Abstract
Poly(lactic acid) (PLA) and wood flour/PLA composites were prepared and blended with two antimicrobial agents, triclosan and silver-substituted zeolite (Zeomic), using a twin-screw extruder. The mechanical and thermal properties, antimicrobial activity, and biodegradation performance were investigated. The addition of wood and Zeomic was found to increase the Young’s modulus of the composites, whereas the tensile strength, elongation at break, and impact strength dropped. However, the mechanical properties of PLA and wood/PLA loaded with triclosan did not show any definite trends. Differential scanning calorimetry data indicated that the glass transition temperature value of neat PLA was 63°C, whereas those of wood/PLA composites were lower. When wood and Zeomic were incorporated, PLA exhibited double melting peaks. Triclosan (1.0 and 1.5 wt%) demonstrated antibacterial activity against Staphylococcus aureus, as determined by plate count agar technique, whereas Zeomic did not. Biodegradation tests of neat PLA and wood/PLA composites showed that after a 60-day incubation period, the biodegradation rate of wood/PLA was higher than that of PLA. PLA and wood/PLA-containing Zeomic were found to degrade more quickly, suggesting that wood and Zeomic acted as biodegradation promoters. On the other hand, triclosan could be considered a biodegradation retarder since no biodegradation was observed for any triclosan-loaded samples during the initial 20 days of incubation, while neat PLA and wood/PLA composites began to degrade within the first few days.
Keywords
Introduction
Because of health and environmental concerns, many researchers have focused on the modification of antibacterial packaging made from different materials. Various thermoplastics have been used for these applications, for example, poly(vinyl chloride), polyethylene, polypropylene, polyurethane, and polystyrene. 1,2 To produce active packaging, many techniques have been employed, such as melt mixing of antimicrobial agents with polymers, applying a coating on polymer surfaces, and immobilizing polymers by covalent bond linkages. 3 After their service life, however, these types of nonbiodegradable packaging become plastic garbage, which ultimately is colonized by microorganisms. Thus, the use of a biodegradable polymer such as poly(lactic acid) (PLA) has received much attention. Since PLA has sufficient strength, high modulus, and the ability to be fabricated, it has become one of the most popular biodegradable polymers for packaging applications. 4
Triclosan and Zeomic are known as broad-spectrum antibacterial agents that have been widely used in many applications, mainly food packaging. 5,6 Triclosan was demonstrated to retard the growth rate of Escherichia coli by up to 83.40% when incorporated with pure PLA, and the reduction percentage increased to 96.78% when 10% wood was added. 7 Zeomic/PLA composite film was also found to retard the growth rates of Staphylococcus aureus and E. coli. 6 These findings indicate that triclosan and Zeomic are suitable options for producing antibacterial biomaterial packaging.
Wood flour, a biodegradable reinforcing material, is one of the most widely used materials for producing wood–polymer composites because it possesses several advantages, for example, high modulus, low density, reduced material use, and low cost. 8 Wood/PLA composites are of great interest because wood and PLA are not only environmentally friendly materials but also expected to degrade completely after disposal at the end of their service lives. However, the incompatibility between wood and PLA is a major problem that must be overcome, usually by means of chemical modification. 9,10
Many techniques are employed to study the biological degradation behavior of “green” biomaterials, including visual observation, weight loss measurement, and detection of changes in mechanical properties, carbon dioxide (CO2) evolution, and clear zone formation. 11 Among these, CO2 evolution is generally preferred because the results of the test are usually more accurate. Using this technique, PLA powders with sizes of 60 and 120 mesh were found to be degraded by up to 91% after 35 days at 58°C in controlled compost. 12 However, the degree of biodegradability of PLA loaded with antimicrobial agents remains unclear. In this work, it was assumed that PLA could lose some of its natural biodegradability after blending with antibacterial agents because the agents might also kill microorganisms that play a key role in the biodegradation process.
Therefore, this study primarily focused on the effects of the incorporation of two different antibacterial agents on the antibacterial properties and biodegradability of PLA and wood/PLA composites. Triclosan and Zeomic were selected as representative organic and inorganic antibacterial agents, respectively. Antibacterial activity against S. aureus was evaluated by plate count agar (PCA) method. CO2 evolution technique was used to determine the biodegradation performance of PLA and wood/PLA composites. Mechanical properties of PLA and wood/PLA composites were measured by a universal testing machine and an impact tester. Thermal properties were studied using differential scanning calorimetry (DSC) technique.
Experimental
Materials and chemicals
PLA (5–7 g/10 min of melt flow index, 210°C/2.16 kg; PLA 2002D; NatureWorks LLC, Minnetonka, Minnesota, USA) was used as a biodegradable polymer matrix. To produce wood/PLA composite samples, wood flour (V.P. Wood Co. Ltd, Bangkok, Thailand), having an average particle size of 100–300 μm, was incorporated into PLA at two different concentrations (5 and 10 wt%) using a twin-screw compounder. All wood particles were chemically surface treated with a coupling agent, N-(2-aminoethyl)-3-aminopropyltrimethoxysilane (KBM 603; Shin-Etsu Chemical Co. Ltd, Tokyo, Japan).
Two different antibacterial agents were incorporated into PLA and wood/PLA composites at loadings of 0–1.5% by weight: 2,4,4′-trichloro-2′-hydroxydiphenyl ether, or triclosan (United States Pharmacopeia grade; Koventure Co. Ltd, Samut Prakan, Thailand) and silver (Ag)-substituted zeolite, or Zeomic (Yamamoto Trading Co. Ltd, Samut Prakan, Thailand), having a structural formula of MX2/nO·Al2O3·YSiO2·ZH2O—where M is Ag, sodium (Na), or other ions, and X, Y, and Z are the ratio of components, in moles—organic and inorganic biocides, respectively.
S. aureus (ATCC 25923) was used as a testing bacterium for antibacterial evaluation.
Specimen preparation
KBM 603 silane coupling agent (1%) was used for chemical surface treatment of wood flour by blending in a high-speed mixer. The treated wood flour was left in an oven at 80°C for 3 days to remove all moisture content. Before the high-speed mixing process with triclosan or Zeomic, the treated wood/PLA was dried in an oven at 70°C overnight. The list of all composite formulations used is given in Table 1. All used materials and antibacterial agents were melt blended at the same time using a twin-screw extruder (HAAKE™ Rheomex CTW 100P; Thermo Fisher Scientific, Waltham, Massachusetts, USA), having temperature profiles from feed to die zone of 170, 180, 180, 180, and 170°C, respectively, with 50 r min−1 screw rotating speed. The extrudate was pelletized and then dried in an oven at 70°C before compression molded to give specimen films. The compression temperature and pressure used for 1- and 3-mm specimens were 160°C and 150 kg cm−2, respectively, under 5-min preheating and 3-min holding times before cooling down to room temperature with 10-min cooling time. Mold spacers were used to produce composite specimens with different thicknesses (1- and 3-mm specimens). The specimens with 1 mm thickness were cut into rectangular pieces (2.5 × 5.0 cm2) for biodegradation and antibacterial testing. Three-millimeter-thick specimens were made for tensile and impact strength testing.
