Abstract
Nowadays, herbal materials are applied extensively in fibrous structures for application as a wound dressing. In this study, catechin (Cat) as the green tea extract with antibacterial characteristics has been loaded in gelatin (Gel)/poly (lactic acid) (PLA) fibrous structure by double-nozzle electrospinning technique. For this, PLA-Cat from one nozzle and Gel-Cat solution from another were injected, and fabricated Gel/PLA, Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat hybrid fibers were gathered onto a rotating collector. In order to verify the application of these scaffolds in bio applications, the morphological, chemical, wettability property, and biological features of fibers were analyzed using SEM, contact angle analysis, antibacterial, and cell attachment tests. The viscosity of spinning solutions increased with the addition of Cat to them that resulted in an increase of fibers diameter. Fourier transform infrared spectroscopy highlighted the presence of PLA, Gel, and Cat in the final structure. The results exhibited that the presence of Cat improved the antibacterial activity. Furthermore, cell attachment studies with L929 fibroblast cells demonstrated that incorporation of catchin increased the cell viability without any toxicity.
Introduction
Chronic wounds that are severely damaged are initially in need of rapid coverage with a dressing that protects them. Moreover, acute wounds have the potential to move from acute position to chronic.1,2 Today’s wound healing products are usually designed to create a suitable environment for rapid wound healing by absorbing excess exudates, providing thermal insulation, oxygen and fluid exchanges, acting as a barrier to microbes, and preventing allergic reactions, toxicity, and scaring. 3 Wound dressings are available on the market in the form of hydrogels, ointment, films, foams, and woven materials. 4 Electrospinning has been investigated as an effective technique for the production of flexible nonwoven fibers. The fibers obtained from electrospinning consist of a high surface area to volume ratio resulting in various unique physical and chemical properties favorable for many applications. 5 With the appearance of electrospinning, fibrous structures containing micro and nanofibers are constructed for wound dressing applications from either synthetic or natural polymers.6–8 Recently, using two-nozzle electrospinning technique has attracted a lot of attention in fiber production. 9
Poly (lactic acid) (PLA), as a conventional and environmentally friendly thermoplastic polymer, is a biodegradable, bioabsorbable, and renewably derived polyester extensively investigated over the last several decades.10–15 However, the weak hydrophilicity of this polymer should be overcome via various physical or chemical methods. Combining of synthetic polymers with natural polymers is a promising way to develop scaffolds with improved mechanical and biological properties. 16
As another potential biomaterial, gelatin (Gel) is a natural biodegradable, biocompatible polymer derived from collagen by controlled hydrolysis which is highly applicable in tissue engineering due to its lower cost and relatively low antigenicity compared with collagen.15,17–19
It has been well documented in the literature that herbal extract–loaded fibers help in accelerating the wound-healing process.20,21 Catechin (Cat) from the flavonoid family is obtained from the extract of gambier and leaf green tea, which is a popular beverage since ancient times. 22 This natural material with nontoxic nature shows antioxidant, anti-inflammatory, anti-tumor, and anti-bacterial characteristics.23,24 It should be noted that, Cat is not stable under conditions such as pH (alkaline or neutral states), increase of temperature and oxygen, enzymes, and light.25,26 Electrospinning method was used as an effective and single-step method for protection of Cat from undesirable conditions with minimum damaging effects. 27 Nanofibers containing poly(ɛ-caprolactone)/multi-walled carbon nanotubes composites with various values of green tea polyphenols (GTP) were successfully designed via an electrospinning method to preserve the chemical structural stability of GTP for application in cancer therapy. 28 Green tea extract (GTE) has successfully been loaded into polyvinylpyrrolidone (PVP) to form composite PVP/GTE nanofibers. The antioxidant activity of the produced PVP/GTE nanofibers increased with reducing the average fiber diameter because of the enhancement in their surface area. 29 Shao et al. 30 (2018) reported production of tea polyphenols loaded carboxymethyl cellulose and pullulan mixture structures by electrospinning for application in fruit preservation. Cat incorporated PCL nanofibers were produced, and by immersing the as-spun fibers in a water-soluble eggshell membrane (S-ESM), S-ESM was introduced into them through hydrogen bonding. Water contact angle test suggested that the presence of S-ESM/Cat improved the hydrophilicity of PCL nanofibers. 