Abstract
Poly(lactic acid) (PLA) was thermally blended with adipate ester (AE) to enhance the toughness of PLA. All specimens which were biodegradable materials were prepared using a plasticorder. Differential scanning calorimetry and Fourier-transform infrared spectroscopy indicated that the PLA structure was looser because of the presence of the AE additive. The elongation at break and biodegradable property increased substantially with increasing amounts of AE. The results reveal that excessive amounts of plasticizer would cause the exudation of AE from the PLA/AE composites, which was ascribed to the plasticizer migration phenomenon.
Introduction
Poly(lactic acid) (PLA) is a most essential biodegradable polymer which is produced from natural sources such as corn starch. 1,2 PLA can function as a biomedical polymer because of its biocompatibility. 3,4 Nonetheless, the brittleness of PLA restricts its potential applications such as in packaging or textiles, 5 -7 although it exhibits sturdy mechanical properties. As to the processability of PLA for spinning or film blowing, blending PLA with other polymers, 8 -12 fillers, 7,11,13,14 or plasticizers 15 -18 has been reported as an effective way to improve its processability to produce many products.
Suchaiya and Aht-Ong 19 studied a banana leaf sheath microcrystalline cellulose (BSMCC), wherein they used a modified banana leaf sheath as a compatibilizer. They observed that the compatibilizer could improve the mechanical properties of the PLA/BSMCC composite at an optimum compatibilized content, althought the tensile strength and Young’s modulus decreased. Kim et al. 20 used atmospheric plasma and silane treatments to improve the interfacial adhesion between PLA and acetate taffeta fabric (ATF); the peel strength and tensile strength of the PLA/ATF composite increased after the plasma surface treatment of ATF. Lai and Li 21 modified silica by melt blending it with polyurethane to initiate the nucleation of PLA; the crystallization peak temperature and optical transmittance of the modified silica/PLA composite were higher than those of the unmodified silica/PLA composite. Tsou et al. 22 synthesized a new nanomaterial (nanosilver-doped multiwall carbon nanotube) to enhance the mechanical property, theromostability, and antibacterial properties of PLA. Moreover, Tsou et al. 18 indicated that acetyl tributyl citrate as a plasticizer could enhance the elongation at break of PLA/tapioca composites.
The use of adipate ester (AE) as a plasticizer for PLA has not yet been reported; thus, in this present study, AE was chosen as a plasticizer to enhance the flexibility, processability, and biodegradability of brittle PLA. This present study is the first to report that a plasticizer would migrate out of a polymer during the water absorption test. The proposed AE content in PLA stemmed from the result of water absorption analyses.
Experimental
Materials and preparation
The PLA used in this study, with a trade name of Nature Green 4032D, was purchased from Cargill-Dow (USA). PLA was dried in a vacuum oven at 80°C for 8 h to remove the water. AE was supplied by Golden Chemical Co. Ltd (Taiwan). PLA/AE at varying weight ratios were melt blended using a plasticorder. During the blending process, the plasticorder was operated at a temperature of 190°C and a speed of 120 r min−1 for 3 min. All prepared specimens were then hot pressed at 190°C and 8 MPa for 3 min, and finally cooled in air at 28°C. All specimens were dried in a vacuum oven at 80°C for 8 h before they were hot pressed. Table 1 summarizes the compositions of all specimens prepared in this study. Figure 1 illustrates the processing and characterization of the PLA/AE specimens.
Composition of PLA and PLA/AE specimens.

PLA: poly(lactic acid); AE: adipate ester.

