Abstract
Due to the ongoing pandemic, various types of facemasks such as certified N-95, non-woven fiber and fabric/textile masks are being used as an essential protective measure to reduce the risk of spread of the SARS-Cov-2 virus. The aerosols size-dependent filtration efficiency and breathing resistance of these masks were tested before and after sterilization by five different methods for two flow rates (20 and 90 L/min) conditions corresponding to regular breathing rate and moderate/strenuous exertion, in the particle size range 0.3–10 µm. Sterilization techniques used here are autoclaving (30 and 60 minutes), dry air oven heating (30 and 60 minutes), ionizing irradiation (15 and 25 kGy), hot water washing with and without detergent and immersing in a 10% concentration of liquid hydrogen peroxide for 30 minutes. Further, the filtration efficiency of each type of masks is also studied with laboratory generated two-order higher aerosol concertation. The certified mask has the most outstanding filtering efficiency among all the other type of facemasks. The ionizing radiation causes a significant reduction in filtration efficacy, so that it is not recommended for sterilization purpose. The best method to sterilize certified N-95 masks without affecting their performance is by using dry air heating with temperature ranging from 70–80 °C. The performance of the cloth and surgical masks is found to be comparable for both flow conditions. As an affordable sterilization method, hot water washing is recommended, which does not deteriorate the fabric masks efficiency and can be used by the common people. The recommended masks for the general people are textile/fabric masks which serves fit for the purpose than non-woven masks because it can help to reduce non-biodegradable waste (facemask) and prevent respiratory droplet transmission. The non-woven mask can be sterilized with dry heat, hot water wash and autoclave.
Introduction
Since the outbreak of the novel Coronavirus Disease 2019 (COVID-19), one of the essential personal protective wear used in this pandemic is facemask, which offers protection against COVID-19 through droplet and aerosols transmission. Starting from just a few cases of COVID-19 in December 2019, as of November 2020, have more than 55 million cases and 1.3 million deaths [1], indicating the seriousness. The use of facemasks has been enforced in major countries across the globe after the recommendation from World Health Organization (WHO). Hence, it becomes mandatory to wear a facemask by the public in a common place and front-line workers who maintain the highest quality patient care against COVID-19 pandemic. Various types of Personnel Protective Equipment (PPEs) are being used by the wearers for various emergency circumstances like thermal exposures [2], chemical hazards [3], UV protection [4], liquid penetration [5], insect, virus, smoke [6], and many more. However, in the present pandemic, good quality masks and other preventive measures (social distancing and hand sanitizing) are useful and could prevent the transmission of virus particles from one person to another. There are three kinds of masks that are being used widely in most of the countries against viral infection, namely, certified N-95, non-woven (surgical) and fabric/textile masks. These masks were made of natural (plant, animal and mineral fiber) and manmade (synthetic, regenerated and inorganic) protective fiber/textile. The fabric masks typically include cotton fabrics, clothing, silk, tissue paper, kitchen towels, pillowcase, and tea cloths [6]. The certified N-95 and non-woven masks are made of multiple layers of polypropylene fibers made by the spun-bond and melt-blown process.
Frontline workers worldwide face a shortage of certified masks because of the increase in the number of users. The facemasks have been reused multiple times to overcome this emergency. Hence, urgent efforts are undertaken in various laboratories across the world to progress new strategies or to explore alternative measures encompassing sterilization strategies. This is also true for non-woven masks, where any new design/fabric needs a quality check for the desired features. Although fabric masks are widely being used by general public for which evaluation of its effectiveness is equally significant. Nevertheless, using a second-hand facemask numerous times without killing the pathogens (if any) can result in allowing any virus or bacteria on its surface to enter our respiratory system directly or indirectly. Here, the various non-traditional sterilization/decontamination processes have come into play. Sterilization is the procedure of destroying all microorganisms in or on a given environment to prevent the spread of infection. Under these scenarios, multiple potential methods for sterilization have begun to explore [7–9]. The thermal heat, radiation or chemical agents usually do this. Many potential methods of sterilization have been explored already, and they are based on chemical and physical processes. Chemical methods include liquid/vapor hydrogen peroxide, chlorine dioxide, ethylene oxide, bleach, alcohol, soap solution, ethylene oxide and ozone decontamination. Physical methods include dry/steam heat treatment, microwave oven, hot water, UV light sterilization, electron beam and ionizing radiation [10–12]. All the above sterilization methods have advantages and disadvantages compared to one and others [7,13,14]. Health care facility would likely employ various methods to address the shortage of PPE based on their resources and capabilities. Some sterilization methods have shown promising results; however, degradation of FE is noticed in some other sterilization methods and hence, the effectiveness needs to be assessed to ensure proper protection of the wearer [15–19]. The methods such as soaking/dipping in alcohol, chlorine-based, soap and others damage the fibers of certified facemasks and significantly degrade respirator filtration ability [15,16]. Deterioration of filtration ability happens because many certified masks use fibers with a static charge, which get disoriented by dipping in alcohol/liquids. There are serval other factors that also need to be assessed, like mask handling requirement, throughput, the ability to decontaminate, shadowing effect, cost effectiveness, which require specialized equipment and expertise to perform decontamination process and safety of operating personal [19,20]. The effectiveness of certified N-95, non-woven and fiber/textile masks has been extensively reviewed [6,21,22]. The comparison of various sterilization methods has been compiled based on the FE, breathing resistance, integrity and handling requirements [17,23].
