Abstract
This study focuses on the development of functional nanocapsules via the coaxial electrohydrodynamic atomization (electrospraying) method. These nanocapsules can manipulate nonwoven surface functionality in terms of antibacterial characteristics for medical textile purposes. Electrosprayed nanocapsules were produced from Poly(lactic acid) (PLA) polymer and
Introduction
Antibacterial activity is a necessity for various textiles, especially those used in the medical area. Hence, the increasing interest in personal health and hygiene has created the need to improve textile surfaces' antibacterial characteristics. Antibacterial textile applications cover many different materials and methods. These materials include metal oxide nanoparticles [1], silver modified fibers [2], inorganic nanotubes [3], chitosan [4], silver phosphate [5], natural dyes [6–8], and methods include sol-gel [1], electrospinning [9–11], nanosol coating [12]. Herbal plant extracts have been studied many times as antibacterial textile finishing thanks to biodegradable, nontoxic, and fewer irritant characteristics [13]. Some of these are lotus [14],
The nano/microencapsulation technique can be the next generation of textile finishing. The encapsulation method has been initially practiced in the biotechnology [20] and has many applications in this area, for instance, enzyme immobilization for biosensors, biocatalysts [21], microorganism encapsulation [22,23], bioactive peptides [24], food formulations, virus capsulation for vaccination, and therapy purposes [25]. Nano/microencapsulation technologies have a wide range of applications in other areas too. These are food industry [26–28], drug delivery and therapeutics applications [29–32], heat storage applications [33,34]. Textile encapsulation implementations cover antifungal, antibacterial, UV absorbent, and anti-insecticide applications [35,36]. But microencapsulation has several disadvantages contrary to the nanoencapsulation method, such as severe conditions, complexity, reproducibility, polydispersity, agglomeration, and irremovable toxic chemicals [37]. Although the coaxial (dual capillary) method is a relatively new encapsulation technology, it has gained much attention for significant advantages than conventional physical/chemical processes for nanoparticle preparation. It can help beat up mentioned disadvantages and contribute to achieving controlled particle size production and release mechanism, decelerate initial burst release, mild environmental conditions, and practices without emulsifiers [38].
Various studies have been performed on the coaxial electrospray method. Ghaffarzadegan et al. [39] developed PLA/Berberine herbal alkaloid drug-loaded cancer drugs. Sanders et al. [25] have studied virus encapsulation to prevent inactivation by the immune system for vaccination and therapy. Smeets et al. have researched cardiovascular disease treatment [40] and gastro-resistant polymer encapsulation [41]. Tang et al. [42] have investigated drug delivery for osteosarcoma treatment. Yu et al. [43] have conducted studies for rapid therapeutic effect. Zhang et al. [34] used phase change microcapsules for thermal regulation and energy storage systems. Gómez-Mascaraque et al. [44] developed capsulated bioactive ingredients in zein and gelatin biopolymers to enhance food materials protection. Niu et al. [45] have practiced masking undesired flavors and odors, reducing volatility and evaporation of bioactive compounds.
Poly(lactic acid) or polylactide (PLA) represents the superior choice for encapsulation applications and controlled delivery systems as a member of the aliphatic polyester family [46]. It is not only biocompatible and biodegradable but also cost-efficient [47,48]. It has shown broad applicability to many areas such as oral delivery, sustained delivery of drugs, etc. [49]. El-Naggar et al. [50] encapsulated the curcumin in PLA-PEG copolymers to improve physicochemical, biological features, and efficacy using an emulsion-diffusion evaporation technique. Sun et al. [51] coaxial electrospun PLA and PVP polymers with bioactive molecules for tissue engineering to investigate process parameters and process stability. They enhance water uptake of produced materials, and the PLA shell prevents the core material from dissolving. Liakos et al. [52] produced lemongrass oil embedded PLA nanocapsules via nanoprecipitation method to achieve antimicrobial therapy and drug delivery purposes.
The present study is focused on the formation of biodegradable PLA/
We use the Scanning electron microscope (SEM) and Field emission scanning electron microscopy (FESEM) to analyze nanocapsules' morphology. We also use Dynamic Light Scattering (DLS) technique for size measurements and dimensional characterization of PLA nanocapsules. Additionally, we take the Transmission electron microscope (TEM) images to demonstrate capsule formation and core-shell structure. Fourier transform infrared (FTIR) spectroscopy is used for spectrophotometric examination of nanocapsules. Pick-up ratios and capsule concentration on coated nonwoven fabrics have been determined. And we test antibacterial activity on the nonwoven surface covered with PLA nanocapsules containing the
Materials and methods
Polypropylene nonwoven fabric is formed from 3 layers as spunbond meltblown spunbond (SMS) via calendaring. Spunbond polypropylene layers are located in the top, and bottom layers, and the meltblown polypropylene layer is located in the middle. It was kindly provided by Ritaş Holding Company (Gaziantep/Turkey). This SMS nonwoven fabric has 45 g m−2 basis weight in which each spunbond layer has 18 g m−2, and the meltblown layer has 9 g m−2. Poly(lactic acid) or polylactide (PLA) (CAS No: 26100-51-6, MW∼60,000), chloroform (CAS No:67-66-3), ethanol (CAS No:64-17-5) were all purchased from Sigma-Aldrich. PLA polymer solutions are prepared with solvent content, including 95/5 wt% chloroform/ethanol. PLA concentration in the solution is used as 1.5 wt%.