Poly(lactic acid) and W/poly(lactic acid) composite formulations.a
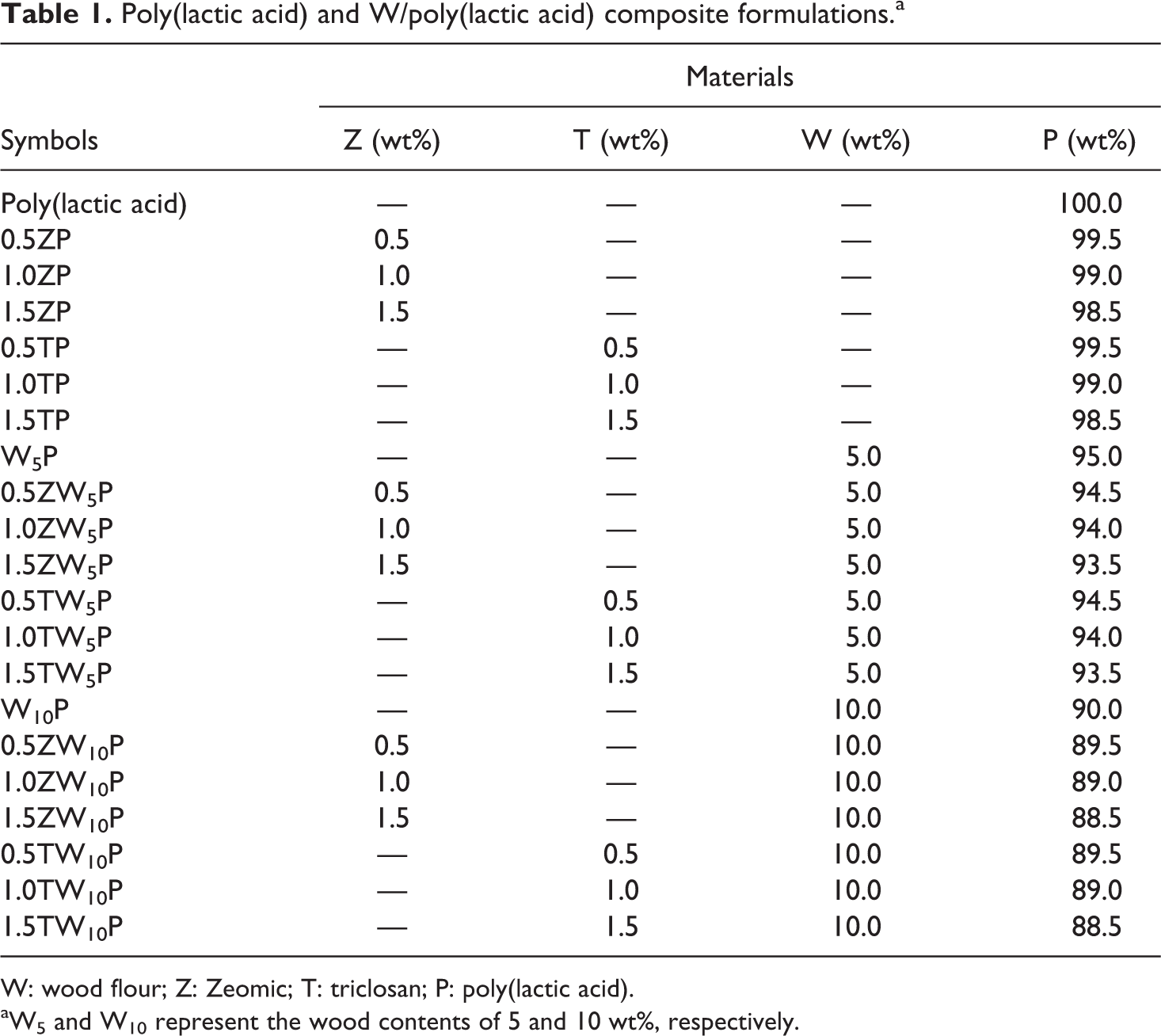
W: wood flour; Z: Zeomic; T: triclosan; P: poly(lactic acid).
aW5 and W10 represent the wood contents of 5 and 10 wt%, respectively.
Mechanical properties
Tensile properties were evaluated using a tensile tester (L Series 50K; Lloyd Instruments, West Sussex, UK), in accordance with ASTM D638 (2008) standard test method, with a crosshead speed of 0.2 in min−1. An impact tester was used to measure the notched Izod impact strength, following ASTM D625 (2006) standard. All mechanical results reported were averaged data from nine independent determinations.
Antibacterial activity evaluation
Antibacterial evaluation of PLA, 5% wood/PLA, and 10% wood/PLA specimens, blended with triclosan or Zeomic antibacterial agents at contents of 0–1.5% by weight, was performed by PCA method following ASTM E2149 (2001) standard. An inoculum of S. aureus was incubated at 37°C overnight before measuring the optical density (OD) values by ultraviolet–visible spectroscopy (DR/4000; Hach, Loveland, Colorado, USA). Peptone solution (prepared with 1 g L−1 peptone in deionized water) was used to dilute each inoculum to OD 0.1. Two pieces of 2.5 × 5 cm2 film specimens (1 mm thickness) were placed in individual flasks filled with 50 mL peptone solution with OD 0.1. A reciprocal shaker was used to shake the flasks at a speed of 100–120 r min−1 at 37°C ± 0.5°C for 0, 60, 120, 180, and 240 min of contact time. In order to find a suitable range of colony-forming units (CFU) of S. aureus, that is, 30 to 300 colonies, 13 a 10-fold serial dilution was applied. For each contact time, 100 μL of bacteria solution was placed on agar in sterilized petri dishes. The inoculated plates were then incubated for 24 h at 37°C. The number of living cell bacterial colonies was counted to determine the antibacterial performance. The percentage reduction of bacterial CFU was calculated using equation (1):

where R is the bacterial reduction (%); A is the number of CFU occurring in specimens without an antibacterial agent, for each contact time (CFU/ml); and B is the number of CFU occurring in specimens incorporating an antibacterial agent, for each contact time (CFU mL−1).
Biodegradation test
Before the biodegradation test, the compost was fermented with fresh leaves for 2 months to enrich the microbial population. The properties of the fermented compost are given in Table 2. Figure 1 shows a diagram of the biodegradation test arrangement, which was modified from the ASTM D5338-11 (2003) standard test method. One-millimeter-thick samples of PLA and PLA-based composites were cut into four rectangular pieces (2.5 × 5.0 cm2) and placed inside a reactor bottle filled with 400 g of compost. Compost containing no specimens served as a blank, which was used to determine the CO2 generated by bacterial activities. The test was controlled at 58°C ± 2 in a water bath for 60 days. Air flow rate of 50 mL min−1 was fed into the system by an air pump (Figure 1, No. 1) during the test to ensure that aerobic conditions were maintained. Barium hydroxide (Ba(OH)2) solution used in the test was 250 mL of 0.0625 M. Silica gel columns (No. 2) and the first Ba(OH)2 solution unit (No. 3) were used to remove moisture and CO2 from the feed air, respectively. The CO2 evolved from the reactor (No. 4) was trapped in Ba(OH)2 solution at the second and third Ba(OH)2 solution units (No. 5). The CO2 reacted with the Ba(OH)2 and precipitated as barium carbonate (BaCO3). The amount of CO2 was measured every 2 days by titration with 0.05 M hydrogen chloride (HCl), using a few drops of phenolphthalein as an indicator to determine the Ba(OH)2 remaining in the solution. The CO2 trapping reactions by the titration of Ba(OH)2 and HCl are shown in equations (2) and (3):
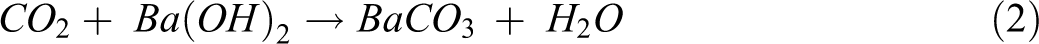
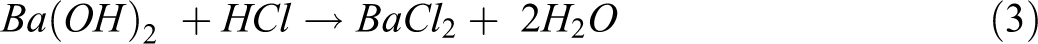
Compost compositions.

