Abstract
An environmentally friendly approach, which consists in coupling a dielectric barrier discharge (DBD) based atmospheric plasma treatment with a coating of an aqueous phase polyurethane dispersion (PUD) containing blocked polyisocyanates, is proposed to improve the adhesion of a thermoplastic polyurethane (TPU) film onto a poly-(ethylene terephthalate) (PET) textile fabric. In this study, a screening digital design of experiment plan (DOE) was developed to determine the influence of process parameters on the adhesion between film and fabric and to evaluate the best possible adhesion value. The process parameters considered are: the dielectric barrier discharge power, the speed of the fabric undergoing the treatment, the concentration of polyisocyanates (NCO) in the PUD and the air gap. The adhesion was measured by a peeling test and further scanning electron microscope observations were carried out. Results showed that an increase of both the processing power and the NCO content in PUD, as well as the decrease in the DBD speed, had a positive effect on the adhesion. In addition, X-ray photoelectron spectroscopy and contact angle measurements demonstrated an increase in the oxygen/carbon atomic percentage ratio between the reference fabric and the treated fabric. Thus, the calibrated oxidation of the PET treated with DBD plasma treatment leads to a greater chemical and physical interaction with the TPU film, which results in better film-fabric adhesion.
Introduction
Coated technical textiles have many possible applications, whether in the automotive, personal protective equipment, inflatable or construction sectors. The fabric is an essential component as it ensures the mechanical strength of the coated structure, in particular, its tensile and peel strength [1]. The coating provides additional properties to the original fabric, such as water and air tightness, protection against external influences like UV radiation or mechanical wear. One of the most commonly used materials for textile coating is poly (vinyl chloride) (PVC) because of its durability and its cost-efficiency [2,3]. Yet, PVC poses recycling and toxicity issues, especially if incinerated, as it is known to release harmful acid hydrochloride when heated to 80°C, which requires specific equipment suited to its treatment capacity [4,5]. Therefore, the environmental, hygiene and safety regulations require its progressive replacement. Thermoplastic polyurethane (TPU) can be a good substitute as part of an end-of-life eco-design approach as it is more environmentally friendly than PVC. This polymer has good mechanical properties including high wear and tensile resistance, high flexibility even at low temperatures and high stretching capacity without the use of plasticizers, in addition to good resistance to oil and grease [6]. Moreover, the thermoplastic nature of TPU facilitates recycling at the end of the product's life. Thermoplastic polyurethane also has several shortcomings, including poor resistance to UV and poor adhesion to low surface energy substrates such as poly-(ethylene terephthalate) (PET) [7,8].
Adhesion refers to the physicochemical mechanisms that occur when two surfaces come into contact. A common example of a chemical modification process to increase the polarity of a fabric substrate is bleaching. This process is efficient, but consumes large amounts of water and energy (especially during the multiple steps of rinsing the fabric), and depends on the use of harmful chemicals [9]. Other solutions include the use of chemical primers, special coatings or adhesives, which create an intermediate layer between the substrate and the coating in the aim of increasing chemical bonding or mechanical anchoring depending on the techniques used [10].
Plasma treatments are an interesting ecological and innovating alternative to modify the surface properties of a given substrate. A plasma is an ionized gas that can have different effects on the processed substrate, including ablation, crosslinking, roughness modification or surface activation [11–16]. Different gases can be used as well as different temperature and pressure conditions to achieve the desired effects. The resulting physical and chemical changes may lead to higher hydrophobicity or improved hydrophilicity depending on the configuration. Surface roughness can also be modified [17,18]. Many authors studied the impact of various gas mixtures (such as nitrogen or oxygen [19,20]) used on the surface roughness of poly-(propylene) or PET textiles induced by the plasma treatment. They found an evolution not only of the surface roughness, but also of the chemical composition of the substrate [21]. These modifications induced a better wettability of the PET textiles. For example, Kan and Yuen [22] measured a capillary rise occurring 2.7 times faster for a PET fabric subjected to plasma at atmospheric pressure in a He/O2 mixture (95/5) compared to that of the untreated reference. A clear increase in the hydrophilicity of the post-treated PET was thus demonstrated, which could then improve its compatibility with a functionalized coating by increasing of the PET substrate polarity.