Pictures of
After harvesting, the following procedure was adopted for extraction: Flowering aerial parts and roots of the plants are dried in the laboratory in a warm environment with a stream of air for 7-10 days. Mechanic grinder is used for fine powder preparation. Powdered flowering aerial parts (287.72 g) and roots (161.49 g) of
Amounts and yield calculations of prepared solvents.

We decided that a considerably small amount of extract concentration as adequate for finishing solution preparation. Previous studies on the literature show that even small amounts of 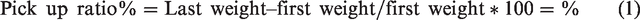
Formation of coaxial electrosprayed PLA nanocapsules
Our coaxial electrospraying set-up is composed of New Era NE-1000X dual pump (comprised from two identical pumps; infusion rate of each pump can change from 0.73 l h−1 to 2100 ml h−1), 0-100 kV-20W ES100P model Gamma high voltage power supply, a grounded collector, and a fabric winder (Figure 2). PLA solution is fed from the outer needle tip while the functional plant extract solution is fed from the inner needle through dual syringe pumps. The coaxial needle tip is connected to the high DC voltage. During the electrospraying, nanocapsules are collected on the surface of aluminum foil and nonwoven fabric which are placed on the grounded collector. SEM and FESEM images are taken from surfaces of these aluminum foils and nonwoven fabrics after electrospraying. Nanocapsules are collected directly on Cu grids for TEM images and collected on an aluminum cup contains water for nanosizer measurements. We make antibacterial assessment on nonwoven fabrics after coaxial electrospraying. The Electrospraying application is carried out for 30 minutes for each run except ones used in nanosizer measurements (10 minutes) and wet-pick up ratio calculations (15, 30 and 60 minutes). Previously optimized processing parameters are used with different flow rates. These parameters are 15 kV applied voltage, 15 cm tip to collector distance, and 22 Gauge needle size (for shell). The applied electric field is 1 kV cm−1 for all flow rates. Different co-fluids flow rates are studied throughout the study. First, total co-fluids flow rates are arranged to 20 µl min−1 (5/15 µl min−1, 9/11 µl min−1, 10/10 µl min−1, and 15/5 µl min−1). After, equal co-fluid flow rates (5/5 µl min−1 and 10/10 µl min−1) are examined for electrospraying. The inner needle number (which core material is passed through) is selected as 26 gauge during all studies. Also, varied core solution concentrations are studied too.

