Abstract
The effect of dispersion technique, reduction method, and the amount of silver nanoparticles on the properties of composite polyacrylonitrile nanofiber containing silver nanoparticles is analyzed using differential scanning calorimetry, scanning electron microscopy, electrical conductivity, tensile testing, X-ray diffraction, and antimicrobial efficiency measurements.
Composite nanofibers reduced by hydrazine hydroxide result in smaller diameter, higher electrical conductivity, higher breaking strength, higher cyclization enthalpy than the samples reduced by xenon arc method. Reduction process results in smaller diameter and higher breaking strength than those of non-reduced nanofiber web containing AgNO3 nanoparticles. Dispersion by ultrasonic homogenizer/bath provides higher breaking strength, electrical conductivity than the samples dispersed by only magnetic stirrer. An increase of silver nanoparticle generally results in an increase of enthalpy, a decrease of both cyclization temperatures and crystallinity. While 1 wt% AgNO3 loading is suitable for high breaking strength, 3 wt% AgNO3 loading is suitable for both high electrical conductivity and antimicrobial properties. Insulator polyacrylonitrile polymer becomes a semiconducting material.
Introduction
Composite nanofiber films loaded with nanoparticles have drawn great attention due to their acquisition of desirable properties such as optical, electrical, and catalytic properties that make them suitable for advanced technological applications [1–3]. As known, polyacrylonitrile (PAN) is a polymer that is also used for the production of composite nanofiber. Furthermore, silver is also used in industries, as it has some benefits such as antimicrobial effect and providing better cosmetic appearance and scarless healing during the medication of burn wounds [4]. Thus, silver (Ag)-incorporated PAN nanofiber webs that have a large surface-area-to-volume ratio can have numerous application areas such as wound-healing application, filtering media and chemical, biological protective materials [5]. Silver-embedded PAN composite nanofiber is also used in air filters or water filters where hygiene and contamination issues are highly important such as in hospitals or water sources [6,7].
Moreover, in many industrial dust collection systems, the electrical property of the filter media has an important role for both filtering efficiency and safety of the system. During processing, static electric charges that can lead to explosions, fires, or personal injury from shock can develop. A conductive fiber whose resistance is lower than 1 × 108 Ω (conductivity 10−8 S/cm) is used to reduce or eliminate the buildup of electrical charges on filtering media, which consisted of several different structures such as nanofiber membrane, nonwoven fabric, woven fabric, etc. [8]. A further advantage of using electrically conductive filter media is to guarantee a complete dust cake release [9]. Thus, electrically conductive nanofiber web such as silver-loaded PAN nanofiber membrane is important for filtering efficiency and safety of the dust collection systems.
The influence of the amount of silver nanoparticles in the polymer matrices is an important issue that attracted attention of various research teams. As known in the published literature, it affects the diameter of the nanofibers, thermal properties, electrical conductivity, and mechanical properties [10–15]. As it is well known in the field, it is very difficult to obtain homogeneous dispersion of the silver nanoparticles in the polymer matrixes. The agglomeration and the high viscosity of the polymer composite material cause difficulties for the dispersion of the silver nanoparticles [13]. Due to their high surface activity, silver nanoparticles tend to generate clusters in the polymer matrix [2,3].
The amount of reduced silver nanoparticles is highly associated with the reduction type of AgNO3. It also affects the final properties of the composite material. Reducing silver nitrate into silver nanoparticles can be done in various methods such as reflux method, treatment with artificial light sources like xenon arc lamp or UV light [3,10]. Also different chemical materials like hydrazine hydroxide, sodium borohydride, and N,N-dimethylformamide (DMF) can be used [1,11,16]. Also DMF was used as a reducing agent to obtain the silver nanoparticles [11]. The effect of reflux method, chemical reduction by sodium borohydride (NaBH4), and heating of PAN/AgNO3 samples at high temperature on morphological and antimicrobial properties of composite nanofibers were investigated in literature [1]. Also, the reduction of the PAN/AgNO3 composite nanofiber by UV irradiation has been studied [10].
From the literature it can be seen that electrical conductivities of silver nitrate-embedded PAN nanofibers are between 10−7 and 10−8 S/cm in different ratios of silver nitrate loading [15,17]. PAN loaded with AgNO3 (between 0.05% and 0.5%) is effective against Staphylococcus aureus with around 72% antimicrobial efficiency [2].