To cope up the growing need of the usage of masks, it is essential that, reusability of these fabric masks should be carried out by adopting a suitable sterilization method and that should be effective at an affordable reach. The filtration efficiency of aerosols by various facemask depends on the aerosol characteristics (concentration, particle size, and charge on filter layer), filter characteristics (solid fraction, porosity, and fiber diameter etc.), environmental conditions (relative humidity and temperature) and flow rate of the air stream [24]. The breathing rate of an average adult under light activity ranges from 13–25 L/min [25], and under strenuous workload, ranges from 80–100 L/min [19,24,26]. The breathing resistance indicates the condition for usage of facemask and the accepted range is 35 and 25 mm of H2O for inhalation and exhalation resistance limit, respectively [27]. Further, the face area of the mask used for testing is taken as 17.34 cm2 [28], 23.75 cm2 [24], 59 cm2 [29], 6.4 cm2 round section of the filter media [19].
Based on the literature, mostly three types of masks are being used widely in most of the countries namely certified N-95 (health care professionals), non-woven fabric (front line workers) and textile masks (publics) and the same are used in the present study. Further, very few studies are found in the literature where the entire face area of facemask tested at a reasonable flow rate. Moreover, the recent importance of understanding the FE of the masks not only pertaining to certified N-95 respirator but also surgical and cloth mask after sterilization. Besides, the usefulness of different fabric masks in blocking respiratory droplets depends on the weave, weft, and number of layers. Earlier studies have also shown that mask efficiency is related to the mesh's closeness and gauge thicknesses [30]. We have taken two variants of fabric masks, one being two-layer fabric mask of the same fabric (cotton) and another two-layer cloth mask of a different material (cotton and gada) The gada textile details can be found elsewhere [31]. The filtration efficiencies of these masks were first tested and then sterilized using the methods mentioned above, and then efficiency is evaluated. Further, the effectiveness of five different types of certified N-95 mask is investigated before and after decontamination.
In the present work, we used some of the common and affordable methods which include autoclaving for 30 and 60 minutes, ionizing radiation (15 and 25 kGy), dry heat (30 and 60 minutes), soaking in 10% concentration of hydrogen peroxide for 30 minutes, hot water washes with and without detergent. The FE and breathing resistance of each type of facemask are evaluated as a function of aerosols size (0.3–10 µm) and laboratory-generated aerosols concentration (104–106/L), flow rate (20 and 90 L/min) for the five sterilization methods. The structure of the facemask also evaluated before and after sterilization. This article presents a detailed experimental process, materials and methods, data analysis, results, discussion, and conclusion.
Materials and methods
The evaluation of Filtration Efficiency (FE) and breathing resistance of different types of masks carried out in a test facility called, HEPA Filter Testing Laboratory at Radiological and Environment Safety Division, Indira Gandhi Centre for Atomic Research (IGCAR). The experimental setup consisting of a stainless-steel cylindrical duct which has provision for fixing the facemask without any air leakage. The upstream and downstream aerosol concentrations are counted, and the mask's FE is determined before and after various sterilization methods. The details of the test setup, types and quantity of facemasks, essential data acquisition systems, sterilization methods, and evaluation of FE of masks for ambient and laboratory-generated aerosols explained in this section.
Experimental setup
The schematic diagram of the experimental arrangement for facemask testing shown in Figure 1. It consists of a cylindrical duct of 8.5 cm internal diameter with upstream and downstream segment and a middle region to connect facemasks media. The total length of the test rig including upstream and downstream length is 190 cm. The concentration measurements in the upstream and downstream are taken at 45 cm away from the connection flange to get uniform concentration along the entire duct. The experimental setup also includes aerosols generator, aerosols diagnostic instruments, velocity meter, differential pressure monitor, relative humidity, and temperature monitoring system of the air stream. The details of equipment’s used in the present studies, their description and main measured characteristics are explained in the Table 1. The test specimen (without cutting the facemask) connected between two flanges fitted with O-rings and mounted in the duct without any air leakage (the mask edges were utterly sealed). The wanted airflow rates are achieved through suction by using an air displacement pump and flow controller. The aerosols are sampled before and after of the specimen when aerosol-air suspension passes through the facemask.

