Abstract
Visible light response PAN@Ag-Ag2O/Sch (PAN@AS, schwertmannite and polyacrylonitrile abbreviated as Sch and PAN) nanofibers with different mass ratios were synthesized by electrospinning technology and pH-induced precipitation reaction. X-Ray diffraction, X-ray photoelectron spectroscopy and transmission electron microscopy analysis showed that the formation of Ag-Ag2O/Sch heterojunction and Ag-Ag2O nanoparticles were evenly distributed on the surface of Sch. The prepared nanofibers have high oxidative removal performance for As(III) under visible light. In the PAN@AS0.10 system, the total As removal percent can reach 90.96% after 120 min irradiation at pH 4.0. The scavenger experiments confirmed that the main active substances of the PAN@AS0.10 system were h+ and •OH. The high oxidation and removal performance of the PAN@AS0.10 composite for As(III) was attributed to the effective separation of photogenerated electron-hole pairs and high adsorption capacity of Sch for As under acidic conditions. This research provides a new material for the oxidation and removal of pollutants in water [such as As(III)] and also provides a research basis for the preparation of recyclable photocatalysts.
Introduction
Arsenic (As) is a toxic and carcinogenic substance. In nature, arsenic usually exists in the form of inorganic As(III) (arsenite) and As(V) (arsenate) 1 ; moreover, As(III) has higher toxicity and mobility than As(V). 2 At pH 3.0–11.0, As(V) mainly exists in the form of H2AsO4− and HAsO42−, while As(III) mainly exists in the form of H3AsO3, 3 which is difficult to remove directly by adsorption and other methods. Generally speaking, As(III) needs to be pre-oxidized to As(V). Photocatalytic oxidation as an efficient and environmentally friendly technology can be a valid alternative. 4 Among many photocatalytic materials, Ag2O can respond under visible light owing to its narrow band gap (about 1.20 eV).5,6 However, Ag2O has strong photo-corrosive properties and can be easily reduced to Ag during light irradiation. 5
Some modified strategies have been developed to improve the stability of Ag2O, including compound semiconductors (such as TiO2 and Bi2MoO6) and doping other materials (such as schwertmannite). During the synthesis of a photocatalyst, Ag2O can be combined with other semiconductor materials to broaden its photoresponse range, making it also have photocatalytic activity in the visible light range. The metal/semiconductor coupling not only facilitates the effective separation of electron-hole pairs (e−-h+), which improves the carrier migration speed, but also prevents Ag2O from further decomposing into Ag, thereby improving the catalytic activity and stability of Ag2O. 7 Chu et al. 8 synthesized the Ag2O/N-TiO2 composite by a simple precipitation method, and found that the presence of Ag2O increases the light absorption range of N-TiO2 and reduces the recombination of electron-hole pairs. The synergistic effect improves the photocatalytic activity. Our previous research 9 has also confirmed that the combination of Ag2O with semiconductor materials (such as TiO2) can broaden its light response range, make it have excellent photocatalytic activity under visible light, and make the oxidation percent of As(III) reach 83.4%. Among many mineral materials, due to its strong adsorption capacity for heavy metals, low price, easy availability and environmental protection, Sch mineral was widely used in water treatment. 10 Keisuke et al. 11 studied the adsorption behaviour of Sch to As(V) at pH = 3.3 and found that the adsorption of As(V) can make the structure of Sch more stable. Therefore, the development of a new type of composite material that can pre-oxidize As(III) to As(V) in the water under visible light, thereby improving the removal percent of total As, has become a research hotspot in recent years.
In order to avoid the potential toxicity risks of the powdered nanomaterials after photocatalytic oxidation, and at the same time to reduce the cost of the powdered nanomaterials in the recovery of water, the most effective method is to fix them on a large carrier or substrate. 12 The polyacrylonitrile (PAN) nanofiber material has become a widely used matrix material due to its large specific surface area, which can expose more contact sites. Yu et al. 13 prepared PAN-based AgI-TiO2 with the aid of electrospinning technology, which facilitates the dispersion of the catalyst on PAN with a large specific surface area, increases the light contact site, and makes separation and recycling easier. As far as we know, studies on the preparation of PAN@Ag-Ag2O/Sch nanocomposites combined with electrostatic spinning for the treatment of arsenic-containing waste water have not been reported.
In this study, the PAN@Sch nanofiber material was prepared by electrostatic spinning, and PAN@Ag-Ag2O/Sch nanofiber membrane was prepared by a simple pH-induced precipitation method. The physical and chemical properties of the composite nanofibers were characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FTIR) spectroscopy, and the surface microstructure was analyzed by scanning electron microscope (SEM). The photocatalytic activity of the obtained nanofibers under visible light irradiation was evaluated by As(III) oxidation, and the removal percent of total As in the water was measured. The influences of pH value of water, catalyst dosage and As(III) concentration on the oxidative activity of aqueous As(III) were also investigated. Moreover, the photocatalytic oxidation mechanism of aqueous As(III) by PAN@Ag-Ag2O/Sch nanofiber was proposed in terms of radical scavenger experiments.