C: Carbon; N: nitrogen.
aTotal solids gained by catching a known volume of compost dried at 105°C for 24 h.
bTotal solids gained by catching a known volume of compost burnt at 550°C for 2 h.
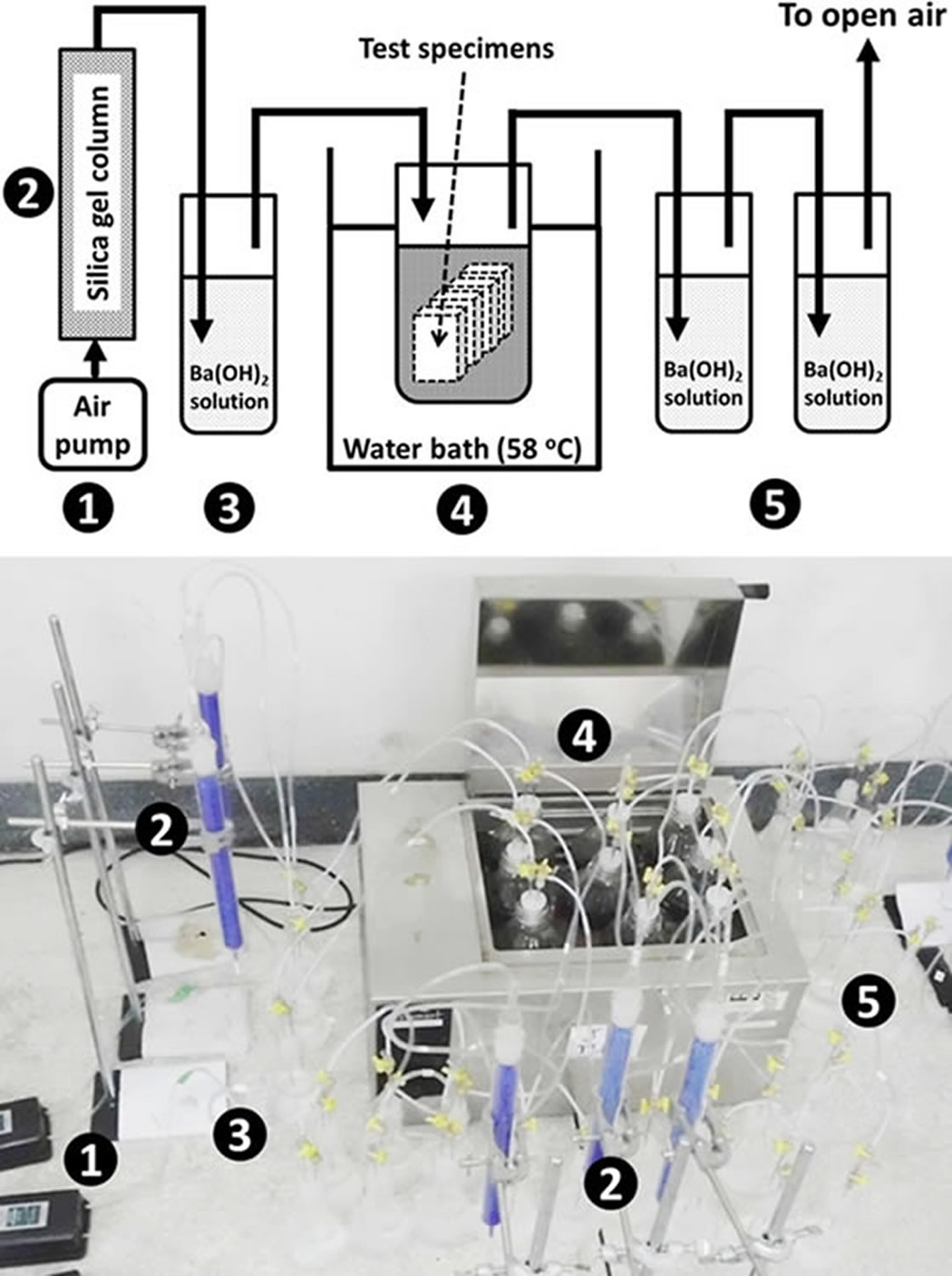
Arrangement of the biodegradation test for PLA and wood/PLA composites (modified from ASTM D5338-1) standard. PLA: poly(lactic acid).
The percentage of biodegradation (Dt) was calculated using equation (4):
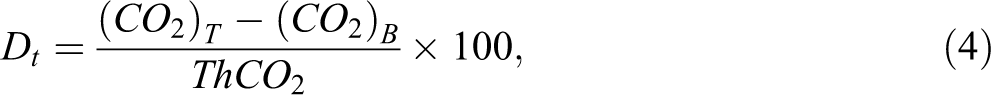
where (CO2)T is the amount of CO2 trapped in the second and third Ba(OH)2 solution flasks occurred from the reactor bottle with test specimens (mg);
(CO2)B is the amount of CO2 trapped in the second and third Ba(OH)2 solution flasks occurred from the reactor bottle without test specimens (mg); and
ThCO2 is the theoretical amount of CO2 from a specific specimen (mg).
Thermal properties characterization
The glass transition temperature (Tg) and crystalline melting temperature (Tm) were determined by DSC (DSC822; Mettler-Toledo, Columbus, Ohio, USA), and the degree of crystallinity (Xc) was determined using equation (5). Each specimen was heated from room temperature to 200°C, cooled down to 30°C, and then reheated to 200°C. Heating and cooling rates used were 10°C min−1 under nitrogen atmosphere.

where ΔHm is the sample enthalpy (J g−1);
w is PLA weight fraction in composites.
Results and discussion
Mechanical properties and thermal analysis
Mechanical properties of PLA and wood/PLA composites (5% and 10% wood contents) with different types and contents of antibacterial agents are given in Figure 2(a) to (d). It was observed that, compared with neat PLA, the Young’s modulus tended to increase with the presence of wood and Zeomic (Figure 2(a)). This was due to the greater rigidity of wood and zeolite particles (in Zeomic) in comparison with neat PLA. Low-rigidity thermoplastic was usually found for this effect. 14 –16 The tensile strength, elongation at break, and impact strength (Figure 2(b) to (d)) of PLA decreased with increasing wood content. The reduction in these mechanical properties was due to the incompatibility of the wood and PLA. 17 –19 Similar to the effect of wood, the mechanical properties of the specimens declined with the addition of Zeomic, especially in the case of PLA with 10% wood content. For example, the tensile strength of PLA, 5% wood/PLA (W5P), and 10% wood/PLA (W10P) decreased from 49.48, 48.16, and 41.77 MPa to 48.61, 43.8, and 11.01 MPa, respectively, when 1.5% Zeomic was loaded. This was likely caused by the degradation of the PLA matrix as a result of a hydrolysis reaction between PLA and water molecules in the zeolite structure and also because water molecules were absorbed by Si–OH bonding of zeolite and the hydroxyl groups in the wood. This was reported by Kaali et al. 20 who revealed that Ag+-loaded zeolite could initiate a hydrolysis reaction for both polyester polyurethane and silicone rubber because the Si–OH bonding, as a function of zeolite, induced the adsorption of water molecules. The effect of triclosan on the mechanical properties of PLA and wood/PLA did not show a clear trend. This could be attributed to the fact that the triclosan did not involve in the hydrolysis reaction. Another possible reason is that the dosage of triclosan used was relatively small.