Although there are several studies associated with the effect of the amount of silver nanoparticles and very limited studies associated with the effect of reduction method on composite nanofiber containing silver nanoparticles, most of these studies were done to analyze just only thermal properties (differential scanning calorimetry (DSC)) and mechanical properties or just only electrical properties [13,14,18]. Also, there are several studies associated with just only antimicrobial properties [1,2]. As seen from the published literature, no studies appeared to be published on the investigation of the effect of dispersion and mixing technique of silver nanoparticles during the preparation of polymer composite solution with the subsequent influence on the properties of the resulting composite nanofiber. In this study, a significant contribution to the published literature was made by carrying out comparative studies thereby focusing on the effect of silver loading, reduction methods, and dispersion technique. Characterization of the PAN composite nanofibers containing silver nitrate was carried out by studies using antimicrobial testing, mechanical testing, X-ray diffraction (XRD) analysis, thermal analysis (DSC), electrical conductivity, and morphological studies.
Materials and methods
Materials
PAN (Mw = 150,000 g/mol), was purchased from Sigma Aldrich. DMF and hydrazine hydroxide (N2H5OH) were obtained from Merck and 99.9995% of AgNO3 was obtained from Alfa Aesar Premion. Chemicals were of analytical grade and were used without further purification.
Methods
PAN (7 wt%) was dissolved in DMF at 60℃, mixed by a magnetic stirrer at the rotational speed of 400 r/min for 1.5 h. AgNO3 (1 wt%, 3 wt%, and 10 wt%) was added to the solution and stirred for 1 h (the amount of silver nitrate was adjusted according to the amount of PAN). The bakers that contain silver composite solutions were covered with an aluminum foil to prevent from the negative effects of sunlight.
PAN nanofibers containing AgNO3 were produced by electrospinning method.
A high-voltage power supply was used to generate an electric field in the electrospinning system. Silver nitrate including polymer solution was loaded into a 10 ml syringe And the solution was purged by the syringe pump to the needle tip that is used as spinneret. A positive voltage was applied to the rotating drum collector that was covered with aluminum foil. The negative voltage was connected to the needle tip. A nonwoven mat was covered on the aluminum foil for collecting the produced nanofibers. The feeding rate of the solution is 1 ml/h under the 15 kV voltages and the collecting distance between the needle tip and the collector was 10 cm (Figure 1).
Scheme of the electrospinnig system.
AgNO3 was induced into silver metallic nanoparticles with two different techniques namely hydrazine method and xenon arc solution method. The chemical reduction by hydrazine method was done with the aqueous solution prepared by volume ratio of distilled water/hydrazine hydroxide (ratio 20:1). After the electrospinning, nanofibers were laid into an aqueous solution for 30 min, then rinsed with distilled water twice and dried in an oven at 40℃ for 2 h and then two days at room temperature. In xenon arc solution reduction method, Suntest CPS Heraeus Original Hanau Xenon Strahler xenon lamp was used, and it was adjusted to 4 photo intensity for 30 min. After reduction of composite solution, electrospinning was done by using the reduced solution (Figure 2).
Scheme of the reduction methods.
To investigate the effect of dispersion method, another method was applied for the dispersion of nanoparticles, i.e., AgNO3 added to the solvent (DMF) was processed for 10 min by ultrasonic homogenizer, then for 45 min by an ultrasonic bath. Then PAN was added into the solution and mixed by a magnetic mixer until the PAN dissolved. The electrospinning of the final solution was prepared using the same conditions as mentioned before.
The morphology and structure of the reduced PAN/Ag nanofibers were investigated by scanning electron microscopy (SEM) Carl Zeiss EVO MA10. The SEM tests were applied at 5-kV voltage. The samples were coated with gold to prevent the sample charging effects. At least 50 measurements were done to obtain an average value of nanofiber diameter, and Image J software was used to measure nanofiber diameters.
TA Instruments Universal Analysis DSC Q10 (temperature range between 30 and 350℃) was used for thermal analysis at a heating rate of 20℃/min under nitrogen atmosphere.
Microtest LCR Meter 6370 (0.01 mΩ–100 MΩ) with two probe-four wire is used to measure the electrical conductivity. Integrated thickness meter was used to measure the thicknesses of the samples. At least seven measurements were done to obtain an average value electrical conductivity and thickness of nanofiber web. The volume resistance of the samples was measured and the conductivity of the samples was calculated according to geometric dimensions of the nanofibers. The electrical conductivity of nanofibers was calculated in S/cm according to following equation (1) as indicated in American Society for Testing and Materials (ASTM) standards [19,20].
where: γ v = volume conductivity, S/cm, Rv = volume resistance, Ω, A = area of the electrodes, cm2, and t = distance between the electrodes, cm.