Materials and methods
Materials
Herein, all reagents were of analytical grade and used directly without any treatment. Polyacrylonitrile powder (PAN, average Mw = 90,000) was obtained from Spectrum Chemical Manufacturing (California, USA). Anhydrous N, N-dimethyl formamide (DMF, 99.5%) was obtained from Jiangtianhuagong Technology Development Co. Ltd. (Tianjin, China), which was chosen as a solvent. Fe2(SO4)3 was obtained from Kairuisi Technology Development Co. Ltd. (Tianjin, China). Silver nitrate (AgNO3, 99.8%) was (purchased from Aladdin Chemistry Co., Ltd.) used as a precursor to synthesize PAN@Ag-Ag2O/Sch composites. As(III) stock solution (5 g L−1) was prepared by dissolving NaAsO2 with deionized water. As(III) working solution was freshly prepared by diluting corresponding stock solutions with deionized water.
Preparation of schwertmannite
First, a flask containing 500 mL of deionized water was placed in a thermostat water bath at 85°C for preheating. After the water temperature got stabilized, 2.6 g of Fe2(SO4)3 solids were dispersed in the flask. At the same time, electric stirring was turned on, and the reaction was carried out at a constant temperature of 85°C for 1 h. After natural cooling, the supernatant was discarded. The obtained pellets were washed with deionized water and then centrifuged at 3500 r/min for three times. After freeze-drying, Sch [Fe8O8(OH)8-2x(SO4)x, (1≤x≤1.75)] solid powder was obtained for further experiments.
Fabrication of polyacrylonitrile@schwertmannite electrospun nanofibers
First, 0.2 g (the mass ratio of Sch to PAN fixed at 10 wt%) Sch powder was dissolved in 18 g of DMF solution and sonicated for 2 h to make the dispersion sufficiently. PAN powder (2 g) was dissolved in the above solution to prepare a spinning solution (PAN@Sch) at a concentration of 10 wt%. It was stirred at room temperature for 6 h to obtain a uniform and transparent polymer solution for electrospinning. The spinning solution was placed in a 10 mL plastic syringe pump, and the syringe pump was connected to a needle having an inner diameter of 0.8 mm. Electrostatic spinning equipment (JDF05) (China Changsha Nayi Instrument Co., Ltd.) was used for electrostatic spinning. During the spinning process, a high voltage of 20.0 kV was used. A roller receiving device was used. The distance between the needle and the receiving roller was 15 cm. The syringe pump was advanced at a speed of 0.8 mL h−1. After spinning, the prepared composite film was placed in a vacuum oven and dried at 60°C for use.
Fabrication of polyacrylonitrile@Ag-Ag2O/schwertmannite composite nanofibers
PAN@Ag-Ag2O/Sch composites were prepared by pH-induced precipitation reaction. The dried PAN@Sch membrane was placed in a 15 cm diameter petri dish and completely immersed in distilled water, and the pH was adjusted to 8.5. Then, a certain proportion of a 0.2 M AgNO3 solution was dropped into the suspension, and the resulting mixture was immersed in the dark for 30 min to reach Ag+ adsorption equilibrium on the PAN@Sch surface. Then, the 0.5 M NaOH solution was slowly added to the Ag+ pre-equilibrium mixture, and the molar ratio of NaOH to AgNO3 was 10. After standing at room temperature for 12 h, the mixture was taken out and washed repeatedly three times with deionized water. The obtained fiber membrane (PAN@Ag-Ag2O/Sch) was then dried in a vacuum drying box at 60°C and marked as PAN@ASX (X = 0.05, 0.10 and 0.20), where X represents the mass ratio of Ag-Ag2O to Sch. For comparison, a PAN@Ag2O composite (without Sch) was prepared by the same method.
Characterization
X-Ray diffraction patterns of the synthetic composite nanofibers were recorded on a JapanD/Max 2200PC Rigaku X-ray diffractometer with CuKa radiation (λ = 1.5406 Å). Scanning electron microscope (Gemini SEM500, Germany) was employed to record their morphologies. X-ray photoelectron spectroscopy analysis was carried out using K-alpha electron spectrometer. Thermogravimetric (TG) analysis of samples was performed in a nitrogen environment with a flux rate of 60 mL min−1 using a TG analyzer (PTC-10A, Rigaku, Japan). Samples were heated from 50°C to 800°C at heating increments of 10°C min−1, after which the thermal loss was observed. Fourier transform infrared spectroscopy (Vertex80, Germany) analysis was recorded to verify the molecular structure. Mechanical strength of the films was determined by a tensile testing equipment (YG005) at an elongation speed of 10 mm min−1. Prepare the film samples into a 40 mm × 5 mm rectangle and fix it on a 20 mm × 5 mm fixture. Five samples were tested for each measurement. The water contact angle (JC2000DM, Shanghai Zhongchen Digtal Technology Apparatus, China) at five different locations of the sample was measured and recorded.
