Abstract
Protection is obligatory for the safety of people in certain occupations where they might be exposed to hazardous chemicals. This review provides an overview of chemical protective clothing, along with its necessity during industrial and military operations as well as in response to acts of terror. Moreover, chemical protective or barrier suits are illustrated and explained including their types, selection processes based on chemical hazards, working environments, and various materials available for the fabrication of effective barrier clothing. Additionally, this review elucidates current research gaps, while underscoring the challenges facing recently developed chemical protective clothing, by compiling relevant research onto a single platform. Besides, this review includes and delineates future trends in chemical protective outfits based on electro-spun nano-fibre technology involved in both detection and decomposition of poisonous chemical agents that come in contact with clothing material, and the integration of selectively permeable membrane technology to discriminately block hazardous chemicals.
Keywords
Introduction
Protective textiles are defined as garments and other fabric-related items that are used primarily for their protection performance or functional characteristics, rather than their aesthetic or decorative purposes [1]. Clothing materials being used as personal protective equipment is now a significant application for textiles and they are considered as technical or industrial textiles [2]. Personal protective textiles are classified as industrial protective textiles, agricultural protective textiles, military protective textiles, civilian protective textiles, medical protective textiles, sports protective textiles and space protective textiles based on their specific applications [3]. The main purpose of barrier textiles is to protect the human body against external hazards such as chemical, thermal, mechanical, biological and radiation while maintaining a safe and comfortable micro-climate around the skin. Among various hazards, protection against chemicals and chemical warfare agents (CWAs) is currently considered as a most crucial issue. Frequent terrorism or biochemical attacks in some countries result in numerous injuries or deaths that make it imperative to employ chemical barrier equipment for various regular and specialised civilian and military occupations.
Chemical protective clothing (CPC) is generally used by military personnel, law enforcement officials and civilians in various wartime and peacetime operations, including chemical warfare, industrial chemical production, chemical spillage accident, riot and counter-terrorism activity [4]. A protective clothing system includes a respirator, a hood, an overgarment, overboots along with gloves. It is continually being developed to offer effective protection against exposure to toxic chemicals [5]. The gloves, which typically have five fingers, are produced in an array of materials, thickness as well as styles and designed for the safe handling of hazardous chemicals, including corrosive acids and bases. Various polymeric materials such as butyl rubber, nitrile rubber, neoprene, natural rubber latex, poly(vinylacetate) (PVA), and poly(vinyl chloride) (PVC) are commonly used for the fabrication of protective gloves [6] and the selection of these materials is mainly based on the chemical nature of the solvent that needs to be handled during an operation [7]. Besides, a chemical-biological mask intended for protecting a wearer from chemical and biological contaminants is generally employed in emergency situation where highly toxic substances are in the atmosphere. The mask forms a sealed cover over the nose and mouth, but may also cover the eyes and other vulnerable soft tissues of the face.
CPC containing halogenated polymers have been widely used through laminating or coating on a fabric to form a surface that is repellent to water and oil [8]. These coated fabrics have low surface energy, they may exhibit a lotus effect or self-cleaning properties to withstand water and organic liquids [9]. Moisture permeable protective clothing, embedded with a breathable microporous membrane has been developed to provide resistance against liquid chemicals while permitting penetration of air and water vapour [10,11]. Currently, activated carbon-based textiles are extensively used for military and civilian purposes as chemical barrier clothing. Textile combined with activated carbon allow the transmission of corporal heat and dissipate perspiration from the skin as well as provide effective protection against hazardous chemical agents through adsorption on textile and activated carbon surfaces [12,13].
Based on the level of protection, along with the operating environment, clothing systems vary in design, use of shell and liner-fabrics, assembly of various protective materials and their interfaces/closures [14]. Since protective material is the main component of CPC, it is necessary to understand different concepts and protection capabilities of current protective materials as well as their compatibility and integration while designing a chemical barrier uniform against harmful chemicals and microorganisms [15]. Additionally, it is desirable that CPC should interfere minimally with release of moisture from the human body and the passage of air, so fabrics need to be air and moisture permeable. To achieve these two apparently contradictory goals, a maximum protection without any compromise on comfort is the main challenge in developing CPC and thus a rigorous study is required in the field of chemical protection. Therefore, the present review is aimed at contemporary research on chemical protective materials, emphasizing current trends and protective capabilities. The organization of this article begins with chemical protective materials and their selection, followed by design of CPC with different materials, as well as characterising their performance against hazardous chemicals and CWAs. Various continuing developments to enhance the comfortability and several new technologies for advanced CPC are discussed in detail.
CPC
CPC refers to apparel and other fabric-related materials that are intended to protect the wearer from potentially hazardous and toxic chemicals as well as to lower the risk of injury or illness. CPC is considered the last line of defence in any chemical-handling operation [16] and the utmost requirement for a CPC is as a barrier to harmful liquid penetration to minimise or eliminate human contact with chemical hazards. The routes of chemical entry into the human body are oral, respiratory and dermal (skin), and the dermal route constitutes nearly 90% of chemical exposure [17]. Moreover, dermal exposure is considered the primary mechanism of chemical entry through the blood towards internal organs of the human body [18].
Typical chemical warfare agents that are hazardous to dermal exposure and inhalation [24].

CWA: chemical warfare agent.
Chemical barrier materials are categorised as encapsulating and non-encapsulating ensembles based on the style of wearing and specific requirements [15]. The encapsulating systems cover the whole body and include respiratory protection equipment. They are mostly used where a high level of chemical protection is required. Non-encapsulating clothing is assembled from separate components and the respiratory system is not a part of the CPC. Many well-designed and constructed CPCs have been developed. However, it is important to note that a one-size-fits-all approach is not really applicable in the case of a CPC, as there is no single protective material that can fulfil all the requirements of protection against all types of chemicals. Several experimental studies have shown that toxic chemicals can easily pass through or damage some protective clothing materials, come into contact the skin, and eventually become absorbed into the blood [26]. Therefore, considerable ongoing research has been carried out comprehensively by research organisations, manufacturers and users of CPC to explore improved and efficient materials that are best suited for specific chemical challenges.
Selection of CPC
Chemical barrier clothing that provides protection in unique situations or severe hazardous conditions should include critical specifications for the clothing selection process. Determination of the hazard level and likely type of exposure in the threatened environment is important in the selection of CPC. In all cases, the CPC must protect the user from the deleterious effects of contact with hazardous chemicals. Thus, chemical barrier outfits and total encapsulating suits are two key options that are selected as standard ensembles for effective personal protection. Personal protective equipment is divided into four categories based on the various levels of protection as defined by the United States Environmental Protection Agency (USEPA), ranging from minimal dermal and respiratory protection, level D, to maximal protection, level A [27], as explained below.
Level A: Fully encapsulating suit with self-contained breathing apparatus. The suit should be worn when the highest level of respiratory, skin, and eye protection is required [28]. The clothing system is comprised of long underwear along with chemical resistant gloves and boots, plus other safety equipment. The totally encapsulating garment is readily decontaminated, as there are fewer places for contaminants to be trapped and they may be used in oxygen-deficient atmospheres.