There are many kinds of plasma treatments, which can be organized depending on the type of electrodes used or the energy and the atmosphere of the discharge. Dielectric barrier discharge (DBD) consists in creating an atmospheric or cold plasma (temperature of plasma gas flow lower than 250°C) by applying a sufficiently high voltage between two electrodes, one of which is covered by a dielectric material [13,23]. It is one of the simplest approaches to increase the polarity of a substrate. Furthermore, cold plasma treatment is carried out in ambient air and at atmospheric pressure. Indeed, this presents the advantage of being easily transferable to a continuous textile process without significantly reducing its productivity. The modified surface properties therefore allow for a better wettability of the substrate.
Esena et al. [24] worked about the surface modification of poly-(ethylene terephthalate) film (PET) after the exposure to an atmospheric pressure air dielectric barrier discharge (DBD) and showed a roughness change in both amplitude and peak spacing. These surface modifications cause both optical property changes and adhesion enhancement to coating and depositions. Liu et al. concluded that the longer the processing durations (low processing speeds and a high number of treatment cycles) and the higher the DBD plasma powers, the higher the surface wettability of the PET [25]. In this context, amine groups were grafted on the DBD plasma treated PET surface with ammonia used as a monomer by Li et al. [26]. The measurement of the water contact angle confirmed the grafting effect by the more hydrophilic behavior of the treated surface. Finally, Kostov et al. [27] studied the surface modification of common commercial polymers such as polyethylene terephthalate (PET) and polyurethane (PU) by an air dielectric barrier discharge (DBD) at atmospheric pressure. An AFM analysis reveals that the plasma treatment roughens the material surface. Due to these structural and morphological changes, the surface of DBD-treated polymers becomes more hydrophilic resulting in enhanced adhesion properties.
The aim of the present study is then to investigate the influence of DBD process parameters at atmospheric pressure in ambient air on the adhesion of a novel thermoplastic polyurethane based dispersion coating containing varying amounts of blocked polyisocyanates (NCO) onto a polyester fabric. The originality of this work lies in the coupling of a reactive plasma treatment and a polyisocyanate additive treatment in the aqueous phase which could make it possible, thanks to the experimental design, to limit the use of PUD while maximizing the interfacial adhesion and therefore the peeling strength of the PET/TPU textile.
Materials and methods
The entire process of treatment and characterization of the PET substrate is shown schematically in Figure 1. The PET fabric was supplied by Chomarat (France) specifically for this study (marine application) and ready to use, with less than 0.1% surface impurities. It has a basis areal weight of 105 g/m2 and its main technical characteristics are given in Table 1 according to the PET fabric datasheet.

Method of processing and characterization. (a) Digital design of experiment plans used for plasma parameters screening. (b) Atmospheric pressure DBD treatment in ambient air of the PET textile. (c) Coating of the PUD paste onto the textile, followed by an over drying step. (d) TPU film deposited by an extrusion-cast technique. (e) Final structure of the textile. (f) Characterization methods used, details of the peeling test are given.
PET fabric datasheet.

Dielectric barrier discharge plasma treatment
The dielectric barrier discharge plasma treatment (Ahlbrandt, Germany) was carried out in ambient air at atmospheric pressure and was delivered by a curtain having a width of 60 cm on the PET textile (Figure 1(b)). Plasma treatment was delivered as an online process and treatment duration was less than 1 second. Processing power limits are set by the equipment used and can vary between 300 W and 1200 W depending on the desired process parameters required by the design of the experiments. The electrodes were 0.8 m long and 0.5 mm thick and were made of alumina ceramic. The counter electrode was a rotating cylinder with a diameter of about 30 cm covered with a silicone insulator about 0.5 mm thick and over which the textile ran directly. The air-gap, or distance between the electrode and the dielectric barrier, was fixed at either 0.5 or 1 mm. The speed of the process was limited by the machine capability to 2 and 5 m/min. The energy E of the DBD is measured in J/cm2 and is given by equation (1) [28]
Aqueous phase polyurethane dispersion and coating
An aqueous phase aliphatic ester polyurethane dispersion (PUD) of 60% dry weight was used for textile coating (U216, Alberdingk Boley, Germany), having good hydrolysis resistance and a pH between 7.5 and 9. A thickening agent (Rheovis PU 1214, BASF, Germany) was added to obtain a coating paste applicable with the air-blade system used. Blocked polyisocyanates (Imprafix 2794 XP, Covestro, Germany) were also added to the solution as cross-linking agent, activating only at temperatures above 120°C. The product used is 38% of dry weight, contains 12.7% of isocyanate and two concentrations of 1% and 9% were compared. Stirring was done by a magnetic agitator at 750 rpm. This intermediate layer is intended to promote mechanical bonding with the PET textile on the one side, and chemical bonding with the TPU film on the other side.