Photocatalytic activity test
Oxidation of As(III) under visible light irradiation was used to evaluate the activities of the prepared samples, and experiments were carried out using a Model XPA-VII photocatalytic reactor (Xujiang Electromechanical Plant, Nanjing, China). The visible light was from a 500 W Xe lamp (λ≥420 nm) which resides in a double-glazing jacket where circulating cool water flows. In a typical experiment, a dark adsorption process was carried out in advance. Afterwards, the Xe lamp was switched on and the 1.5 mL sample was withdrawn according to the experimental intervals of time. The suspensions were centrifuged, after which the supernatants were taken out and diluted. The molybdenum blue method was used to measure the concentrations of aqueous As(V) and total As at the wavelength of 880 nm via a UV-Vis spectrophotometer (Mapada, UV-1800, Shanghai, China). The concentration of aqueous As(III) was the difference between As(V) and total As. All kinetic experiments in this study were conducted in duplicate and the mean data were recorded (Figure 1). Schematic illustration for the preparation of PAN@Ag-Ag2O/Sch composite nanofibers.
Results and discussion
Morphology and microstructure of the polyacrylonitrile@Ag-Ag2O/schwertmannite nanofibers
As shown in Figure S1(a) and Figure 2(a), PAN nanofibers have a smooth surface, uniform thickness (average diameter of about 238 nm) and good spinnability. When Ag2O nanoparticles were loaded on the surface of PAN@Sch, the surface particles of the obtained PAN@AS0.05, PAN@AS0.10 and PAN@AS0.20 materials increased, and the respective diameters became thicker at 255 nm, 260 nm and 268 nm (Figure S1(c)–(e)). In addition, Figure 2(e) shows the surface morphology of the nanofiber membrane after 100K magnification of PAN@AS0.10. The surface chemical composition of the prepared PAN@ASX samples was further determined by EDX measurement. Figure 2(f) showed that the PAN@ASX composite was composed of C, N, O, S, Fe and Ag. However, the PAN@Ag2O nanofibers showed significant agglomeration (Figure S1(f) and Figure 2(c)), which increased the fiber diameter to 394 nm, may be due to the poor dispersion of Ag2O. PAN nanofiber membranes were often used as substrates for electrospinning due to their excellent spinnability, chemical properties and mechanical properties.
13
SEM images of (a) PAN, (b) PAN@Sch, (c) PAN@Ag2O, (d,e) PAN@AS0.10 nanofibers and (f) EDX of PAN@AS0.10 nanofibers. Note: SEM: scanning electron microscope.
The XRD patterns of PAN, PAN@Sch, PAN@Ag2O and three different PAN@ASX composite nanofibers are shown in Figure 3(a). As confirmed by our previous research,
8
the cubic phase of Ag2O (JCPDS No. 41–1104)
14
was detected in the curve of PAN@ASX composite nanofibers, which the diffraction peaks at 2θ of 33.0°, 38.3°, 55.1° and 66.1° corresponded to the (111), (200), (220) and (311) crystal planes, respectively. In addition, the curve of PAN@ASX composite nanofibers also shows the crystal phase of elemental Ag (JCPDS No. 04–0783).
15
The diffraction peaks at 2θ of 38.1° and 44.3° corresponded to the (111) and (200) crystal planes16,17 of elemental Ag, respectively, confirming the coexistence of Ag2O and Ag in the synthesized PAN@ASX composite. The characteristic peaks of Sch (JCPDS 47–1775) are at 2θ of 18.2°, 26.6°, 35.0°, 46.6°, 55.3° and 61.4°.
18
In this study, as shown in Figure 3(a), the peak shape was particularly obvious at 2θ of 35.0°, which corresponded to the (212) crystal plane. (a) XRD, (b) FTIR patterns of the PAN@Sch, PAN@Ag2O and PAN@ASX (X=0.05, 0.10 and 0.20) nanofibers; XPS spectrum: (c) the survey scan of PAN@AS0.10 nanofibers and (d) proportion of Ag and Ag2O in PAN@AS0.10 nanofibers. Note: XRD: X-ray diffraction; FTIR: Fourier transform infrared spectroscopy; XPS: X-ray photoelectron spectroscopy.
