Abstract
In order to introduce multifunctional properties into the recycled polyphenylene sulfite (PPS) nonwoven fabrics, highly conductive and hydrophobic fabrics with excellent Joule heating performance were fabricated by depositing in-situ polymerized polypyrrole (PPy) onto PPS nonwoven fabric and followed by a silicone coating process. The resultant PPy-coated nonwoven fabric exhibited high electrical conductivity. The surface resistivity of PPy-coated nonwoven fabric was less than 8.8 Ω/sq. The high conductivity of PPy coating provided the fabric with excellent electrical heating properties in conjunction with a photothermal heating properties. The saturation temperature of the samples can get to 125.6°C with 4V DC power. The thin silicone coating can help to resist the oxidation of PPy and endow the nonwoven fabrics with excellent water-resistant features. This study provided a simple and effective method to transform the recycled PPS nonwoven fabric into a multifunction material.
Introduction
In some cold areas, pipes, greenhouses and concrete need to be insulated from freezing. Some kinds of thermal insulating textile such as pipe insulation sleeve and Greenhouse insulation blanket were used to avoid the freezing damage. The concept of textile insulation at sub-zero temperature involves two techniques. Firstly, using a material with low thermal conductivity, and secondly, restraining the heat loss due to radiation. 1 The nonwoven fabrics usually were used as the thermal insulating material due to the high-bulk structure with high volume of air. 2 However, the normal textiles are negative thermal insulating materials. They could not help in some extreme cold conditions that require heating. Therefore, a material which could perform the dual role of thermal insulation and active heating as needed has gradually become the research focus.
Heating textiles can warm the protected target actively for working in a low-temperature environment, which include solar heating textile, chemical heating textile, phase change material heating textile and electric heating textile. 3 Electric heating fabric uses electric energy to drive the heating element inside the fabric. The thermal energy could be generated actively when the electrical current passing through a heat-generating conductor. The fabric-based heating element has advantages of light weight, good flexibility and durability, which is one of the most important components of the electric heating fabrics. The fabric-based heating element is composed of conductive materials and substrate materials.
Conductive materials can be divided into metallic and non-metallic materials. Metal materials include metal wire, metal fibre blended yarn4,5 and metal coated yarn6,7 However, there are many limitations for the metallic materials which associated with wire breakage, processability, weight burden and inflexible nature. Non-metallic materials include inherently conductive polymers (ICPs) 8 and carbon materials. 9 These materials do not affect the performance of the fabric because of the light weight property. Among these materials, ICPs are well-suited for the fabrication of the conductive textiles since the great properties of the materials such as high conductivity, flexibility, simple chemistry and lower cost. Electro-conductive textiles have expanded rapidly due to their potential applications in the fields of wearable electronics, sensors, electromagnetic shielding and heating fabrics.10–12
Polypyrrole (PPy) has been the mostly used material compared to other conducting polymers, which is a promising conductive polymer for commercial applications. However, PPy has poor mechanical properties. Therefore, PPy could be coated on strong and flexible textile substrates such as PET, nylon, cotton and lycra fabric.13–15 Hao et al. 16 used the in situ polymerization method to produce PPy-coated cotton fabrics. The surface resistivity of PPy-coated fabric was 303 Ω/sq. The maximum equilibrium temperature can reach to 28/56/76/83°C at the applied voltage of 3/6/7. 5/9V. Wang et al. 17 fabricated a multi-functional textile by depositing in situ polymerized polypyrrole modified MXene sheets onto PET textiles. The saturated temperatures reached to 57 and 79°C at the voltage of 3 V and 4 V, respectively. Kaynak et al. 18 studied the heating effects of polypyrrole-coated PET-Lycra fabrics. The surface resistance of PPy-coated fabrics ranged from approximately 150–500 Ω/sq. The resistance of conductive fabrics increased about 10% in 72 h when subjected to a constant voltage of 24 V. Shang et al. 19 used gas combination method to deposit PPy coating on the polyester (PET) fabrics. The maximum equilibrium temperature of PPy-coated fabric (250 Ω/sq) reaches to 59.2°C at the applied voltage of 30 V. The temperature decreases slightly when a DC voltage is applied for 120 min, which could lead to the oxidative degradation of PPy coating. The disadvantage of PPy coating is the poor stability under ambient conditions, which restricts its commercial applications. Thus, improving the heat stability is important for PPy-coated fabrics.