The functionalized aqueous phase polyurethane was then coated onto the PET textile with an air blade system (Rollmac, Italy) (Figure 1(c)), which consists in applying a certain stress to the fabric thanks to two guide rollers between which the aqueous phase polyurethane paste is spread by a straight blade. The process speed was fixed at 1 m/min, and once coated, the textile passed through a drying oven at 100°C so as not to activate the blocked poly-isocyanates added to the coating paste. In total, 22 g/m2 of PUD was applied upon the textile, accounting for a 6 ± 1 µm coating thickness.
TPU film coating by cast extrusion
An aromatic-polyether thermoplastic polyurethane film was extruded and cast upon the pre-treated PET textile (plasma treatment and PUD coating). The polyether grade was chosen to avoid hydrolysis in contact with water. It was produced by Huntsman (USA), with an intermediate hardness (A87), a density of 1.12 g/cm3 and a melt flow index of 42 g/10 min at 190°C and 10 kg. The TPU granulates, which were first oven dried at least three hours at 50°C prior to extrusion, were converted by a single screw extruder (Amut Group, Italy) at 180°C and the melted polymer was delivered through a flat die before being cast upon the coated textile as shown in Figure 1(d). The thickness obtained was 180 µm. The temperature at the exit of the extruder was measured at 160°C. High pressure (40 bar) was applied during calendaring as the created soft composite was pressed under two calendaring rolls maintained at 40°C to optimize film-fabric adhesion (Figure 1(e) and Figure 2(a) and (b)).

Final structure of the textile (SEM image – (a) magnification x 250 and (b) magnification x 500)).
Design digital of experiment plans (DOE)
A screening digital Design Of Experiment plans (DOE) was established using the software NEMRODW®. The aim of this type of DOE is to determine the relative influence of the chosen process parameters. The software enables to visualize the response (adhesion between the TPU film and the polyester fabric). The experimental area was created according to the technical limits of the machines used. Therefore, the dielectric barrier discharge power is either 300 W or 1200 W, the speed of the textile undergoing the treatment is either 2 m/min or 5 m/min, and the air gap is either 0.5 or 1 mm. Finally, the concentration of poly-isocyanates in the coating paste is either 1% or 9%. Also, the study of factors consists in determining, among a previously chosen set of study factors, those that are statistically influential on the area of interest. This study makes it possible to quantify their effects, their sign and to compare them with an analysis of variance statistical test (ANOVA), as by example the multilinear regression coefficient R2, which is a right adjustment indicator and the p-value, which is a decreasing indicator of the sample’s reliability.
Also, the mathematical model using the DOE was given by the following equation based on Plackett and Burman’s model (equation (2)).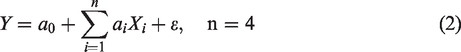
Peeling test
The adhesion between the components was measured by a peeling test based on the ISO 2411 standard. Samples of 200 × 50 mm2 were cut from the coated textile in the warp and in the weft direction. To avoid any eventual border effects following the TPU extrusion and calendaring step, samples were cut at the center of the fabric. TPU coating was manually peeled off the fabric on 50 mm to initiate the peeling. TPU film and the polyester fabric ends were clamped between the two pneumatic aluminum jaws of the tensile testing bench (Zwick Roell Group, Germany). This avoided any twisting of the samples and the tension was evenly distributed between the PET fabric and the TPU film (Figure 1(f)). Peeling was then carried out at 100 mm/min without any pre-load, and finally the variation of the force applied to separate the film from the textile was measured in N/50 mm by a 1 kN load cell.