The FTIR spectra of PAN, PAN@Sch, PAN@Ag2O and PAN@ASX (X = 0.05, 0.10 and 0.20) nanofibers are shown in Figure 3(b). The literature indicates that the characteristic absorption band at 2243 cm−1 is the nitrile group of PAN (-C≡N).19,20 The peak at 1732 cm−1 may be due to the vibration of the C=O bond or the residual of DMF due to the hydrolysis of PAN nanofibers.17,19 Compared with PAN, the FTIR spectrum of PAN@Sch has a wider absorption band around 432 cm−1, which corresponds to Fe=O vibration, 18 confirming the existence of Sch. The peak at 700–900 cm−1 can be attributed to the Ag-O-Ag stretching frequency. The FTIR results further indicate the formation of Sch and Ag-Ag2O heterostructures on the surface of PAN nanofibers.
The samples were characterized by XPS to further confirm the composition and interface characteristics of the prepared PAN@AS0.10 nanofibers. X-ray photoelectron spectroscopy analysis of PAN@AS0.10 sample was shown in Figure 3(c). As expected, C, N, O, Ag, Fe and S elements were found in the XPS spectrum, and no impurity peaks were observed (Figure 3(c)). Figure 3(d) shows the Ag 3d spectrum of PAN@AS. The Ag 3d3/2 peak has two different peaks 21 at 373.5 eV and 373.9 eV, while the Ag 3d5/2 peak has two peaks 22 at 367.6 eV and 367.9 eV. Peaks at 367.9 eV and 373.9 eV indicate the presence of Ag0, while the peaks at 373.5 eV and 367.6 eV indicate the presence of Ag2O. 23
The TG analysis of PAN-based composite nanofibers was shown in Figure S2(a). The heating process initiates from 50°C to 800°C, and the quality of the composite material decreases as the temperature rises. After heat treatment, the residual mass of pristine PAN was 48.23%. However, due to the addition of Sch and Sch/Ag-Ag2O nanoparticles, the residual mass of PAN@Sch and PAN@AS0.10 nanofibers increased to 49.03% and 64.07%, respectively. Previous studies have shown that the mass change in the range of 100–200°C was caused by water loss, while the sharp change in mass in the range of 280–300°C was caused by the carbonization of organic matter.9,24 When the heating temperature exceeds 500°C, the PAN@AS0.10 composite nanofibers have no obvious decomposition, exhibiting good thermal stability. All the synthesized nanofiber membranes showed good mechanical properties (Figure S2(b)), meeting the application requirements in water. 25 In addition, in order to evaluate the hydrophilicity of the nanofiber membrane, its contact angle was tested. As shown in Figure S3, after 30 s interaction with water, the contact angles of the synthesized PAN, PAN@Sch, PAN@AS0.10 and PAN@Ag2O nanofiber membrane were 44.5°, 98.5°, 15.5° and 16.5°, respectively. Specifically, the change in contact angle of PAN@AS0.10 nanofiber membrane within 6 s interaction with water was shown in Figure S2(c). With the increase of time, the contact angle of water changed from 44° to 15.5°, almost completely wetting, indicating that the synthesized nanofiber membranes have good hydrophilicity and were suitable for application in water.
Photocatalytic oxidation of As(III) and removal of total As
The dark adsorption of As(V) and As(III) on PAN@AS0.10 at pH 4.0 was conducted. Figure 4 shows high adsorption capacity of As(V) and As(III) on PAN@AS0.10. As(III) oxidation under visible light (λ> 420 nm) was conducted to evaluate the photocatalytic activity of PAN@Sch, PAN@Ag2O and PAN@ASX composite nanofibers. The calibration curve of As(V) and As(III) is shown in Figure S4. In the dark condition of 30 min, the content of As(V) was lower than 0.047 mg L−1 in all systems, indicating that there was almost no As(III) oxidation in the absence of light. After 120 min of visible light irradiation, the conversion of As(III) in the PAN, PAN@Sch and PAN@Ag2O systems were only 0.95%, 1.00% and 2.90%, respectively (Figure 5(a)). With the introduction of Ag2O nanoparticles, all PAN@ASX composite nanofibers showed higher photocatalytic activity, and the respective As(III) conversion was 10.50%, 19.00% and 7.40% in the system of PAN@AS0.05, PAN@AS0.10 and PAN@AS0.20 nanofibers. It can be seen that the content of Ag2O plays a decisive role in the oxidation of As(III) by the PAN@ASX composite material, and the best oxidation activity can be obtained when X = 0.10. Dark adsorption capacity of As(V) and As(III) on PAN@AS0.10 nanocomposite at pH 4.0. Photocatalytic oxidation activity of As(III) under visible light. Concentrations of aqueous (a) As(V) and (b) total As in the presence of PAN, PAN@Sch, PAN@Ag2O and PAN@ASX (X = 0.05, 0.10 and 0.20) nanofibers at pH = 4.0.

