Abstract
In this study, the liquid phase oxidative polymerization method was utilized to prepare polypyrrole conductive three-dimensional spacer fabrics. By controlling pyrrole solution, oxidant FeCl3 solution, dopant p-toluene sulfonic acid concentration and reaction time, the optimal process for the preparation of conductive spacer fabrics was obtained. This led to the best preparation process of polypyrrole-coated three-dimensional spacer conductive fabric (polypyrrole/three-dimensional spacer fabrics) being obtained. The results showed that the conductive properties of polypyrrole/three-dimensional spacer fabrics were the best when 0.10 mol/L pyrrole, 0.40 mol/L oxidant FeCl3 solution, and 0.40 mol/L dopant p-toluene sulfonic acid were prepared within a 2-h reaction time. The properties of polypyrrole/three-dimensional spacer fabrics were analyzed using the results from surface resistance, Fourier transform-infrared spectroscopy, mechanical properties, and stability tests. The results showed that polypyrrole was well attached to the three-dimensional spacer fabric surface, and the concentration of polypyrrole in the fabric exhibited an inverse correlation with changes in surface resistance. The mechanical properties of polypyrrole/three-dimensional spacer fabrics after treatment exhibit superior performance, with consistent changes in the meridional and zonal electrical properties during tensile testing and can maintain excellent long-term electrical stability in atmospheric environments.
Introduction
Three-dimensional spacer fabrics (3DSF) have excellent properties such as high strength, good air permeability, and good compression performance.1,2 With the development of new materials and the continuous development of technology, researchers are attempting to combine conductive materials with knitted 3DSF to give spacer fabric new functions, adopting various preparation processes to endow the spacer fabric with conductivity, making it widely applicable in fields such as flexible sensors and health care.
At present, the preparation methods of conductive 3DSF can be roughly divided into two ways, one of which is to directly weeve with conductive yarn to prepare a conductive spacer fabric, or to mix with conductive yarn. Hong et al. 3 used polyurethane yarns and conductive yarns to weave conductive fabrics with different densities, with excellent linearity, sensitivity, and repeatability under small strains. Although the conductivity is excellent, there are problems such as difficulty in spinning, poor braiding performance, hard feeling, and low wear comfort.
Another way is to treat the fabric with metal materials, carbon-based materials, and conductive polymers to give the fabric electrical conductivity. Metal materials are often combined with textiles by electrochemical polymerization4–7 or spraying; 8 conductive fabrics retain the mechanical properties of traditional polymers and the good electrical properties of metals. However, the obvious disadvantages of metal-coated fabrics are low flexibility, limited fastness to washing, and poor wear resistance. The use of carbon-based materials such as carbon nanotubes,6,9 graphene,10,11 and MXene12,13 to impart electrical conductivity to conductive fabrics has also been studied and used, but the high cost of carbon-based materials, complicated preparation processes, and poor air permeability limit their wide application. The excellent flexibility, processability, and low cost of conductive polymers have attracted extensive attention in the preparation of conductive fabrics.14,15 Common conductive polymers include polyaniline (PANI), polyacetylene (PA), polythiophene (PT), and polypyrrole (PPy). Wu et al. 16 polymerized pyrrole (Py) on nylon Lycra fabrics and used it in for wearable biomechanics such as intelligent knee pads to realize the real-time monitoring of human movement.
PPy conductive fabrics can be used in microwave shielding, 17 electromagnetic interference shielding,18,19 sensors, 20 biomedicine, and other fields.21–23 In addition, PPy-coated textiles are soft and easy to fit, and they are ideal for use as wearable biomechanical sensors that can be used for a range of applications that monitor human movement. At present, PPy is mainly combined with fibers and other two-dimensional structural fabrics to give it a certain electrical conductivity, which is mostly used in human activity detection.24,25 However, these types of textile materials usually have poor elasticity, permeability after conductive treatment, and poor recovery after use, which greatly affect the user’s experience when applied to smart textiles. The 3DSF has good permeability, elasticity, and elastic recovery, and the conductive material prepared with it as the base can be comfortable and wearable, and has broad application prospects in human activity monitoring, smart textiles, piezoelectric sensors, and so on. At present, the widely used electromagnetic shielding materials are often made from metal materials, which have disadvantages, such as having high weight and cost, and complex preparation.26–28 The 3DSF is commonly used as a buffer material, and the spacer fabric is used as a base to prepare conductive materials, which have good mechanical properties, high flexibility, and excellent compressibility, and are essential in the application scenarios such as lightweight electromagnetic shielding materials and antistatic packaging materials.
In this study, we prepared the conductive fabric by a dip-coating method, which is simple to operate and can maintain the flexibility of the fabric to the greatest extent. The performance of the prepared PPy conductive fabric was investigated through factors with Py solution, oxidant FeCl3 concentration, dopant p-toluene sulfonic acid (PTSA) concentration, and reaction time during the preparation of conductive fabric, and explored the optimal process of conductive fabric preparation. After successfully preparing PPy-coated 3D spacer conductive fabric (PPy/3DSF) with good conductive performance, the performance of the prepared conductive fabric was tested. 29 The properties of PPy/3DSF were analyzed by experience results from surface resistance, Fourier transform-infrared (FT-IR) spectroscopy, mechanical properties, and stability tests. The results showed that PPy was well attached to the 3DSF surface, and the concentration of PPy in the fabric exhibited an inverse correlation with changes in surface resistance. The mechanical properties of PPy/3DSF after treatment exhibit superior performance, with consistent changes in the meridional and zonal electrical properties during tensile testing and can maintaining excellent long-term electrical stability in atmospheric environments. It provides a reference for the application of 3DSF in human motion monitoring, smart home, piezoelectric sensor, and electromagnetic shielding.