In recent years, recycling has been an attractive issue for researchers considering the sustainability and the wastes arising from different engineering applications. 20 The usage of the recycled nonwoven filter bag in thermal insulation and heat generation contribute to the decrease in environmental pollution and the prevention of the decrease in petroleum supplies. The polyphenylene sulfite (PPS) fibre with outstanding thermal stability, inherent flame resistance and excellent chemical resistance is widely used in filter bags. 21 The nonwoven filter bag made of PPS has been widely used to remove the particulate pollutants, such as PM2.5 in the industrial flue gas. 22 The nonwoven fabrics have more porosity and higher filtration efficiency that are preferred for air filter applications. When the filtration efficiency cannot meet the requirements, hundreds of filter bags will be replaced. These used filter bags would be sent to the landfill sites and remain there for ages as they are not biodegradable.
In this paper, we used the recycled polyphenylene sulfite (PPS) nonwoven filter as the substrate material to prepare PPy-coated nonwoven fabric with remarkable performance by using the improved chemical polymerization technique. The PPy-coated nonwoven PPS fabric (PCPF) has shown superior electro-conductive and heat generation performance. This study provided a simple and practical method for transforming the recycled nonwoven filter into a heat-generating material.
Experimental section
Material
The PPS needle-punched nonwoven filters were recycled from the coal-fired power stations, which had been used for 2 years for flue gas filtration. The nonwoven fabrics were made of 100% PPS staple fibre and the base fabric. The base fabric is the woven fabric made by PPS filament with an area density of 85 g/m2. The areal density of the nonwoven fabric is 588 g/m2 and the thickness is 2.25 mm.
Pyrrole (97%) monomer was procured by Sigma Aldrich China. Ferric trichloride hexahydrate (FeCl3·6H2O), sodium 5-sulfosalicylate (NaSSA), Cyclohexane, and absolute ethyl alcohol were purchased from Sinopharm Chemical Reagent Co. Ltd (Shanghai, China). Silicone oligomer (Polydimethylsiloxane PDMS) was purchased from Dow Corning Co. Ltd.
Fabrication of PPy-coated PPS nonwoven fabric
An improved in situ solution polymerization method was used to fabricate PPy-coated PPS nonwoven fabric (PCPF).10,23 The PPS filter bags were washed by water to remove the dust. Then the filter bag was cut along the suture site to get the PPS nonwoven fabric (Figure 1(a)). The PPS nonwoven fabric were cut into the required size (square:30×30 mm and rectangle: 300×50 mm) and horizontal placed in a polypropylene box. The samples were washed by absolute ethyl alcohol to make easily infiltrate. The aqueous solution was prepared by dissolving sodium 5-sulfosalicylate and ferric chloride in de-ionized water. The aqueous solution was added drop wise until the fabrics were completely immersed in the solution. Then, the PP box was frozen for 3 h. Subsequently, an oil-phase solution was prepared by dissolving pyrrole monomer in cyclohexane. The oil-phase solution was added on the frozen fabric. Then, a two-phase system was placed in champers at low temperature (0–5°C) and remained stationary for a certain time. The polymerization samples were taken out and thoroughly washed in distilled water, and vacuum dried at 80°C for 12 h before measurement. (a) Photos of recycled filter, nonwoven fabric and PPy-coated fabric. (b) Schematic view of preparation of PPy-coated nonwoven fabric.
In order to make it resistance to oxidation and water-resistant, silicone oligomer (Dow Corning 184) was used to coat the PCPF through a dip-coating process. The weight fraction between the components A:B was 10:1. The silicone oligomer solution was diluted by cyclohexane, and the mass ratio was 1:2. The PPy-fabric was impregnated by the diluted solution and the excess solution was removed. Then cured at 100°C for 30 min in the vacuum oven to complete the hydrosilylation reaction.