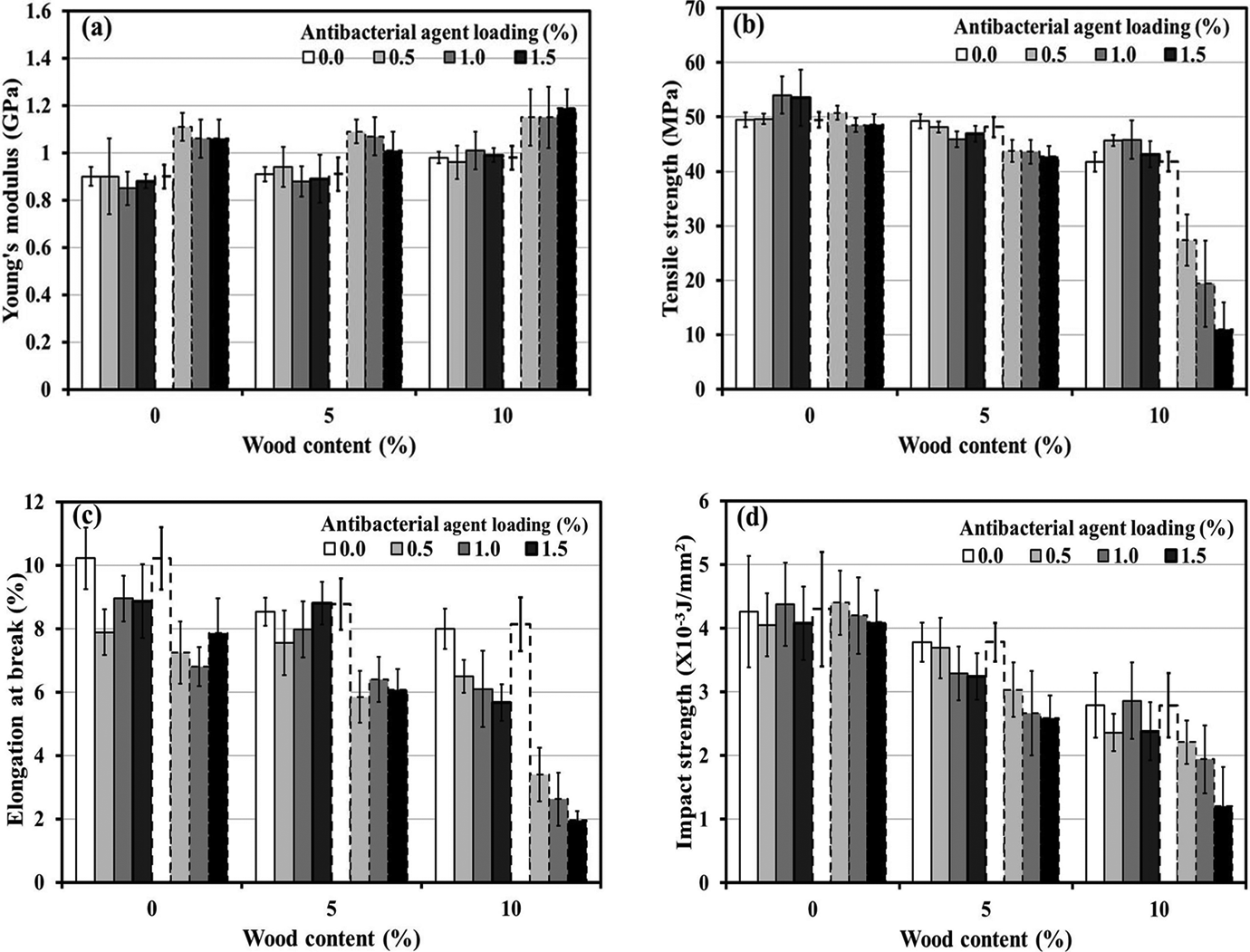
Mechanical properties of PLA and wood/PLA composites with different wood contents and containing different types and amounts of antibacterial agents: (a) Young’s modulus, (b) tensile strength, (c) elongation at break, and (d) impact strength (solid line: triclosan system; dashed line: Zeomic system). PLA: poly(lactic acid).
The DSC patterns for PLA, W5P, and W10P with and without triclosan or Zeomic loadings are given in Figure 3, showing the Tg, Tm, and Xc. The Tg of neat PLA was 63°C, whereas the Tg values of all other PLA composites were lower. The reduction in the Tg value is thought to be connected with the hydrolysis reaction initiated by the additives used (especially in the case of wood/PLA systems), which causes the PLA to have a lower molecular weight. A reduction in the Tg value of the PLA matrix with the presence of fibers was also observed by Mohamed et al. 21 With the incorporation of wood and Zeomic, the PLA composites displayed double melting peaks. It is possible that wood and Zeomic may perform as rigid foreign particles that create crystalline regions. The first melting peak could be attributed to PLA crystals, whereas the second could be associated with imperfect PLA crystals occurring as a result of the addition of wood and Zeomic, these being referred to as cold crystallizations. 22 The occurrence of double melting peak behavior of PLA composites was also found by Shi et al. 22 These double melting peaks indicate that Zeomic was responsible for raising PLA crystallinity levels—from 7.9% for neat PLA to 31.9%, 34.4%, and 36.3%, when 1.5 wt% Zeomic was loaded into PLA (1.5ZP), 5% wood/PLA (1.5ZW5P), and 10% wood/PLA (1.5ZW10P), respectively.
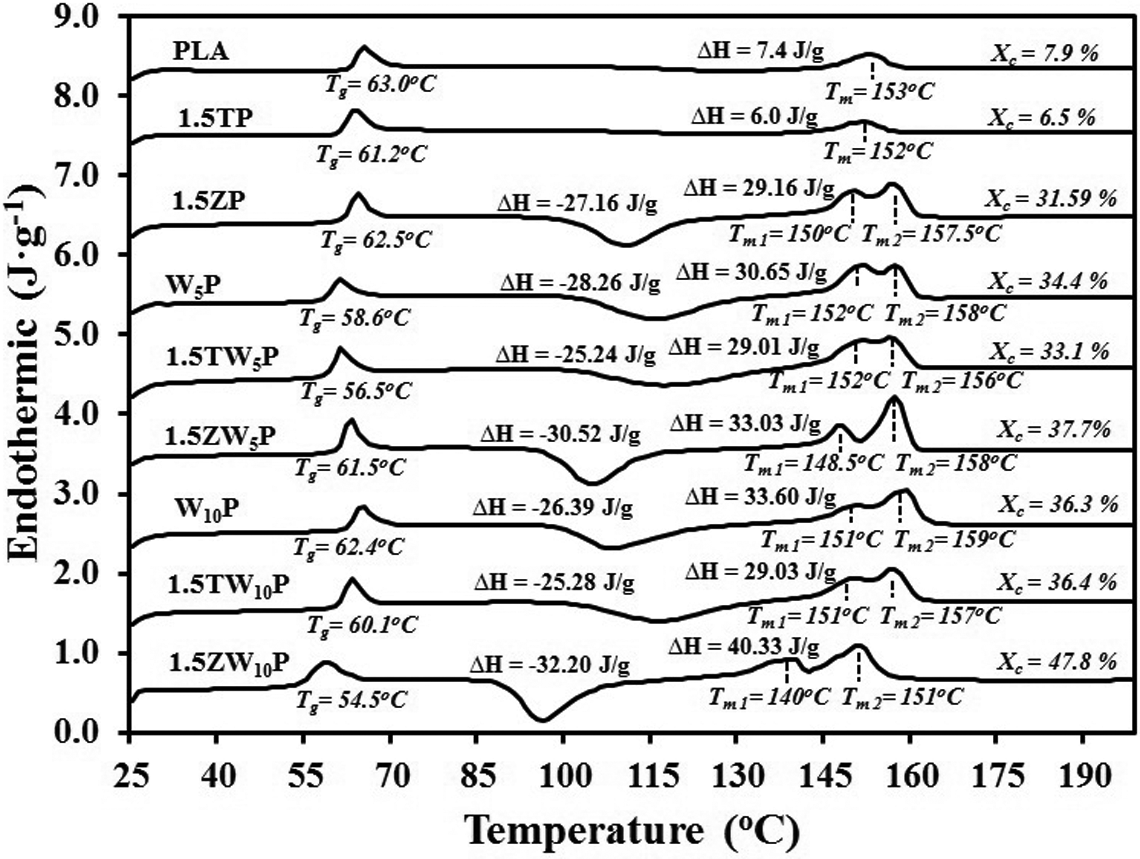
DSC thermogram of poly(lactic acid), W5P, and W10P composites with and without the addition of 1.5 wt% triclosan or Zeomic. DSC: differential scanning calorimetry; W: wood flour; P: poly(lactic acid).