Materials and Methods
Reagents and Materials
Reagents: Analytical grade Py was purchased from Merck; FeCl3 solution (purchased from Merck), PTSA (purchased from Tianjin Damao Chemical Reagent Factory), ethanol, sodium hydroxide, and dilute hydrochloric acid are purchased from Sinopod Chemical Reagent Factory; deionized water laboratory was self-produced.
Four kinds of 3DSF were used in the experiment; their specific parameters are shown in Table 1. The three-dimensional spacer fabrics used in the experiment were all purchased from China Changshu Meiya Ge Home Textile Co., Ltd.
List of samples and their parameters.
Preparation Process
3DSF Pretreatment
3DSF was ultrasonically cleaned in ethanol solution (95%) for 30 min to wash, dry, and preserve it. The purpose was to remove the stains and grease remaining on the fabric surface during knitting, processing, and transportation. The surface structure and internal spacer fibers of the 3DSF are made of polyester fiber; the surface of the polyester fiber is smooth with no hydrophilic groups, resulting in poor adhesion to the surface of the spacer fabric during the polymerization reaction in Py solution, greatly affecting the electrical conductivity of the fabric, with 0.75 mol/L NaOH solution in water bath at 80°C for 30 min. Under the combined action of high temperature and an alkaline environment, the molecular chains on the polyester fiber break cause grooves to appear on the fabric surface to increase the fabric surface activity and make Py more polymerized to the fabric surface. As shown in Figure 1(a), the surface morphology of the polyester fiber before and after treatment under a scanning electron microscope showed that the surface of the untreated polyester fiber was smooth without grooves. As shown in Figure 1(b), after NaOH solution treatment, grooves and microholes appeared on the surface of the polyester fabric, which indicated that the roughness of the surface of the spacer fabric increased after NaOH solution treatment.

SEM pictures of polyester fiber before and after NaOH solution treatment: (a) SEM picture of polyester fiber before treatment and (b) SEM picture of NaOH solution-treated polyester fiber. SEM: scanning electron microscopy.
Preparation of PPy/3DSF
Due to the poor electrical conductivity of the intrinsic PPy, dopants need to be added to improve the electrical conductivity during polymerization. The addition of oxidizing agents and dopants is required in Py to polymerize and be conductive. In this experiment, a chemical polymerization method was selected to prepare PPy/3DSF. Among the many oxidants in the oxidative polymerization of Py, FeCl3 provides the best electrical conductivity for PPy polymerized conductive fabrics. 30 Among many dopants, the reaction of PTSA is slow, which is conducive to the formation of a smooth membrane of PPy and can be more evenly distributed on the surface of 3DSF.31–33
The 3DSF treated with NaOH solution is immersed in Py solution, keeping the material-to-liquor ratio as 1:50, to ensure that the Py in the solution can penetrate into the interior of the fabric, and the process takes 30 min at room temperature. Then it is configured with different concentrations of oxidation dopants while preparing without adding dopants as a control. The condition range was selected based on the previous research on PPy conductive fabric by other researchers.34–36 The mixture solution of oxidizer and dopant is added to the Py solution, and the ratio of Py solution to the oxidation solution is 2:1. 37 After the addition of the oxidation, Py will undergo oxidation polymerization and deposit on the surface of the 3DSF, and different reaction times are set as a control. The prepared conductive PPy/3DSF was repeatedly washed with deionized water to remove the residue of PPy and oxidation dopant that was not completely adsorbed on the fabric surface, and then dried in the oven at 60°C for use. The preparation process of PPy/3DSF is shown in Figure 2. Table 2 shows the 3DSF after the conductive treatment of PPy, compared with the untreated 3DSF in Table 1. It can be cleary observed that there are significant changes of the fabric before and after the conductive treatment, the color of the fabric has changed, and the adhesion of PPy can be obviously observed on the surface of the fabric.
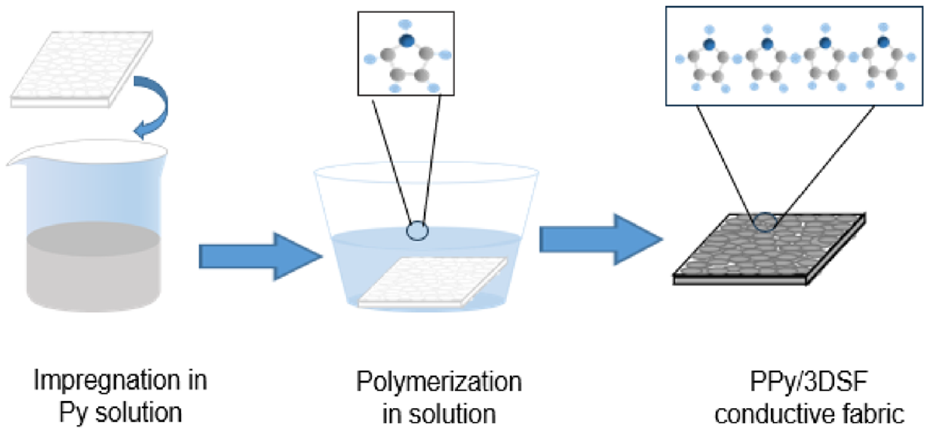
PPy/3DSF preparation process. PPy: polypyrrole; 3DSF: three-dimensional spacer fabrics.
3DSF after PPy conductive treatment.

PPy: polypyrrole.