Graphs obtained represent the peel force as a function of the displacement of the movable clamping jaw. Once the curve has stabilized, the adhesion is calculated as the average of the three highest and three lowest load values recorded.
Scanning electron microscopy (SEM)
The fracture surface was observed with a Quanta FEG 200 scanning electron microscope commercialized by FEI. Samples were cut from the peeled test piece and metallized in high vacuum sputter coater Bal-Tec CED 030 Balzers in order to avoid their charging during analysis. Then, the prepared fracture surface was analyzed using a sample holder and micrographs were obtained under high vacuum at a voltage of 12.5 kV, with a working distance of 10 mm at a x60, x250 and x500 magnification.
Surface energy
The modification of the polyester textile surface energy with the intensity of DBD treatment was evaluated by contact angle measurements. Varying powers of DBD treatments were analyzed: 300 W, 600 W and 1200 W, while the treatment speed and air-gap stayed constant at 2 m/min and 0.5 mm respectively in order to evaluate the effects of the energy of DBD in the more severe conditions. A droplet of 2 µL of the two different experimental liquids (ethylene glycol, diiodomethane) and water (as liquid test) was delicately placed onto the textile substrate in ambient environmental conditions using a Krüss goniometer K100 SF, and the contact angles were measured by image analysis and compared to that of a reference non-treated fabric. Given the texture of the substrate, the Fowkes’ method is chosen to calculate the surface free energy from the contact angle with the three liquids mentioned above and the solid. The surface energy and its two components (polar and dispersive) were calculated with a precision of 2 mN/m [30,31].
According to Young’s equation, there is a relationship between the contact angle θ, the surface tension of the liquid σL, the interfacial tension σSL between liquid and solid and the surface free energy σS of the solid (equation (3))
In order to be able to calculate the surface free energy from the contact angle, the second unknown variable σSL must be determined. In the Fowkes method, the interfacial tension σSL is calculated based on the two surface tensions σS and σL and the similar interactions between the phases. These interactions are interpreted as the geometric mean of a dispersive part σD and a polar part σP (not described by Fowkes in more detail) of the surface tension or surface free energy (equation (4))
The surface free energy of the solid is determined from the contact angle data in two steps: The dispersive part is first calculated with one purely dispersive liquid (diiodomethane in the study). The polar part is then determined with at least one liquid with polar parts (two other liquids from ethylene glycol and water (liquid test) in this study).
X-ray photoelectron spectroscopy (XPS)
The evolution of the chemical rearrangement of the fabric’s surface after different DBD treatments were analyzed by an ESCALAB 250 (Thermo Electron) X-ray Photoelectron Spectroscopy (XPSAs for contact angle measurements, textiles treated with varying powers of DBD (300 W, 600 W and 1200 W) but constant speed (2 m/min) and air-gap (0.5 mm) were analyzed. Samples were cut from the central part of the fabric and placed onto graphite tape in a vacuum chamber. A 400 μm electron spectra was delivered by an Al Kα (1486.6 eV) monochromatic anode and scans were carried out with a 0.1 eV resolution between 0 and 1200 eV. The carbon peak was set at 284.7 eV for the
Results
Peeling test
Peeling tests were performed to evaluate the efficiency of different treatment parameters established with the DOE and results are given in Table 2. The initial adhesion between the reference polyester textile and the TPU film is of approximately 21.4 N/50 mm when the textile has undergone no DBD plasma treatment and the PUD coating contains no poly-isocyanates (NCO). The adhesion slowly increases with duration of DBD plasma treatment (i.e. lower speed), DBD power as well as concentration of NCO, from 30 N/50 mm to about 45 N/50 mm for a DBD power of 300 W, an air gap fixed at 1 mm, a treatment speed of 5 and 2 m/min respectively, and concentrations of NCO of 9% and 1% respectively. The adhesion is multiplied by more than three to its maximum, when the DBD treatment is carried out at 1200 W, at 2 m/min with an air gap of 1 mm and 9% NCO in the coating paste (experiment number 5 in Table 2).