A high Ag2O content may prevent visible light from reaching the surface of the composite material, resulting in a reduction of photogenerated carriers, thereby inhibiting the oxidation activity of the PAN@ASX composite material. At the same time, we found that the addition of Sch helped reduce the total As content in the water (Figure 5(b)). After the PAN@AS0.10 nanofiber membrane was exposed to visible light for 120 min, the total As content in the water was 0.45 mg L−1, and the removal percent was as high as 90.96%. At this time, the remaining As(V) content in the test solution was 0.44 mg L−1, that is, almost no As(III) was present. It shows that Sch has good adsorption of As(V) in an acidic environment and the adsorption amount of As(V) decreases with the increase of the environmental pH. However, the As(V) content of the PAN@AS0.05 and PAN@AS0.20 nanofiber membranes after exposure to visible light for 120 min was 0.33 mg L−1 and 0.30 mg L−1, and the content of total As was, respectively, 2.07 mg L−1 and 2.73 mg L−1 indicating that both As(III) residues are present in the water.
Comparison of As(III) conversion and total As removal with different photocatalysts.

pH plays an important role in the study of water. The effect of pH on catalytic activity in water is shown in Figure 6. As(III) is in the form of H3AsO3 in the range of pH 3.0–9.2.
30
When the pH value is greater than 9.2, it exists as H2AsO3−.
21
Therefore, this paper mainly studies the changes of As(III) and As(V) between pH 3.0 and 7.0. At pH = 3.0, the concentration of As(V) in the water is 0.38 mg L−1, and the total As concentration is 0.74 mg L−1, suggesting that a small amount of As(III) has not been oxidized. Experiments show that pH 4.0 is more conducive to the oxidation of As(III) and the removal of total As. The content of As(V) is 0.44 mg L−1, and the remaining amount of total As is only 0.45 mg L−1 at pH = 4.0. However, the catalytic activity was significantly inhibited at pH = 7.0, and the content of As(V) was 0.09 mg L−1, while the remaining amount of total As was 3.87 mg L−1. The oxidation activity was not high, which may be related to the pH value in the water. Photocatalytic oxidation activity of As(III) under visible light. Concentrations of aqueous (a) As(V) and (b) total As as a function of pH in the presence of PAN@AS0.10 nanofibers.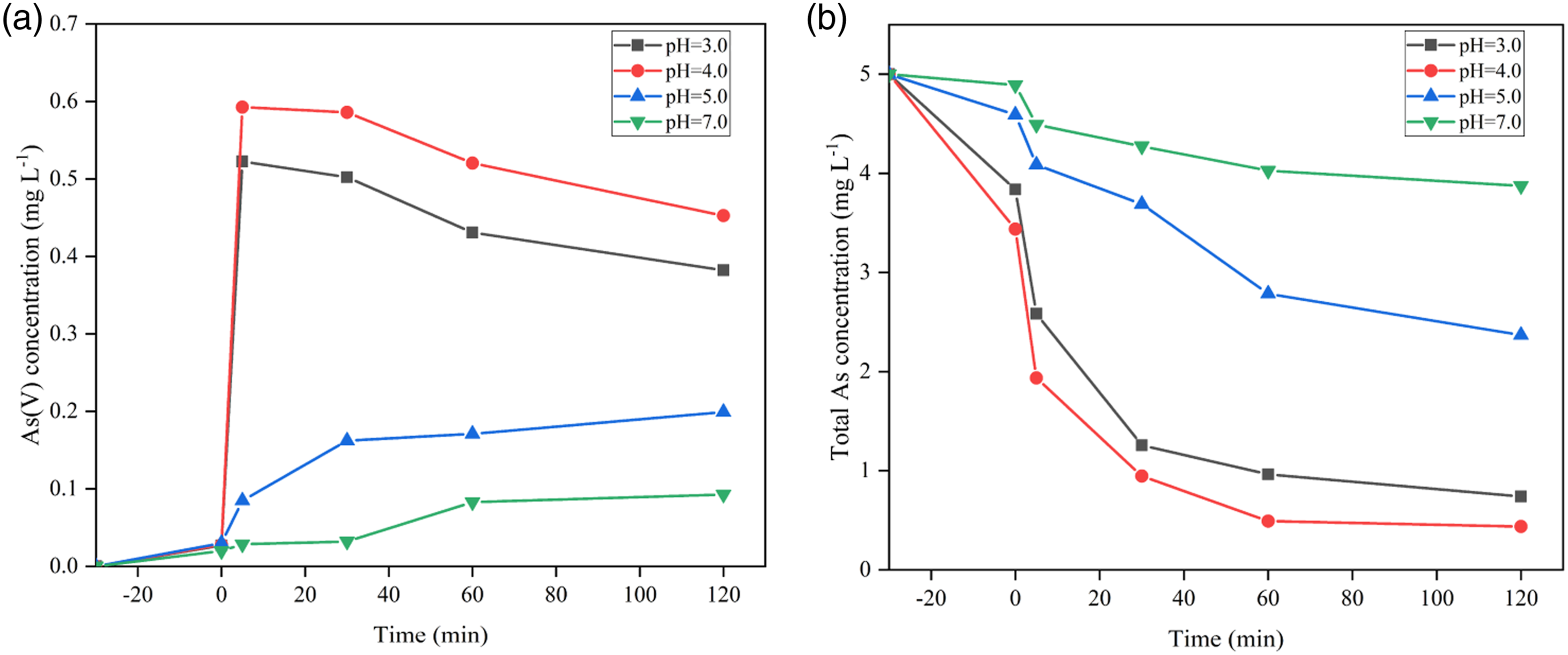