Testing and Characterization
Surface Resistance of PPy/3DSF
Since the thickness of 3DSF is much larger than that of PPy coatings, it is inaccurate to characterize the conductivity of conductive fabrics by conductivity. 35 According to AATCC 76-2005 “Experimental Methods for Surface Resistance of Textiles,” the surface resistance of the fabric is used to characterize the conductive ability of the conductive fabric. At a relative humidity of 65% and a temperature of 24°C, When the length and width of the conductive fabric are both 1 cm, the surface resistance of the fabric is measured by a multimeter (VC86D, Xi’an Shengli Instrument Co., Ltd.), the conductivity of the PPy/3DSF is measured 10 times in different positions, and the conductivity of the PPy/3DSF is characterized by the average resistance value.
Quality Increase Rate of PPy/3DSF
After treatment, it can be clearly observed that the adhesion of PPy makes the surface of the spacer fabric turn black, and the attachment of PPy to the surface of the fabric will cause the weight of the fabric to increase. The degree of polymerization of PPy on the fabric can be characterized by the increase in the quality of the PPy/3DSF.
Scanning Electron Microscopy Analyses
Morphological analysis of PPy/3DSF was performed by scanning electron microscopy (SEM) (Carl Zeiss, MERLIN Compact, Germany). The surface and cross-section of the inner fiber of the conductive fabric were prepared by placing the sample in conductive glue, and the surface morphology was observed under the scanning electron microscope.
The FT-IR Spectroscopy of PPy/3DSF
FT-IR spectroscopy (IRAffinity-1 Shimadzu Fourier Transform-infrared Spectrophotometer, Shimadzu Manufacturing Company, Japan) was used to test PPy/3DSF and PPy powder to determine the chemical composition changes of fabrics before and after conductive treatment.
Mechanical Property Test of PPy/3DSF
According to GB/T3923.1-2013 “Tensile properties of textile fabric-Part 1: Determination of breaking strength and elongation at break (strip method),” equivalent to ISO 13934-1:2013 Textiles—Tensile properties of fabrics—Part 1: Determination of maximum force and elongation at maximum force using the strip method, Determination of maximum force and elongation at maximum force using the strip method on the electronic universal testing machine (Autograph AGS-X100KN, Shimadzu, Japan), the changes in mechanical properties of 3DSF before and after conductive treatment were explored.
Stability Test of PPy/3DSF
Refer to GB/T3921.1-1997 “Textile Color fastness test Color fastness to washing” to evaluate the washing fastness of the PPy/3DSF. To avoid possible chemical test results between PPy and the washing solution, we chose to use deionized water as the washing solution here. The PPy/3DSF was placed in an ultrasonic cleaning machine (SB-3200DTD, Ningbo Xinzhi Biotechnology Co., Ltd), and the bath ratio of the conductive fabric was controlled to be 1:50, and the temperature was 30°C, and the resistance change of the PPy/3DSF was tested after different washing times to characterize its washing resistance.
To simulate the change of electrical resistance of conductive fabric in atmospheric environment, the environmental stability test of PPy/3DSF was carried out in an atmospheric environment with a relative humidity of 65% and a temperature of 24°C, and the change of resistance value after exposure to the atmospheric environment for different time was recorded to explore its air stability.
Results and Discussion
Study on the Best Preparation Process of PPy/3DSF
The optimum preparation process of PPy/3DSF was optimized by single factor analysis. The s-1 fabric was selected as the base to explore the optimal concentration of the prepared solution and the surface resistance was used to characterize the conductive property. Figure 3 shows the effects of Py solution concentration, oxidizer concentration, dopant concentration, and reaction time on the electrical conductivity of conductive fabrics.
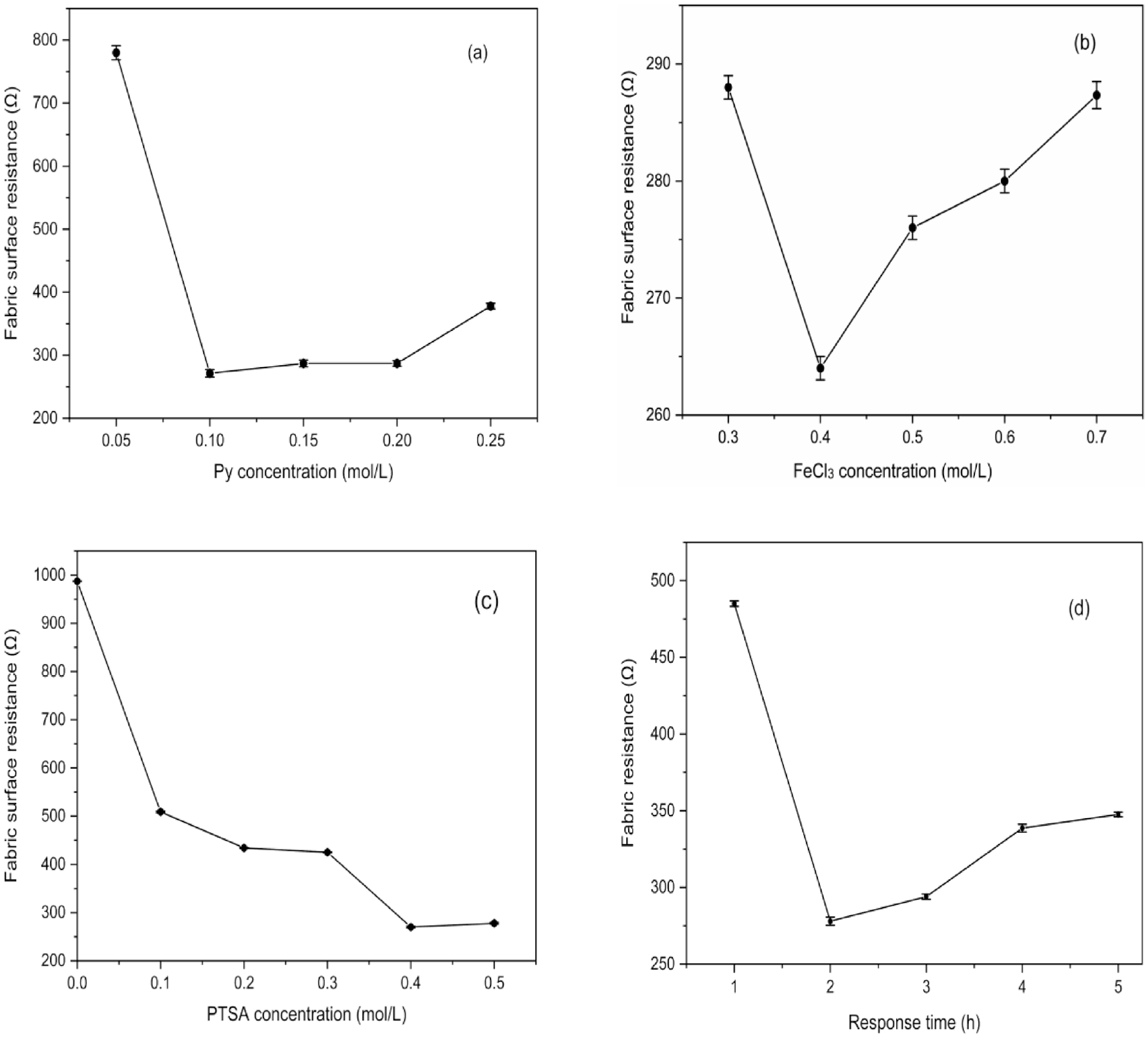
Effects of different factors on the resistance of PPy/3DSF: (a) effect of Py concentration on surface resistance of fabric; (b) effect of catalyst FeCl3 concentration on surface resistance of fabric; (c) effect of dopant PTSA concentration on surface resistance of fabric; and (d) effect of reaction time on surface resistance of fabric. PPy: polypyrrole; 3DSF: three-dimensional spacer fabrics; PTSA: p-toluene sulfonic acid.