Results of the peeling test from the screening design of digital experiment plan.

It is possible to see on Figure 3 that the adhesion between the polyester fabric and the TPU film evolves similarly depending on the concentration of NCO as well as with the increase of the energy of DBD, suggesting a strong relationship between these two parameters.

Evolution of adhesion depending on the energy of DBD and the concentration of polyisocyanates. Dashed lines represent an exponential fit as guide for the eyes.
Design of digital experiment plans and study of the influence of DBD parameters: screening study
The Nemrodw® analysis of the screening study showed that the polynomial without interaction model terms X1, X2, X3, X4 are significant (with a significance p-value of 1.52, the normal plot “Henry’s plot” is found as a straight-line evolution). The coefficient of multiregression equation was calculated at 0.966 (close to 1) and the data was fitted to a first order polynomial equation without interaction between the terms Xi. Two check points for i) a power of 300 W, a speed of 2 m/min, an air-gap of 1 mm and an isocyanate concentration of 1% and ii) for a power of 1200 W, a speed of 5 m/min, an air-gap of 1 mm and an isocyanate concentration of 9% confirm the correlation between model and experiments due to a very low residue between the result of the experiment and the predicted model.
Only the X3 term (identified as the air gap) was not significant. Therefore, the adhesion response Y can be expressed by the following regression equation (equation (5))
On the one hand, it was found that both the terms X1 (Dielectric barrier discharge power) and X4 (concentration of poly-isocyanates in the coating paste) had a positive sign reflecting an increase in the response Y (adhesion) when their value changes from the relative variable range (−1 to +1) from 300 W to 1200 W and from 1% to 9% for X1 and X4 respectively. On the other hand, the value X2 (Speed of the textile undergoing the treatment) caused the Y response to drop as they increase from 2 mm/min to 5 mm/min.
In addition to that, the coefficients reflecting the effect of the factors on the response Y (adhesion) showed that the coefficient associated with the term of the dielectric barrier discharge power (X1) had a preponderant effect twice as large as the significant coefficients X2 and X4 of speed of the textile undergoing the treatment and concentration of poly-isocyanates in the coating paste respectively.
In order to maximize the adhesion between the TPU film and the polyester fabric, the values of the four parameters X1, X2, X3, X4 must be adjusted to a respective level of 1200 W, 2 mm/min and 9% of isocyanate for dielectric barrier discharge power, speed of the textile undergoing the treatment and concentration of poly-isocyanates in the coating paste. For the X3 parameter (air gap), as the term coefficient is not significant, it can be fitted to a practical value. In a case of the optimization of the parameters, the air gap will be set at the minimum value of 0.5 mm.
SEM observations
The fracture surface of the peeled samples were observed by SEM in order to identify the fracture mode (adhesive or cohesive) between the two components (Figure 4). Essentially all of the PUD coating was peeled off with the TPU film, aside from a few remains at the intersection of the fabric’s yarns, where a better mechanical bond occurred (Figure 4(a)). It was also possible to see some PUD coating leftovers between the filaments of the yarns (Figure 4(b)). The DBD treatment caused a better compatibility between the polyester fabric and the PUD coating to increase the penetration depth of the coating in the textile, but this mechanical bond has not significantly increased compared to the original material.

SEM observations of the PET textile after peeling. View of the textile after the peeling of the coating (a) without plasma treatment and (b) with plasma treatment).
Contact angle measurements
The determination of contact angle as function of the power of the DBD treatment and the nature of solvent is given in Table 3. The evolution of the PET surface free energy calculated from contact angle measurements is shown in Figure 5.
Results of the contact angle evolution of untreated and treated PET-based fabric.