Level B: Hooded chemical-resistant clothing (long-sleeved jacket; one or two-piece chemical-splash suit; disposable chemical-resistant overalls) with a self-contained breathing apparatus. They offer the highest level of respiratory protection but a lesser level of skin protection [18]. These ensembles are lighter and less bulky than those of level A.
Level C: This category is of pressing need when the concentration(s) and type(s) of airborne substance(s) are known and the criteria for using air-purifying respirators are met. Level C ensembles consist of a full face or half-mask air purifying respirator including hooded chemical-resistant clothing as in level B with other safety equipment [27]. The ensemble is lighter than Level B ensemble due to their “coverall” design and thinner fabric as well as no provision for air supply is required.
Level D: A work uniform providing minimal skin protection and usually used only for nuisance contamination. No respiratory protection is required, but gloves, boots, boot coverings and other related safety equipment may be used optionally [15]. Level D protection should be employed when the atmosphere contains no known hazard and work activities preclude splashes, immersion potential for unexpected inhalation, or contact with hazardous levels of any chemicals.
Physical resistance is another important factor which must be considered for the selection of CPCs. The important physical properties that are required to evaluate the performance of barrier clothing include tear strength, tensile strength, cutting resistance, puncture resistance as well as abrasion resistance [26]. It is desirable that the best chemically resistant material should not be affected substantially by any physical forces such as tear, cutting, puncture or other damage. However, many materials provide significant resistance against perilous chemicals, but exhibit poor mechanical properties; hence, they cannot be used alone for CPCs. Besides, seam strength and seam slippage, bursting strength and flexibility are other notable mechanical properties that must be considered for the selection of articles of protective clothing. The exact desired properties depend on the working environment and the requirements of the activity to be performed.
Factors determining the resistance of CPC
The chemical resistance of CPC is mainly determined by permeation, penetration, and degradation (Figure 1) [18]. Permeation is the diffusion of chemicals through the protective clothing at the molecular level, causing direct contact with the body by absorption [29]. This movement of chemical through the protective clothing may not be readily noticeable since it occurs at a molecular or microscopic level with no obvious signs of physical degradation of the material (Figure 1(a)). For many chemicals, permeation is the most important factor in determining chemical resistance and the permeation of protective clothing is determined by two important parameters, breakthrough time and permeation rate [30]. Breakthrough time refers to the total time it takes for contagions to pass through the protective material until it is first detected by an analytical instrument [31]. A normalized breakthrough time refers to the time at which the chemical under evaluation is first detected at the inner surface of the test material [32].
Schematic representation of (a) permeation (b) penetration and (c) degradation mechanism of chemicals moving through barrier clothing.
The rate at which a chemical moves through the clothing material and reaches break through, is called the permeation rate [33]. The permeation rate depends on the solubility and the diffusion coefficient of the chemical in the material and it can be measured directly using a permeation cell or it can be calculated from solubility and diffusion coefficient data [32]. The higher the permeation rate, the faster the chemical will move through the material, and a lower permeation rate of CPC is always desirable under the conditions of use. The rate of permeation of chemicals through CPC is influenced by different factors such as the type of chemical and its concentration, the thickness of fabric, environmental humidity, temperature and pressure applied on clothing material.
Apart from permeation, another important factor of protective clothing is penetration, which is defined as the bulk flow of chemical through physical spaces of the barrier material (Figure 1(b)) [29]. Penetration may occur when a chemical leaks through the seams, zippers, pinholes, tears, rips and other imperfections in the clothing. Resistance to degradation against highly corrosive chemicals such as acids and alkalis is another important parameter to determine the performance of CPC. Degradation refers to the change in chemical structure of the clothing material as a result of adverse effects of the chemical contaminant (Figure 1(c)). The consequences of degradation may cause the material to soften, swell, shrink, stretch, dissolve, or to become hard and brittle. It is expected that an appropriate chemical barrier material must exhibit no penetration, higher breakthrough time than the duration of the task being undertaken as well as low permeation rate along with no significant degradation upon contact with the poisonous chemicals exposed.
Materials for designing CPC
The design of CPC requires multidisciplinary skills for the development of new products with high technological content. Hence, a successfully designed protective suit must have all specific functional requirements and all components optimised in their mutual system in use, as well as maximising the protection and eliminating or minimising the risk propensity effectively [34]. Moreover, the CPC must balance functional elements with both structural design and aesthetic considerations, along with the important subjective element of comfort. As a result, the design of CPC involves using various types of materials including impermeable, semi-permeable and air-permeable materials for a wide range of applications [4].
Impermeable materials
Critical surface tension values of some polymers used in impermeable clothing materials [36].

Being impermeable, these polymers hinder the transmission of perspiration, leading to obstacles against the normal cooling processes of the human body (Figure 2) [37]. Consequently, the prolonged use of impermeable CPC in warm/hot climates significantly aggravates the danger of body heat stress due to inhibition of sweat evaporation and dry heat loss [38]. Nevertheless, improved toxicological agent protective (ITAP) ensembles and the self-contained toxic environment protective outfit (STEPO) systems developed by the U.S. Army employed impermeable clothing for soldiers as well as civilians during emergency operations involving chemical spills, toxic chemical handling and cleanups in severely contaminated environments, having higher risk concentrations for short-term use [16]. A microclimate cooling system is an integral part of these protective clothing systems to compensate for their inability to permit moisture permeation evaporative cooling. The embedded microclimate cooling system enables reduction of heat stress of impermeable clothing by allowing vapour permeation through the fabric; however, these systems are expensive and increase bulkiness of the suit after donning [14].
Schematic representation of water vapour resistance of impermeable protective clothing [16].
Semi-permeable materials
Semi-permeable barrier materials like porous membranes have been successfully used in the accomplishment of selective and efficient operations, providing the opportunity to incorporate various encouraging aspects of membrane engineering in some strategic industrial sectors, including protective textile clothing [29,39,40]. Membranes act as a barrier towards toxic and undesirable materials without inhibiting perspiration together with no significant reduction of chemical protection. The membranes work as a filter with ability to allow controlled water vapour permeability.
CPC assembled with semipermeable membranes (Figure 3) of different pore sizes, ranging from macro-pores (pores of micrometres or tens of nanometres in size), micro-pores (nano-sized pores), to ultra-pores (molecular sieving pores) [4]. Macro-porous membranes, that are resistant to liquid water penetration and allow convective flow of air, aerosols, and vapours transmission through their large pores, have become key materials for many practical applications such as protective clothing and shoes, filters and separation media, and medical supplies [41]. The fascinating performance of waterproofness and breathability results from the porous structure formed by hydrophobic raw materials, which could block water droplet penetration and serve as interconnected passageways for vapour transmission simultaneously [11].