It is shown in Figure 3(a) that when the concentration of Py is 0.10 mol/L, the surface resistance of PPy/3DSF is small and stable; when the Py concentration is less than 0.05 mol/L, the content of PPy polymerized on the fabric surface is low, and the conductive ions formed on the surface are relatively small, it is difficult to form a complete conductive path on the surface of the fabric, and the resistance of the fabric is large. When the Py solution is between 0.10 and 0.20 mol/L, the adhesion of PPy on the surface of the fabric increases, forming a complete conductive path, which significantly reduces the surface resistance of the fabric and improves the conductive property. Later, with the increase in Py concentration, when the concentration of Py is greater than 0.20 mol/L, the excess Py on the fabric surface could not be completely reacted, and the excess Py attached to the surface of PPy limited the movement of conducting ions and caused the surface resistance to increase.
In Figure 3(b), with the increase in the FeCl3 solution concentration, the surface resistance first decreases and then tends to be stable; when the concentration of oxidizer is low, Py cannot be completely oxidized; with the increase in the Py and FeCl3 solution concentration, the fabric resistance decreases, and then with the increase in the oxidizer concentration, the PPy/3DSF surface resistance increases significantly.
In the Py polymerization process, the addition of dopants can improve the disadvantages of poor conductivity of the constitutive state PPy. The addition of dopants will not only affect the conductivity of conductive fabrics, but also affect the microscopic morphology of the formation of PPy,36,38 which plays an important role in the application and improvement of physical and chemical properties of PPy. 31 In Figure 3(c), the surface resistance of the PPy/3DSF without PTSA is high, and the surface resistance of the fabric decreases significantly after PTSA is added at the beginning. When the PTSA concentration is 0.4 mol/L, the surface resistance of the fabric tends to stabilize.
Figure 3(d) shows that as the reaction time increases, the surface resistance of the PPy/3DSF first decreases and then increases. When the reaction time is short, the deposition content of Py on the fabric surface is less, the reaction with oxidation dopant is not complete, and the surface resistance of the fabric is larger. At 2 h, the resistance of the fabric is as small as 278 Ω. At this time, the polymerization degree of Py monomer is increased, and PPy forms a continuous phase on the surface of the fabric,39,40 and the electrical conductivity increases. Later, with more reaction time, other ions in the solution will be deposited on the fabric surface, causing the surface resistance of the fabric to increase.
The above experiments show that the conductivity of PPy/3DSF depended on the concentration and reaction time of corresponding solution. The optimal process for the preparation of conductive spacer fabrics was obtained when the conductivity of the 3DSF prepared using 0.10 mol/L Py, 0.40 mol/L oxidant FeCl3 solution, and 0.40 mol/L dopant PTSA within the reaction time of 2 h. PPy/3DSF with good electrical conductivity was successfully prepared.
Performance Test Result of PPy/3DSF
The optimal preparation process of PPy/3DSF was obtained according to the above research, and different types of fabrics were used as the base for conductive treatment to explore its properties.
Mass Increase Rate of PPy/3DSF
Py polymerization on the fabric surface will increase the weight of the fabric and explore the effect of increasing the quality of conductive fabric on its conductive property. The curves of mass increase and surface resistance change after Py conductive treatment are shown in Figure 4, the mass increase in fabrics with different structures after polymerization varies, and the content of PPy loaded on different types of fabrics varies, which is due to the different thickness, number, and diameter of spacer fibers of different types of fabrics, resulting in different levels of PPy deposition on different fabrics, and PPy loading rate is inversely proportional to the resistance of the fabric. When the PPy load increases, PPy/3DSF surface resistance decreases, and the conductivity of the fabric increases.