31 Nanoencapsulation of Cat in nanofibers from Azivash gum-poly(vinyl alcohol) (PVA) was done via electrospinning for application in food packaging or pharmaceutical purposes. The results exhibited that, Cat improved the thermal resistance of nanofibers because of the interaction with the polymer solution through hydrogen bonds and increased adhesion between molecular chains. 27 Fabrication of composite nanofibers composed of poly (lactic acid)/tea polyphenol (PLA/TP) with four different blend ratios of PLA/TP 5:1, 4:1, 3:1, and 2:1, by electrospinning was investigated. PLA/TP-3:1 structure with good mechanical property, exhibited the antioxidant and antimicrobial characteristic. 32 Bilayer film with internal layer consisted of Cat/gelatin with excellent biocompatibility and the external layer contained a PCL layer formed using air-jet spinning was produced for application in wound healing with preventing harmful external factors and helping wound healing with excellent antioxidant ability. Cytotoxicity and cell proliferation evaluation showed that the films did not exhibit much toxicity. 33
In the current research, our goal is to fabricate Cat incorporated hybrid composite electrospun fibers–based skin dressing for improved wound healing with antibacterial properties and proper biological properties. According to our knowledge, fabrication of Cat incorporated Gel/PLA fibers using double-nozzle electrospining technique has not been investigated before. For this, Gel from one syringe and PLA from another one were injected and fabricated fibers were collected on the rotating drum. Then Cat with antibacterial and antioxidant characteristics was loaded in PLA or Gel or both of them. The morphological, antibacterial, and wettability properties of scaffolds were assessed. Also, the cell proliferation and attachment of fibroblast cells on the produced structures were analyzed.
Experimental
Materials
Gelatin powder from bovine skin (type B) and Cat were bought from Sigma Aldrich and used without purification. PLA type D3251 produced by NatureWorks USA with Mn and Mw 29,000 and 52,000 g/mol was used. Dichloromethane (98%) and glacial acetic acid (99.99%) were also applied to dissolve PLA and Gel, respectively.
Preparation of Gel/PLA microfibers
18% (w/v) of gelatin solution (Gel) and 12% (w/v) of ploy (lactic acid) (PLA) were prepared by dissolving the polymers in acetic acid and dichloromethane, respectively. Mechanical stirring was used for 24 h to get homogenous Gel and PLA. Before the electrospinning process, the viscosity of Gel, PLA, PLA-Cat, and Gel-Cat solutions were measured by a viscometer (model: DV-II+pro, Brookfield, Massachusetts, USA) at 25°C. This test was repeated three times for each sample.
A horizontal electrospinning apparatus (Figure 1) was used (ANSTCO, Iran) to produce electrospun microfibers. For fabrication of Cat-loaded fibers, 5 wt. % of Cat was used and to improve the Cat particles dispersion in Gel or PLA, the solutions were sonicated for 10 min before being processed by electrospinning.
34
For fabrication of Gel-Cat/PLA-Cat structure, 2.5 wt. % of Cat was incorporated in PLA solution and 2.5 wt. % of it was loaded in gelatin solution. Needle-to-collector distance, feed rate, and applied voltage were set at 13 cm, 1 mL/h, and 15 kV, for PLA solution and 13 cm, 0.7 mL/h, and 15 kV for Gel solution, respectively, to reach the structure with Gel/PLA 50:50. Polymeric solutions from gelatin and PLA with or without Cat were simultaneously electrospun onto a rotating drum (250 r/min with an external diameter of 50 mm) to obtain fibrous membrane. Different compositions containing Gel/PLA, Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat were produced. A horizontal electrospinning apparatus for fiber production.
Characterization of fibers
The morphology of the electrospun fibers was analyzed by using a scanning electron microscope (SEM) (Seron technology, AIS2300 C, Korea). Fibrous mat was sputtered with a thin layer of gold and then analyzed by SEM. The average diameter of the fibers was calculated from 100 individual fibers from the SEM images for each sample using image analysis software (Image J, National Institute of Health, Bethesda, MD). Histogram of fiber diameter distribution was presented for each sample.