Schematic presentation of the experimental setup.
The details of equipment’s used in the present studies, their description and main measured characteristics.

Test aerosols generation and aerosols diagnostic instruments
All types of facemasks were tested for FE with ambient and laboratory-generated aerosols. The aerosols suspended in ambient air are continuously introduced at the inlet of the test rig. Poly Alfa Olefin (PAO) aerosols are generated using Laskin-Nozzle aerosol generator and used as polydispersed test aerosols to test the facemasks. The aerosol sample is taken from the upstream and downstream simultaneously using two Optical Particle Counter (OPC). The OPC works on light scattering principle and measures the particle diameter in the size range of 0.3–20 µm. The OPC's reproducibility is ± 3% for the entire measuring range of OPC (0.3 to 20 µm). Hence, the acceptable accuracy in the measurement of aerosols number concentration from OPC is ± 3%.
Types of protective facemasks
Three types of facemasks N-95 (certified facemasks), non-woven fabric (equivalent to surgical mask) and self-made double-layer cotton fabric mask (public choice during the pandemic) have been used in the present studies. The types of face masks and their description along with their image is summarized in Table 2. Five types of certified N-95 facemask commonly used by healthcare professionals are selected to examine whether they maintain their performance and integrity after single time sterilization. The certified masks have two variants viz. (i) three layers (outer, filter and inner) and (ii) four layers (outer, filter, middle and inner layer). The outer and inner layer of mask are made of spun-bond polypropylene fiber, filter layer is made of melt-blown polypropylene fiber, and middle layer is made of modacrylic fiber [6]. The effectiveness of non-woven fabric masks taken for the study consists of 3 layers of spun-bond polypropylene fiber (20–25 GSM) [32]. The textile masks that are taken for the study consists of two variants viz. (i) having same fabric for both outer and inner layers; that are stitched from a bundle cloth made of plain weave (one warp by one weft thread) having 80 × 52 counts per square inch and (ii) having different fabric with outer layer is made from plain weave (90 × 56) and inner is a gada cotton fabric (∼ 180 GSM) with plan weave (52 × 44).
Description and types of facemasks along with their optical image.

The structure of same and different fabric masks is pleated and flat, respectively. When these masks fitted to the sampler holder, care has been taken that masks should be flat but not stretched, and there should be no air leakage from the edges. The material area density of textile (fabric mass per unit area) is determined by weighing a specified area 1 inch2 (2.54 × 2.54 cm2) of punch of material. The fabric weight has been calculated based on the punch of all layer and flatted fabric surface. An analytical microbalance with an accuracy 0.1 mg used for the weighing of masks and the error in measurement of the length of cut facemask specimen is 0.1 cm. Further, macro and microscopic analysis of the physical structure of each kind of facemasks were examined by the optical microscope.
Sterilization/decontamination methods
The sterilization methods employed in this study are ionizing irradiation, dry heat, autoclave (steam), soaking in H2O2 liquid and hot water wash with and without detergent for various types of facemasks and included in Table 1. The antibacterial and virus decontamination test has been performed many laboratories across the world simultaneously and few researchers have been reviewed and compiled the literature [33–35]. Nevertheless, the published relevant antibacterial test and virus deactivation studies relevant to the scope of the paper has been reviewed and included in the Table 3. The facemask sterilization methods and duration of the process are selected based on the literature pertaining to the deactivation of bacterial and virus test with different sterilization methods. The sterilization methods have been preferred for each kind of masks based on the availability of sterilization infrastructure at an affordable condition for the various community of people. The certified N-95 masks are sterilized by gamma radiation, autoclave, dry heat and H2O2 liquid soaking. The non-woven masks are sterilized by gamma radiation, autoclave, dry heat, H2O2 liquid soaking and hot water wash (with and without detergent). The fabric/textile masks are sterilized by gamma radiation, autoclave, and hot water wash (with and without detergent).
There are five sterilization methods used in the present studies and their antibacterial and virus deactivation test with sterilization time is summarized from the literature.