Characterization
The morphology of PPy on PPS fabrics was characterized by the field emission scanning electron microscopy (Nova NanoSEM 450, USA) equipped with energy dispersive X-ray spectroscopy (EDS). The accelerating voltage of 15 kV was used. The electrical conductivity of PPy coated fabrics was measured by a four-point probe measurement system (RTS-8, Guangzhou, China) at 25 ± 2°C temperature and 65% relative humidity. A DC power supply (GPS-3030DD, GWINSTEK, China) was used to test the electrical and thermal properties of the PPy-coated fabric. Thermal images were taken by using a forward-looking infrared radiometer IR imaging camera (FLK-TIR32 9HZ, Fluke, USA). The water-resistant performances were tested by a contact angle measuring instrument (Powerach®JC200D3, Shanghai, China). In addition, the average water contact angles were obtained by measuring the same sample at least in five different positions.
Results and discussion
Preparation of PCPF
In the previous research, we used an improved in-situ solution polymerization method to prepare various conductive fabrics, 10 conductive yarns 24 and conductive sponges, 25 and obtained multifunctional fabrics, yarns and energy storage materials. In this experiment, we used an improved in-situ solution polymerization method to prepare PPy-coated nonwoven PPS fabrics for heat generation application.
In the experiments, the oxidant ferric chloride can infiltrate the entire PPS nonwoven fabric. Then, the solution was frozen. Subsequently, the cyclohexane solution containing pyrrole was added onto the surface of the frozen solution. Pyrrole was slowly phase-extracted owing to the gravity and gradually infiltrated the aqueous solution. After that pyrrole would slowly diffuse into the porous structure of PPS nonwoven fabric, undergoes oxidative polymerization and depositing on the inner wall of the porous structures, and finally the conductive PPS nonwoven fabrics were fabricated (Figure 1(b)). The improved in situ polymerization technique can lower the reaction rate with low temperature and gradually phase-extracted of pyrrole, which improves the order degree of the molecular structure of the polypyrrole. The slower progression of polymerization ensures joining of pyrrole units to growing chain preferably at 2-5′ position with 180° rotation. 26 At the same time, reduced the chemical defects occurring in PPy polymerization. PPy with a planarity and linearity of chains yields higher conductivity characteristics.
In order to find out the individual effects of the oxidant (FeCl3), dopant (NaSSA), and the monomer (pyrrole) on the sheet resistance of the coated fabric, concentrations of the reactants were varied. The sheet resistance of PCPF is shown in Figure 2. The PPy-coated nonwoven fabrics were prepared with different ratios of n(Py)/n(FeCl3), the sheet resistance initially decreases with the increase of the concentration of FeCl3. The lowest sheet resistance of n(Py)/n(FeCl3) is 1:2. There is a gradual increase in the resistivity with further increase in the concentration of FeCl3. The effect of dopant concentration on the fabric resistivity is shown in Figure 2(b). The sheet resistance of PPy-coated fabric decreased with the increasing in n(NaSSA)/n(FeCl3) ratio. The resistivity decreased rapidly by increasing in dopant concentration until a plateau is reached at the concentration of 0.18 mol. The change of resistivity is not obvious with further increase in NaSSA concentration. Similar observations have been reported on the effects of dopant concentration on the electrical conductivity.
27
(a) Resistance versus ferric chloride concentration (Pyrrole: 0.18 mol, NaSSA:0.12 mol, Duration: 12 h); (b) Resistance versus NaSSA concentration (Pyrrole: 0.18 mol, FeCl3: 0.36 mol, Duration: 12 h).
