with PU only. The decrease of surface energy because of adding the hydrophobic PU membrane to the fabric surface enhanced the overall repellency of the fabrics after coating. Analogous water repellency was also noticed for the fabric (PS1) coated with PU–SAP with a low concentration of SAP solution. However, PU–SAP coated fabrics (PS2) with a higher SAP concentration exhibit a slightly decreased water repellency rating (ISO 3), as shown in Figure 5(b). The incorporation of hydrophilic SAP with PU during the coating created a negative impact on the hydrophobicity, which leads to the low water repellency rating of the fabric. This water repellency rating of fabrics was almost similar and did not change considerably after laundering. The water repellency of fabrics treated with fluoropolymer decreased slightly and exhibited ratings of 90. However, before washing, the fabric specimens PF, SF1 and SF2 exhibited the maximum repellency grade of 100 (ISO 5). The low surface energy of fluoropolymer after padding imparted a maximum hydrophobicity to the coated surface. Thus, the fabric specimens demonstrated the highest water repellency rating (100) with no sticking or wetting on the top surface of the material. Accordingly, the fabrics with the maximum repellency rating can be employed to develop protective garments for reliable protection against penetration of water through the clothing.
Oil and aqueous liquid repellency
The test of fabric oil and aqueous liquid repellency was conducted to determine the resistance of textiles to wetting against a selected series of liquid hydrocarbons and water/alcohol solutions of different surface tensions. Both oil and aqueous liquid repellency of fabrics were assessed using a grade number with numerical values from 0 to 8. A fabric with no repellency to the liquid is assigned to the grade zero (0), while the maximum grade of eight (8) is referred to as no wetting against the highest numbered test liquid within a specific time.
The uncoated cotton fabric did not show any repellency against oil and aqueous liquid solutions because of its high surface energy, which leads to the zero (0) repellency grade against the solutions (Figures 6(a) and (b)). The surface energy of the fabric has been reduced considerably after coating with the PU; therefore, PU-coated specimens demonstrated an improved repellency grade of 3.5 against oil (n-tetradecane) and 5.5 for aqueous liquid solutions (60:40/water: isopropyl alcohol). However, a slight decrease in repellency shown by PU–SAP coated specimens against both liquids was due to the addition of hydrophilic SAP in the coating. Protective performance concerning oil and aqueous liquid repellency of fabric: (a) aqueous liquid repellency, and (b) oil repellency grade of uncoated and coated cotton fabrics.
A considerable improvement in oil and aqueous liquid repellency was observed after the treatment with fluoropolymer. Both the PU and PU–SAP coated fabrics after padding demonstrate excellent repellency against aqueous liquids (PF = 8.0; SF1 = 7.5 and SF2 = 7.5), as shown in Figure 6(a). The low surface energy of the coated surface after padding played the key role in this resistance to aqueous liquids. The surface tension of the water/alcohol (98:2) solution was 59.0 dyn/cm, which decreased to 24.0 dyn/cm (40:60) with the increment of the water/alcohol ratio (vol:vol) in the solution (Supplemental Table S3). However, the omniphobic fluoropolymers had the surface tension of 16.2–18.2 dyn/cm, 12 which was considerably lower than aqueous liquid solutions. This low surface energy of fluoropolymer incorporated superhydrophobicity on the coated surface after padding, and therefore, exhibited excellent repellency against aqueous liquids. Accordingly, the aqueous liquids formed an almost sphere-shaped droplet on the fabric surface, as revealed in the photographs of Supplemental Figure S1.
In the case of oil repellency, the fabric specimens exhibit a slightly inferior repellency grade against the oils, as shown in Figure 6(b). This was due to the lower surface tension of hydrocarbon solutions compared with the aqueous liquid solutions supplemental (Table S3). The oil solutions with higher surface tension were repelled by the fabrics treated with fluoropolymer (Supplemental Figure S2). However, among the hydrocarbon solutions, n-heptane has the lowest surface tension of 19.8 dyn/cm, which is very close to the surface energy of coated fabrics, and therefore, not repelled by most coated specimens. Nevertheless, the treatment of fluoropolymer contributed to the significant reduction of the surface energy of the coated surface, which is sufficient for repelling oils and aqueous liquid solutions satisfactorily. This repellency property of the surface confirmed the development of an omniphobic barrier layer on the fabric that can resist the penetration of oils and aqueous liquids through the clothing.
Resistance to liquid chemical penetration
Chemical resistance concerning repellency and penetration of chemicals through fabric.