Antibacterial activity
The antibacterial performances against S. aureus of PLA and wood/PLA composites incorporated with triclosan or Zeomic at different concentrations and for varying contact times are given in Figures 4 and 5, respectively. For triclosan systems, the viable cell counts of triclosan-loaded PLA and wood/PLA (Figure 4(a) to (c)) for all contact times were lower than those of specimens without triclosan. Nevertheless, S. aureus bacteria still grew at a slower rate when the contact time was increased. This suggested that triclosan could kill or inhibit S. aureus bacterial growth for PLA and wood/PLA composites. For Zeomic systems, an increase in contact time and/or Zeomic content did not affect the reduction of S. aureus CFUs (Figure 5(a) to (c)). With longer contact time, more CFUs occurred, even when Zeomic was added at the maximum dosage of 1.5%. This suggested that Zeomic did not have the ability to retard bacterial growth.
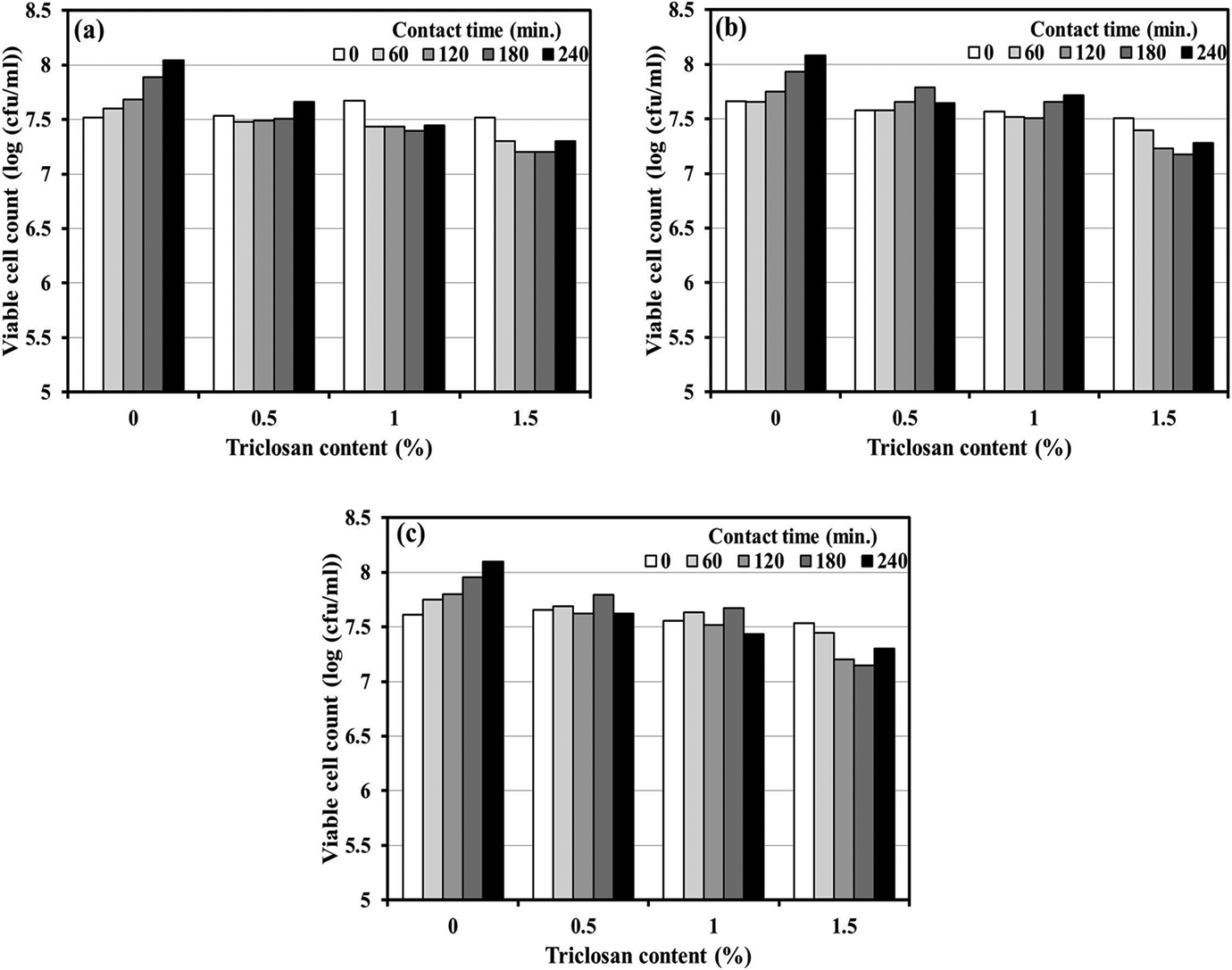
Viable cell count of S. aureus with triclosan loadings of 0.5–1.5 wt% for different contact times: (a) poly(lactic acid), (b) W5P composites, and (c) W10P composites. S. aureus: Staphylococcus aureus; W: wood flour; P: poly(lactic acid).