The effect of the initial concentration of As(III) on its photocatalytic oxidation was also studied, as shown in Figure 7. After 30 min of adsorption in the dark, as the initial concentration of As(III) increased, the content of oxidation to As(V) also increased. Specifically, when the initial concentration of As(III) was 8 mg L−1, the total As after 120 min visible light irradiation was still 4.37 mg L−1, and the removal percent of arsenic was only 45.38%. For the 5 mg L−1 As(III) system, after 120 min visible light irradiation, the final total As amount was only 0.45 mg L−1, and the arsenic removal percent was 90.96%. Photocatalytic oxidation activity of As(III) under visible light. Effect of photocatalyst dosage on the production of aqueous (a) As(V) and (b) total As by PAN@AS0.10 nanofibers at pH 4.0.
The effect of the content of PAN@AS0.10 nanofibers on the photocatalytic oxidation of As(III) was also studied, as shown in Figure 8. As the content of nanofibers increases, the photocatalytic conversion of As(III) and total As removal percent increased first and then decreased. When PAN@AS0.10 nanofibers increased from 0.15 g L−1 to 0.20 g L−1, the light absorption was reduced.
14
Therefore, the oxidation performance of As(III) decreased significantly, and the total As removal percent was also decreased from 90.96% to 61.40%. Photocatalytic oxidation activity of As(III) under visible light. Effect of initial As(III) concentration on (a) the production of aqueous As(V) and (b) the removal of total As by PAN@AS0.10 nanofibers at pH 4.0. Reaction conditions: [As(III)] = 5 mg L−1, [photocatalyst] = 0.15 g L−1.
After 30 min and 120 min photoreaction, the nanofiber film was sampled for XPS analysis to check the valence state of As on the surface of the nanofiber film. Previous studies have shown that the binding energies of As(III) and As(V) in arsenic oxides are always in the range of 44.3–44.5 eV and 45.2–45.6 eV, respectively.
19
Figure 9(a) and (b) show the As 3d spectrum of the nanofiber membrane after photocatalytic oxidation of As(III). The curve fitting results show that As has two oxidation states. The binding energies at 44.31 eV and 45.20 eV are As(III) and As(V), respectively. After the dark reaction, no significant arsenic peak was detected. At this time, the total As in the solution was 3.86 mg L−1, and there was only a small amount of adsorption. In addition, using the XPS peak 4.1 software analysis, it was further concluded that after 30 min of photoreaction, the surface of the nanofiber membrane was As(V) accounting for 84.65%, while As(III) accounting for 15.35%. However, As(III) was hardly detected on the surface of the nanofiber film after the 120 min photoreaction was completed. Therefore, we can conclude that arsenic mainly exists as As(V) on the photocatalyst surface after photocatalytic oxidation of As(III) by PAN@AS0.10 nanofibers. XPS spectra of As 3d after (a) 30 min and (b) 120 min visible light irradiation in the system of PAN@AS0.10 nanofibers at pH 4.0.