Figure 3 has shown the fabric resistivity and weight add-on percentage of experimental trials performed by varying reaction time. As can be seen from Figure 3, the surface resistivity of PPy-coated fabric decreased with the increasing in the reaction time. With time increasing, the α-site of pyrrole at the interface is more likely to come into contact with other pyrroles at the α-site. This facilitates more α–α polymerization of pyrrole molecules, which improves the planarity and linearity of the polypyrrole molecular structure and ultimately improves the conductivity of polypyrrole. In the experiments, it was found that the best electrical conductivity of polypyrrole could be obtained with a reaction time of 24 h. Continuing to extend the reaction time, the conductivity of pyrrole was not further enhanced, probably because the polypyrrole had already precipitated out of water by this time and could not further increase its molecular weight and orderliness, but the loading would increase with time. Resistance and add-on percentage versus reaction time (Pyrrole: 0.18 mol, FeCl3: 0.36 mol, NaSSA:0.12 mol).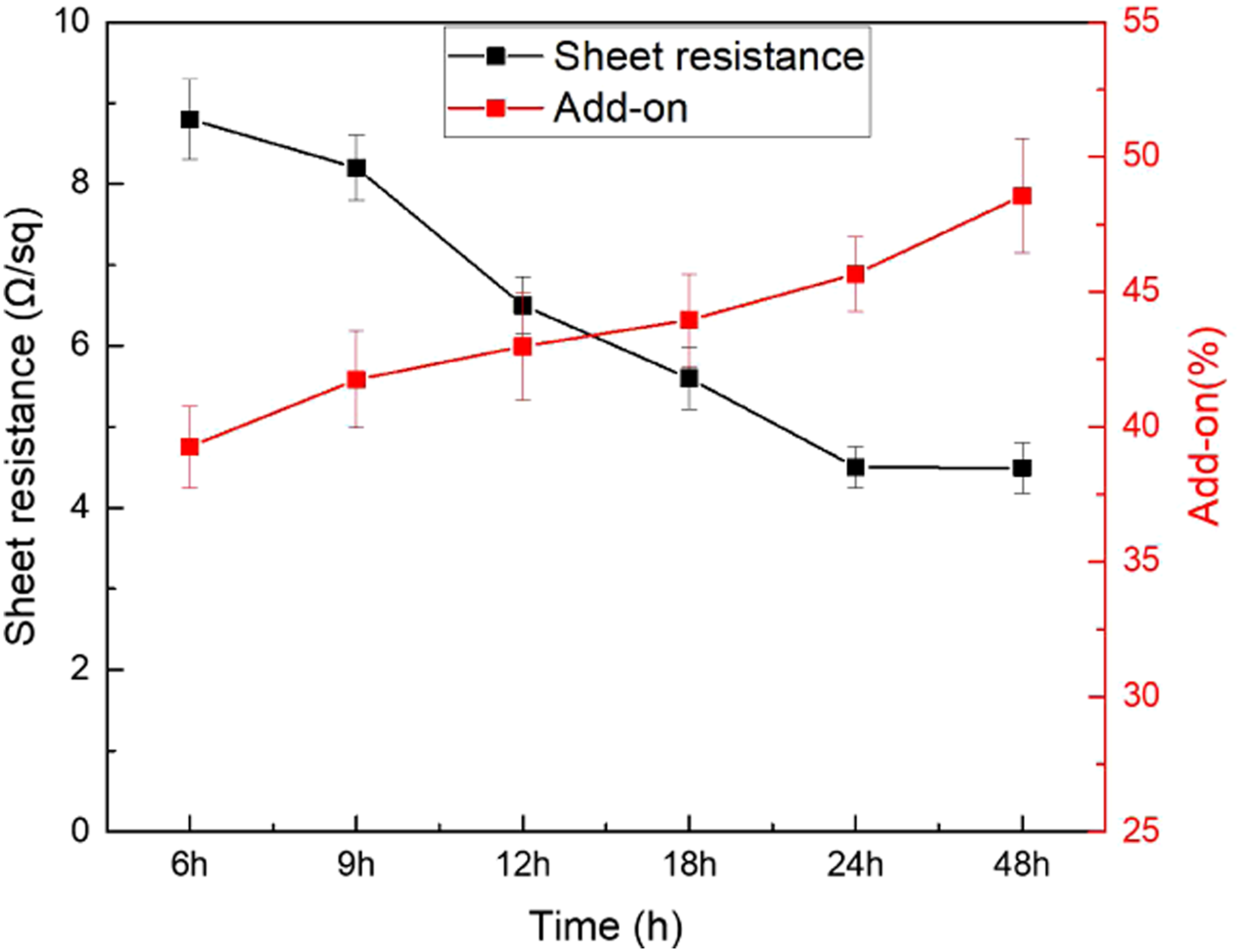
The add-on rate also depends on the experimental conditions and the fabric structure. The weight add-on rate of PPy-coated fabric increased from 39% at 6 h to 48.5% at 48 h. The weight add-on rates of nonwoven fabric are higher than that of flat woven fabrics. 12 Needle-punched nonwoven fabric has more bulky and porous structure than that of woven fabric. Therefore, needle-punched nonwoven fabrics would hold more polypyrrole between fibre interstices.