The sterilization by ionizing radiation of different kind of masks achieved by using gamma irradiation for the desired dose level (15 and 25 kGy). The typical radiation dose for medical equipment sterilization is 24 kGy and deactivation of virus (SARS–1) is around 10 kGy [36]. The dry heat decontamination of facemasks for 30 and 60 minutes is achieved by hot air oven with air temperature ranging from 70–80 ± 3 °C. The deactivation of SARS-CoV-2 is achieved by three order in 60 minutes at 70 °C air temperature [40,44]. Steam sterilization is carried out by autoclave chamber and sterilization process takes place at a pressure of 103.4 kPa and at a temperature of 121 °C. The facemasks sterilized for 30 minutes and 60 minutes by autoclave. Autoclave decontamination is 100% effective in eradicating Bacillus spores [16] and SARS-CoV-2 [41]. The facemask sterilization with 10% concentration of H2O2 (diluted with distilled water) is carried out by soaking for 30 minutes. The liquid H2O2 sterilization method virtually eliminates SARS-CoV-2, H1N1 virus and spores of Geobacillus stearothermophilus [40,45,46]. A common man approach of sterilization of facemask (textile/fabric and non-woven) is by washing using hot water with and without detergent by soaking it for 30 minutes. The water temperature kept in the range of 70–80 °C, which is required to deactivate coronavirus [37]. The photograph of gamma irradiator, hot air oven, autoclave sterilization chamber, H2O2 sterilization are given in supplementary material (Figure A1). The facemask, which is in liquid submersion, soaking in H2O2 and the autoclave has been dried overnight before performing the FE testing in the room environment, where the temperature and humidity are in the range of 25–30 °C and 60–80%, respectively.
Measurements of differential pressure across the facemask, face velocity, relative humidity, and temperature of the air stream