Coaxial electrospraying set-up.
Characterization of coaxial electrosprayed PLA nanocapsules
Electrosprayed nonwoven fabrics and aluminum foils are examined with SEM (JEOL JSM-6390) and FESEM (Zeiss-Supra 55) images determining nanocapsule morphology in their surfaces. Before SEM investigation, samples are coated with Au/Pd in SC 7620 Sputter Coater, and samples are coated with Pt/C for FESEM. SEM pictures are taken at 5-20 kV accelerating voltage, and 12-15 mm working distance with magnifications between 100 and 5000. TEM (FEI Tecnai G2 F30) is used for taking the high-level resolution images of collected nanocapsules on Cu grids. MALVERN Zetasizer Nano ZS using the Dynamic light scattering (DLS) technique is utilized to determine PLA nanocapsules' distribution and dimension. We collect electrosprayed PLA nanocapsules into an aluminum cup containing distilled water for this measurement. The entered refractive index value and absorption rate of PLA polymer are 1.44 [58] and 0.1. Three measurements are taken for each sample at 25 0C. As a result of this test, PLA nanocapsules' number, size, and volume distributions are obtained. In addition to these, nanocapsules' spectrophotometric investigation is conducted on electrosprayed nonwoven fabrics using Shimadzu IRAffinity-1S FTIR spectroscopy.
Antimicrobial activity assessment
The antibacterial activity is determined according to the quantitative test method ASTM E2149-01 “Standard Test Method for Determining the Antimicrobial Activity of Antimicrobial Agents Under Dynamic Contact Conditions” (2015, Turkey). 
“A” means number of bacteria recovered from the inoculated test specimen swatches in the flask incubated 24 hours. “B” means the number of bacteria recovered from the inoculated test specimen swatches at ‘0' contact time.
Results and discussion
Effect of flow rate on nanocapsule formation
The effect of flow rate on the size of electrosprayed capsules was studied for co-fluids. The core/shell flow rates are varied, while other parameters stay the same as mentioned before. Firstly, coaxial electrospraying was performed for the 20 µl min−1 total co-fluid flow rate. The studied flow rates are 5/15 µl min−1, 9/11 µl min−1, 10/10 µl min−1, and 15/5 µl min−1. We observe the feeding rate’s effect on nanocapsule formation with TEM images (Figure 3). Results show that all nanocapsules have a distinctive core/shell structure. But 10/10 µl min−1 flow rate gives the best morphology. Other feeding rates show undesired formations such as separation or aggregation from place to place. Also, molecular interactions between core and shell solutions have critical importance in encapsulation [46,59] and stable Taylor cone [60]. Low interface tension between these solutions positively affects coaxial electrosprayed jet [46,59]. Xu and Hanna [46] coaxial electrosprayed PLA and the bovine serum albumin (BSA) solutions. They observed that concentration increase in the PLA concentration (core solvents) contribute to interfacial tension decrease between PLA and BSA solutions. The high viscosity of PLA and the low interfacial tension of both solutions favor stable cone-jet formation. The achieved results fit these studies. Shell material fails to wrap core material for the lowest shell feeding rate (15/5 µl min−1). In contrast, a high shell flow rates contribute to capsule formation.

TEM images of coaxial electrosprayed nanocapsules at different flow rates.
After that, 5/5 µl min−1 and 10/10 µl min−1 flow rates were studied, and size measurement on electrosprayed nanocapsules was carried out (Figure 4). Our results show that particle size increases with the flow rate increase from 5/5 µl min−1 (132.6 ± 18 nm) to 10/10 µl min−1 (271.2 ± 13 nm). This result is coherent with previous studies on the literature [61]. Flow rate increase leads to a rise in the amount of the delivered solution to the capillary tip, so there isn’t enough drying time and stretching force before reaching the collector. Li and Wang [62] and Xie et al. [63] reported that these phenomena cause to thicker diameter like our study. Another finding is electrosprayed nanocapsules show polydispersity at 5/5 µl min−1 flow rate and narrow size distribution at 10/10 µl min−1 flow rate. This situation is attributed to the stretching force has more effect on the lower amount of solution, which makes instability on the sprayed jet cause to polydispersity.

Size measurements of coaxial electrosprayed nanocapsules at different flow rates (Number distribution).
Figure 5 shows that photo and SEM images of electrosprayed aluminum foils to investigate the spread of jet. The SEM images show a very thin film formation of electrosprayed nanocapsules on the aluminum foil surface coated with a 5/5 µl min−1 flow rate. The reason for thinner coating is considered as electrostatic force has a higher impact on lower flow rates at the same electric field. We also don’t observe any fully spherical capsule morphology at this lower flow rate. The studies show that the flow rate is a critical parameter for coaxial electrospraying [64,65]. Luo and Edirisinghe [66] observed higher charge density for the lower flow rates. They found that fluids with higher charge density can easily overcome the surface tension, and therefore, these co-electrosprayed particles can move faster, which triggers the size reduction.

SEM images and pictures of coaxial electrosprayed aluminum foils at different flow rates.
Figure 6 shows the SEM image of the nonwoven fabrics before and after co-electrospraying. The same thin film formation achieved on aluminum foil at 5/5 µl min−1 flow rate is also observed on nonwoven fabric. At 10/10 µl min−1, nanocapsules are successfully applied and widely spreaded to the fibers on the fabric's surface. Also, before and after photographs of electrosprayed nonwoven fabrics are given in Figure 7. The co-electrospraying application doesn’t cause any apparent change in fabric for both flow rates according to these photographs.

SEM images of coaxial electrosprayed nonwoven fabrics at different flow rates.