Retention of liquid chemicals by polyurethane (PU) and PU–superabsorbent polymer coated fabric after the evaporation loss.

Clothing thermal comfort
Dry and evaporative heat resistance and air permeability
The change of fabric dry heat resistance before and after the coating is shown in Figure 7(a). The PU-coated fabric exhibited the least thermal resistance; however, the thermal resistance increased gradually after adding the SAP in the coating solution. The heat resistance of clothing is mainly influenced by fabric construction and its porosity, along with the thermal conductivity of constituent fibres.
40
An increase in heat resistance was predicted after the coating, because an entire covering of fabric pores by a continuous layer on the surface prevents easy transferring of heat through the fabric. However, in practice, uncoated cotton specimens demonstrate a greater heat resistance compared with the PU–cotton fabric (Figure 7(a)). This might be due to the formation of strong interfacial bonding between PU and the cotton fabric after coating, as well as the significant difference in thermal conductivity of PU (177 mW.m−1. K−1)
41
to the cotton fibre (71 mW.m−1. K−1).
42
Accordingly, the very high thermal conductivity of polyurethane outweighed the predicted thermal resistance of coated material by improving the dry heat flow via the interface of PU–cotton fabric because of strong interfacial adhesion.
9
As a result, an overall decrease in thermal resistance was observed for PU-coated specimens. In contrast, fabrics coated with PU–SAP demonstrate an increased dry heat resistance, as shown in Figure 7(a). The high thermal resistance of SAP, due to its containing metallic ions (Na+ and K+) in the structure of cross-linked polyacrylate,
43
contributed to this improved dry heat resistance; therefore, thermal resistance of fabric increased gradually with the increment of SAP concentration. However, no significant change in thermal resistance was found after padding the specimens with fluoropolymer solution, and the coated specimens demonstrated almost similar dry heat resistance before and after the padding. Thermal comfort of clothing: (a) dry heat resistance (R
cf
), (b) evaporative resistance (R
ef
), (c) moisture permeability index (i
mt
), and (d) fabric air permeability.
The evaporative resistance (R ef ) in Figure 7(b) shows an abrupt change in moisture vapour transmittance of fabric after coating. The uncoated specimens have the minimum evaporative resistance, and the maximum was noted for the fabrics coated with PU only. Conversely, a considerable decrease in evaporative resistance can be observed after adding SAP with PU in the coating (Figure 7(b)). The coated fabrics with the high SAP concentration (PS2) demonstrated a minimum moisture vapour resistance among the coated fabric specimens. The key mechanism behind the fact is the presence of carboxylate anions (–COO–) and sodium cations (Na+) in the cross-linked network of sodium polyacrylate, as mentioned earlier. Hence, SAP showed strong hydrophilicity owing to the natural interactions between carboxylate anions and water molecules. This hydrophilic nature of SAP facilitated the transfer of water molecules through the sodium polyacrylate due to the free movement of sodium ions (Na+) in the cross-linked structure. 44 Accordingly, SAPs became activated when they encountered the moisture vapour and initiated the transfer of the water molecules through the coating by diffusion. This diffusion of liquid moisture leads to the gradual reduction of evaporative resistance of coated fabrics and, consequently, the greater thermal comfort of the clothing.
A slight increase in evaporative resistance of the coated specimens treated with fluoropolymer in Figure 7(b) was resulted from the development of surface hydrophobicity on both sides of the fabric after padding. This hydrophobicity created an initial resistance to moisture vapour transmission via the inner side to the face side of the material. Therefore, the fabrics with padding (PF, SF1 and SF2) demonstrated an increased evaporative resistance compared with the fabrics without padding (PC, PS1 and PS2). Nevertheless, the PU–SAP coated fabrics with the maximum SAP concentration (SF2) showed adequate low moisture vapour transmittance (Figure 7(b)), which lies within the acceptable limit of the fabric evaporative resistance (0–30). 45 Accordingly, the high evaporative transmittance together with low thermal resistance leads to an increased moisture permeability index (i mt ) of fabrics coated with PU–SAP (Figure 7(c)). This improved moisture permeability index can create a favourable thermal comfort microenvironment within the apparel–skin in hot and humid conditions. 46
In the case of fabric air permeability, uncoated specimens demonstrate the maximum breathability (6.2 mL.cm−2.s−1), as presented in Figure 7(d). An abrupt decrease of permeability was noted for the fabric after coating. The breathability of textile materials primarily depends on fabric porosity, that is, the cover factor and the structure of the fabric. 47 Accordingly, the numerous holes in the uncoated fabrics provided adequate porosity that allowed the transfer of air through the clothing and therefore exhibited a high permeability. On the other hand, the significant reduction of breathability after coating was to continuous coverage of the fabric surface, which filled in fabric pores and hindered the transfer of air through the clothing.