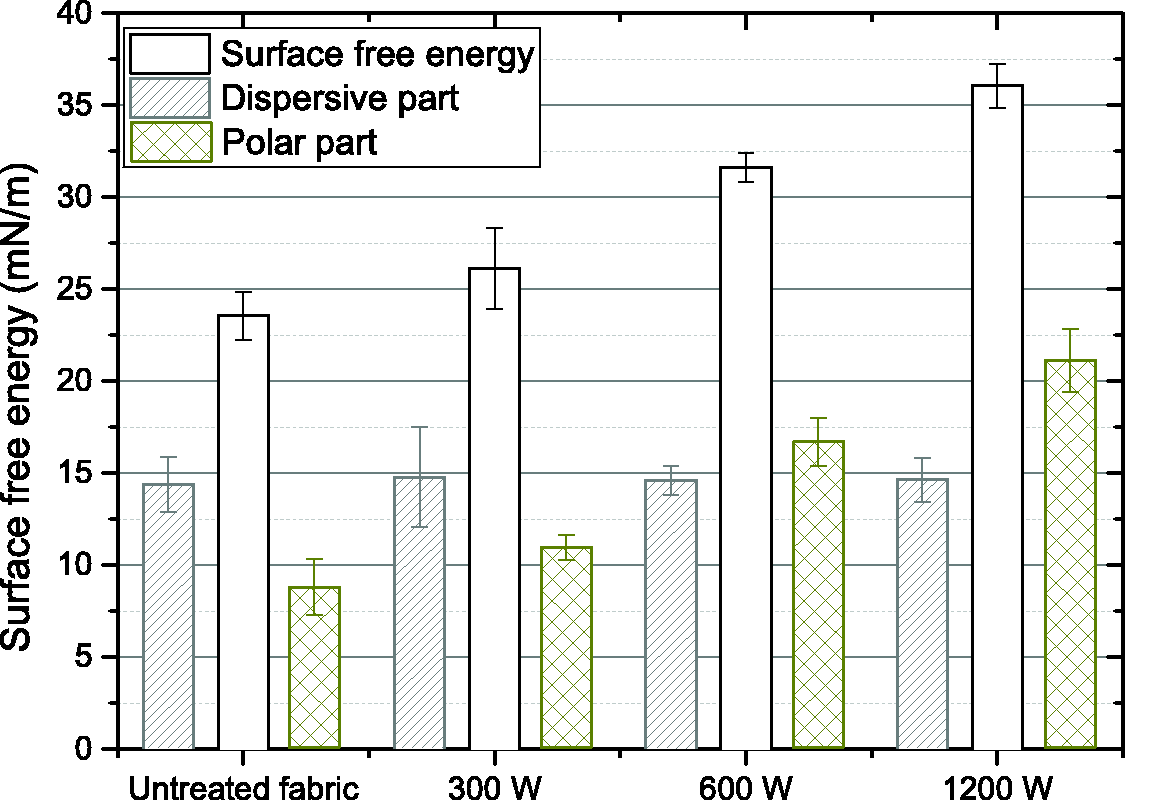
Evolution of the free surface energy of the polyester textile with the evolution of the polar and dispersive parts.
The evolution of the contact angle, from 66° to 46° for ethylene glycol, demonstrates the effect of the DBD treatment on the PET fabric, which became more polar. On the other hand, for diiodomethane, no modification of the contact angle was observed indicating that the dispersive component was not impacted by the DBD treatment. Thus the compatibility of the fabric with the TPU coating should be improved.
The surface energy measured for the reference PET textile is much lower than that found in the literature (usually around 40–45 mN/m [33]). The accurate measurement of contact angles from a woven structure is complicated due to its porous nature, inducing a wicking effect and a decrease in the measured angles. Measures were taken immediately following the deposition of the liquid drop. These results will therefore be analyzed in a qualitative manner as they are nevertheless interesting to understand the global surface energy increase following plasma treatment. It is possible to see in Figure 5 that the surface free energy increases almost linearly from 23.6 mN/m initially for the reference textile to 36.1 mN/m for a 1200 W DBD plasma treatment. As contact angles were measured with both polar and dispersive liquids (ethylene glycol, water and diiodomethane), the dispersive and the polar parts of the surface energy were calculated based on the Fowkes’ equation (equations (3) and (4)) [34]. While the dispersive part stays constant at approximately 14.5 mN/m, the polar part evolves linearly with the power of DBD from 9.2 mN/m initially to 21.4 mN/m for the highest DBD power at 1200 W, being therefore responsible for the substrate’s increase of surface energy.