Photographs of coaxial electrosprayed nonwoven fabrics at different flow rates.
As a result of the flow rate investigations, nanocapsule formation was achieved with the desired narrow distribution range instead of film formation at 10/10 µl min−1 flow rate. Thus, a 10 µl min−1 flow rate is chosen as an optimum flow rate for the co-electrospraying applications.
After the electrospraying application, the consumption rate of chemicals (wet pick-up ratios) and capsule concentration (gcapsules/kgfabric) on the electrosprayed nonwoven fabric were calculated. The wet pick-up ratios started from a very low value (0.8%) and reached up to 4.18% for 15 minute and 60 minute applications. During the electrospraying process, a great deal of solvent evaporates, and most of the plant extract reaches on to the fabric surface. Therefore, wet pick-up ratios increase with increasing either concentration or application time. The concentration of capsules on the nonwovens were 8.06 g kg−1 for 15 minutes, 22.27 g kg−1 for 30 minutes, and 41.83 g kg−1 for 60 minutes. This increase is expected since more chemicals accumulates on to fibers with the time [61]. FTIR experiments are performed with raw and electrosprayed nonwoven fabric to examine the existence and type of interfacial interaction in the nonwoven fabric surface (Figure 8). The resulted peaks indicate the PLA polymer presence. It was reported that C = O stretching and C–O–C stretching peaks are around 1750 cm and 1180 cm in PLA spectra [67]. Our result is coherent with the literature and showed PLA nanocapsules’ existence on nonwoven fabric.
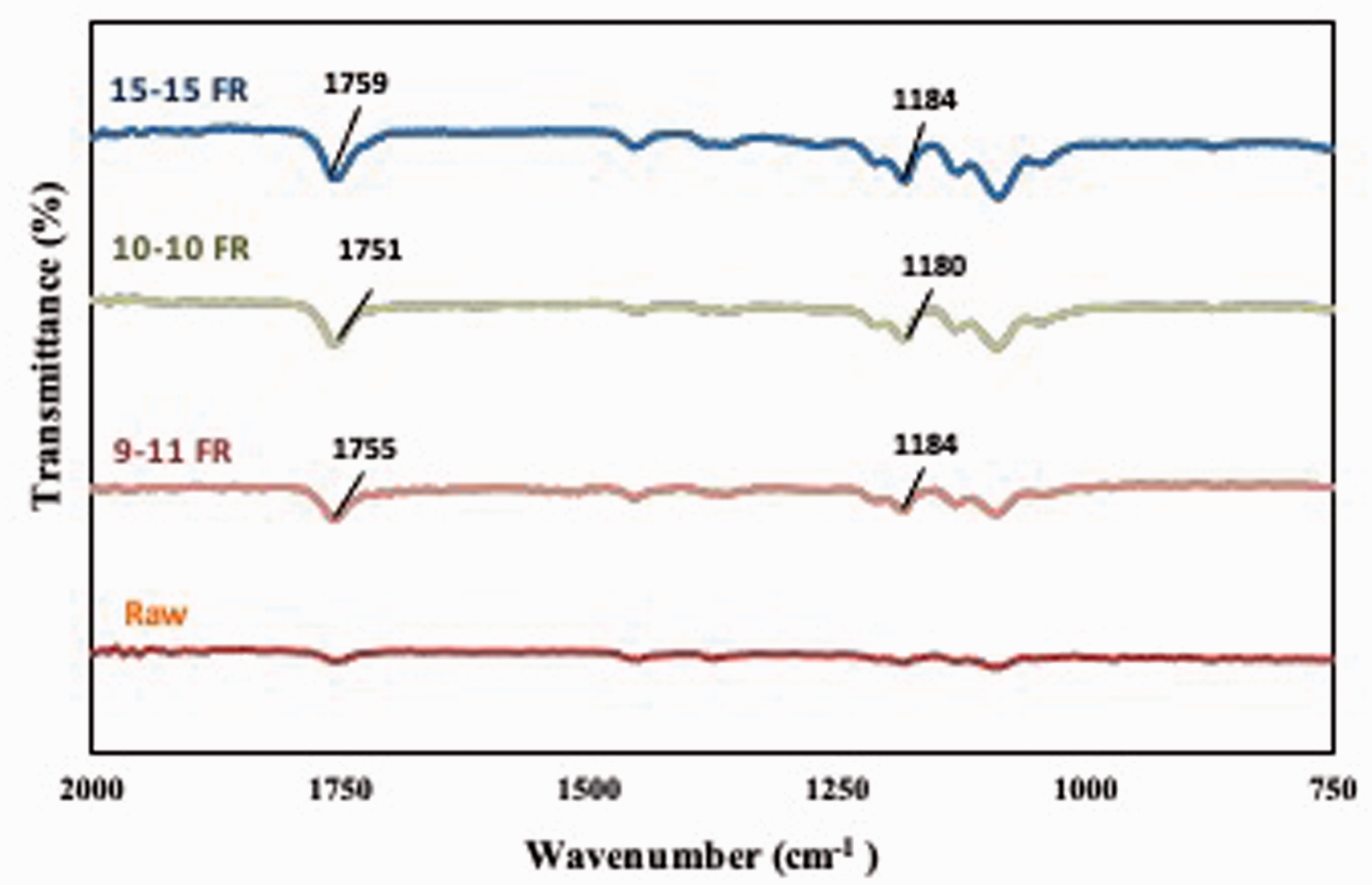
FTIR results of coaxial electrosprayed nonwoven fabrics at different flow rates.
Effect of extract concentration on nanocapsule formation
The