Processing and characterization of PLA/AE specimens. PLA: poly(lactic acid); AE: adipate ester.
Tensile properties
PLA/AE specimens at 25°C were characterized using a Shimadzu (Japan) tensile testing machine (AG-10KNA model) with a speed of 50 mm min−1. Dog-bone-shaped specimens were prepared according to ASTM D638 Type IV standards. The data for tensile properties, based on the average tensile results of at least six specimens, were obtained.
Differential scanning calorimetry
The thermal properties of specimens were determined using differential scanning calorimetry (DSC) (TA Q100, TA Instruments, USA). DSC scans were all at a heating rate of 10°C min−1 and under a flowing nitrogen at 50 mL min−1. The thermograms were obtained from the second heating scan after the initial quench cooling to avoid the effects of heat history. The instrument was calibrated using pure indium. Samples (0.5 mg) were placed in standard aluminum pans for glass transition temperature (T g) and melting temperature (T m) determination.
Water absorption analysis
Five specimens (10 × 10 × 0.5 mm3) were measured for their water absorption. After the specimens were conditioned in desiccators for 3 weeks, they were weighed, and the weights recorded. The samples were immersed in distilled water at room temperature for 24 h. Then, the excess water on the suface of the specimens were wiped off with a tissue. The water absorption was calculated using equation (1)

where W 0 was the original weight of the dry sample and W 1 was the weight of the sample immersed in distilled water for 24 h. The data on water absorption, based on the average water absorption of at least five specimens, were obtained.
Enzymatic hydrolysis test
The biodegradative evaluation of specimens through hydrolysis was conducted at 27°C with the help of an aqueous solution containing 50 mg starch enzyme (0.025 MNa2HPO4 + 0.025 MKH2PO4). The specimens with dimensions of 5 × 5 cm2 were immersed in the aqueous solution at varying periods of time (18, 36, 72, and144 h), after which they were washed with distilled water and dried in a vacuum oven at 80°C for 8 h. The degree of degradation, based on the specimen’s weight loss, was calcuated using equation (2)

where W 0 was the dry weight before the degradation and Wt was the dry weight at time t.
Fourier-transform infrared spectroscopy
A PerkinElmer spectrometer (Spectrum One model; PerkinElmer Inc., Waltham, Massachusetts, USA) provided the Fourier-transform infrared (FTIR) spectral data. Fifteen FTIR scans were averaged to produce the specimens’ spectra; the wavenumber range was 2250–3750 cm−1, and the resolution was 2 cm−1.
Results and discussion
Tensile properties
Figure 2 plots the tensile strength and elongation at break of PLA and PLA/AE specimens. The elongation at break of PLA was only 3.7%. After blending PLA with AE, the elongation at break of the formed PLA/AE specimens exhibited a substantial increase. AE improved the ductility of PLA. By contrast, the tensile strengths (Figure 2) and tensile moduli (Table 2) of PLA/AE specimens decreased substantially with increasing amounts of AE. These results indicate that the brittleness of PLA was synergistically enhanced after AE was added to PLA.

Tensile properties of PLA and PLA/AE specimens. PLA: poly(lactic acid); AE: adipate ester.
Tensile modulus and data for PLA and PLA/AE specimens.

PLA: poly(lactic acid); AE: adipate ester; T g: glass transition temperature; T cc: cold crystallization temperature; T m: melting temperature; H m: melting enthalpy.
Thermal properties
The thermal behavior of PLA and PLA/AE specimens of different compositions was investigated. The molded specimens were cooled rapidly, and the cold crystallization peak appeared during the second heating cycle. Figure 3 depicts the DSC curves of PLA and PLA/AE specimens which were melt quenched. The crystallization and melting enthalpies indicated that PLA was nearly amorphous after melt quenching. The DSC curves of PLA with plasticizer whose content ranged from 0 wt% to 30 wt% indicated a single T g and cold crystallization (T cc) temperatures which decreased with increasing amounts of AE. This result agrees with a previous study by Tsou et al. 18 who analyzed PLA/tapioca composites with plasticizer. PLA/AE specimens indicated a decreasing trend of T g and T m with increasing AE content which is due to the longer distance between the molecular chains after AE was added as a plasticizer to PLA. 23 Therefore, PLA could be melted easily.