Schematic representation of water vapour transfer mechanism through textiles embedding a breathable barrier membrane [16].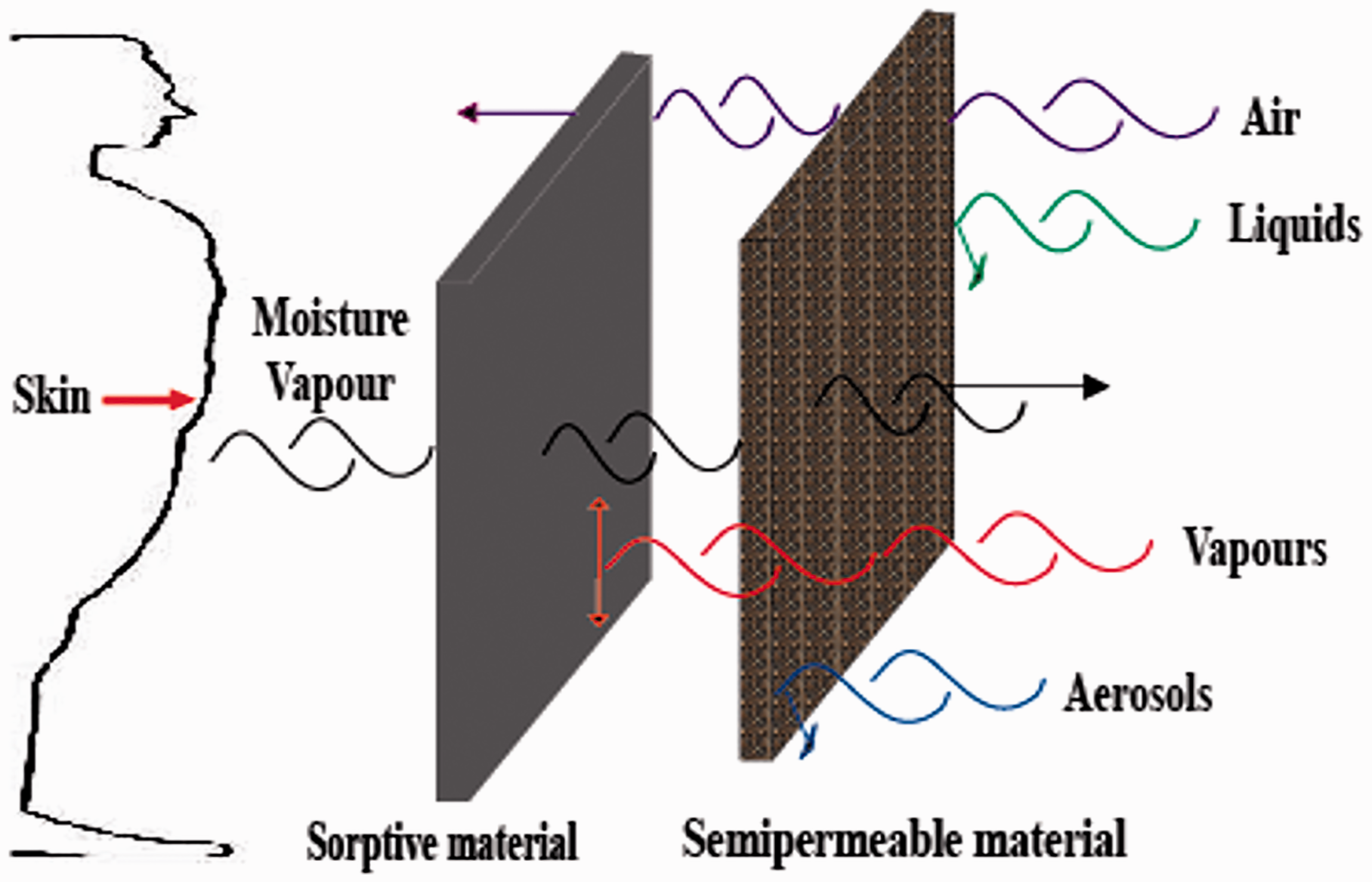
For a micro-porous membrane, lighter gas molecules are allowed to preferentially diffuse through nano-sized pores by following Knudsen diffusion [16]. This diffusion occurs when the scale length of a system is smaller than the mean free path of the particles involved. Many types of micro-porous membranes with different chemical compositions have been employed in protective clothing systems. However, choice of polymer materials depends on the desired structure of membrane. Generally, hydrophobic materials are preferred for manufacturing micro-porous membranes, where a barrier function against liquid water is required [11]. A micro-porous semipermeable membrane combined with an activated carbon containing foam is being used by the USA Army for improved chemical protection along with better comfortability [2]. Protective clothing imbedded with an ultra-porous membrane is typically employed to exclude large molecules from the pores by virtue of their size. Ultra-porous membranes along with macro-porous and micro-porous membranes employed in CPC are mainly manufactured from various polymers, including expanded PTFE, sulfonated poly(phenylene sulphide) (S-PPS), sulfonated poly(styrene-isobutylene-styrene) (S-SIBS) and sulfonated tetrafluoroethylene (Nafion) [42–44].
Selectively permeable membrane
A selectively permeable membrane allows the permeation of water-vapour molecules, but resists to the permeation of organic molecules. This selective permeability for water-vapour is interesting for medical and agricultural applications and can be applied in the apparel industry, especially in protective clothing systems [45]. Being commonly known as solution–diffusion membrane [46], this type of membrane usually follows Fickian diffusion theory and contains different types of polymers having the combined properties of impermeable and semipermeable materials [16]. As shown in Figure 4, the SPM technology permits selective permeation of moisture vapour from the body of the wearer through protective clothing layers by continuous evaporative cooling, and consequently, a gentle decrease in the temperature of the skin and the tissue underneath [47]. It simultaneously provides optimum protection against toxic chemicals along with reduced heat stress, weight and bulkiness for the wearer [40].
Mechanism of selective vapour transport through selectively permeable membrane in a 3-layer protective clothing [48].
A high level of reliable protection of SPMs is provided by a moisture-activated functional film through a selective solution/diffusion process, while existing carbon-based protective clothing fabrics rely on the adsorption process associated with porous-activated carbon materials [14]. SPMs consist of multi-layer composite polymer systems developed by using various base polymers depending on the nature of targeted chemical hazards to be handled [4]. These polymers include poly(vinyl alcohol), cellulose acetate, cellulosic cotton and poly(allylamine), and many of them are already being widely used throughout the chemical industry in gas separation, water purification, and in medical/metabolic waste filtration [49–51].
Air permeable materials
Permeable chemical barrier clothing is typically made of a woven shell fabric, which is permeable to gases, liquid, aerosol as well as vapour from water and chemical agents [4]. This permeability allows liquids to penetrate easily through the fabric at low hydrostatic pressures; therefore, various functional finishes are usually applied to the outer-shell fabric to provide liquid repellency as well as waterproofness (Figure 5(a)) [16]. To overcome problems associated with heat stress, a layer of sorptive material (Figure 5(b)) such as porous activated carbon, which is permeable owing to its open porous structure, is included to assist removal of perspiration from the protective suit, along with creating a barrier against CWAs by adsorbing liquid chemicals.
Permeable protective clothing with (a) different layers of fabrics and (b) schematic representation of water vapour transfer mechanism through porous fabrics [16].