Visual observation
The images of PPS nonwoven fabrics and PCPF are shown in Figure 1(a). The colour of nonwoven-filter changed from brown to deep black post polymerization, indicating good distribution of PPy polymer deposition onto the fabric surface. The surface morphologies of the pristine and PPy-coated PPS fibres are shown in Figure 4. The neat and smooth surface of the pristine PPS fibres are shown in Image a. The surface of PPy-coated PPS fibres were characterized as shown in Images b-d in Figure 4, where there is a homogeneous, thin, and dense coating along the surface of the fibre. The thickness of PPy layer is about 2±0.5 μm. The PPy-coated fibres exhibited a uniform coating. Polymerization of pyrrole took place simultaneously both on the fibre surface and in the solution as bulk polymerization. The coating process was attributed to a physical adsorption mechanism due to large surface area of fabric substrate. Some PPy particles were deposited on the surface of the fibres. These PPy particles were loosely deposited onto the surface of the fibre, which can be washed off the surface. But, a mass of PPy particles were observed in the interstices of the filter owing to the nature of bulky and porous of the nonwoven structure. The elemental mapping images of the coating are shown in Figures 4(e)–(i). From the elemental mapping images, it can be seen that carbon from PPy and NaSSA, nitrogen from PPy, oxygen from NaSSA, as well as sulphur and chlorine from NaSSA and FeCl3 doped in PPy are distributed uniformly along the fiber surface, indicating that the improved technique helped to distribute a uniform conductive polymer coating on the surface of PPS nonwoven fabric. Photographs of (a)–(d) SEM image of PPy-coated fabric. (e)–(i) elemental mapping images.
Surface resistance
Comparation of sheet resistance of PPy-coated fabric.

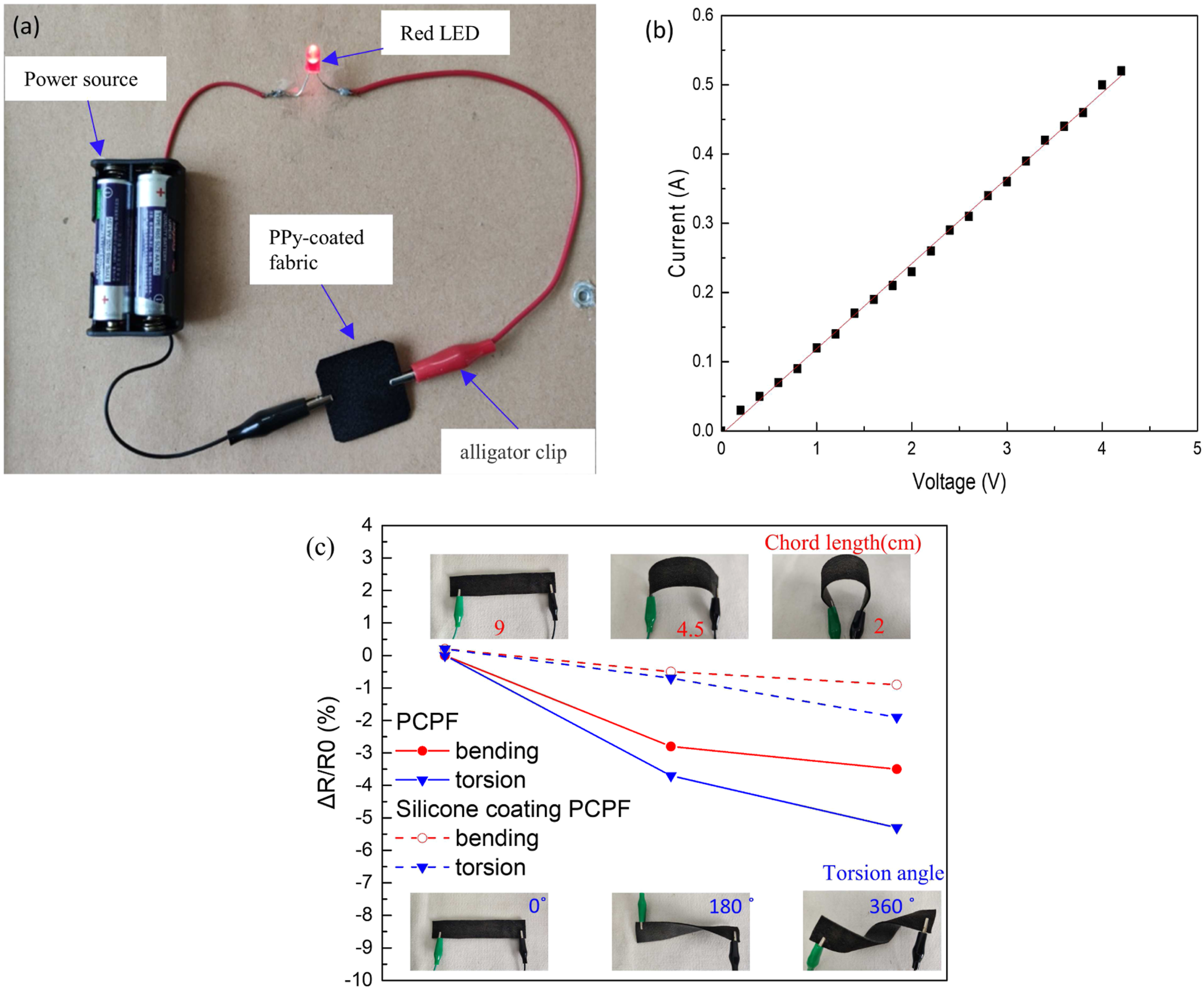
(a) Photo of PPy-coated fabric as a connection wire in a closed circuit. (b) Current-voltage curves of PPy-coated fabric. (c) Normalized relative resistance changes during bending and torsion tests.
The nonwoven fabric has satisfactory mechanical flexibility and structural stability, which can withstand complicated deformations during practical applications. However, whether the electrical conduction of PCPF can withstand folding, bending, and torsion is unknown. Figure 5(c) shows the electrical resistance changes (ΔR/R0×100%) of PCPF (100 mm×20 mm) and its silicone-coated counterpart under bending and torsion. It is interesting that the PCPF reduced the electrical resistance under bending/torsion. The fibres were squeezed together during the bending/torsion, the adjacent individual fibres tend to contact each other to form tight conductive network. Therefore, the