DSC for (a) PLA, (b) PLA9 5AE5, (c) PLA9 0AE10, (d) PLA85AE15, (e) PLA80AE20, (f) PLA75AE25, and (g) PLA70AE30 specimens. PLA: poly(lactic acid); AE: adipate ester; DSC: differential scanning calorimetry.
Water absorption analysis
The water absorption of PLA/AE specimens in Figure 4 indicated that the water absorption rate increased from 0.7% to 0.91% as the AE content increased from 0% to 5%. But the water absorption rates decreased substantially (0.91–0.43%) with increasing amounts of AE (5–30%). This interesting result could be due to the exudation of AE as the AE content increased from 10% to 30%. Furthermore, the water absorption increased with the AE content (5–15%) (this increase was higher than that for the neat PLA) which was attributed to the AE’s capacity to increase the free volume in PLA. Therefore, the water molecules infiltrated PLA easily, because the AE content increased the mobility of PLA molecules. This behavior is evidenced by the DSC analysis (Figure 3): T g decreased with increasing AE content. Hence, PLA could absorb water molecules easily as the AE content increased the mobility of PLA molecules. The proposed AE content in this study was 10 wt% because of the minimal plasticizer migration phenomenon, but the elongation approached 250% from 3.7%.

Water absorptions for PLA and PLA/AE specimens. PLA: poly(lactic acid); AE: adipate ester.
Hydrolysis
Plots of the weight loss of PLA and PLA/AE specimens at varying periods of enzymatic hydrolysis time (Figure 5) denoted a common result: the percentage weight loss for all series increased with the hydrolysis time. Notably, the weight loss of PLA/AE increased from 0.02% to 10.23% as the AE content increased from 0 wt% to 30 wt% within 144 h of hydrolysis time. Water could attack the molecules of PLA easily when PLA was incorporated with AE which could not only enhance the process of hydrolysis but also impart biodegradative properites to PLA. The result of the hydrolysis analysis can be a useful data for biodegradable polymer industries.

Weight loss of PLA (^), PLA95AE5 (△), PLA90AE10 (□), PLA85AE15 (◇), PLA80AE20 (☆), PLA75AE25 (+), and PLA75AE25 (×). PLA: poly(lactic acid); AE: adipate ester.
Spectral characteristics and the interaction between PLA and AE
Figure 6 represents the FTIR spectra of PLA and PLA/AE specimens. The PLA spectrum indicated O–H stretching vibration at 3661 and 3509 cm−1, and that of PLA/AE at 3659 and 3506 cm−1. Three absorption bands were located at 2990, 2945, and 3508 cm−1, corresponding to C–H aliphatic stretching. The FTIR spectra of PLA/AE specimens indicated molecular interactions between PLA and AE. This interaction might be attributed to the H bond between the C=O group in AE and a few terminal hydroxyl groups in the main chain of PLA. 24 The FTIR spectrum of pure PLA indicated peaks at 2990 and 2945 cm−1, corresponding to the OH group stretching of PLA. These peaks decreased gradually with increasing amounts of AE. The OH group could be due to the probable terminal hydroxyl groups of PLA which was released during the interaction between PLA and AE. Figure 7 illustrates the proposed molecular interaction between PLA and AE. Enzymes or water broke the structures of PLA easily, as the amount of AE increased during hydrolysis. 18