Tensile tester was used for the evaluation of mechanical properties. At least seven measurements were done to obtain average value of mechanical properties of fibers. The specimens were cut in 5 mm (width) × 35 mm (length) dimensions for tensile testing. The crosshead speed was 20 mm/min and the gauge length was 15 mm.
The crystallinity of the samples was investigated by XRD analysis. Bruker® AXS D8 Advance X-ray diffractometer system was used to obtain wide-angle XRD traces, under the conditions of 40-kV voltage and 40-mA current. The observed equatorial X-ray scattering data were collected in reflection mode in the 5–40° 2θ range.
Antimicrobial test against S. aureus for 24 h was carried out according to ASTM E2149-10 standard.
Results and discussions
Morphology of the nanofibers
Figure 3 shows the SEM images of nanofibers. The diameter of the samples is presented in Table 1.
SEM images of different amounts of AgNO3 addition. (a) 100% PAN nanofiber web without treatment, (b) 100% PAN nanofiber web with hydrazine (c) 100% PAN nanofiber web xenon arc solution treatment, (d) 1 wt% AgNO3-loaded nanofiber reduced by hydrazine method, (e) 10 wt% AgNO3-loaded nanofiber reduced by xenon arc solution method, and (f) 10 wt% AgNO3-loaded nanofiber reduced by hydrazine method. The diameters of composite nanofibers. Note: PAN: Polyacrylonitrile, CV: Coefficient of variation.

Silver nanoparticles generally can have two different effects on the diameter of nanofiber, i.e., they can increase the fiber diameter due to an addition of new material into polymer matrix or agglomeration of nanofiller and they can also decrease the fiber diameter because of an increase of conductivity of jet during electrospinning leading to thinner nanofiber. As seen from Table 1, the presence of AgNO3 (not reduced) results in an increase of diameter of nanofiber compared to the diameter of pure PAN nanofiber; however, reduced nanofibers by hydrazine method have less diameters than those of samples that are not reduced possibly due to the diffusion of metallic silver atoms into inner parts of the nanofiber web forming coordination bonds with nitrile (C≡N) groups [16] and reduction of AgNO3 into metallic silver nanoparticles that do not have groups of NO3. It is reported that the formation of coordination bonding between nitrile groups of PAN and silver atoms reduced by hydrazine method causes an even and homogeneous distribution of silver nanoparticles on the nanofiber web and prevents the formation of agglomeration [16].
Treatment by hydrazine hydroxide or xenon arc lamp results in a slight decrease of the diameter of pure PAN nanofiber web. However, the highest diameter of nanofiber was observed on the samples reduced by xenon arc solution method. This may be due to more agglomerated nanosilver particles in the solution before electrospinning.