Size measurements of coaxial electrosprayed nanocapsules with different concentrations of the

SEM images of coaxial electrosprayed aluminum foils with different concentrations of the

SEM Images of coaxial electrosprayed nonwoven fabrics with different concentrations of the
Also, SEM images of electrosprayed aliminium foils (Figure 10) and nonwoven fabrics (Figure 11) support this inference about size increase at 0.01 g l−1 concentration compared to 1 g l−1 and 0.5 g l−1 concentrations. When the solution with 1 g l−1 concentration is diluted as 100 times to 0.01 g l−1 concentration, the solvent rate increases excessively. Higher liquid presence causes immense surface tension force on the sprayed jet. So, molecules tend to accumulate and take spherical form. Electrostatic force should overcome higher surface tension force for spraying. It gets harder to break jets to smaller droplets during electrospraying, so particle size gets bigger [68]. This situation is considered one of the fundamental reasons for bigger particle formation. Another reason is seen as an excessive amount of acetone usage in 0.01 g l−1 concentration. The high volatile characteristic of acetone causes very rapid evaporation of the solvent after spraying; the concentration suddenly increases. This sudden concentration change induces size increase on nanocapsules. Costa et al. [69] also reported that acetone usage generates a rapid rise in the electrosprayed fluid concentration. Even it can result in electrospinning instead of electrospraying at very low concentrations. This phenomenon is also observed for previous studies in the literature. It was explained that polymer concentration influence the surface tension, which effects particle size [65]. Sun et al. [51] reported that surface tension shows an increase for higher concentrations. Therefore, the particle size increases. Solvent evaporation has crucial importance for both core and shell solutions in terms of achieved morphology [70,71]. Especially core solvent has a determinative role in core-shell structure [70]. When the core solvent evaporates rapidly, it triggers thin layer formation at the core/shell interface. This interface layer eases to trapping of core material in the shell as well as prevents mixing core and shell solution and unstable tailor cone formation result of possible mixing [70,71].
Figure 12 shows photographs of electrosprayed surfaces of aluminum foils and nonwoven fabrics. According to these photographs, there isn’t any appearance change on nonwoven fabrics after application except 0.01 g l−1 concentration at which larger particles presence are produced. Taken together with all findings, the application is successful, and nanocapsules are widely spread on nonwoven fabrics' surface fibers as desired.

Photographs of coaxial electrosprayed surfaces of aluminum foils and nonwoven fabrics with different concentrations of the
Characterization of the nanocapsules
Coaxial electrospraying was performed with the same process parameters used in the flow rate study with a 10/10 µl min−1 flow rate and 1 g l−1

TEM images of coaxial electrosprayed nanocapsules.

FESEM image of coaxial electrosprayed surface of an aluminum foil.
Antibacterial efficiency of nanocapsules on medical nonwovens
Coaxial electrosprayed nanocapsules are collected on nonwoven fabrics. The aim of the nanocapsule application on the nonwoven fabric is to achieve antibacterial functionality. The results are shown in Table 2. The fabric coated with nanocapsules showed antibacterial efficiency up to 80% for
The antibacterial activities after 24 hours of incubation.

The concentration of bacteria was adjusted to 5.00 * 10 4 (log 4.70) cfu*/ml.
7.00 × 10 5 (log 5.85) cfu*/ml for each 1 g sample.
Conclusions
In the present study, nonwoven surfaces are coated with functional poly(lactic acid) nanocapsules containing
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was fully supported by the Scientific and Technological Research Council of Turkey (TUBITAK).