FTIR of (a) PLA, (b) PLA9 5AE5, (c) PLA9 0AE10, (d) PLA85AE15, (e) PLA80AE20, (f) PLA75AE25, and (g) PLA70AE30 specimens. PLA: poly(lactic acid); AE: adipate ester. FTIR: Fourier-transform infrared.
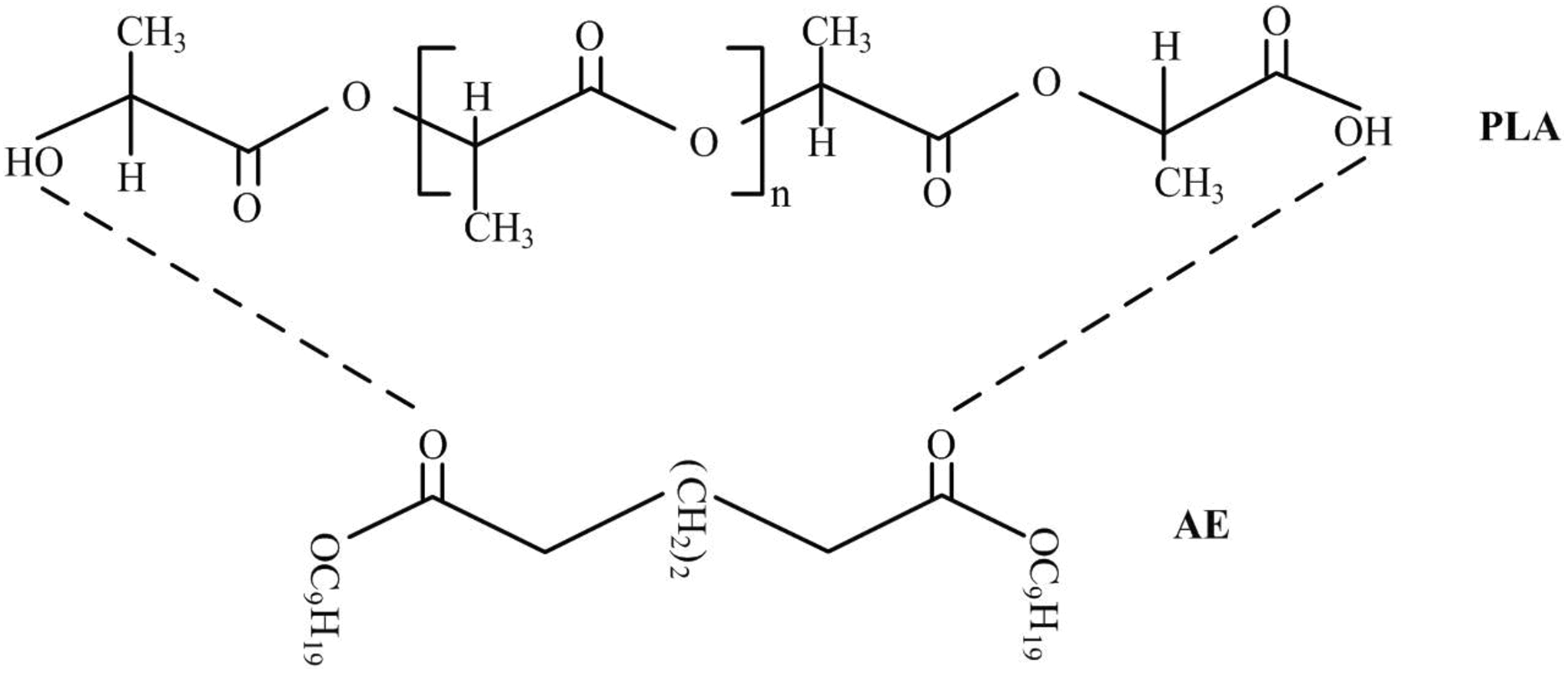
Proposed molecular interaction between PLA and AE. PLA: poly(lactic acid); AE: adipate ester.
Conclusions
Increasing amounts of AE improved the elongation at break but reduced the tensile strength. Furthermore, the addition of AE (0–30 wt%) lowered the glass transition and cold crystallization of PLA and PLA/AE specimens, as demonstrated by their DSC curves. The maximum in the water absorption curve was a phenomenon associated with the AE exudation from PLA. During the hydrolysis test, the weight loss resulting from a decrease in the AE content was attributed to the less dense structure of PLA, causing the PLA to absorb water molecules and be degraded easily. From the results and discussion about tensile properties and water absorption, the AE content of PLA at 10 wt% was recommended for PLA/AE specimens.
Footnotes
Acknowledgment
This research was supported by the National Natural Science Foundation of China. The authors also express their appreciation to Wuliangye Group Co. Ltd, Apex Nanotek Co. Ltd, the Ratchadapisek Sompote Fund for Postoctoral Fellowship (Chulalongkon University), the Sichuan Province Science and Technology Support Program, the Zigong City Science and Technology office (2017XC16), the National Science Council and the Ministry of Science and Technology (Taipei City, Taiwan, R.O.C.), (MOST 103-2622-E-244-001-CC3) for financial support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Rachadapisek Sompote Fund for Postdoctoral Fellowship, Chulalongkorn University.