For analyzing the wettability property of the fabricated materials, a sessile drop method was applied in which 0.5 μL deionized water was pipetted gently on the surface of the rectangular shape (10 mm × 10 mm) fibers and images from the droplet were taken from droplets. The wettability value of samples was reported from the average of both right and left side of angles. The image of drop shape on the surface of nanofibers was captured by video equipment model CAG10 9610IL58300 made by JIKAN CAG 10 company, and the contact angles were evaluated using Image J Software. These experiments were performed at room temperature. The average of three measurements was reported as the average water contact angle.
Fourier transform infrared spectroscopy (FTIR) analysis (Thermo Nicolet, Nexus 670 made in the USA) was used to study the chemical characteristics of different structures.
Cell culture
L929 fibroblast cells from National Cell Bank of Iran (NCBI; Pasteur institute) were cultured in Dulbecco’s Modification of Eagles Medium (DMEM; Gibco, Scotland) containing 10% fetal bovine serum (FBS; Gibco, Scotland), 1% Pen-Strep (100 U/mL Penicillin and 100 μg/mL Streptomycin; Sigma–Aldrich, USA) in a humidified atmosphere of 5% CO2 at 37°C. Before the cell seeding, the electrospun scaffolds on to coverslip with a 15 mm diameter were cut into the round disc with 10 mm diameter and were disinfected through keeping in ethanol 70% for 45 min and then in PBS for 3 times and each time 15 min.
Cell attachment study
To examine the cell adhesion on the fabricated mats, L929 cells were cultured on 24 well plates (20,000 cells/well) containing sterilized Gel/PLA, Gel-Cat/PLA-Cat, Gel-Cat/PLA and Gel/PLA-Cat fibers. After the cells were cultured on the fibers for 12 h, the culture medium was removed, samples were fixed with 4% (v/v) glutaraldehyde solution in PBS at 4°C for 30 min, and finally dehydrated in a series of ethanol/distilled water solution (10% ethanol increments; each step 10 min). The morphology of the attached cells on dried electrospun scaffolds was observed by SEM (Seron technology, AIS2300 C, Korea) at an accelerating voltage of 2.00 kV. For this, the samples were coated with gold using sputter coating for the observation of cell morphology.
Cell Adhesion studies Using DAPI
The adhesion potential of L929 fibroblast cells on Cat incorporated fibers was analyzed by using DAPI staining method. For this, 10,000 cells were cultured on 24-cell plates containing fibers. For staining nuclei of cells, the cultured cells on the fibers were washed using phosphate-buffered saline (PBS) after 12 h and then were fixed with 4% formaldehyde. After permeating the membrane with 0.5% Triton X-100(0.3%) for 20 min, cells were incubated with DAPI (4′, 6-diamidino-2-phenylindole; 1:1000) for nuclear staining. The cells were imaged with a fluorescence microscope (Olympus CKX41) which was equipped with blue and UV filters. The number of cells/mm2 was reported quantitatively by using Image J software (National Institute of Mental Health, Bethesda, Maryland, USA).
Antibacterial activity
The antibacterial activity of the Gel/PLA and Gel-Cat/PLA-Cat electrospun materials with different formulation against both Gram-positive Staphylococcus aureus (S. aureus, ATCC 12,600) and Gram-negative Escherichia coli (E. coli, ATCC 9637) was reported by using disk diffusion and liquid medium microdilution techniques. In the disc diffusion method, the fibers were inserted into the nutrient agar plates containing bacteria cells and then the plates were incubated at 37°C for 24 h. Gentamycin was considered as a negative control (standard discs for both Gram positive and Gram negative bacteria) and samples without any antibacterial agent is considered as positive control. The zone of inhibition around the discs was calculated to determine the relative antibacterial effect of the scaffolds. 35 The quantitative analysis of the antibacterial characteristic was done according to the liquid medium microdilution assay. 36 For this, to produce the standardize bacteria solution, fresh colonies (3–4) from E. coli and S. aureus strains were added to nutrient broth medium and incubated for 24 h at 37°C. Then, the suspensions were diluted to prepare absorbance value of 0.8 at 625 nm. After that, the disinfected fibers (10 mg) (Gel/PLA and Gel-Cat/PLA-Cat) were added to the prepared standardize bacterial suspensions and incubated at 37°C with gentle stirring for 24 h. The absorbance value of all produced solutions was read at 625 nm using UV-vis spectrophotometer (Lambda 25).