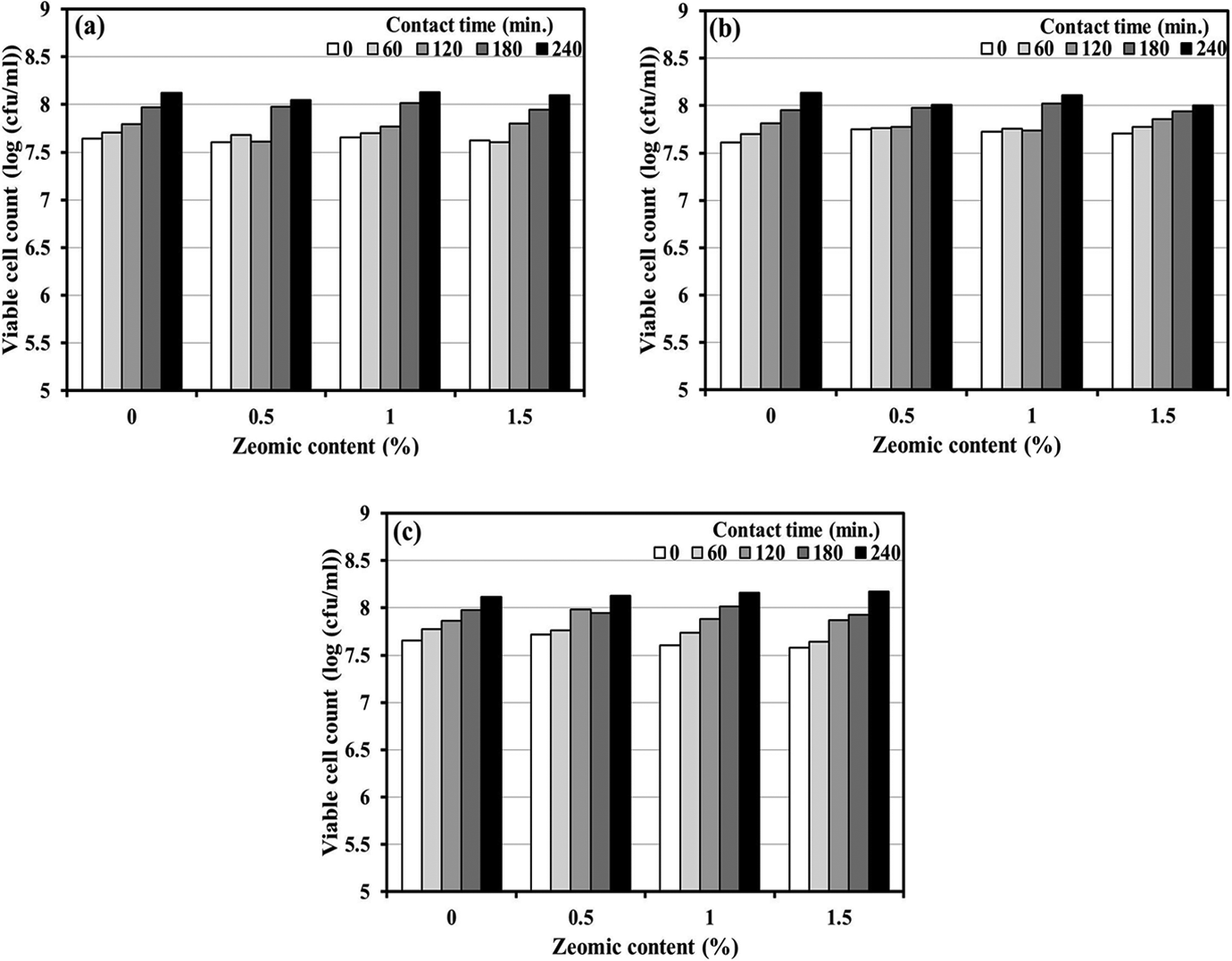
Viable cell count of S. aureus with Zeomic loadings of 0.5–1.5 wt% for different contact times: (a) poly(lactic acid), (b) W5P composites, and (c) W10P composites. S. aureus: Staphylococcus aureus; W: wood flour; P: poly(lactic acid).
In order to further illustrate antibacterial effectiveness, the percentage of bacterial reduction was calculated using equation (1). Table 3 shows the percentage of bacterial reduction of S. aureus for PLA and wood/PLA composites containing triclosan or Zeomic loadings of 0.5–1.5 wt% at different contact times. With one exception (0.5TW10P at 180 min), the higher the triclosan concentration and/or the longer the contact time, the greater the percentage of bacterial reduction. For example, at 60-min contact time, the antibacterial performance of PLA containing 0.5% triclosan was enhanced from 25.00% to 50.00% when the triclosan content was raised to 1.5%, and the percentage reduction also increased up to 58.18% when the contact time was extended to 240 min. The optimum antibacterial performance of PLA, W5P, and W10P (81.82, 84.17, and 84.80%, respectively) occurred with 1.5% triclosan loading and 240-min contact time. On the other hand, Zeomic did not show any antibacterial property. Higher Zeomic dosages and longer contact times did not affect the antibacterial activity of the composites. The bactericidal effectiveness of Zeomic depended upon the amount of Ag ions generated by ion-exchange reactions between Ag particles and positive ions, such as Na ions from water molecules. 23 However, thermal analysis indicated that Zeomic created crystalline regions in PLA that could interrupt the generation of Ag ions, thereby limiting the bactericidal effect against S. aureus. Fernández et al. 6 also discovered that the Xc of PLA was the cause of the drop in antibacterial performance of Zeomic/PLA composites.
Percentage of bacterial reduction at different contact times for poly(lactic acid) and W/poly(lactic acid) composites with triclosan and Zeomic loadings of 0.5–1.5 wt%.
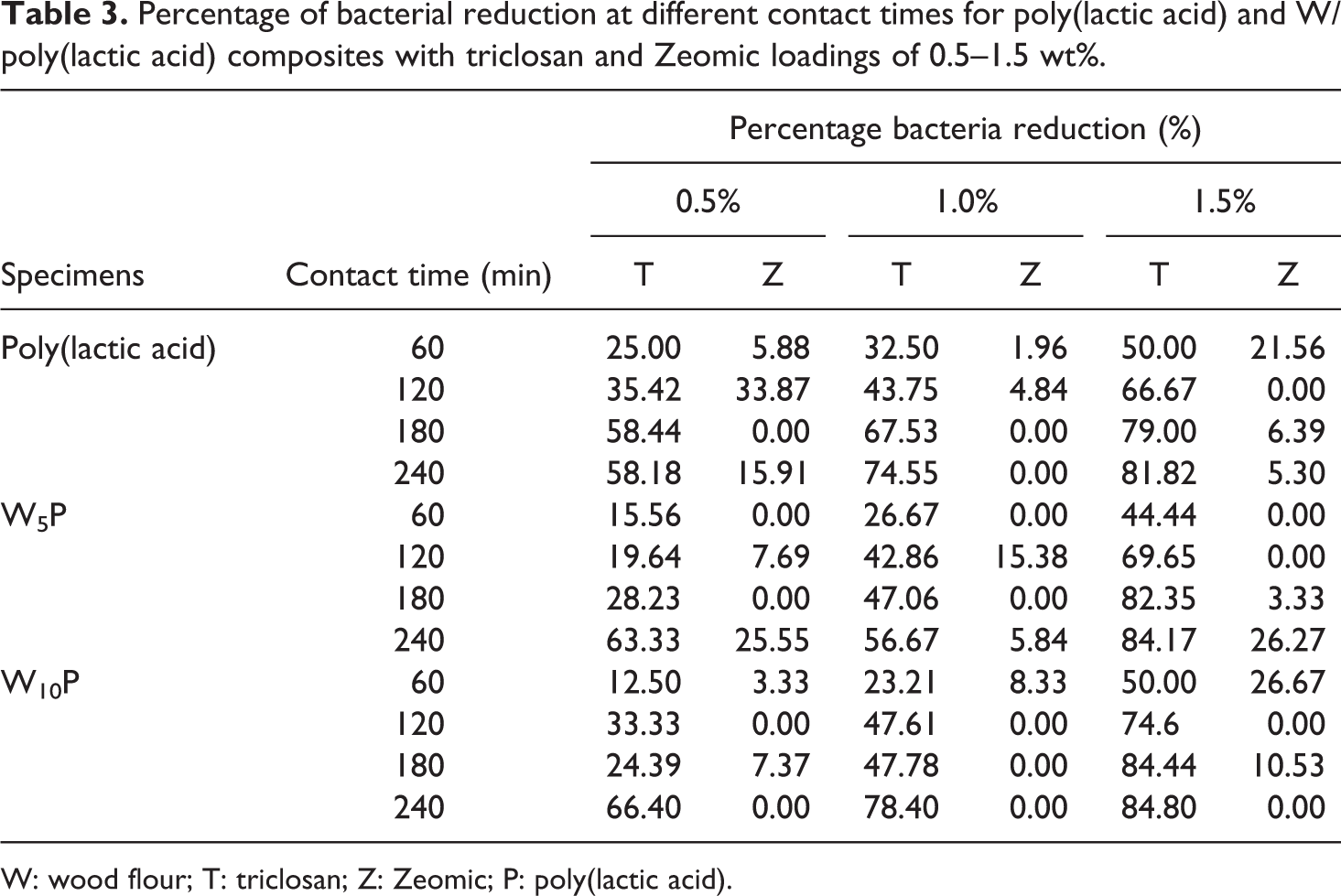
W: wood flour; T: triclosan; Z: Zeomic; P: poly(lactic acid).