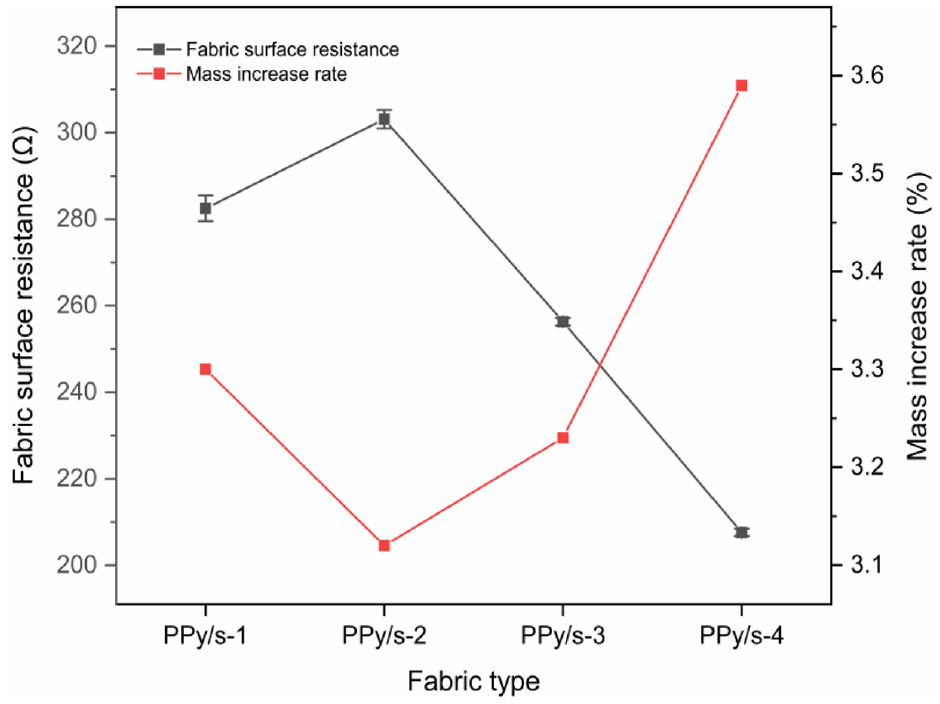
Mass increase and surface resistance change of fabric after conductive treatment of PPy. PPy: polypyrrole.
The FT-IR Spectroscopy Test Result of PPy/3DSF
Under ambient conditions, the Shimadzu spectrometer uses the KBr particle method to record FT-IR spectroscopy in the wave number range of 500–4000 cm−1. The infrared curves of different fabrics are shown in Figure 5. The infrared curves of 3DSF in 1620, 1273, and 1411 cm−1 have C=O, C-O-C, and a benzene ring in place of the stretching vibration characteristics, and are polyester fiber absorption peaks’ characteristics. In the PPy/3DSF infrared curve, the characteristic peaks of the polyester fabric are weakened or disappear to different degrees, at 3471, 1056, and 1188 cm−1, and so on. After conductive treatment, characteristic peak of 3DSF is weakened to different degrees, and it is related to the bending vibration of Py. Moreover, the wave crest of the fabric and the peak performance of PPy after conductive treatment are largely similar, which can confirm that PPy can adhere well to 3DSF.
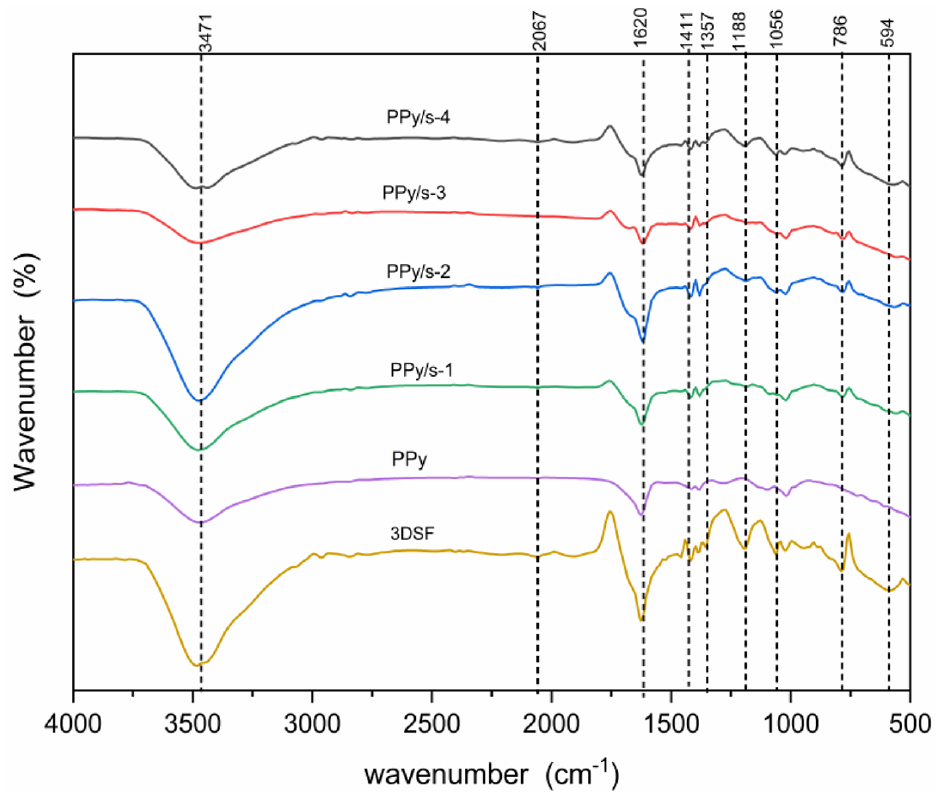
FT-IR spectra curve of PPy/3DSF. FT-IR: Fourier transform infrared; PPy: polypyrrole; 3DSF: three-dimensional spacer fabrics.