Statistical analysis
Data from contact angle tests were reported as the mean ± standard deviation of triplicate determinations. Statistical difference was assessed by a one-way analysis of variance (ANOVA) followed by Dunett’s test. SPSS software (SPSS Inc., Chicago, IL, USA) was applied and p<0.05 shows statistically significant result.
Results and Discussion
Morphology of fibers
SEM micrographs and the diameter distribution histograms of different samples were shown in Figure 2. Reported diameter for Gel/PLA, was about 0.95 ± 0.02, 1.11 ± 0.03, 1.32 ± 0.06, and 1.76 ± 0.06 μm, respectively. With addition of the Cat to the Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat polymeric solutions, the resulting fibers were uneven and inhomogeneous compared to Gel/PLA sample. Viscosity measurements of Gel, PLA, Gel-Cat, and PLA-Cat revealed values about 398 ± 3, 438 ± 4, 449 ± 3, and 479 ± 4 cP, respectively (Figure 3). The increase of the viscosity of polymeric solutions could be due to the increase of solution concentration as well as the hydrogen bonding interactions between PLA, Gel, and Cat. This increase in viscosity resulted in production of thicker fibers. Many studies have been implied that enhancing solution viscosity, higher molecular entanglement, and increased resistance to jet elongation at the point of flight in the electrospinning method are associated with the production of large diameter fibers.27,31 SEM images and histograms of samples; A) Gel/PLA; B) Gel/PLA-Cat; C) Gel-Cat/PLA; and D) Gel-Cat/PLA-Cat. Viscosity of different polymeric solutions for application in hybrid fiber production.

Contact Angle of Samples
The hydrophilicity of samples is one of the most important factors in medical applications. An appropriate structure should simulate the morphological property of the natural ECM, provide physical support for cells and increase cell–surface interactions.
37
Hydrophobic scaffolds such as PLA or PCL are unfavorable from the point of view of tissue engineering. For medical applications, contact angle values ought to be below 100.
38
In this regard, appropriate hydrophilic materials in the structure of fibers that improves these features is thus necessary. In this work, the contact angle for Gel/PLA, Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat was about 87 ± 3, 65 ± 2°, 62 ± 3°, and 36 ± 1°, respectively (Figure 4). Cat is natural hydrophilic antioxidant material with hydroxyl groups in its structure used extensively in medicines.
39
Our results for contact angle measurement confirmed that by adding Cat in fibers formulation (Gel-Cat/PLA-Cat), contact angle decreases significantly (p ≤ 0.05) compared with Gel/PLA, Gel/PLA-Cat, and Gel-Cat/PLA samples. For Gel-Cat/PLA-Cat fibers, this angle is the lowest and this sample is entirely hydrophilic. Contact angle of different samples; A) Gel/PLA; B) Gel/PLA-Cat; C) Gel-Cat/PLA; D) Gel-Cat/PLA-Cat.
FTIR Analysis
Figure 5 demonstrates the FTIR images of Cat, Gel/PLA, and Gel-Cat/PLA-Cat samples. Characteristic peaks of Cat were observed at 3372 (O-H vibration), 1610 (C=C stretching in the aromatic rings), and 1033 (C-O absorption) cm−1 (Figure 5(a)).
40
In PLA/Gel structure (Figure 5(b)), the characteristic stretch vibration of the carbonyl group (C=O) (1751 cm−1) in the backbone of PLA, amide I (1662 cm−1), amide II (1535 cm−1), amide III (1452 cm−1), and N–H stretching of secondary amide at 3292 cm−1 in the structure of gelatin can be observed. It is observable that, Gel/PLA and Gel-Cat/PLA-Cat matched with each other but some shifts or disappearance of peaks in the region of 1080–1184 cm−1 in the spectra of Cat containing sample shows the existence of both polymers and Cat in substrate formulation. Also, the addition of Cat into fiber composition shifted the bands of 3292 and 1751 cm− 1 to the lower wavenumber values which could be attributed OH, NH, and C–O vibration indicating the formation of hydrogen bonds between polymeric fibers and Cat.31,41 FTIR analysis of samples; A) catechin,B) Gel/PLA, and Gel-Cat/PLA-Cat.