Electrical conductivity of composite nanofibers
Electrical conductivity of composite nanofiber web.

Mechanical behavior of composite nanofibers
Mechanical properties of composite nanofiber.
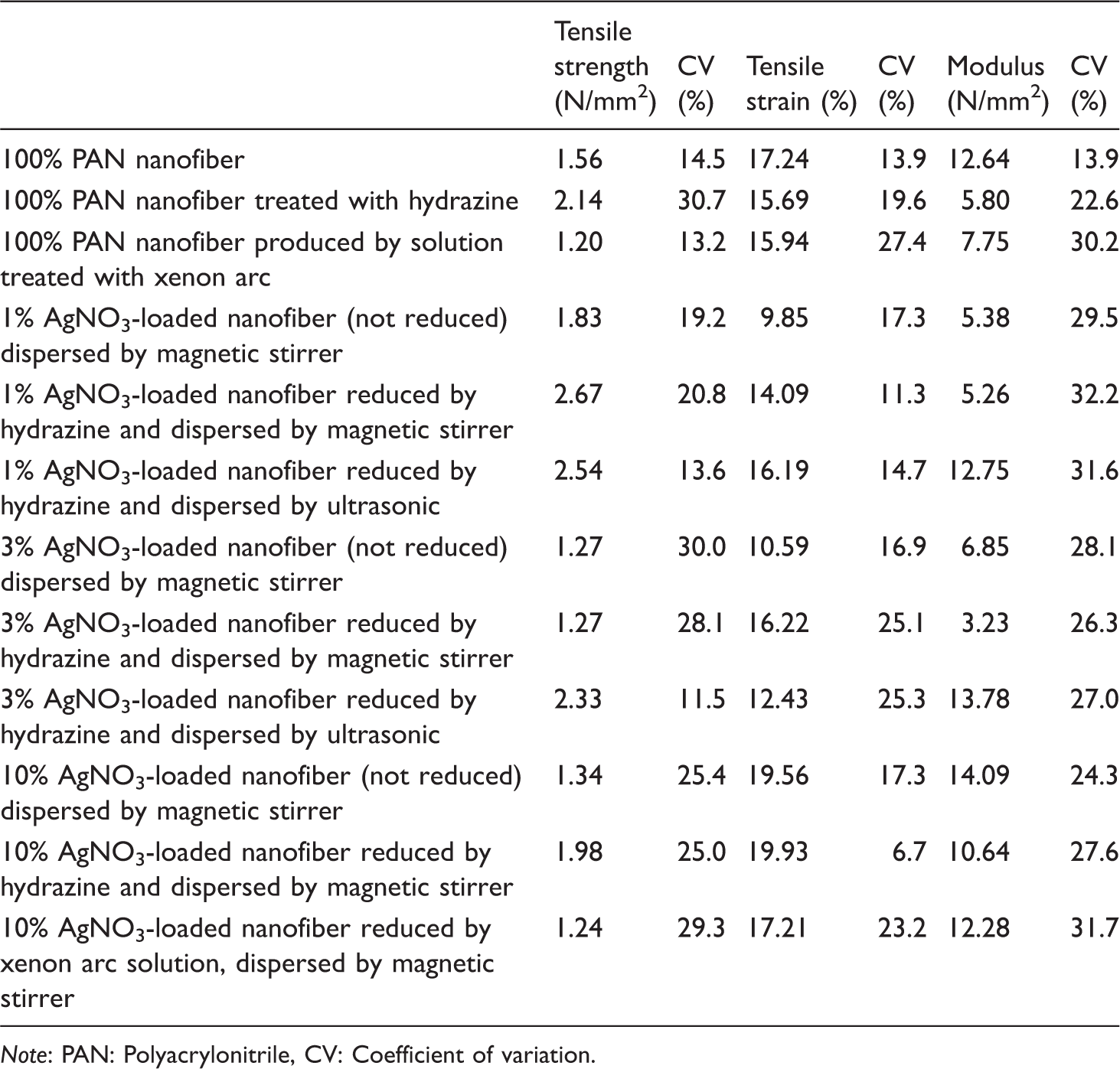
Note: PAN: Polyacrylonitrile, CV: Coefficient of variation.
When compared to the hydrazine method, samples treated by xenon arc lamp have less breaking strength than the samples treated with hydrazine due to degradation effect of xenon arc light on polymer solution [23,24]. Thus, while the strength of 100% PAN nanofiber treated with hydrazine hydroxide is higher than that of 100% PAN (not treated), the strength of 100% PAN reduced by xenon arc solution is less than that of 100% PAN (not treated). Composite nanofiber reduced by xenon arc solution method has less strength than that reduced by hydrazine method. An increase of AgNO3 or reduced silver nanoparticles generally results in a decrease of breaking strength due to an increase of agglomeration tendency. As known, optimum amount of nanofillers generally increases the strength of composite polymer material due to decreasing molecular mobility and sometimes loading stress [25,26]. Thus, it has been seen that 1 wt% AgNO3 provides higher breaking strength than others. Reduction of AgNO3 particles into silver nanoparticles results in an increase of breaking strength of nanofiber web due to a possible formation of complexation between metallic silver atoms and nitrile groups of acrylonitrile groups. Dispersion by ultrasonic homogenizer and bath generally provides higher breaking strength than only magnetic stirrer due to better dispersion of particles.
The results of elastic modulus and breaking elongation are very complex, since its breaking behavior is not as simple as any metallic rod; there are many parameters that affect each other such as pore size and distribution through the nanofiber web, the orientation, and placement of nanofiber.
As seen from Table 4 and Figure 4 and Figure 5, the XRD trace on samples containing 10 wt% AgNO3 shows the presence of a very strong crystalline Ag(111) diffraction peak indicating high concentration of silver nanoparticles. The presence of high concentration of silver nanoparticles (10 wt% AgNO3) decreases the crystallinity of PAN nanofiber (100% PAN). The agglomeration of the silver nitrate may cause an obstacle for the formation of some crystalline structure in the PAN chains. Thus, the presence of high concentrations of Ag+ ions and reduced silver (Ag0) nanoparticles in the structure disturbs the crystalline structure causing the reduction of the degree of crystallinity [27].