Morphology of PPy/3DSF
The morphology of the treated fabric was analyzed by scanning electron microscope, and the SEM picture obtained by the test is shown in Figure 6. As shown in Figure 6(a), the surface of the nonalkali treated fabric has less PPy adhesion after conductive treatment, mainly due to the smooth surface of the polyester fabric and the poor adhesion of PPy to it, and the conductivity of the prepared conductive fabric is also poor. After NaOH solution treatment, there are defects on the surface of polyester fabric, and there are many adhesion sites when Py polymerizes on the surface of the fabric. It can be observed from Figure 6(b) that a large number of PPy particles exist on the surface of the conductive fabric prepared after NaOH solution treatment, and the conductive fabric also has better conductive properties. The comparison of the cross-sections before and after the conductive treatment of polyester fiber in Figure 6(c) and (d) shows that the conductive treatment of polyester spacer fabric with Py is applied, the polymerization of Py mostly occurs on the surface of polyester fiber, and the existence of PPy is hardly observed in the interior of the fiber. This indicates that the polymerization of Py in the conductive fabric prepared by the optimal preparation parameters explored in this study only occurs on the surface of the fabric.

SEM pictures of conductive fabric: (a) SEM picture of conductive fabric without NaOH solution treatment; (b) SEM picture of conductive fabric with NaOH solution treatment; (c) SEM picture of NaOH solution treated polyester fiber cross-section; and (d) SEM picture of conductive treated polyester fiber cross-section. SEM: scanning electron microscopy.
Mechanical Property Test Result of PPy/3DSF
Testing the tensile properties of the samples before and after conductive treatment, the tensile elongation at break and breaking strength of the fabric in Table 3 shows that the strength and strain of the fabric after conductive treatment have increased to varying degrees. This is attributable to PPy attachment; it plays a plasticizing role on the fabric41,42 and increases the strength of conductive fabric to a certain extent.
Test results of tensile strain and strength of fabric.
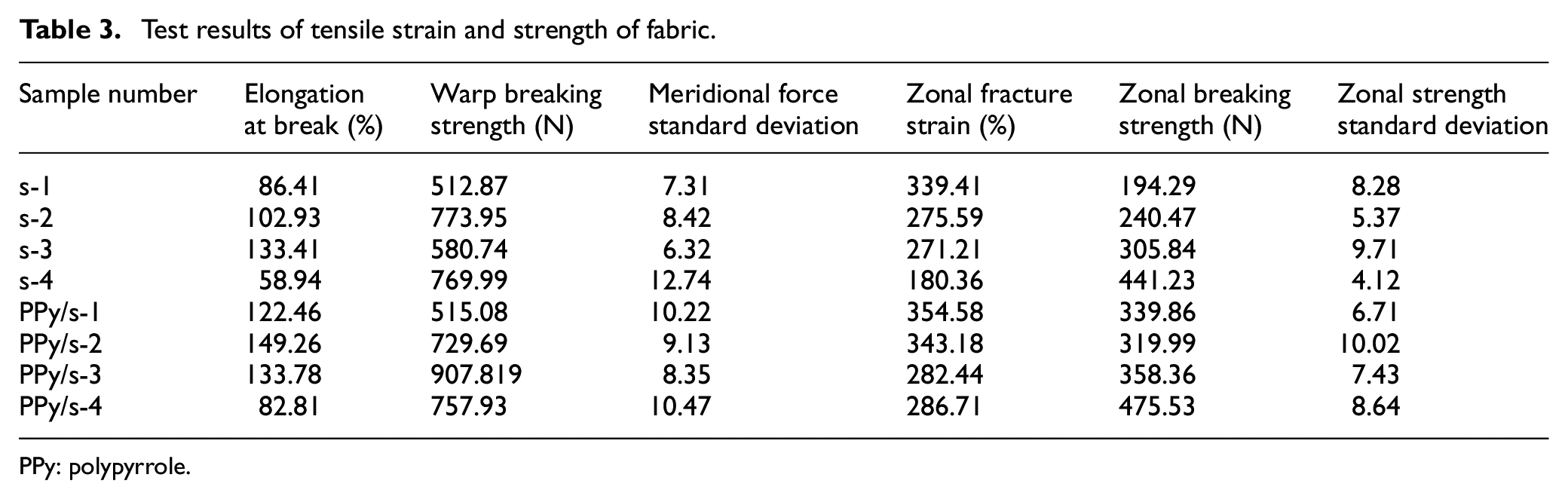
PPy: polypyrrole.
The changes in strain and the surface resistance of different conductive fabrics during stretching are shown in Figure 7. At the beginning of drawing, conductive fabric exhibits stable resistance changes. With the increase in tensile strain, the connection distance between the conductive 3DSF surface structure and the spacer wire increases, the overall conductive path of the fabric is longer, and the resistance begins to increase gradually. Until the fabric is stretched to breaking, the conductive path of the conductor of the fabric itself will be broken to different degrees, making the resistance of the fabric show unstable and irregular changes. On the contrary, when the fabric is stretched, the polymerized Py on the fabric surface falls off, resulting in an increase in surface resistance. The combined effect of two factors leads to strain instability in the electrical mechanical properties of PPy/3DSF.