Cell attachment study
The morphology of the attached cells on dried Gel/PLA, Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat electrospun scaffolds was observed by SEM (Figure 6). It can be observed that the fibroblast cells spread along the surface of fibers. The spreading of cells was observed in different directions because of the orientation of fibers in diverse directions. Gel-Cat/PLA-Cat fibers demonstrated higher amount of cell growth (Figure 6) and L929 cells completely covered the surface of this substrate. Improved hydrophilicity because of the presence of Cat with OH groups in Gel-Cat/PLA-Cat structure can justify the same behavior. Webb et al
42
studied the highest level of NIH 3T3 fibroblasts cell attachment at hydrophilic surfaces, and the best results were observed for surfaces with contact angles in the range of 20–60°. SEM images of L929 cells: A) Gel/PLA; B) Gel-Cat/PLA; C) Gel/PLA-Cat; and D) Gel-Cat/PLA-Cat.
Fluorescence staining
Cell adhesion behavior of Gel/PLA, Gel/PLA-Cat, Gel-Cat/PLA, and Gel-Cat/PLA-Cat substrates was evaluated by DAPI staining (Figure 7). Fluorescent microscopy images of stained nucleus showed increased cell adhesion in Gel-Cat/PLA-Cat structure, so that highest cell attachment was occurred on this structure. The cell number obtained from the observed graphs for the Gel/PLA, Gel-Cat/PLA, Gel/PLA-Cat, and Gel-Cat/PLA-Cat samples was 300 ± 40, 358 ± 46, 783± 89, and 960 ± 109 cells/mm,
2
respectively. These results confirm our hypothesis that the incorporation of catechin in to the microfibrous scaffolds structures improves their cell adhesion. Fluorescent microscopy images of cell nucleus stained with DAPI after 12 h of culture on: A) Gel/PLA; B) Gel-Cat/PLA; C) Gel/PLA-Cat; D) Gel-Cat/PLA-Cat.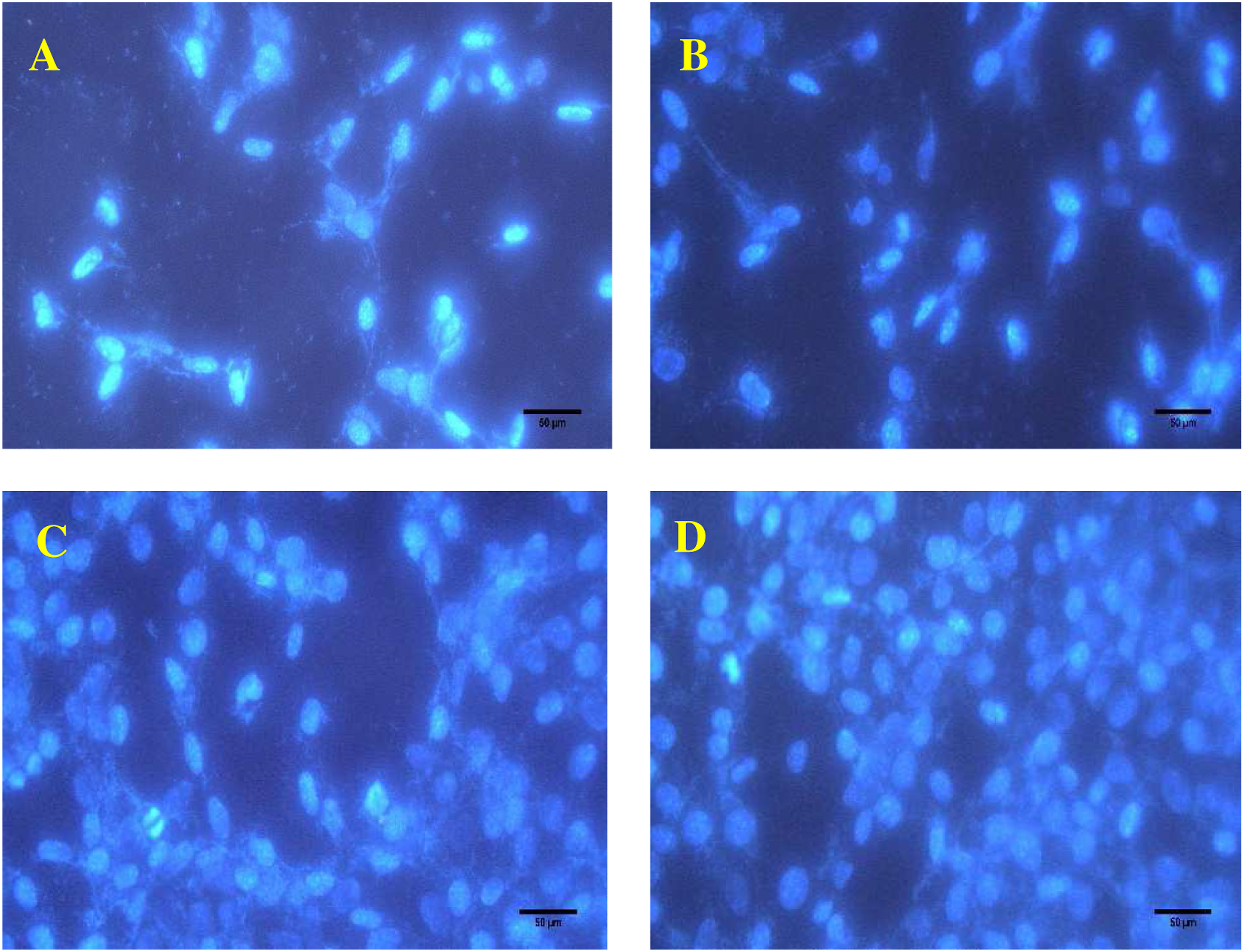
Antibacterial property
The most important advantages of the fabricated wound dressings are remarkable antibacterial characteristics as well as significant and rapid wound-closure capability.
43
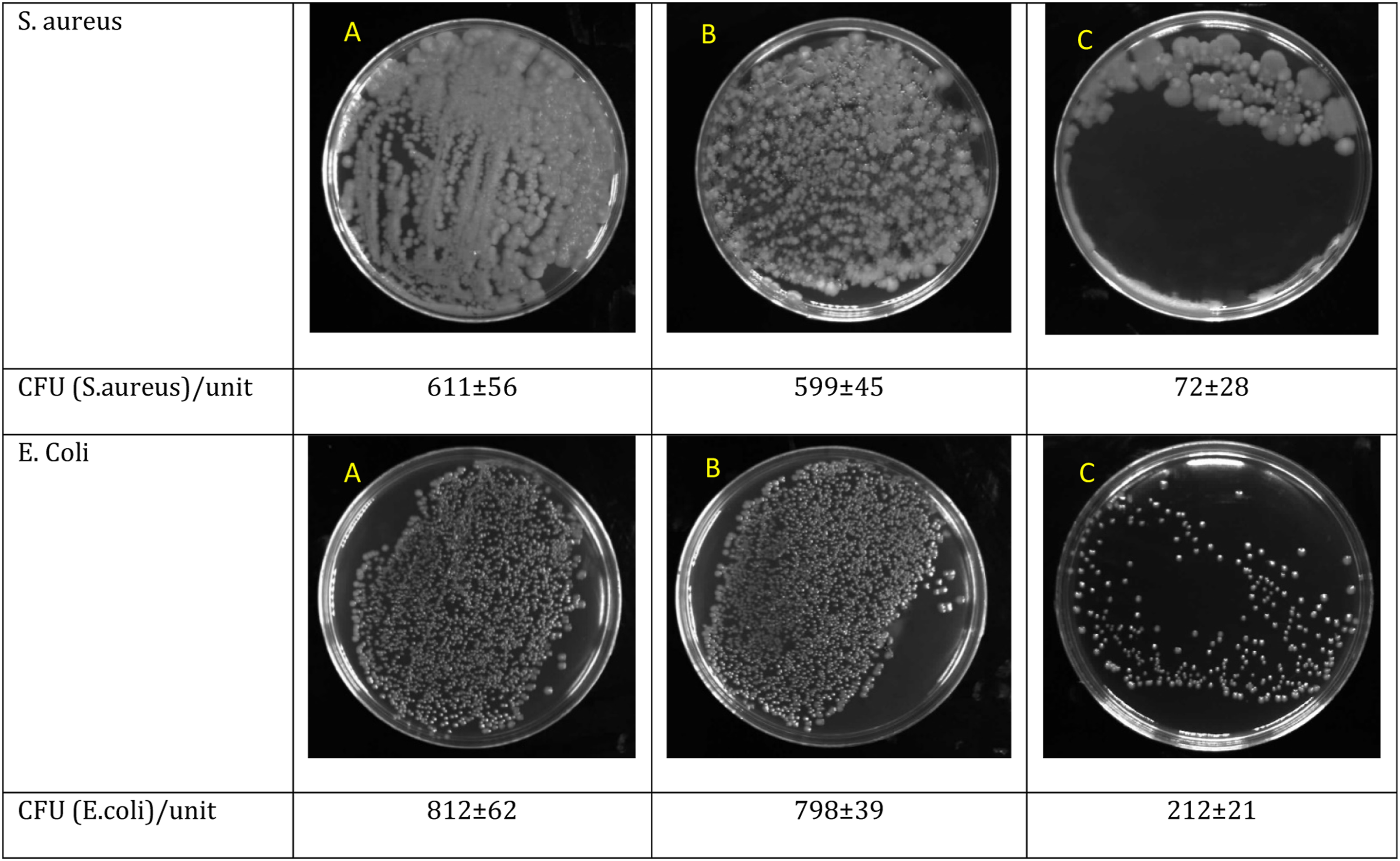
The antibacterial properties of the Gel/PLA and Gel-Cat/PLA-Cat samples toward Gram-positive Staphylococcus aureus (S. aureus, ATCC 12,600) and Gram-negative Escherichia coli (E. coli, ATCC 9637) bacteria were evaluated. Figure 8 demonstrates the clear inhibition zone around the Gel-Cat/PLA-Cat fibers against E. coli and S. aureus after 24 h into the agar plate at 37oC. Gel/PLA hybrid fibers exhibited no antibacterial efficiency against all bacterial strains used in this study. After the addition of Cat to PLA and Gel (Gel-Cat/PLA-Cat), the size of inhibition zones for E. coli and S. aureus bacteria reaches 1.34 ± 0.1 mm and 1.88 ± 0.1 mm, respectively. Figure 9 illustrates that the number of viable bacteria (CFU/mL) for Gel-Cat/PLA-Cat mat was lower than values related to Gel/PLA sample against bacterial strains. The better antibacterial activity of Gel-Cat/PLA-Cat against Gram-positive bacteria (e.g., S. aureus) than Gram-negative bacteria (e.g., E. coli) was also observable. The relatively lower antibacterial property of Cat against Gram-negative bacteria is due to the protection of this bacteria by the outer membrane and lipopolysaccharides.
44
Cat is a flavonoid (flavan-3-ol) found, for example, in tea, demonstrates antibacterial activity. The antimicrobial characteristics of tea have been reported for over a century and its bacteriostatic and bactericidal effects have been analyzed.
39
The bactericidal activity of Cat is because of its hydrogen peroxide generation.
45
Presence of polyphenolic Cat damages the bacterial cell wall and membrane of bacterial cells and causes lethal damage to bacterial cells by the production of hydrogen peroxide.
41
Gopal et al.