An activated carbon sorptive material layer generally consists of activated carbon impregnated foam or an activated carbon-loaded nonwoven felt, and a liner fabric [15]. Many consumers like activated carbon embedded permeable clothing due to convective airflow through the clothing, which cools the body by evaporative action. However, several shortcomings are associated with protective clothing made from breathing materials including contamination by fuels, oils, solvents, poisons, penetration of aerosols and chemical vapour, as well as decrease in protection ability of the clothing within a few days of use without any exterior contamination [52]. Nevertheless, charcoal containing permeable chemical protective suit was developed rapidly as chemical protection equipment since 1960s and was largely provided to the army personnel in many countries including the USA, UK and Canada [16].
Activated carbon-based permeable CPC
Activated carbon, also known as activated charcoal, is a non-hazardous, processed carbonaceous product with an intricate micro-porous structure and it possesses an internal surface area available for adsorption or chemical reactions [53]. Activated carbon materials are unique and versatile adsorbents ranging from granular to powdered form, and now they are being prepared in spherical, fibrous and cloth forms (Figure 6) [54]. They are extensively used as adsorbents among other materials having adsorption properties including silica gel and activated alumina, zeolites (molecular sieves) and some synthetic polymers [55]. Many desirable characteristics such as large surface area, rigid porous structure, adsorptivity, high thermal conductivity as well as high stability towards chemicals give them undeniable advantages, which lead to a wide range of applications including permeable chemical barrier clothing. Protective textile materials combined with activated carbon are considered to provide two main functions; the wearer protection against the chemical hazards, and allowing transmission of water vapour produced from the human body through the clothing materials to maintain the thermophysiological comfort [4].
Activated carbon in different forms employed in permeable CPC (a) granular activated carbon (b) activated carbon fibre and (c) carbon cloth.
Activated carbon materials both in granular and fibre form are generally used in permeable chemical barrier clothing employed to resist the penetration of liquid chemicals and vapours through adsorption. The adsorption of toxic chemical gases is accompanied by accumulation of vapour molecules at the interface of the adsorbent due to the operation of chemical interactions, including van der Waals interactions, and consequently the penetration or diffusion onto the surface of the solid when it comes into contact with carbon [56]. Activated charcoal contains different sizes of pores including micro-pores (<2 nm), meso-pores (2–50 nm) and macro-pores (>50 nm) (Figure 7(a)) and their adsorption process is largely controlled and affected by the size of pores and their distribution [57]. Moreover, each type of pore has a specific function to adsorb small to large organic molecules in their adsorption processes (Figure 7(b)).
Structural schemes for (a) typical activated carbon with different types of pores and (b) adsorption of small and large organic molecules in different pore sizes.
The manufacturing technique of charcoal fibre intended for chemical protection includes development of amorphous carbon through pyrolysis of carbonaceous raw materials at about 1000℃, followed by carbonization below 800℃ in an inert atmosphere, along with chemical activation of the carbonized product between 950 and 1000℃ [54]. The production of activated carbon fibre embraces various organic precursors such as polyacrylonitrile (PAN), poly (vinylidene chloride) (PVDC), phenolic resin textiles as well as cellulose acetate and rayon fibres [58,59]. However, rayon-based charcoal fibre is still employed in current chemical barrier clothing materials though fibres from other precursors are easier to prepare as well as economically viable.
Granular activated carbon is generally coated in nonwoven pads, cotton flannel and polyurethane foam to apply in protective clothing as an inter-liner material. However, concerning the thermo-physiological comfort, needle-punched fabrics coated with activated carbon have better thermal comfort properties [60] compared with polyurethane foam that typically has low air and water vapour permeability [61]. In addition, granular activated carbon exhibits a lower level of adsorption capability than activated carbon in fibre form in protective clothing materials. As mentioned earlier, the adsorption characteristics of activated carbon are mostly influenced by surface area and pore volume. The specific surface area of the available active carbon should be in between 500 m2/g and 1500 m2/g as well as pore volume in the order of 0.20 to 0.60 cm3/g to provide good protection against chemical agents [52]. Activated carbon fibres have a large surface area and contain comparatively higher large pore fraction that provides direct access of the absorbable molecules to the micro-pores (less than 2 nm) openly exposed on their surface. Conversely, the granular form consists of particles from 0.5 to 10 mm. It has a large internal surface area and small pores. Therefore, the absorption kinetics of activated carbon fibre is 2 to 20 times faster than those of granular activated carbon, resulting in high absorption capacities of liquid chemicals and vapours in CPC [4]. Nevertheless, the development of a charcoal-containing permeable chemical protective suit is rapid due to its substantial resistance to chemical and gas permeation, high liquid holding capacity besides good breathability.
Activated carbon clothing materials have some intrinsic limitations, such as high density and poor mechanical properties, especially brittleness, low abrasion resistance and low tensile strength compared with traditional textile materials [62]. In addition, poor moisture absorption ability coupled with inherent bulkiness and thermal insulative properties makes it unable to evaporate a large volume of perspiration from the human body, resulting in poor thermophysiological comfort to the wearer [37]. Moreover, the use of activated carbon in the protective mask might affect user inhalation due to absorption of oxygen from the environment by moist-activated carbon [4]. The adsorption ability of the activated carbon is selective, only nerve and mustard agents are adsorbed; therefore, various additive coatings are usually required for the adsorption of toxic chemicals as well as dynamic detoxification of the chemicals adsorbed on the fibres [59,63].
Current CPC system for military and civilian
The key purpose of CPC is to protect the wearer against the effects of toxic or corrosive products including vapours, liquids, or solids that could enter the body or cause tissue damage upon contact with the skin. However, a single protective material is not compatible with every chemical. Therefore, the design of chemical barrier clothing to provide desirable chemical resistance is critical. It requires contributions from multi-skills including a suitable design of clothing, development of efficient barrier materials for effective protection along with adequate knowledge on toxic chemicals and their capabilities for mass destruction. Hence, constant efforts have been enduring to provide military as well as civilians the best suit(s) with various desirable properties such as lightweight, comfortable, durable, low cost, and easy care. Also, they should require little maintenance and be compatible with current individual equipment. Some existing protective clothing systems for civilian and military purposes are mentioned below.
Military application
Chemical protective combat clothing known as Joint Service Lightweight Integrated Suit Technology (JSLIST) over-garment is used by the USA military for protection against CB chemical and biological agents, radioactive fallout particles and battlefield contaminants [40]. The JSLIST over-garment is a lightweight, front-opening outfit that can be worn as a primary uniform over underwear. The system comprises an integral hood, high-waist trousers along with adjustable waistband, adjustable suspenders and a waist-length jacket to impart comfort as well as maximizing compatibility with individual user equipment [64]. Laminated activated carbon spheres are incorporated as a liner in the JSLIST system to absorb chemical agents. The garment features an advanced chemical protection as well as allowing more flexibility for the wearer, and can be laundered three times and is disposable after 45 days of wear [16]. It is fully compatible with extreme cold weather clothing, including improved chemical and biological protective gloves and multipurpose over-boots. Similar activated charcoal-based breathable chemical protective over-garments include the UK Mark IV NBC as well as the French Paul Boyé’s NRBC Protective Suit.