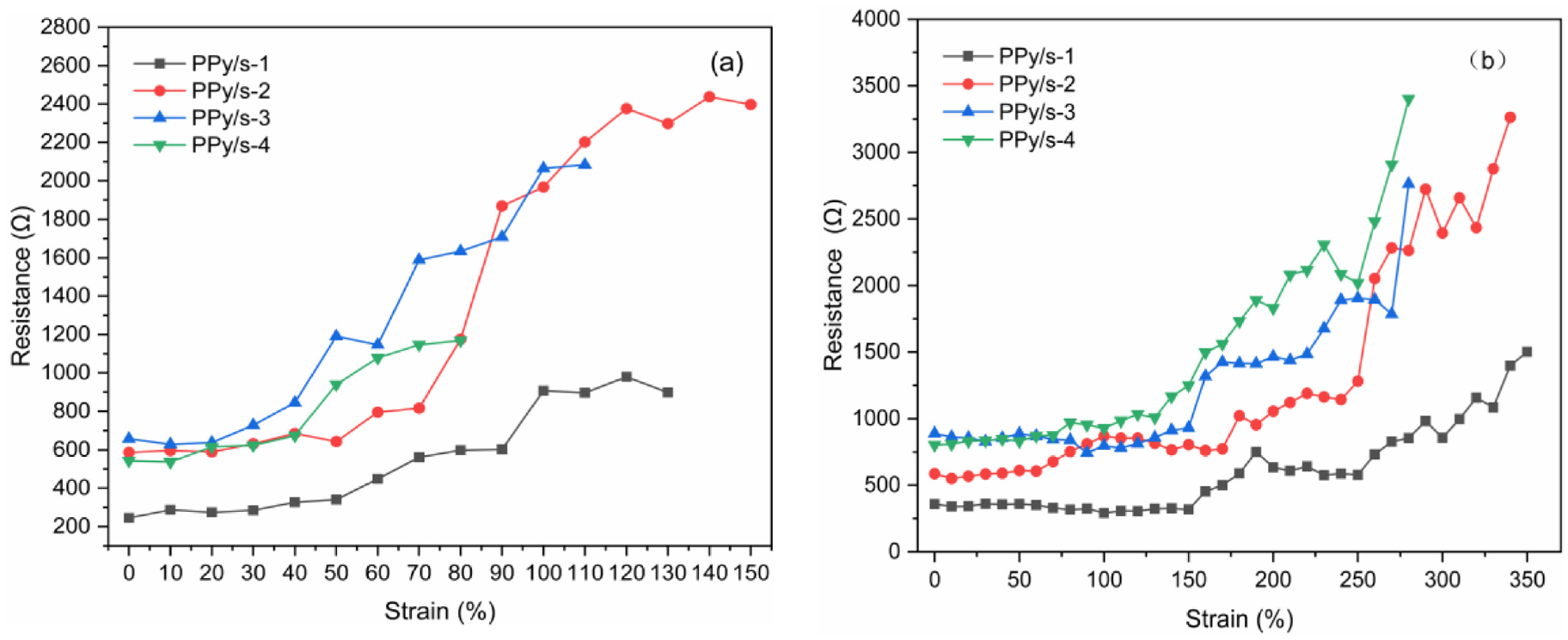
Strain-resistance diagrams of PPy/3DSF at warp and weft tensile: (a) PPy/3DSF warp drawing strain-resistance diagram and (b) PPy/3DSF weft drawing strain-resistance diagram. PPy: polypyrrole; 3DSF: three-dimensional spacer fabrics.
Stability Test Result of PPy/3DSF
The conductive stability of PPy conductive fabric is easily affected by the external environment, resulting in a decrease in conductivity, and its conductivity is easily affected by air and PPy shedding. The stability of PPy/3DSF was characterized by measuring the change of surface resistance of conductive fabric after washing in atmospheric environment and water.
The resistance of PPy conductive fabrics with different structures showed a consistent change rule after air stability test as shown in Figure 8(a). First, the resistance of the fabric exposed to the atmospheric environment increases at the beginning, because the oxygen in the air enters the inside of the PPy, and the oxidation destroys the conjugate structure on its main chain, resulting in the increase in the resistance of the fabric. When the main chain structure does not change after full contact with the air, the resistance begins to stabilize, and the surface resistance of the PPy/3DSF tends to stabilize after 20 days.