Air permeability is considered as one of the primary fabric characteristics concerning thermophysiological wear comfort, and this parameter is commonly used in assessing the breathability of textile materials. 48 However, it can be mentioned that protective clothing with resistance to liquid penetration must be impervious for reliable protection, and any porosity of the fabric for the permeation of air could be catastrophic for chemical protection. Hence, the development of a chemical protective outfit via the continuous PU–SAP coating with a low air permeability and enhanced moisture vapour transmittance (Figure 7(b)) can be considered an effective mechanism for thermal comfort without declining the barrier performance of clothing.
Moisture management performance
Moisture management performance profile of test specimens.

OWTC: one-way transport capability; OMMC: overall moisture management capacity

Transfer of liquids from the inner (top) side to outer (bottom) side of fabric.
The moisture management profile of fabrics was altered completely after coating. The continuous layer of coating made the fabrics impervious in both directions and restricted the liquid penetration through coating due to the coverage of open pores in the fabric. Therefore, coated fabrics showed zero absorption and spreading of liquids to the outer surface. Consequently, the specimens show a negative value of OWTC and zero overall moisture management capacity OMMC, as presented in Table 4. The padding of fabrics with fluoropolymer incorporated superhydrophobicity on the coating that limited the spreading of water on the inner side of the fabric (Figure 8) and made the material more impermeable to liquid transfer.
The moisture management profile of a fabric indicates its ability to absorb the liquid sweat from the body and diffuse it to the air to retain the thermal balance of the wearer. 49 The impermeable nature of the coating created a negative impact on the performance of moisture management due to inhibiting the liquid transfer that resulted in the inferior comfort of clothing relating to wet clinginess. However, a positive view of such resistance confirmed the formation of a stable layer on the cloth surface, providing reliable protection against liquid chemicals.
Conclusion
The development of omniphobic PU–SAP–fluoropolymer surface coating on the cotton fabric, and its resistance against water, oil and liquid chemicals, as well as permeability to moisture vapour for thermal wear comfort, were investigated. The morphological study of coated cotton fabrics revealed the formation of a continuous and stable layer on the fabric surface owing to the durable interfacial adhesion between polyurethane and the constituent cotton fibres. Increased thickness and weight and a change of fabric handle properties were observed for the coated fabrics. The coating on cotton fabric with PU and treatment with fluoropolymer incorporated low surface energy with substantial hydrophobicity, which leads to a high-water contact angle with a maximum water repellency rating of 100. A satisfactory oil and aqueous repellency grading with no penetration of liquids through coating suggested the formation of a stable protective layer on the fabric that can be employed for reliable protection against water, oil and liquid chemicals. Concerning thermophysiological comfort, improved thermal resistance with moisture vapour transmission and consequently a high moisture permeability index of PU–SAP–fluoropolymers coated fabrics are expected to provide a certain degree of thermal comfort in hot and humid conditions by transferring metabolic heat from the body to the environment. The reduction of fabric air permeability with an unaltered moisture management profile indicated that a continuous and hydrophobic barrier layer on the fabric was formed to resist the transfer of air and liquid sweat through the fabric. This study demonstrated a prospective application of a PU–SAP–fluoropolymer coating on the cotton fabric surface for functional textiles with simultaneous barrier performance and thermal comfort. This will further benefit the development of protective clothing that can be employed in any emergency operation with minimal body heat stress to the user in a hot and humid environment.
Supplemental Material
sj-pdf-1-jit-10.1177_15280837221078535 – Supplemental Material for Omniphobic polyurethane – superabsorbent polymer – fluoropolymer surface coating on cotton fabric for chemical protection and thermal comfort
Supplemental Material, sj-pdf-1-jit-10.1177_15280837221078535 for Omniphobic polyurethane – superabsorbent polymer – fluoropolymer surface coating on cotton fabric for chemical protection and thermal comfort by M A Rahman Bhuiyan, Lijing Wang, Zinia Anjuman Ara, Tanushree Saha and Xin Wang in Journal of Industrial Textiles
Footnotes
Acknowledgments
M. A. Rahman Bhuiyan is thankful to RMIT University, Australia for scholarship support towards his PhD study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