The pressure drops or differential pressure (ΔP) was measured using an instrument across the tested masks. The measuring range and error in the measurement of ΔP are -381 to +381 and ±1% of reading + 0.13 mm of H2O. The differential pressure across the mask is an indicator of the condition of comfort and breathability. The air face velocity in the test section is measured with an air velocity meter. The measuring range and error in the measurement of face velocity are 0–50 m/s and ± 3% of the reading. The K type thermocouple and humidity sensor are used to measure temperature (T) and relative humidity (RH) of the air stream. The measuring range of T and RH with error are 10 to 60 ± 0.3 °C and 5 to 95 ± 3% respectively.
Data acquirement and analysis
The FE of three different types of facemask is calculated using the following formula:
Results and discussion
Qualification of an experimental setup and typical experiment parameters
To begin with, the experimental test rig is tested for zero efficiency condition using ambient aerosols. The upstream and downstream counts are recorded without a facemask. The average upstream and downstream aerosol counts as a function of aerosols size are given in Figure 2. The difference in the upstream and downstream counts are found to be 10–30 #/L for the aerosol size 0.3–0.7 µm and after that difference is significantly less (1–3 #/L). The average total number of particles in the upstream and downstream in the test rig without a face mask for 20 and 90 L/min flow rate is given in Table 4. The zero efficiency of the test rig with uncertainty was tested using ambient aerosols and found to be 2.07 ± 0.55% and 2.35 ± 0.65% for 20 and 90 L/min, respectively.
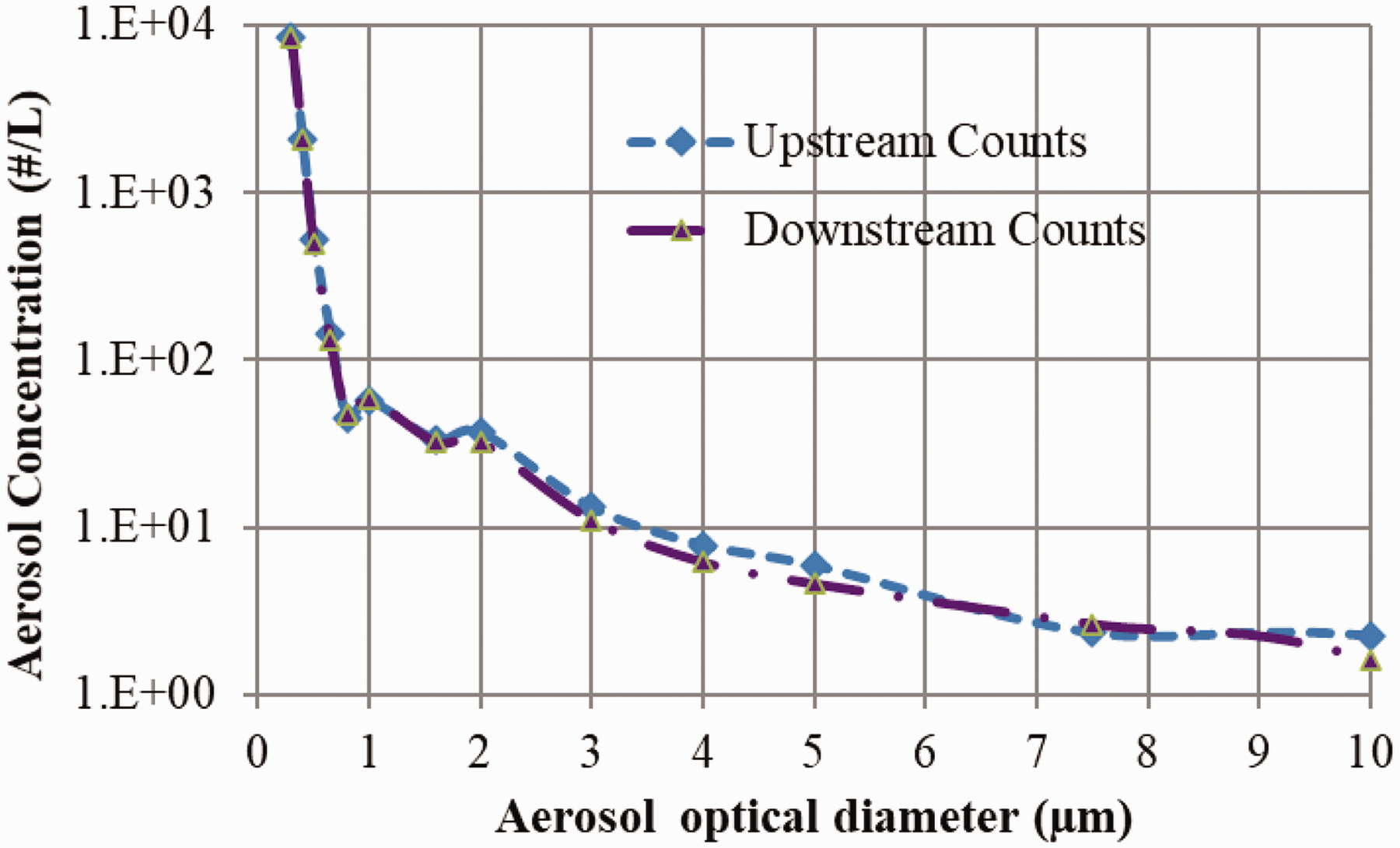
Upstream and downstream average aerosols count as a function of optical diameter measured by OPC.
The zero-efficiency test of the test rig for ambient aerosols.
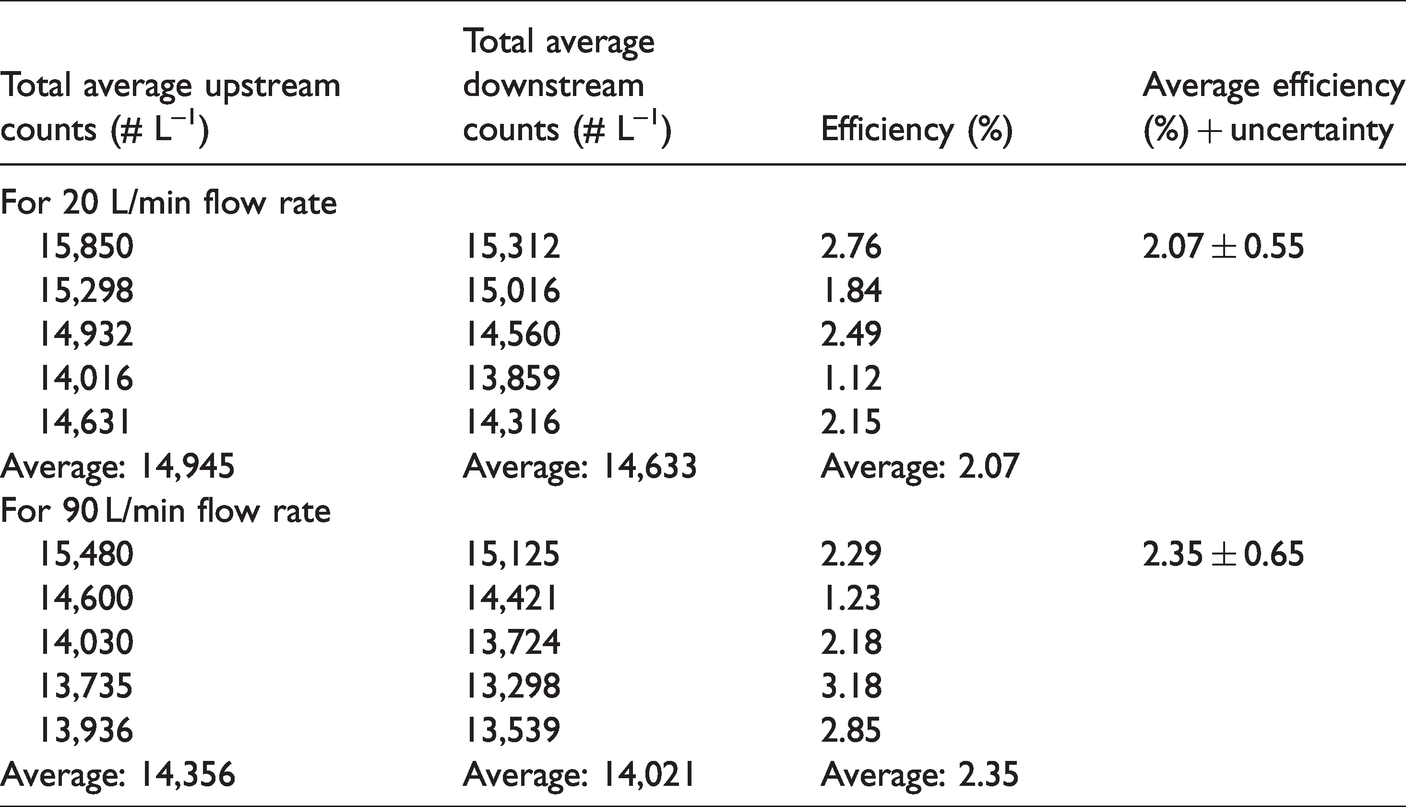
A correction factor for zero efficiency is included in the estimation of uncertainty in FE of the tested facemask. The measured average aerosol concentration at the upstream and two-flow rates of 20 and 90 L/min for ambient and laboratory-generated aerosols are presented in Figure 3(a) and (b), respectively. A significantly lower aerosol concentration is observed in the upper size range for both flow conditions, that is, for aerosol greater than 10 µm. Data excluded above this threshold (10 µm) for all the results presented in the paper due to the low concentration, which may upsurge statistical error.