electrical resistance of PCPF decreased. The silicone-coated PCPF presents a higher resistance than that of the neat PCPF due to the slight detrimental effect of the insulating silicone on the formation of conductive networks. The silicone coating reduces the resistance change sensitivity of the textile under bending/torsion. The insulating silicone layer hinders further contact of PPy-coated fibres, leading to less resistance change.
Joule heating performance of PCPF
The PCPF fabricated by the improved in situ polymerization technique has the higher electrical conductivity as shown in Table 1. According to the joule’s law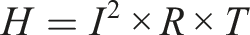
DC voltage was applied onto PCPF. The electric heating was tested at various voltages. Four different levels of low voltage were chosen such as 1, 2, 3, and 4 V. At a fixed level of applied voltage, the surface temperature of the fabric was measured. The results are shown in Figure 6. It can be seen from Figure 6(a) that the initial temperature rises sharply during the first 100 s, then followed by a levelling-off stage up to saturation temperature. For example, the saturation temperature of PCPF was 125.6°C when 4 V was applied. During the initial stage, the electrical energy input into the fabric exceeds the total thermal energy losses so that the temperature increased. As the temperature rising, the heat loss increases. The temperature stabilizes until the generated and lost heat reached a balance. The infrared camera images in Figure 6(b) indicated a steady temperature response with all different voltages that have been tested. (a) Time–temperature curve at different voltages (b) thermal camera images of fabric after the application of different voltage.
Comparisons of the saturation temperature with those of other report.

Beside the electric heating behaviour discussed above, the photothermal performance of PCPF was also studied. Firstly, the PPy-coated PET nonwoven fabric was cut into different shapes and patterns. As shown in Figure 7(a), ‘YC’ was cut as a pattern from the fabric. The cut PCPF were exposed to the sunlight. The temperature of the fabric rises with the increase of the exposure duration and get stable at the saturation temperature. After 8 min exposure to the sunlight, the saturation temperature of PCPF stabled at 57.3°C (Figure 7(b)). The electric heating and photothermal heating behaviour of PCPF make it very suitable to be applied as a heating element. The current will flow through fabrics without any wiring, which produces more even flow of heat throughout the fabric. At the same time, heat generating was not affected by holes or cuts in the fabric. (a) Photo of PPy-coated fabric (b) thermal camera images in natural sunlight.