46
demonstrated the effectiveness of Cat in inhibiting bacteria. The antibacterial activity evaluation of control, Gel/PLA and Gel-Cat/PLA-Cat against; A) S. aureus; B) E. coli by disk diffusion method. The antibacterial activity evaluation of different samples against S. aureus and E. coli: A) Control (without fiber); B) Gel/PLA; C) Gel-Cat/PLA-Cat.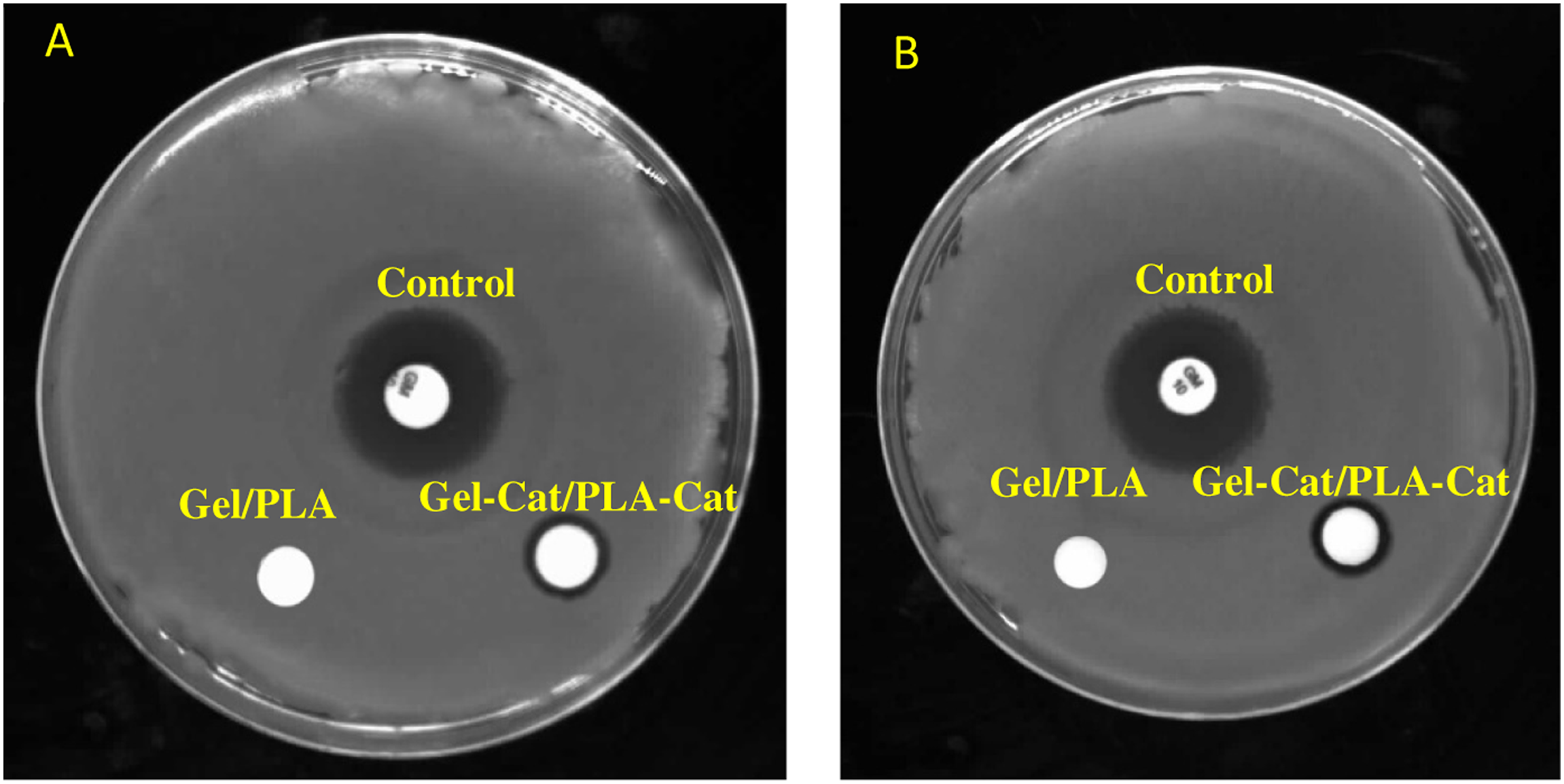
Conclusion
Cat has some limitations because of its instability under processing or storage conditions that can be protected by encapsulation. This paper conducted in the field of encapsulation of Cat via electrospinning process to protect this compound. In this study, electrospun Gel/PLA fibers containing herbal Cat were produced by double nozzle electrospinning method. Cat increased the viscosity of polymeric solutions and the diameter of fibers. Cat decreased the contact angle of samples and for Gel-Cat/PLA-Cat, its value was about 36±1°. The antibacterial properties of the samples toward S. aureus and E. coli bacteria showed that presence of Cat increased inhibition zone diameter and for Gel-Cat/PLA-Cat, this diameter was about 1.34 ± 0.1 and 1.88 ± 0.1 mm for S. aureus and E. coli, respectively. Cell attachment study by L929 fibroblast showed more cell adherence and attachment onto the Gel-Cat/PLA-Cat fibers compared to other structures. The Gel-Cat/PLA-Cat fibers with hydrophilic nature, higher cell attachment and antibacterial properties could have potential application as wound dressing in medical field.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by University of Bonab (9816).