Aerosol’s concentration as a function of aerosols optical diameter, two flow rates, (a) ambient and (b) laboratory-generated aerosols.
The non-woven (pleated) and fabric masks (pleated and flat) are mostly rectangular (length greater than breadth), and the certified mask are cup/pouch (duckbill) shape (it was neither rectangular nor circular). Hence, it ensured that the most exposed portion of the facemask is fitted in the test rig and tested. The effective tested area of the various masks was 56.74 cm2. The face area used for testing of facemask/filter is in line with previous research work viz. 6.4 cm2 [19]. 17.34 cm2 [28], 23.75 cm2 [24], 59 cm2 [29] and 78.5 cm2 [48].
The FE also depends on the environmental conditions (RH % and temperature) apart from air stream flow rate, aerosol characteristics, and filter fiber characteristics. It is customary to mention at which environmental condition the experimental work has been performed. The facemask shall be tested at temperature ranges from 16–32 °C (23 ± 2 °C) and the relative humidity of the air at 45 ± 15% [49]. The measured ambient air temperature during the experiments is 27 ± 0.3 °C, and RH % is 60–70 ± 3% which is the predominated atmospheric condition in our region. Hence, testing at the ambient environment condition and that of the standards is nearly same. Interestingly, it is worth mentioning here that, the masks are conditioned at 85 ± 5% RH to simulate the actual usages condition during breathing, because breathing creates high humidity [50,51]. The aerosols were blown through the mask with a face velocity of 5.8 ± 0.2 and 26.4 ± 0.9 cm/s corresponding to 20 ± 0.2 and 90 ± 1.0 L/min flow rates, respectively. The European standard is followed where penetration of material is evaluated at a flow rate of 95 L/min [49]. Many researchers in different countries have used the flow rate in the range of 20–30 L/min and 80–100 L/min [19,24,26]. Before starting the experiment, the tested facemask is conditioned for about 30 minutes in an environment where temperature and humidity are 25–30 °C and 60–80%, respectively.
Differential pressure drops across the facemasks
The measured pressure drops (ΔP) across the various facemasks before and after sterilization is given in Table 5. As expected, the ΔP across all type of facemasks increases with an increase in face velocity (flow rate). The average pressure drops of certified masks ranges from 3.2 ± 0.22–4.27 ± 0.24 (Magnum), 1.02 ± 0.14–1.54 ± 0.18 (Venus and Lot No. 210307) mm of H2O for 20 L/min and 24.46 ± 0.43–26.4 ± 0.47 (Magnum), 5.33 ± 0.21–8.10 ± 0.25 (Venus and Lot No. 210307) mm of H2O for 90 L/min.
The measured pressure drops across the facemasks before and after sterilization.