Determination of reactive oxidative species
As we all know, •OH, O2•− and h+ are the most common active substances in the photocatalytic oxidation process.31,32 In order to study the role of different active substances in the oxidation of As(III), Na2C2O4, CH3OH and superoxide dismutase (SOD) were used as scavengers of h+, •OH and O2•−, respectively. Figure 10(a) shows the respective concentration of As(V) in the water is 0.14 mg L−1 and 0.61 mg L−1 after 120 min reaction in the presence of 20 mM Na2C2O4 and 100 mM CH3OH, both significantly lower than that in the control group. The results showed that the main active substances involved in the photocatalytic oxidation of As(III) by PAN@AS0.10 were h+ and •OH. (a) The plots of photogenerated carriers trapping during As(III) oxidation over PAN@AS0.10 composite nanofibers under visible light; (b) the variation of As(III), As(V) and total As during the photocatalytic oxidation process by PAN@AS0.10 nanofibers under optimal experimental conditions; (c) circulating runs in the remove of total As by PAN@AS0.10 nanofibers under visible light. pH = 4.0, [As(III)] = 5 mg L−1, [photocatalyst] = 0.15 g L−1.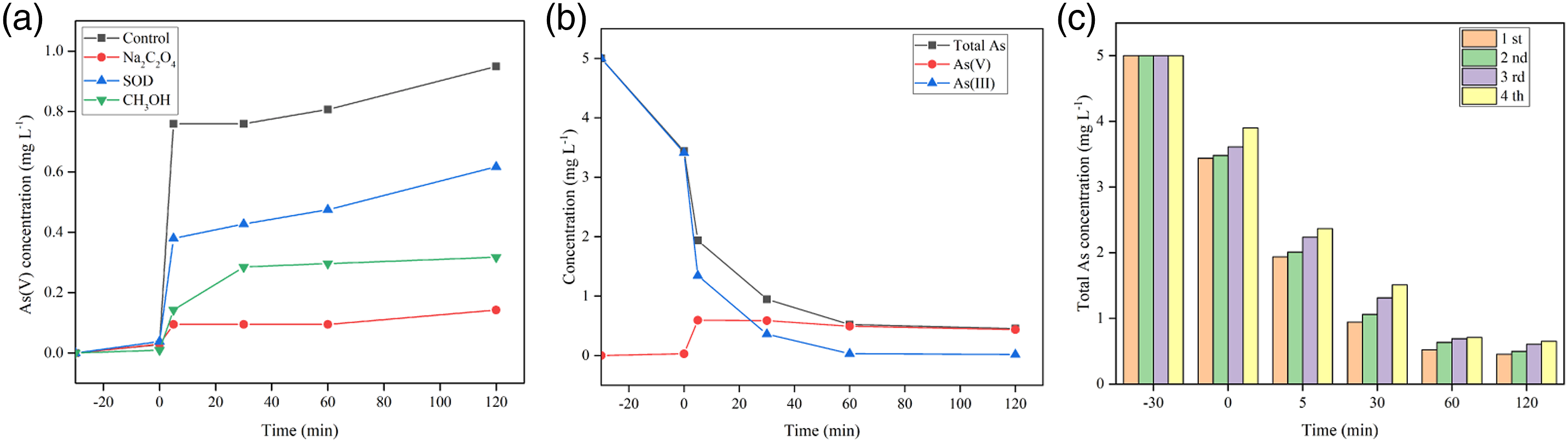
We studied the changes of As(III), As(V) and total As during the photocatalytic oxidation process under the optimal experimental conditions (Figure 10(b)). After 120 min exposure to visible light, the total As concentration in the water decreased from 5 mg L−1 to 0.45 mg L−1, and the total As removal percent by the PAN@AS0.10 composite material reached 90.96%.
Whether the PAN@ASX composite nanofiber membrane can be reused depends on its stability. As shown in Figure 10(c), PAN@AS0.10 composite nanofibers were used for four cycles of total As removal. During the cycle experiment, the removal percent of total As did not decrease significantly. After repeated use for four times, under visible light irradiation, the total As in the water was 0.64 mg L−1, and the removal percent was still 87.20%. The results confirm that the PAN@ASX composite nanofiber membrane prepared in this research has good stability and reusability. In addition, the decrease in the total As removal percent was attributed to the slight mass loss of the Ag-Ag2O/Sch composite material repeatedly washed and dried during the recovery process.
Photocatalytic conversion mechanism
In the first stage, under visible light irradiation, due to the narrow band gap (1.20 eV) of Ag2O nanoparticles, they can be excited by visible light to generate photogenerated electrons (e−) and holes (h+).
33
The electrons will be captured by O2 absorbed on the surface, forming O2•− and active oxygen. At the same time, O2•−, •OH and O2 also have strong oxidation ability, which can oxidize pollutants. Therefore, an effective separation of electron-hole pairs is achieved, which promotes the photocatalytic oxidation of pollutants. With the extension of light time, Ag2O may decompose into Ag0. Therefore, in the newly constructed system, due to their surface plasmon resonance under visible light irradiation, electron-hole pairs can also be formed in Ag0. Considering the local electromagnetic field and polarization effects caused by the negatively charged Ag2O surface, the photogenerated electrons and holes of Ag0 are migrated to completely different directions. The PL spectrum was useful for revealing the mitigation, transfer and recombination processes of the photogenerated h+-e− pairs in a semiconductor. It was generally believed that a higher PL intensity always indicated a faster recombination of photogenerated electrons and holes. As shown in Figure 11, PL spectra of PAN@Sch, PAN@Ag2O and PAN@AS0.10 composites suggested that the presence of Ag-Ag2O apparently weakens the recombination of h+-e-. PL spectra of the PAN@Sch, PAN@Ag2O and PAN@AS0.10 composites. Proposed mechanism involved in the photocatalytic oxidation of As(III) by PAN@As0.10 composite nanofibers under visible light irradiation.