Active-carbon spheres encapsulated Saratoga protective textiles have gained much attention among the forces of USA and NATO. The garment consists of outer shell polybenzimidazole (PBI) and Nomex (20/80) blended fabric weighing approximately 150 g/m2 with spherical adsorbers having 25 mesh and 120 g/m2 density to offer additional protection and comfort [52]. The key features of Saratoga system include a low heat stress and high air permeability, ability to laundering repeatedly, resistance to perspiration and petroleum products, good strength and lightweight [65].
Chemical protective undergarment (CPU) is another clothing system originally designed for the United States Special Forces. The production of CPU involved polymerically encapsulated carbon, integrated with Nylon/Lycra tricot fabric as a composite [66]. The fabric also provides exceptional mobility and comfort due to its unique stretch characteristics along with excellent air permeability promoting evaporative cooling that leads to reduced heat stress. In addition to the above, another charcoal-containing cotton flannel, the inner layer of M-82 permeable chemical protective suit was developed for the Chinese army [67]. One side of the cotton flannel was finished with fluorochemicals and another side was finished with active charcoal-polyacrylate emulsion to play the role of adsorbent and oil repellent, respectively [52].
Civilian application
Protective clothing system includes the STEPO and the ITAP ensemble that are used for soldiers as well as civilians for special purposes, such as domestic emergency operations for chemical spills along with the maintenance of toxic chemical and clean-up in a challenging environments [16]. The STEPO protective ensemble consists of a fully encapsulating impermeable, butyl rubber-coated Nylon suit to provide protection against chemical/biological agents, missile/rocket fuels, petroleum, oils and lubricants (POL) and industrial chemicals. Moreover, the ensemble incorporates 4 h of self-contained breathing and cooling apparatus as well as being capable of decontamination for reuse [68]. Although the STEPO system is more costly, it provides greater protection and has better cooling capabilities than toxicological agent protective (TAP) suit that has been employed for level-A protection to protect individuals from toxic chemical agents [69].
ITAP ensemble is designed to provide protection against hazardous chemicals during peacetime and wartime for short-term operations. ITAP can be engaged in immediately dangerous to life and health (IDLH) toxic chemical environments for up to 1 h as well as in emergency and incident response and initial entry monitoring [16]. The ITAP system includes an impermeable suit totally encapsulating the body except for the facial area and the outer shell of the ensembles consists of light grey PTFE and Nomex material [70]. The suit is self-extinguishing and capable of being decontaminated for a minimum of five-reuses after vapour and particulate contamination. It has a minimum of five-year shelf life.
Suit contamination avoidance liquid protection (SCALP) and TAP suits are designed for military and civilians in low threat environments [14]. The TAP system is impermeable to water and has a high level of insulation, causing marked heat stress for the users [70]. It has an adjustable collar, double sleeves, trouser cuffs along with adjustable belt and offers liquid splash protection. On the other hand, SCALP is conceived to protect its user from gross liquid contamination for a short period of time. The SCALP consists of a jacket with hood and trouser is made of PE-coated impermeable materials and worn over the Battledress overgarment. The SCALP is not intended to be decontaminated, reimpregnated, or laundered, and should be properly discarded after exposure to chemical agents. Besides, there are various commercially available suits that are actively being marketed for use in events or incidents involving the use of chemical and biological agents.
Testing and evaluation of CPC
Clothing comfort and protective function are key parameters usually required for evaluation of chemical barrier materials, and the performance of a protective suit is absolutely vital. Therefore, standard test methods for evaluation of chemical resistance of a hazmat suit are indispensable for practical applications. However, assessment of chemical resistance is complex and difficult to correlate with practical conditions of chemical protection using laboratory tests [52]. In the practical field, toxic chemicals may be a mixture of various chemical compounds or mixed with solvents and the effects of these mixtures on the penetration characteristics of protective materials are unknown. Mixed chemical species may enhance the degradability of solvents making CPC less resistant to penetration of toxic compounds. Therefore, various methods have been developed by several international, national and industrial organizations including International Organization for Standardization (ISO), European Committee for Standardization (CEN), British Standard (BS), National Fire Protection Association (NFPA), the American Association of Textile Chemists and Colourists (AATCC) and American Society for Testing and Materials (ASTM) for testing and standardization of chemical protective materials [71].
Standard test methods associated with the assessment of chemical resistance property of protective clothing.

ASTM: American Society for Testing and Material; ISO: International Organization for Standardization.
Standard test methods associated with the comfort, fit and permeability of chemical protective clothing.
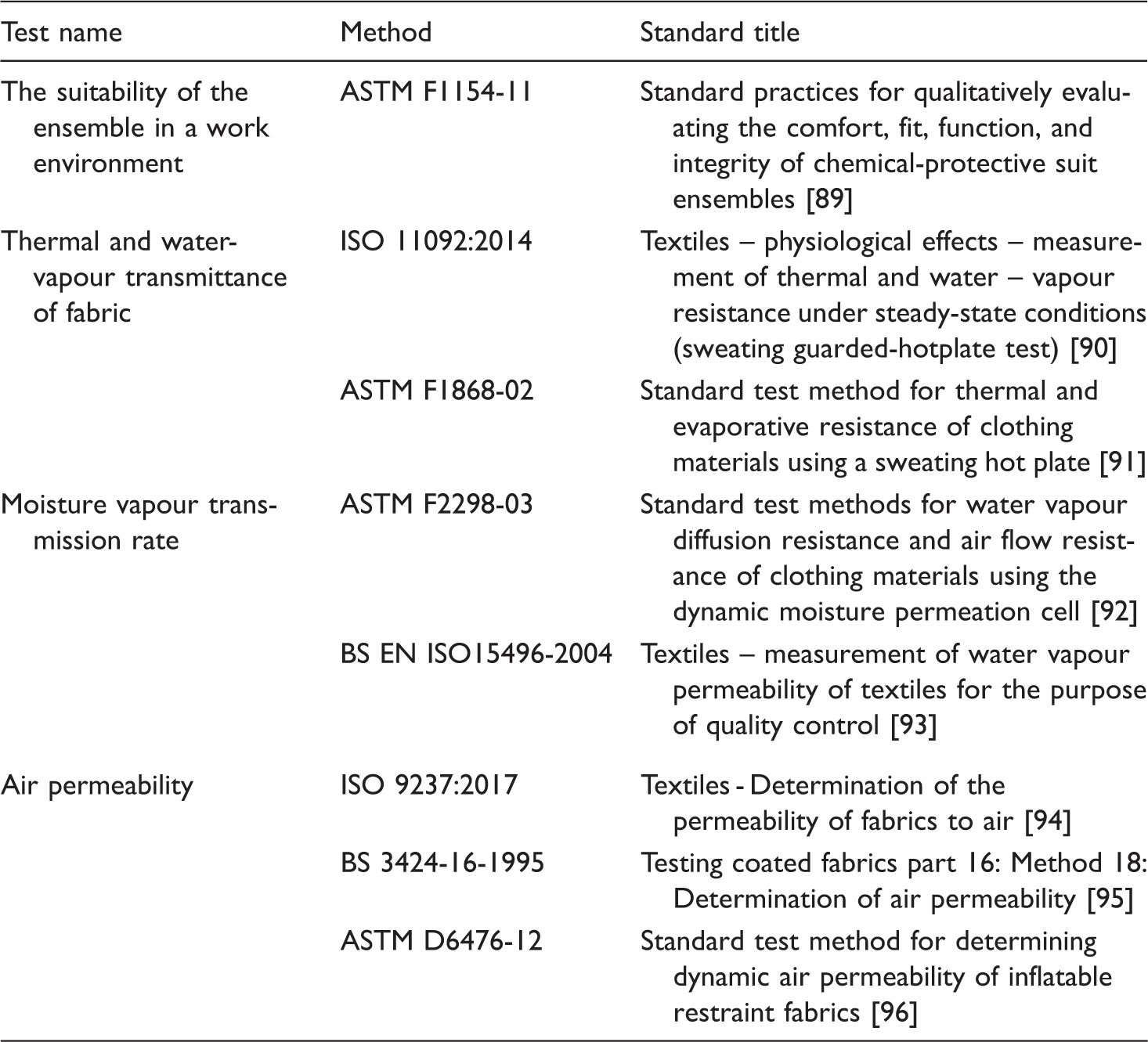
ASTM: American Society for Testing and Material; ISO: International Organization for Standardization.