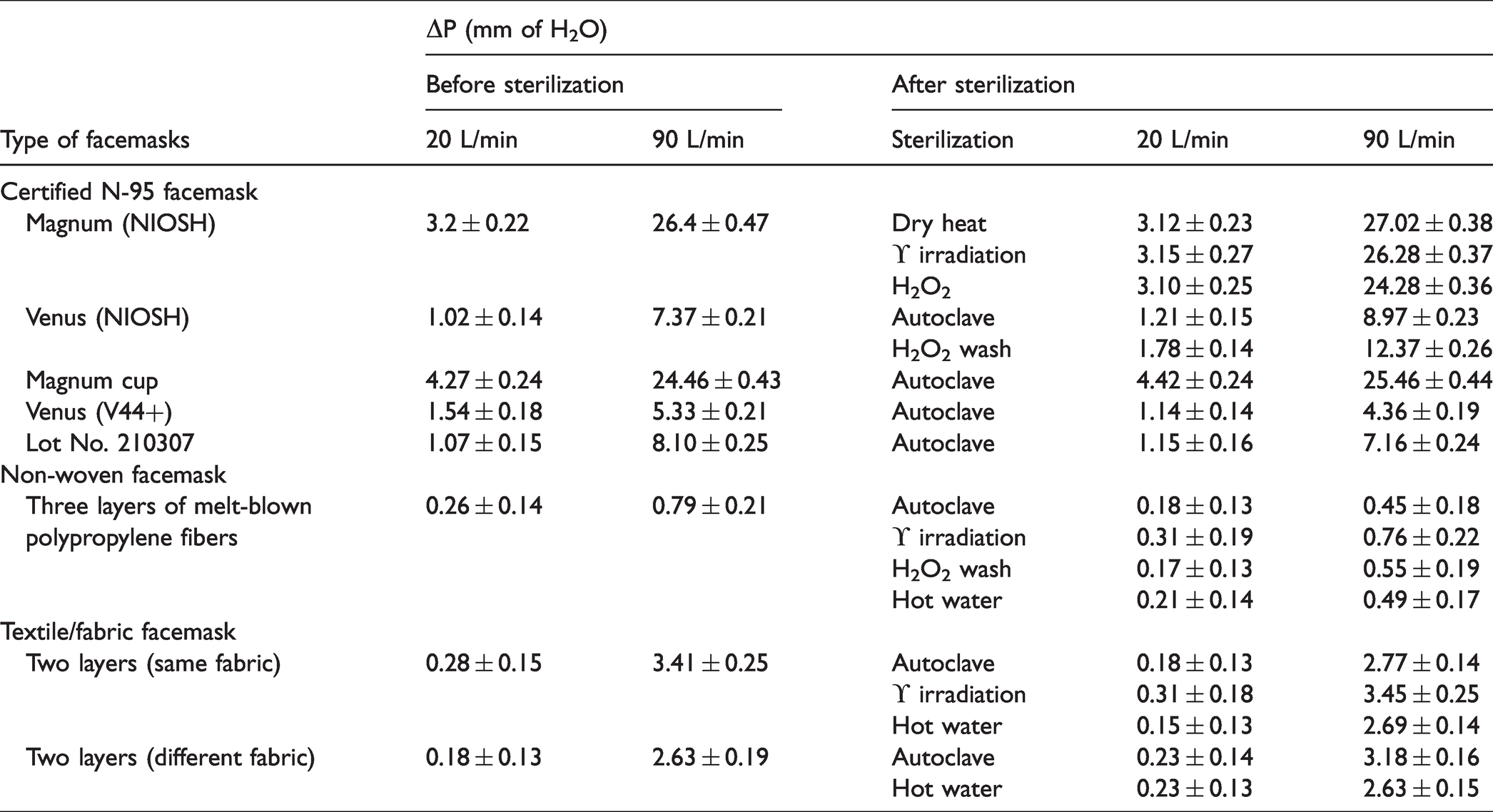
The pressure drop for Magnum mask is found to be higher than other types of certified mask for both flow conditions. The ΔP across Magnum (NIOSH) has remained almost same after dry heat, gamma and H2O2 sterilization (significant difference p < 0.05) while for Magnum cup-shaped mask, the ΔP slightly increased after autoclave sterilization (p < 0.05). For Venus (NIOSH) masks, the ΔP is slightly increased (p < 0.05), while for V-44+ facemasks, ΔP is decreased (p < 0.05) for both flow conditions. The ΔP slightly increased for certified masks (Lot No. 210307) for low flow rate while slightly decreased for high flow rate than the control masks. The average pressure drops of the control non-woven mask found to be 0.26 ± 0.14 and 0.79 ± 0.21 mm of H2O for 20 and 90 L/min flow rate. The average ΔP of the non-woven mask after sterilization ranges from 0.17 ± 0.13–0.31 ± 0.19 and 0.45 ± 0.18–0.76 ± 0.22 mm of H2O for 20 and 90 L/min flow rate, respectively. The average pressure drop of the non-woven mask is found to be same after gamma sterilization, while, for other decontamination it is slightly reduced for both flow conditions (p < 0.05). The average ΔP of control fabric masks (each layer has same fabric) is found to be 0.28 ± 0.15 and 3.41 ± 0.25 mm of H2O for 20 and 90 L/min flow rate, respectively. The average ΔP of control fabric masks (each layer has a different fabric) is found to be 0.18 ± 0.13 and 2.63 ± 0.19 mm of H2O for 20 and 90 L/min flow rate, respectively. The pressure drops across both fabric type of facemask showed that there are no significant measurable variations observed after decontamination. The change appears within the statistical error, i.e., there is no physical change in bulk material density (fabric mass per unit area) of face mask and threads counts after decontamination. The average pressure drop for non-woven and fabric masks is less than certified masks for both flow conditions. The breathing resistance indicates the condition for usage during breathing and found to be in the accepted range, i.e., 35 and 25 mm of H2O for inhalation and exhalation resistance limit, respectively [27]. The average pressure drop with uncertainty across the various control face mask is compared for 20 and 90 L/min flow rate and shown in supplementary material (Figure A2).