Curve fitting of equatorial X-ray diffraction trace of electrospun nanofibers. (a) Pure PAN, (b) 1 wt% of AgNO3-loaded nanofiber (unreduced), (c) 1 wt% of AgNO3-loaded nanofiber (reduced by hydrazine method). Curve fitting of equatorial X-ray diffraction trace of electrospun nanofibers. (a) Pure PAN, (b) 10 wt% of AgNO3-loaded nanofiber (unreduced), (c) 10 wt% of AgNO3-loaded nanofiber (reduced by hydrazine method). Crystallinity of pure and composite PAN nanofibers from XRD. Note: PAN: polyacrylonitrile; XRD: X-ray diffraction.


Thermal analysis
The thermal analyses of the produced samples were examined by DSC analysis in the temperature range between 30 and 400℃ with a heating rate of 20℃/min.
DSC results of the samples.

Note: PAN: polyacrylonitrile; DSC: differential scanning calorimetry.
The lowest enthalpy was obtained from the samples reduced by xenon arc solution method; this may be due to agglomeration tendency of silver nanoparticles and degradation of polymer solution due to xenon arc light treatment.
Antimicrobial test
Antimicrobial efficiency results for the samples.

Conclusions
The following conclusions were drawn:
Treatment by hydrazine hydroxide or xenon arc lamp resulted in a slight decrease of the diameter of pure PAN nanofiber. The presence of AgNO3 (unreduced) resulted in an increase of diameter of nanofibers compared to the diameter of pure PAN nanofiber due to an addition of new material into polymer matrix or agglomeration of nanofiller. Nanofibers containing reduced silver nanoparticles were found to have less diameters than those of the samples which contained unreduced silver. Nanofibers reduced by xenon arc solution method were found to have the highest diameter. This may be a direct result of agglomerated silver nanoparticles in the solution before electrospinning. Agglomeration of silver nanoparticles may have caused a decrease of electrical conductivity; thus, 10 wt% silver nitrate-loaded PAN was found to have less electrical conductivity than 3 wt% silver nitrate-loaded PAN nanofiber. Highest electrical conductivity was obtained from 3 wt% silver nitrate-loaded PAN nanofiber. Insulating PAN polymer (10−12 S/cm) became semiconducting material and obtained static electric dischargeability (10−7 S/cm). The electrical conductivity of samples reduced by hydrazine method was found to be higher than that of the samples reduced by xenon arc solution. This may be due to more reduced silver nanoparticles that are available on the surface for the samples reduced by hydrazine method than the samples reduced by xenon arc solution method. It was found that the samples prepared by ultrasonic homogenizer and bath were found to have higher electrical conductivity and breaking strength than the samples prepared by magnetic stirrer. This was ascribed to better dispersion of silver particles by ultrasonic homogenizer and bath than just only magnetic stirrer. The strength of the 100% PAN increased when it was treated with hydrazine hydroxide. This was ascribed to the formation of compact structure arising from dimensional shrinkage of nanofiber web and also due to the effect of crosslinking occurring between PAN chains. Samples treated by xenon arc lamp was found to have less breaking strength than the samples treated with hydrazine hydroxide due to the degradation effect of xenon arc light on polymer structure. Composite nanofiber reduced by xenon arc solution method had less strength than that of reduced by hydrazine method due to both agglomeration tendency and degradation of polymer. It was observed that 1 wt% AgNO3 provided higher breaking strength than other loadings due to optimum amount of nanofillers which provided better dispersion and less agglomeration tendency leading to a decrease of molecular mobility and sometimes loading stress during breaking. Reduction of AgNO3 particles into silver nanoparticles resulted in an increase of breaking strength of nanofiber web. The presence of high concentration of silver nanoparticles (10 wt% AgNO3) decreased the crystallinity of PAN nanofiber. Reduction process either by hydrazine hydroxide or by xenon arc solution resulted in a decrease of enthalpy and an increase of cyclization temperatures (Tc) of 100% PAN nanofiber web. An increase in the amount of silver nanoparticles reduced by hydrazine method generally results in an increase of enthalpy required for cyclization reactions and a decrease of cyclization temperatures (Tc). The lowest enthalpy was obtained from the samples reduced by Xenon arc solution method. Samples having 3 wt% and more percentage of AgNO3 reduced by hydrazine method acquired very effective antimicrobial properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TUBITAK supported this study (project 112M877).