Standard test methods for evaluation of physical properties of chemical protective clothing materials.

FTMS: Federal Test of Material Standard; ASTM: American Society for Testing and Material; ISO: International Organization for Standardization.
Physiological assessment of chemical barrier materials is usually performed using live subjects (volunteer soldiers) to determine the effects of each CPC on the user after wearing. At the outset, insulative value of each suit is measured on a thermal manikin to get a baseline which gives an idea of the degree of heat stress that the live participants will encounter when they don the suits. Consequently, heat stress, core temperature, and other physiological signs are measured on each participant wearing the various protective suits. Besides the evaluation of clothing system designs, durability and user comfort while wearing are conducted in limited field experiments that typically range from one week to as long as four weeks of test time in selected locations based on the intended environments and climates [14]. Control clothing systems are included in the test procedure for comparative purposes. The assessment program also comprises the structured questionnaires that are used to interview soldier volunteers at the end of the testing.
Novel technologies for advanced CPC
Increased concern over exposure to hazards of venomous CWAs due to an intentional or accidental release spurs further improvement in countermeasures to provide a reliable protection for military personnel and emergency responders. An unremitting research effort is continuing globally to introduce advanced fabrics for consistent chemical protection [109–112]. The development of protective clothing systems embedded with innovative technologies is envisioned with novel features including the detection of harmful chemical agents on the fabric surface, chemical demolition of toxic materials that come into contact with the fabric, and the ability to selectively block hazardous chemicals. Novel materials being integrated into CPC for an enhanced chemical protection comprise reactive nano-fibres and nano-particles, conductive-polymer indicators on optical fibres as well as enzyme-based biocatalysts [113–117]. New-generation protective clothing being developed by incorporating these novel materials and methods will implicate barrier protection and detection, trapping decontaminating toxic particles, liquids and vapours, when contacting these advanced fabrics.
Nanomaterial-based protective textiles
The term nanotechnology is defined as the understanding, manipulation, and control of matters on a length scale of the nanometre, i.e. nanotechnology deals with structures that are sized between 1 and 100 nm and involves the development of materials or devices having dimension within that size range [118]. The application of nanotechnology in the field of textiles is booming in an unprecedented way, due to its unique and versatile aspects in addition to its tremendous impact on other fields. With the latest buzz of nanotechnology, researchers across the globe are involved in finding solutions intended for advanced materials concerning chemical protection.
Recent research on nanomaterials includes nano-particles, nano-rods, nano-tubes, nano-wires, nano-structures, and nano-composites [119–121]. The interest in nanoparticles mainly stems from their unique properties, which differ considerably from those of bulk materials [122]. Various metallic nano-particles such as Ag, Au, Pd, Cu, Ni, and metal oxide nano-particles including MgO, CaO, ZnO, TiO2, Al2O3, MnO2 and Fe2O3 are reported in the scientific literature for their various catalytic applications [123–125] and may serve as an effective catalyst against CWAs leading to reliable protection in current CPC via concurrent adsorption and decomposition of toxic materials [126–128].
Metal oxide nano-particles are usually synthesized from a number of precursors using different methods, including sono-chemical preparation, sol-gel transformation, gas-phase condensation, microwave-assisted synthesis and aerogel technique [129–132]. Nano-particles prepared by the aerogel technique are widely considered for use in protective clothing due to their high surface area (>400 m2/g) along with a large number of pores (>85%) [129,133]. Aerogel materials possess a low conductivity and low density and have many defect sites, together with the ability to exhibit high surface chemical reactivity compared with most conventionally prepared materials [134]. Ultrahigh surface area and high surface concentrations of reactive defect sites make these nanoparticles unique “destructive adsorbents,” being capable of converting hazardous chemical agents into harmless species through irreversible decomposition [37].
CWAs that can be decomposed by metal oxides include blister agents, nerve agents, insecticide model compounds [135–138] as well as perilous biological agents [139,140]. Sadeghi et al Proposed mechanism for the decontamination of CEPS on the ZnO nano-particles [141]. CEPS: chloroethyl-phenyl sulfide.
Another recent study demonstrated an almost complete decomposition of dimethyl methyl phosphonate (DMMP), a nerve-agent simulant by using graphene oxide (GO)-MnO2 nanocomposites in an aqueous media [142]. Adsorption of DMMP is initiated by hydrogen bonding leading to complex formation on metal oxide surfaces via O–P–P bridging species along with gas-phase methanol (Scheme 2). Several other researchers established MgO, TiO2, CaO, Al2O3 nano-particles and their composites as destructive adsorbents being capable of detoxifying toxic chemicals and CWAs through simultaneous adsorption and irreversible decomposition [143–146].
The proposed mechanism of destructive adsorption of DMMP nerve-agent simulant onto the GO-MnO2 surface with a possible formation of surface complexes [142]. DMMP: dimethyl methyl phosphonate.
The detection of CWAs and their simulants via sensor nanomaterials has been demonstrated in some contemporary research studies. Sayago et al
Metal oxide nano-particles can impart functional properties to textile fibres through surface modification. Protective textile materials together with metal oxide nanoparticles can decompose organic matters such as odorous molecules and harmful bacteria, and destroy poisonous chemicals through photocatalytic action [151,152]. Modification of textile fibre surfaces with metal oxides has some innate limitations. The agglomeration of particles during application in a surface coating makes the process difficult, and the particles are not permanently on the surface of the fabric; therefore, various methods are needed to stabilise the dispersed particles [37]. Treatment of textiles with nano-particles may reduce flexibility and abrasion resistance, besides deterioration in appearance of the fabric.