Biodegradation test
Biodegradation test results for PLA and PLA composites are shown in Figure 6(a) to (c). The results were divided into three categories: (i) PLA and wood/PLA (Figure 6(a)), (ii) PLA and wood/PLA incorporating Zeomic (Figure 6(b)), and (iii) PLA and wood/PLA incorporating triclosan (Figure 6(c)). Each category was further subdivided into three different periods of incubation time: zone 1 (1–20 days), zone 2 (21–40 days), and zone 3 (41–60 days). In order to obtain a better understanding of the biodegradation behavior of PLA and PLA composites, the biodegradation rates for different time periods were calculated and are given in Table 4.
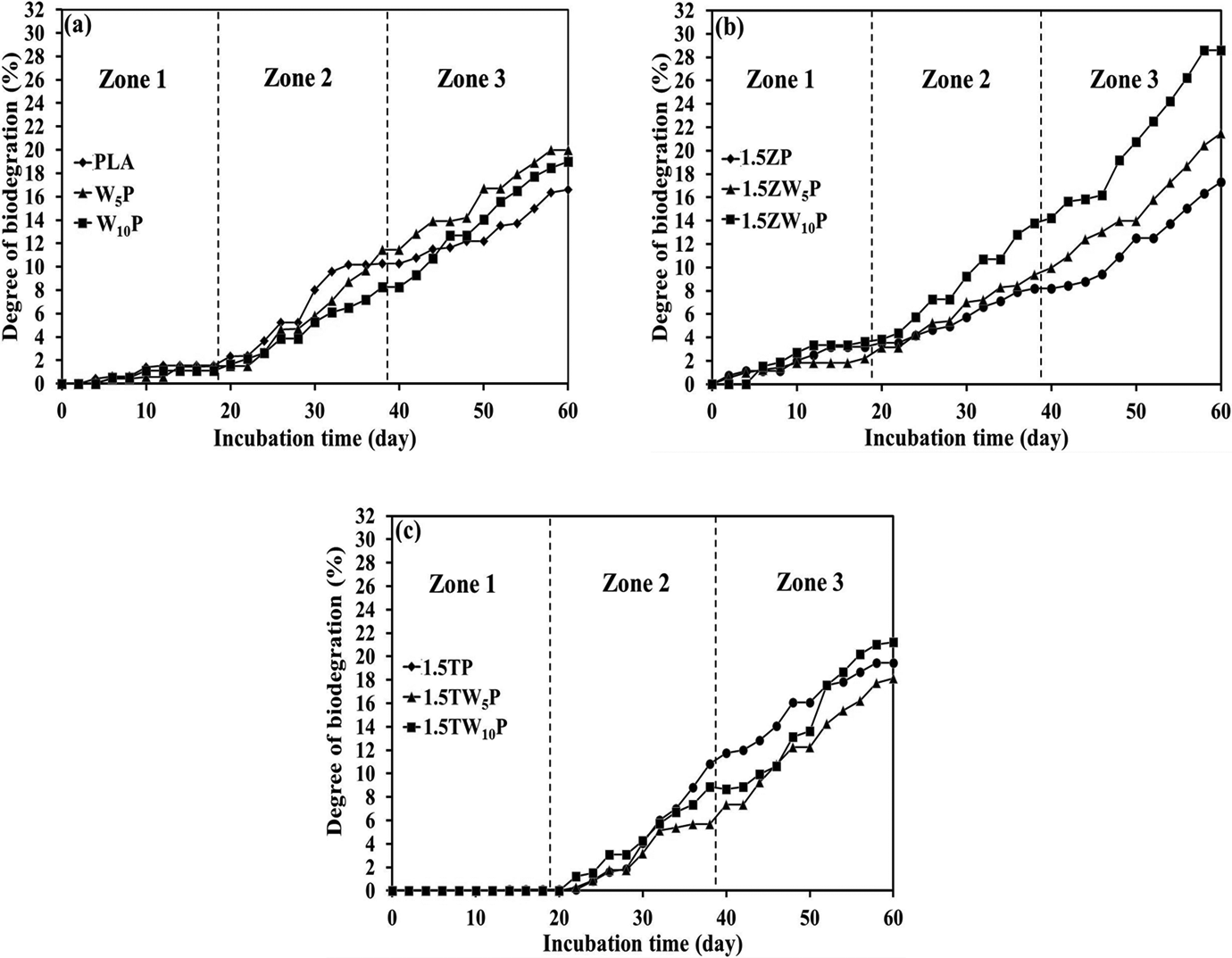
Degree of biodegradation: (a) PLA and wood/PLA composites, (b) PLA and wood/PLA composites with Zeomic, and (c) PLA and wood/PLA composites with triclosan. PLA: poly(lactic acid).
Biodegradation rates for poly(lactic acid) and W/poly(lactic acid) composites with and without the addition of Zeomic or triclosan (1.5 wt%).