Atmospheric aging of PCPF
As a heating material, the thermostability is important. The PPy-coated fabric was investigated by observing the surface resistance changes upon heat treatment at 130°C for 1–12 h, respectively. The accelerated aging was also tested in vacuum to verify the atmospheric oxidation effect. Figure 8(a) shown that the surface resistivity was increased sharply with the time in the open atmosphere. The surface resistivity increased to 549.1 Ω/sq after 12 h, which was 62 times higher than the initial surface resistivity. As a contrast, the surface resistivity of the samples in the vacuum remained stable after 12 h. The atmospheric degradation kinetics of PPy has been investigated by many scholars. The reports suggested that the oxygen attack was the major cause of the degradation.
31
The α, β-unsaturated carbonyl groups were formed by the reaction of oxygen on PPy backbone, which were believed to act as an electron trap.26,32 Therefore, chemical degradation of PPy disrupts the continuity in conjugation length and subsequently hindered the charge movement. (a) Accelerated aging in 130°C for 1–12 h (b) Temperature-stability of the silicone-coated nonwoven fabric in heating/cooling cycles.
The loss of conductivity of PPy seems unavoidable in the atmosphere. In order to make it resistance to oxidation, silicone was used to encapsulate the PPy layer to minimize the contact with air. To assess the heating stability and recyclability, the heating performance stability was evaluated by performing cyclic heating/cooling cycles on the silicone-coated PCPF under a voltage of 2 V. The silicone-coated PCPF was heating/cooling on the 1st, 20th, and 50th cycles. The Joule heating performances are shown in Figure 8(b). The temperature-time curves of silicone-coated fabric for the 1st, 20th, and 50th cycles are nearly overlapped, which verified the stable electrical conductivity of PPy coating and confirmed the long-term heating stability of the silicone-coated PCPF.
Water-resistant performances of PCPF