Nano-fibre based CPC for chemical protection
Nano-fibres are extremely fine materials having diameters with nanometre-scale, and generally produced by an electrospinning process [153]. Electrospun nano-fibres are considered as a promising tool for versatile applications due to some extraordinary features, such as high specific surface area, high porosity, and low fabric weight [112]. Recent research has been focused on developing nano-fibres from nano-particles, by mixing them with polymer solutions [154,155]. These nano-fibre materials have attracted much attention in various fields including tissue engineering, wound dressing, blood vessel regeneration, composite materials, filtration and chemical sensors [156–159]. A potential application of nano-fibre materials in protective clothing system has been demonstrated in several contemporary research studies [140,152,160,161]. These experimental works revealed that lightweight electrospun nano-fibre clothing materials exhibited greater breathability along with an improved liquid chemical and aerosol particle retention capability compared with current commercially available membranes.
The prospective use of electrospun nano-fibre mats in CPC including protection against CWAs, liquid penetration barriers and clothing materials for agricultural workers has been extensively investigated [39,162]. Electrospun nano-fibrous webs as a barrier to liquid penetration in CPC revealed that a very thin layer of polyurethane nano-fibre web can significantly improve barrier performance for challenge liquids with a range of physicochemical properties [153]. Schreuder-Gibson et al
Relative rates of organophosphorus (OP) hydrolysis by different functionalized nano-fibre membranes [165].

PVC: poly(vinyl chloride).
Chen et al. [112] studied chemical methods to counteract nerve agents and remediate organophosphate (OP) contamination by means of nano-particles, polymers, and nano-fibres functionalized by Reaction of IBA-β-CD with Paraoxon [165].
Recently, Choi et al Decontamination mechanism of Demeton-S-methyl with NCH-PU [166].
Novel ceramic nano-fibre by using zinc titanate (ZnO-TiO2) was developed through the sol–gel method and proposed to use in protective clothing and face masks for shielding against harmful chemicals and other agents [167]. The synthesized nano-fibres had a high porosity with a large surface area and exhibited the decomposition of nerve and mustard agent simulants into non-toxic products via a simultaneous adsorption and detoxification process.
Conducting polymers for chemical detection
Conducting polymers are a relatively new class of materials that possess electronic conductivity. The high conductivity of such polymers leads to a novel and exciting area of material science due to their useful electrical properties and redox potential, along with environmental stability and processability, as well as rapid and reversible absorption kinetics [168,169]. The extraordinary electrical conductivity and low ionization potential of conductive polymers are provided by charge mobility due to delocalization of π electrons along the polymer backbone [170].
Conducting polymers are sensitive towards chemical agents due to electronic changes arising in the polymeric films by a gas-phase adsorption of electronically active vapours [171]. The changes in conductivity in these polymers are generally attributed to the interaction of electronically active analytes with either the polymer backbone itself or dopant molecules being incorporated within the polymer, thereby modulating the mobility and/or the number of available free charge carriers [172]. This exceptional property makes them excellent candidates for sensor applications; therefore, various approaches including conductive nanofibres and development of fibre optic chemical sensors using conductive polymers are presently under consideration regarding their use in advanced CPC [113,173].
Conducting nanofibre materials are generally prepared by an electrospinning process and the development of electrospun nanofibres includes various polymers such as polyaniline (PANI), polypyrrole (PPY) and poly(3,4-ethylenedioxythiophene) (PEDOT) [174,175]. The electronic characterization of these polymers exhibits their responses to a variety of gases including many organic vapours as well as liquid acids and bases [40]. Among conducting polymers, PANI is more apposite due to its suitable conductivity, low cost, good transparency and minimal mechanical change in a matrix and it can readily be tuned by dedoping [176]. The oxidation state of PANI can be reversibly changed from the insulating emeraldine base form (σ < 10−10 S/cm) to the conducting emeraldine salt form (σ > 1 S/cm) by acid or base doping and dedoping (Scheme 5). This large change of conductivity is utilized to develop PANI sensors for detection of toxic industrial chemicals.
Switching between emeraldine base (insulating form) and emeraldine salt (conducting form) by HCl and ammonia, respectively [176].
The interaction between PANI with electrophilic gases attracts electrons from the polymer phase, causing an upsurge in conductivity, whereas the interaction with nucleophilic gases increases the resistance of the polymer [177]. Virji et al Chemical interaction of phosgene with amines [178].
Moreover, the improvement of chemical sensing ability of PANI polymer towards noxious chemical agents was demonstrated in several research studies by incorporating various metal salts (CuCl2, CuBr2, CuF2, NiCl2, FeCl3, and CoCl2) through the formation of PANI-metal salt composite nano-fibres [179–181]. The magnitude of the increase in conductivity suggests the generation of strong acid, including hydrofluoric acid (HF), nitric acid (HNO3), HCl and hydrobromic acid (HBr) due to chemical interaction between toxic gases and metal salt additives (Scheme 7). The acid produced can then dope the PANI resulting in large changes in resistance by reducing the composite metal ion as well as oxidizing the detecting chemicals [181]. Besides, the change electronic charge-transfer process due to adsorption of organic molecules has a solvent-type interaction with the polymer, causing physical swelling of the polymer structure.
Proposed mechanism of interaction of metal salt-polyaniline nanofiber composite material with arsine gas (AsH3) [181].
The change in oxidation state of conductive polymer in the presence of chemical contaminants may alter the optical properties of fibres within a fabric. Conducting polymers, such as PANI and PPY, are accompanied by optical property changes in a fabric when they undergo a reversible change in conductivity upon exposure to chemical vapours [169]. Alteration of optical properties, due to chemical interaction, includes a change in the refractive index and variation in the optical-absorption spectrum of polymer films [40]. Both of these variations modify the intensity of the light transmitted through the optical fibre as a result of changes in the refractive index and interactions with the evanescent field, respectively. This reversible change in conductivity upon exposure to chemical vapours is considered as an effective method for detection of toxic chemical vapours.
Organic conducting polymers (OCPs) are easy to prepare. They show excellent ambient stability, rapid and reversible absorption kinetics, as well as having the ability to be embedded into various textile structures [182]. Thus, fibre optic materials were developed by using chemically responsive conducting polymers such as PANI or PPY through formation of submicron-thick films on fibres in a polymerization process [175]. The newly developed class of fibre optic sensors combined the advantages of optical fibres and conducting polymers with high sensitivity and easy processability for sensing hydrogen chloride (HCl), ammonia (NH3), hydrazine (H4N2), and dimethyl methylphosphonate (DMMP) (C3H9O3P) through chemical interaction [169].
There are many gas sensors using conducting polymers as their sensing elements, with extensive design flexibility and efficient detection capability of lethal chemical vapours through variation in light absorbance. A change of UV-visible absorbance band (40% reduction) of conducting PPY thin films, exposed to DMMP has been revealed in a research study [183]. It can be assumed that DMMP interacts electronically with PPY to increase the amount of free mobile hole charge carriers attributed to changes in bulk conductivity and absorbance properties. Moreover, a change of refractive index from 1.82 (before exposure to hydrazine) to 1.71 (after exposure to hydrazine) as a result of interaction between hydrazine (N2H4) and PPY film has been mentioned [169].