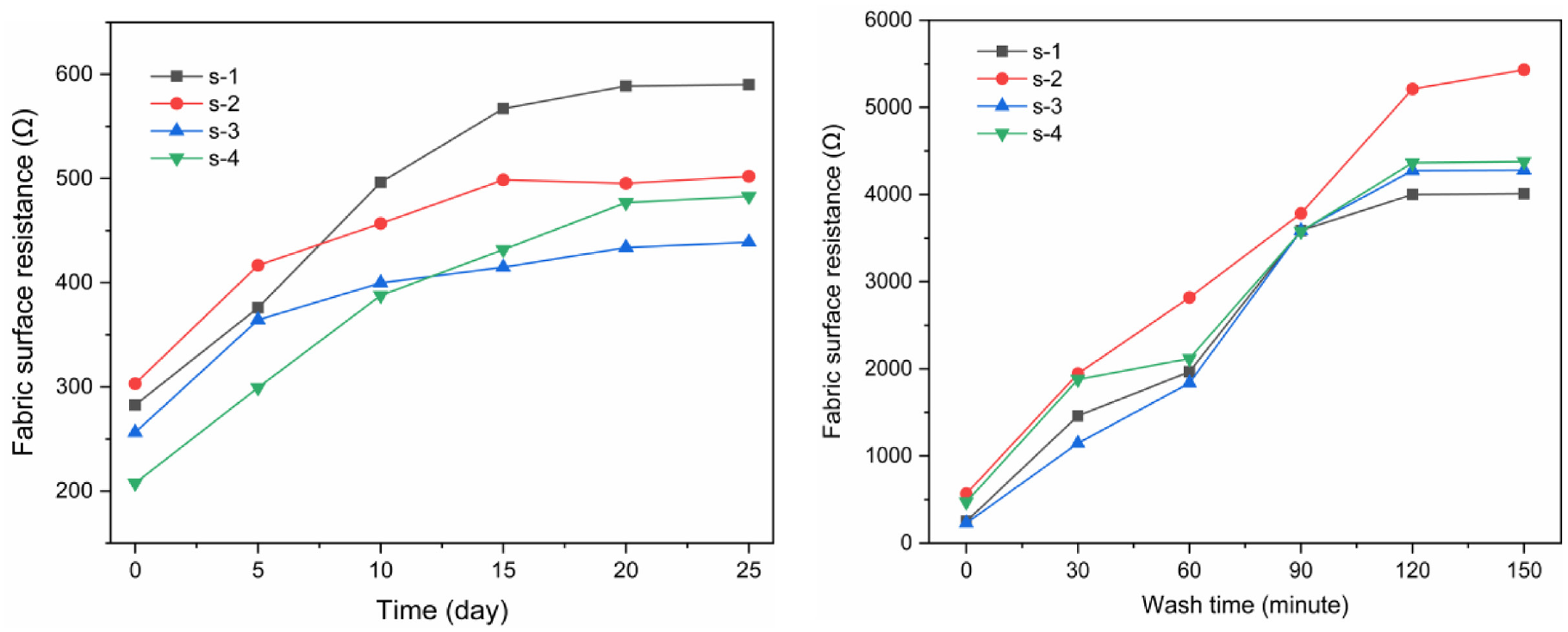
Stability of conductive fabrics: (a) air stability of conductive fabric and (b) washable stability of conductive fabrics.
The change of the resistance of the conductive fabric after washing is shown in Figure 8(b). With the increase in washing time, the surface resistance of the conductive fabric increases, and the falling PPy can be observed from the washing solution. After washing exceeds 120 min, the surface resistance tends to be stable. On the one hand, the resistance increases due to PPy shedding; on the other, PPy dedoping reaction occurs in aqueous solution, destroying the structure of the PPy itself,43,44 making the resistance change greatly compared with the unwashed fabric, and the PPy conductive spacer fabric has poor washable performance.
Conclusion
Based on the extensive application of PPy in flexible sensors, electromagnetic shielding, and other conductive materials and the good mechanical properties of 3DSF, in this study, PPy was polymerized on 3DSF to prepare flexible materials with excellent mechanical properties and good electrical conductivity. We prepared the conductive fabric by dipping coating method, which is simple to operate and can maintain the flexibility of the fabric to the greatest extent. The performance of the prepared PPy conductive fabric was investigated through factors with Py solution, oxidant FeCl3 concentration, dopant PTSA concentration, and reaction time during the preparation of conductive fabric, and we explored the optimal process of conductive fabric preparation. The results showed that the conductive properties of PPy-coated three-dimensional spacer fabric were the best when 0.10 mol/L Py, 0.40 mol/L oxidant FeCl3 solution, and 0.40 mol/L dopant PTSA were prepared within 2 h. The low concentration of polypyridine has good deposition performance on the spacer fabric and can make the fabric have good electrical conductivity. After successfully preparing PPy/3DSF with good conductive performance, the performance of the prepared conductive fabric was tested. The properties of PPy/3DSF were analyzed by the results from surface resistance, FT-IR spectroscopy, mechanical properties, and stability tests. The results show that polypyridine has good adhesion on the 3DSF surface, and the concentration of polypyridine in fabric is inversely proportional to the change of surface resistance. In a certain range, with the increase in the concentration of Py solution, the resistance of the prepared conductive fabric becomes smaller and the conductivity of the fabric becomes better. However, when the concentration of Py solution is greater than 0.20 mol/L, the relationship fails. The results showed that PPy was well attached to the 3DSF surface, and the concentration of PPy in the fabric exhibited an inverse correlation with changes in surface resistance. The mechanical properties of PPy/3DSF after treatment exhibit superior performance, the strength of the PPy/3DSF has increased to varying degrees, but its electrical conductivity has decreased due to the drop of PPy and the lengthening and destruction of the conductive path during the mechanical test, and with consistent changes in the meridional and zonal electrical properties during tensile testing.
PPy/3DSF can maintain excellent long-term electrical stability in atmospheric environments. When the conductive fabric was just placed in the atmospheric environment, the resistance of the fabric gradually increased with time. After 20 days of treatment, the change of resistance tends to be stable, so the electrical properties of the conductive fabric can maintain excellent long-term electrical stability in atmospheric environments. The washing resistance of PPy/3DSF was tested in aqueous solution. With the increase in washing time, the resistance of the conductive fabric increased significantly. When the washing time exceeded 120 min, the resistance of the conductive fabric tended to stabilize, and the electrical conductivity of the fabric decreased, so its washing performance was poor. Based on the instability of washable resistance of conductive fabrics, the washable stability should be improved in the subsequent application research to realize the application of conductive fabrics in more fields.
All in all, the PPy/3DSF prepared in this study shows good performance, which can provide a reference for the preparation, performance research, and application of PPy/3DSF in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Inner Mongolia Natural Science Foundation (grant no. 2020LH05007) and Basic scientific research business expenses of colleges and universities (grant no. JY20230089).