As the industrial textile, water-resistant is a basic requirement in some applications. It is known that PPy layer is hydrophilic and the silicone is hydrophobic. Polydimethylsiloxane (PDMS) with identical terminate groups of methyl is an elastic silicone rubber which possesses excellent strength, wear-resistance, and strong adhesion to fabrics. The silicone coating can help to improve the water-resistant performance of PCPF. The contact angles of fabrics were tested and shown in Figure 9 to illustrate the transition from hydrophilicity to hydrophobicity induced by the silicone layer. The initial water contact angle of PPy coated nonwoven fabric is about 100° due to the rough surface of the nonwoven fabric and PPy nanoparticles on the surface of the fibre. Then, the water droplet begins to bond with PPy coating and quickly expand in the interstices within 30 s (Figure 9(a)). In contrast, the silicone-coated PCPF has a much higher contact angle of 126° due to its hydrophobic feature (Figure 9(b)). Therefore, the silicone coating can not only improve the heating stability by protecting PPy from oxidation but also convert the hydrophilic PPy-coated fabric to hydrophobic. Water contact angle measurements of (a) PCPF and (b) the silicone coated fabric.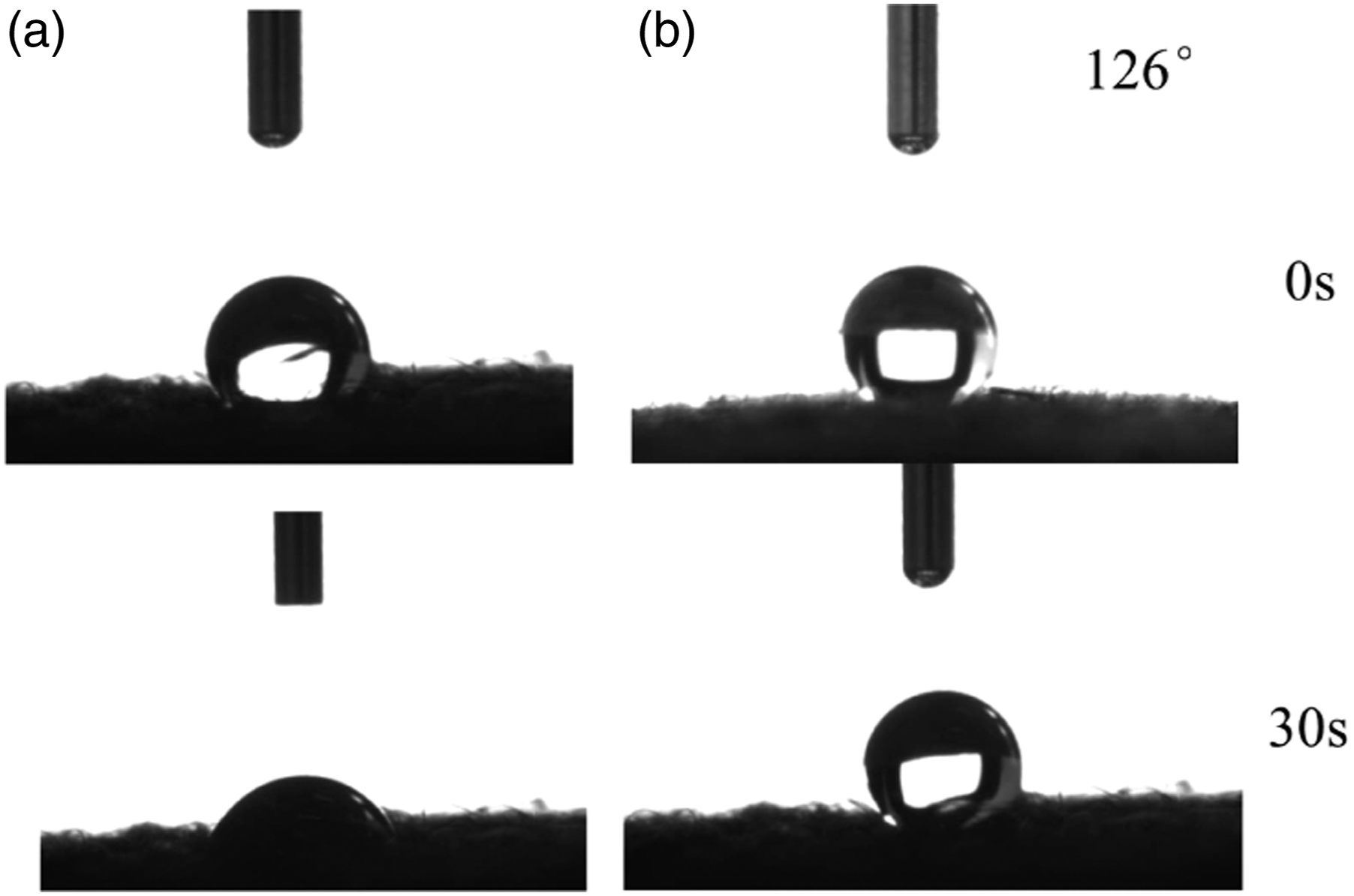
Conclusions
This work introduced an efficient in situ polymerization approach for fabricating highly conductive, hydrophobic, and flexible PPy-coated recycled polyphenylene sulfite nonwoven fabric with excellent Joule heating performance. The sheet resistance of PCPF was controlled to 4.5–8.8 Ω/sq. The saturation temperature can get to 125.6°C when 4 V was applied. At the same time, PCPF has good photothermal heating properties. The aging property of PCPF was tested at high temperature. Atmospheric oxidation of PPy is obvious at 130°C. The silicone was used to coat PCPF for resisting the oxidation of PPy and giving the material a good water-resistant feature. The contact angle of the silicone-coated PCPF was increased to 126°. The silicone coating converted the hydrophilic PCPF into hydrophobic, and improved the long-term stability of the electrical heating performances. The silicone-coated PCPF has demonstrated excellent Joule heating performance with good cycling stability and tunability at satisfactory voltages. Thus, the multifunctional recycled PPS nonwoven fabrics with a unique blend of electrical conductivity, thermal insulating, electrical and photothermal heating performance, and water-resistant features have made it a highly promising material for industrial applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