XPS analysis
The XPS analysis confirms the previous results obtained with the contact angles because a clear increase in the oxygen/carbon ratio was measured. The surface of the fabric has therefore been modified as it evolves from the O/C ratio of 29 atomic percent (at. %) initially (300 W) to 38 at. % for a DBD plasma treatment at 1200 W (Table 4).
XPS analyses regarding atomic percentage of the oxygen/carbon ratio at the surface of the PET fabric for three powers of the DBD plasma treatment (constant speed (2 m/min) and air-gap (0.5 mm)) – (average standard deviation: ±0.1).

The significance of boldface is the ratio between O1s and C1s and reflects the oxidative modification of PET fabrics as a function of the power imposed by the plasma (300 – 1200W).
According to Table 5, the carbon-carbon bonds decrease with the power of treatment, from 56.7 at. % originally to 46.2 at. % at 300 W, and do not evolve significantly with DBD power from 300 W to 1200 W; while the amount of ether bonds stays constant. The carboxyl and hydroxyl bonds, responsible for the polarity of the polyester surface, increase respectively from 7.9 at. % for a 300 W treatment to 9.6 at. % at 1200 W, and 8.3 at. % originally to 12.5 at. % at 1200 W. One should consider that the total amount of each percentage of each peak is not equal to 100% due to the presence of impurity at the surface of the textile. These impurities are mainly due to original fabric preparation.
XPS analyses regarding the atomic percentage of the chemical bond at the surface of the PET fabric in function of the DBD plasma treatment powers (constant speed (2 m/min) and air-gap (0.5 mm)) – (average standard deviation: ± 0.1).

Discussions
Results of the peeling test after the DBD plasma treatment indicate an increase in overall material adhesion, suggesting that the DBD effectively modify the chemical surface of the PET fabric. This is supported by the contact angle measurements with the overall relative increase of 133% of the polar part of the surface energy of the fabric from the initial value of 9.2 mN/m for the untreated one, while the dispersive part remains constant at approximately 14.6 mN/m. Although these results seem to show the favorable hydrophilic effect of the DBD treatment, the values of surface free energy obtained for the untreated PET of 23.6 mN/m is much below the expected values of 40–45 mN/m [34]. A certain lack of accuracy when it comes to contact angle measurements at the surface of porous surfaces such as fabrics may explain the smaller surface free energy values obtained compared to that of the literature [35,36]. In the literature, the surface energy is measured on textile using contact angle method while we used the capillary method to avoid the porous effect of textiles. This could also explain the difference. Results in adhesion from the peeling tests seem to reach and asymptote for high energies of DBD, indicating the presence of a critical Treatment Power (TPc) as was observed by Leroux et al. [28]. This phenomenon results in maximum adhesion which is achieved with treatment at a specific energy of DBD, and above which the adhesion remains relatively constant. Care should be taken with the power or duration of the DBD treatment as critical thresholds of specific machine parameters must be found in order to limit fabric degradation due to excessive treatment powers above the TPc. But while Figure 3 seems to indicate the presence of a TPc, further testing needs to be done at higher DBD processing energies for more precision on the value of this TPc.
The XPS analysis brings necessary additional insight on the surface chemical modification mechanisms following the DBD treatment. The XPS measurements showing a relative increase in carboxyl bonds and a reduction by 10 at. % of the carbon-carbon bonds is thought to be responsible for the fabric’s surface increasing polarity,. The reduction of carbon-carbon bonds may be caused by the scission of the poly(ethylene terephthalate) chain either at the ether or the carboxylic bonds, creating very reactive chain ends that would easily react with oxygen radicals present in the DBD plasma, in turn resulting in the creation of ester and hydroxyl groups [37]. This will then favor a chemical bonding with the NCO into possible urethane groups, as a urethane group is the result of the mixing of hydroxyl and isocyanate groups. The reaction between NCO and hydroxyl groups create strong covalent bond that act positively for the adhesion increase.