As a carrier material, PAN nanofibers also play a good role in dispersing Ag and Ag2O. When the PAN@Ag-Ag2O/Sch composite material was exposed to visible light (Figure 12), Ag2O nanoparticles can be excited to generate electron-hole pairs [equation (1)]. Ag2O nanoparticles were unstable, and can be partially reduced to elemental Ag0 by photogenerated electrons [equation (2)]. As confirmed by previous studies and XRD analysis, as shown in Figure 3(a), new diffraction peaks appear at 44.3° and 38.1°, which are in phase with the (111) and (200) planes (JCPDS, number 04–0783) of metallic Ag0. At the same time, the presence of Ag0 produced by the photoreduction of Ag2O can effectively protect incomplete conversion of Ag2O into Ag0 under visible light irradiation. Previous studies have also shown that once an Ag-Ag2O structure is formed during photodegradation, Ag2O nanoparticles will exhibit self-stability. 34 X-Ray photoelectron spectroscopy analysis confirmed the existence of both Ag0 and Ag+ in the composite. At this time, photogenerated electrons tend to transfer from Ag2O to Ag0, which effectively inhibits the recombination of photogenerated electrons and holes. Under visible light, photogenerated electrons in the Ag2O conduction band combine with O2 to form O2•−; O2•− and H2O ionize H+ to form HOO• [equation (4)]. The instability of HOO• generates H2O2 under the action of excited state e− [equation (5)]. As(III) was oxidized to As(V) under the action of •OH, h+ and O2•−. The oxidized As(V) was adsorbed by schwertmannite and fixed on the surface of the PAN nanofiber membrane.
Therefore, it can be concluded that the PAN@Ag-Ag2O/Sch nanofiber membrane has a good photocatalytic oxidation activity for As(III) and strong adsorption performance for As(V). It can be summarized into three aspects: (i) The PAN nanofiber membrane has a large specific surface area, enhancing the dispersion of Sch and Ag-Ag2O; (ii) The presence of metallic Ag accelerates the separation efficiency of electron-hole pairs; (iii) Sch posses good adsorption capacity for As.
Conclusions
In order to solve the problem that total As is difficult to be removed from the water, new PAN@Ag-Ag2O/Sch nanocomposites with different mass ratios were prepared by introducing Sch through electrospinning technology and pH-induced precipitation reaction. As(III) oxidation and total As removal under visible light were conducted to evaluate the photocatalytic activity of PAN@ASX composite nanofibers. The characterization analysis confirmed that Sch nanoparticles were evenly distributed on the surface of PAN nanofiber membrane. Kinetic experiments showed that PAN@AS0.10 composite nanofibers had the best removal efficiency of total As, and the removal percent of total As after 120 min at pH 4.0 was 90.96%. Scavenger experiments confirmed that the main active substances of the PAN@AS0.10 system are h+ and •OH. The higher oxidation and removal percent of PAN@AS0.10 composite nanofibers are attributed to the higher adsorption of As(V) by Sch and the effective separation of photogenerated electron-hole pairs under acidic conditions. Because of its excellent catalytic and removal properties and reusable properties for As, the PAN@Ag-Ag2O/Sch material developed in this research has great application prospects for the oxidation and removal of As(III) in the water.
Supplemental Material
sj-pdf-1-jit-10.1177_15280837211056985 – Supplemental Material for Oxidative removal of As(III) by polyacrylonitrile@Ag-Ag2O/schwertmannite nanofiber under visible light
Supplemental Material, sj-pdf-1-jit-10.1177_15280837211056985 for Oxidative removal of As(III) by polyacrylonitrile@Ag-Ag2O/schwertmannite nanofiber under visible light by Jing Han, Hai-Tao Ren, Ting-Ting Li, Bing-Chiuan Shiu, Yong-Gui Li, Jia-Horng Lin and Ching-Wen Lou in Journal of Industrial Textiles
Footnotes
Acknowledgements
The authors are thankful for fundings from National Natural Science Foundation of China (No.21806121 and 52070143) and the Young and Middle-aged Teacher Education Research Project (Science and Technology) of Fujian Province (No.JAT200404) and Science and technology projects of Fujian Province (No.2019H6019).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China, Young and Middle-aged Teacher Education Research Project (Science and Technology) of Fujian Province and Science and technology projects of Fujian Province.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