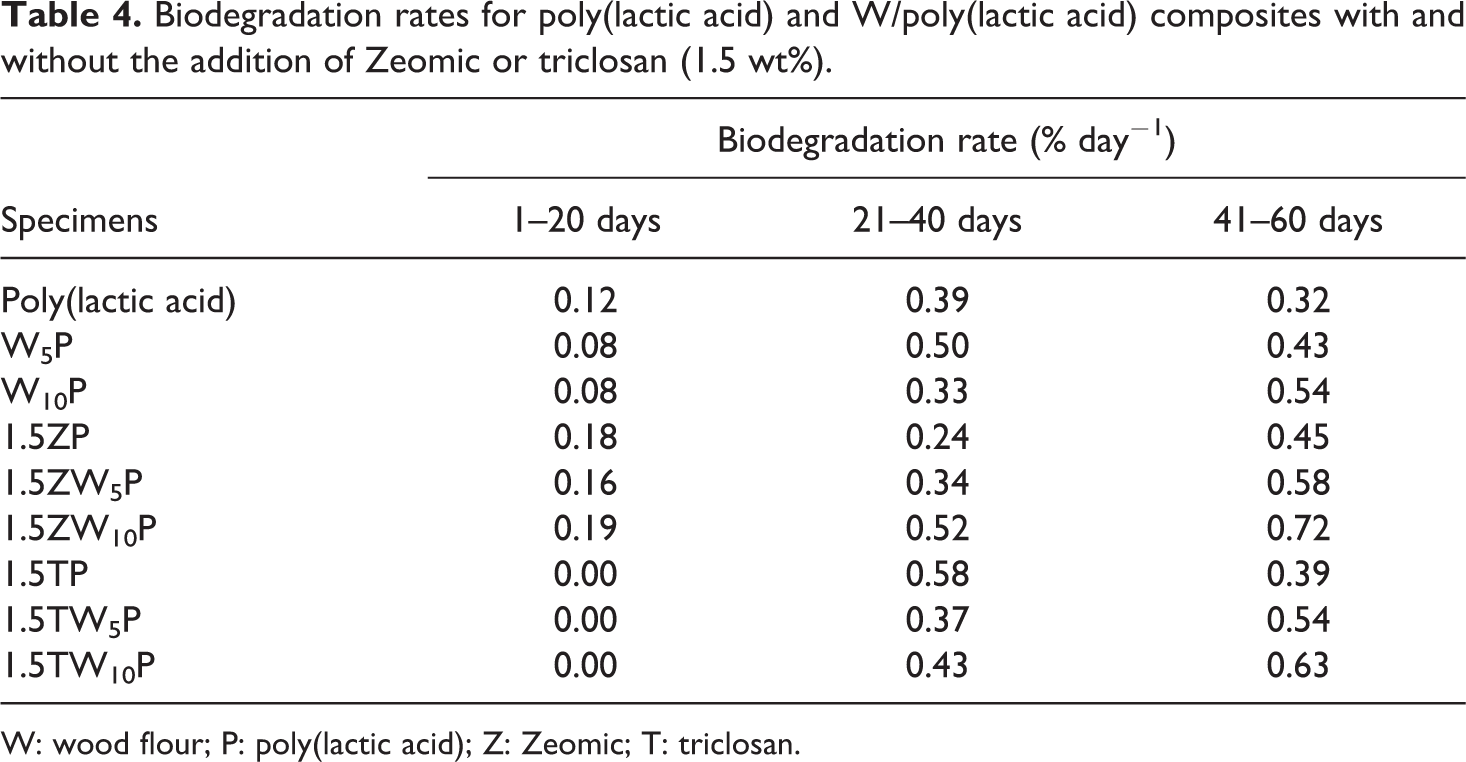
W: wood flour; P: poly(lactic acid); Z: Zeomic; T: triclosan.
For PLA and wood/PLA (Figure 6(a)), the graphs showed a slow increase in biodegradation levels for the first 20 days (zone 1), especially for PLA containing 5% and 10% wood. After 20 days (at the beginning of zone 2), the degradation rates of wood/PLA became faster. Finally, after 60 days of incubation (at the end of zone 3), the wood/PLA systems exhibited better degradation performances than neat PLA, with degradation levels of 19.97%, 18.99%, and 16.59% for W5P, W10P, and PLA, respectively. Table 4 shows that the degradation rates of wood/PLA started at 0.08% day−1, which was lower than the rate of 0.12% day−1 for PLA. As indicated in the DSC thermogram (Figure 3), wood enhanced the crystallization of PLA, and this plays an important role in retarding the hydrolysis reaction, which is the main chemical reaction in the biodegradation process. The crystalline structure of PLA takes a longer time to hydrolyze at the ester linkages and become lower in molecular weight, 24 when it can be more easily digested by microorganisms. The degradation rates increased after 20 days, to 0.39, 0.50, and 0.33% day− 1 (for W5P, W10P, and PLA, respectively) for the second period (zone 2; 21–40 days) of the test. Finally, during the third test period (zone 3; 41–60 days), the degradation rates of PLA, W5P, and W10P increased to 0.32, 0.43, and 0.54% day− 1, respectively. It can be observed that the biodegradation rate of W5P became higher than that of neat PLA in zone 2 and that both W5P and W10P had higher rates than PLA in zone 3. Although wood created crystalline regions in PLA (as indicated by the DSC thermogram, Figure 3), wood particles are also known to be highly hydrophilic, containing hydroxyl groups (–OH) that can absorb water molecules. The absorption of moisture initiated the hydrolysis reaction, which influenced the comparatively higher biodegradation rates of wood/PLA during zones 2 and 3. Therefore, it could be said that wood particles can be considered to be a “biodegradation promoter” for PLA.
For Zeomic systems (Figure 6(b)), biodegradation occurred rapidly, beginning in the first zone. The similar patterns of biodegradation occurred in zones 2 and 3, but to an increased degree, especially for 1.5ZW10P. Ultimately, the biodegradation levels of 1.5ZP, 1.5ZW5P, and 1.5ZW10P after 60 days of incubation were 17.32%, 21.50%, and 28.61%, respectively. Table 4 shows that the biodegradation rates increased sharply throughout the 60-day testing period, particularly with the presence of 10% wood. As discussed earlier, the Si–OH bonding as a function of zeolite, together with the hydrophilic wood particles, could absorb water molecules from the environment, thereby initiating the hydrolysis reaction. Therefore, the biodegradation rates of 1.5ZP, 1.5ZW5P, and 1.5ZW10P after 60 days of incubation were 0.45, 0.58, and 0.72%/day, respectively, which were the highest degradation rates compared with the other systems. These results clearly suggested that Zeomic could act as a biodegradation promoter for PLA.
For triclosan systems (Figure 6(c)), the degree of biodegradation of all specimens was zero during the first 20 days. However, substantial degradation began to occur in zone 2. In zone 3, the biodegradation levels continued to steadily increase until day 60 of the test, when the biodegradation levels of 1.5TP, 1.5TW5P, and 1.5TW10P reached 19.45%, 18.12%, and 21.20%, respectively. Table 4 shows that the biodegradation rates were 0% day−1 during the first 20 days because none of the microorganisms could attack the specimens. Triclosan can kill or inhibit bacterial growth above 80%. Triclosan is released from the PLA surface and prevents the surrounding microorganisms from attacking the PLA. This suggests that triclosan acted as a biodegradation retarder for PLA. However, the degradation rates in zone 2 increased dramatically, from 0 to 0.58, 0.37, and 0.43% day−1 for 1.5TP, 1.5TW5P, and 1.5TW10P, respectively. Although triclosan could protect the PLA from microbial attack, the hydrolysis reaction also occurred simultaneously. Therefore, over time, the concentration of triclosan may not be sufficient to protect PLA from attack by microorganisms, and the biodegradation rate took place abruptly. During days 41–60 of the test, the biodegradation rates were 0.39, 0.54, and 0.63% day−1 for 1.5TP, 1.5TW5P, and 1.5TW10P, respectively, showing an increase only for the samples containing wood.
Conclusion
In this work, two antibacterial agents, triclosan and Zeomic, were compounded with PLA and wood flour using a twin-screw extruder. The mechanical and thermal properties, antibacterial activity, and biodegradation were monitored. Wood and Zeomic enhanced the Young’s modulus of the composites, while the tensile strength, elongation at break, and impact strength were found to decrease. However, PLA and wood/PLA specimens loaded with triclosan did not show any definite trends in mechanical properties. The DSC thermogram showed that, with the presence of wood and Zeomic particles, PLA displayed double melting peaks, which indicated that Zeomic and wood were responsible for the creation of crystalline regions in the PLA. Triclosan demonstrated significant antibacterial activity against S. aureus, while Zeomic was ineffective. Wood and Zeomic were found to promote the rates of biodegradation of PLA and wood/PLA composites, whereas triclosan was found to retard their biodegradation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Office of the Higher Education Commission (OHEC) under the National Research University Program and the Thailand Research Fund (TRF Senior Research Scholar; RTA5580009) for financial support throughout this work.