Measures of contact angles at the surface of the polyester fabric treated with different powers of DBD at 2 m/min with an air gap of 0.5 mm illustrate a rise in the substrate’s surface free energy due to an increase in its polar component, while its dispersive part stays constant. The rise of hydrophilicity of the polyester is confirmed by the XPS analysis. The oxygen/carbon ratio increases by 10% at the surface of the polyester textile due to an increase in the polar chemical bonds such as the hydroxyl groups. The additional hydroxyl bonds created react with the blocked poly-isocyanates of the coating paste when the TPU film is cast-extruded as they activate to form a chemical bond being the reason to a better adhesion between the polyester fabric and the aqueous phase polyurethane dispersion coating.
The increase in the polyester surface polarity is due to a 4.5 at. % increase in hydroxyl groups from the reference PET fabric and that treated with a 1200 W DBD. Therefore, these accessible hydroxyl groups are privileged sites for the creation of urethane functions due to their reaction with the isocyanates of the PUD coating through covalent bonds [38].
The fracture surfaces of samples following the peeling tests between the TPU film and the PET textile show an adhesive fracture between the PUD coating and the PET fabric despite the DBD treatment. This lack of intensive mechanical bonding may be due to the air-blade technique used. Indeed, a rapid study of the amount of deposited PUD coating showed that only 22 g/m2 were applied, accounting for a thin 6 ± 1 µm coating thickness. Nevertheless, this PUD layer allows for a better stress transfer between the TPU film and the fabric because of a favoured chemical adhesion with the TPU film.
Unlike many studies in the literature, no surface roughness was observed by SEM analysis on the PET filaments following their DBD treatment, even for the higher DBD energy of 4.5 J/cm2. But the DBD energies applied to the textile in the present study are relatively lower than that of studies where surface roughness were observed by atomic force microscopy, as was the case for Brzeziński et al. [39] at a plasma energy of 76 J/cm2, or even for Takke et al. [40] for a plasma energy of 12 J/cm2.
The SEM observations of the fracture surface reveal an adhesive fracture mode that does not change drastically from that of the original material. One can still observe some remaining coating on the polyester roving overlap. To improve the adhesion between the components a higher mechanical bonding of the coating in the depths of the polyester textile, by using a more efficient impregnation technique than the air-blade process used for this study, would be necessary.
Thus, the power of DBD and the concentration of poly-isocyanates in the PUD coating have a direct positive impact on the overall adhesion of the material, while the DOE shows the opposite when it comes to processing speed. Obviously, at a slower speed the DBD treatment will have a more efficient effect on the polyester fabric. Finally, the air gap has a negligible influence on adhesion for this study. Indeed, the air gap variation amplitude was of 0.5 mm due to machine restrictions, with a minimal air gap of 0.5 mm.
Conclusion
The present study has demonstrated that the use of a DBD plasma treatment is an effective solution for modifying the surface of a PET fabric by oxidation. This facilitated the adhesion of a polyurethane dispersion coating containing blocked polyisocyanates and in turn greatly increased the adhesion of a TPU-based calendared film extrusion to the treated textile, with a + 151% increase compared to the reference untreated textile.
The peel tests carried out on the flexible composite showed that the adhesion of the components is multiplied by more than 3 between the original material having an adhesion of 21.4 N/50 mm and the final material treated by plasma DBD at 67.9 N/50 mm.
The DOE screening determined the relative impact of the significant influence of DBD treatment on the adhesion of the TPU coating onto the polyester fabric treated by DBD plasma with an intermediate coating in aqueous phase polyurethane dispersion containing polyisocyanates blocked (PUD). DBD power, processing speed, DBD machine air gap and NCO concentration in PUD coating paste were analyzed by verified DOE sieving. The results showed that the treatment power has the most significant influence, followed by the speed of the process and the polyisocyanate concentration, which are both of equivalent influence. The air gap can be neglected under current experimental conditions due to a low relative influence of less than 1%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