Enzyme-based chemical protective textiles
Enzymes are biological substances that are produced by living organisms. They act as a catalyst to bring about a specific biochemical reaction. Enzymes are considered as efficient biocatalyst and the use of enzymes in industrial catalysis continues to grow because of the considerable advantages of natural catalytic systems [184]. A number of enzymes are capable of degrading a wide range of organophosphorus (OP) compounds [185]. Currently, there has been rising interest in the application of these enzymes for the protection of agricultural workers and military personnel against pesticides and CWAs through decontamination or detoxification [186–188]. A wide variety of self-decontaminating surfaces can be developed by incorporating enzyme-containing polymeric materials and be employed for the fabrication of CPC [71].
Among various enzymes, organophosphohydrolase (OPH) is well known for its wide variety of hydrolytic activity against OP pesticides including paraoxon, parathion, coumaphos, and diazinon [186], as well as nerve agents such as sarin (GB), soman (GD), and VX [189]. However, the hydrolytic activity of OPH against the warfare agent VX paraoxon is less than 0.1% relative to its activity against parathion and paraoxon [190]. The detoxification capability of OPH against organophosphorous neurotoxins includes hydrolysing via various phosphorous-ester bonds (P-O, P-F, P-CN and P-S) between the phosphorous centre and an electrophilic leaving group [191].
Serum paraoxonase 1 (PON1) is another type enzyme which is considered as an efficient catalytic bio-scavenger due to its ability to hydrolyse a broad range of OPs, including pesticides and nerve agents [192]. This enzyme is found in all mammalian species and synthesized primarily in the liver and a portion is secreted into the plasma, where it is associated with high-density lipoproteins (HDLs) [193]. PON1 is a calcium-dependent hydrolytic enzyme, the most-studied member of a family of calcium-dependent hydrolases and received its name from its promiscuous activity in hydrolysing the phosphotriester pesticide paraoxon as well as sarin or soman [194,195] (Scheme 8). Structurally, PON1 is a six-bladed β-propeller with a flexible loop fold with two calcium ions in its central tunnel where the catalytic Ca+ lies at the bottom of the active-site cavity [196]. This active site of PON1 shows a remarkable level of networking and versatility as well as allows to bind different substrates promiscuously to catalyse the chemical reactions [197]. Such enzymes and their mutants could be potentially employed as coatings in the development of chemical protective finishes.
Hydrolysis of nerve gases by paraoxonase 1 (PON1) [198].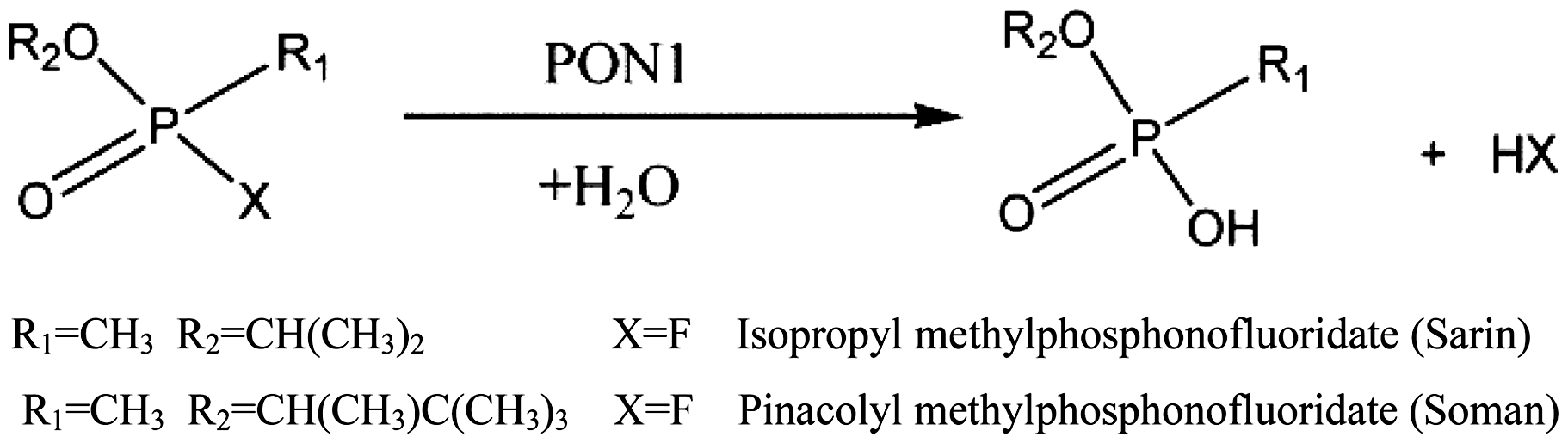
One limitation to enzymatic hydrolysis of the G-agents as well as VX and HD is the creation of hydrolysis products that are acidic, is shown in Scheme 9. The hydrolytic activity of enzyme usually drops significantly at pH values below about 6 [199]. Therefore, a buffer is required to maintain the pH of the hydrolysis catalysts reaction in the neutral to a slightly alkaline range to decontaminate or detoxify relatively large quantities of chemical agent. This buffering capacity is commonly provided by stoichiometric acid-base pairs through the formation of ammonia from urea by urease to neutralize the acidic hydrolysis products [200].
The acidic products of G-agent (soman) after hydrolysis reaction [199].
Conclusions
CPC is generally considered as a last-ditch defence in any chemical-handling operation. It is employed to protect military personnel, law enforcement officials and civilians from contamination by various chemical hazards. Selection of CPCs considers many factors, such as weight, comfort, level of protection and duration of required protection. Permeation, penetration and degradation are three important factors concerning the level of protection against diffusion of chemicals at the molecular level. Any appropriate barrier clothing needs to demonstrate no penetration, higher breakthrough time than the duration of the task and no significant degradation upon contact with harmful chemicals. Impermeable, semi-permeable and air permeable protective clothing systems are developed by integrating various types of materials. Impermeable clothing creates a resistant barrier against penetration of the chemicals in liquid, vapour and aerosol forms, but hampers transmission of perspiration, which creates an obstacle for the normal cooling processes of the human body. To overcome this obstacle, permeable protective clothing embedded with activated carbon allows transmission of water vapour to maintain thermo-physiological comfort, while providing protection against chemical hazards. Some intrinsic limitations include poor mechanical properties, particularly high brittleness, and low moisture absorption ability coupled with inherent bulkiness that hinders the application range of activated carbon. Protective clothing combined with selectively permeable materials may deliver effective protection against specific chemical agents along with maintaining effective moisture-transport properties at a lower weight and bulk than earlier designs. Advanced fabrics integrated with some novel technologies are envisioned to introduce protective, reactive and responsive clothing systems, including some unique features such as the capabilities of detecting and deactivating toxic chemical agents and selectively blocking hazardous chemicals. Self-detoxifying, innovative fabrics incorporating chemically deactivating nano-particles and nano-fibres will provide consistent chemical protection through detection, trapping and decontamination of toxic particles, liquids and vapours. Besides, enzyme-containing polymeric materials will be employed as efficient biocatalyst for the development of self-decontaminating advanced CPC for reliable chemical shielding. Detection by means of conductive polymer materials will provide the wearer with an early warning of environmental hazards. Finally, it can be predicted that with access to aforesaid emerging technologies, new classes of CPC will outperform their conventional counterparts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: M A Rahman Bhuiyan is thankful to RMIT University, Australia for scholarship support towards his PhD study.