Evaluation of FE of control masks for ambient aerosols of size 0.3–10 µm
The FE of various types of control facemasks is determined with ambient aerosols for particle size 0.3–10.0 µm, with five size intervals 0.3–0.5, 0.5–1.0, 1.0–3.0, 3.0–5.0 and 5.0–10.0 µm, for two flow rates and given in Table 6. The FE is least for Most Penetrating Particle Size (MPPS: 0.3–0.5 µm) for all three type of mask and both flow rates. The FE of certified masks is significantly larger than the non-woven and fabric masks for all measured aerosol size ranges. FE is higher for low flow rate (20 L/min) than high flow rate (90 L/min) for all types of certified and non-certified masks. Even the Most Penetrating Particle Size (MPPS) is filtered with an efficiency of more than 97.37 ± 0.69% except for Venus (NIOSH) mask for all size range of aerosols and 20 L/min flow rate. Similarly, the FE is higher than 92.58 ± 1.59% except for Lot No. 210307 facemasks for all size range of aerosols and 90 L/min flow rate. The minimum FE is 93.44 ± 1.49% for Venus (NIOSH) and 82.98 ± 1.50% for N-95 (Lot No. 210307) facemask for 20 and 90 L/min. The certified facemasks have electrostatic filters that are efficient at low airflow velocity (flow rate) rather than high airflow velocity [25]. The FE increases with an increase of aerosols optical diameter and data variability increases with the flow rate rising from 20 to 90 L/min, which is more at larger aerosol optical diameter for non-certified masks. The FE of the fabric mask is less than a non-woven mask for both flow rates. The fabric mask having a different fabric layer is performing with slightly lower efficiency than the same fabric layered masks. The same layered fabric mask has higher thread counts, which help in trapping the particles more efficiently. A comparison of average FE for MPPS (0.3–0.5 µm) with an uncertainty of various certified and non-certified control face mask for 20 and 90 L/min flow rate is given in supplementary material (Figure A3). The statistical variability in FE is more for textile/fabric masks than the non-woven mask and least for certified masks. Further, statistical variability in all facemasks is more for 90 L/min flow rate than 20 L/min.
FE of individual control facemasks (certified, non-woven and fabric) at a flow rate of 20 and 90 L/min for ambient aerosols.

Evaluation of FE of certified facemasks (magnum) after different sterilization
The certified (Magnum NIOSH) masks are sterilized with the help of dry heat for 30 and 60 minutes, gamma irradiation for 15 and 25 kGy dose and 10% H2O2 concentrated liquid soaking; after that FE is evaluated for two flow rates and shown in Figure 4. The FE of the mask remains consistent to that of the control mask within statistical uncertainty after dry heat and H2O2 sterilization for both flow rates. The Magnum NIOSH facemasks offered higher FE (98.53 ± 0.79%) at 20 L/min. At the higher flow rate (90 L/min), the FE found to be about 95.16 ± 0.69% for the ambient aerosols. Further, the FE of Magnum NIOSH facemasks decreases significantly (p < 0.05) for less than 1.0 µm optical diameter after gamma sterilization (15 and 25 kGy) for both flow conditions. The percentage of FE reduction after sterilization with ionizing radiation ranges from 28.71–28.12% and 33.06–32.09% for 20 and 90 L/min flow rate, respectively, for 0.3–0.5 µm particles. The reduction in FE is more for higher flow rate/face velocity. The FE is more for 20 L/min than 90 L/min in all the sterilization conditions.

FE of certified (Magnum NIOSH) facemasks after gamma, dry heat and H2O2 sterilization for 20 and 90 L/min flow rates.
The particle filtration takes place by five basic mechanisms viz. gravitational settling, inertial impaction, interception, diffusion, and electrostatic interaction [21,52,]. The certified mask consists of electrostatic filtration media capable of capturing and retaining fine aerosol through electrostatic interaction and mechanical processes [53]. It is known that the least FE efficiency is associated with particles in the range of 0.3–0.5 µm that is bigger for diffusion and smaller for interception; hence, the 99% filtering efficiency is achieved for this range by electrostatic interaction. When the filter media loses its charges (charge density), the particles are captured only by mechanical processes where the efficiency reduced from 99 ± 0.09% to 63.73 ± 1.09%. In the case of 1.0 µm aerosols, the FE was not reduced even after gamma irradiation. FE of mechanical filters has the minimum efficiency at MPPS and increases with an increase or decrease of aerosol diameter from MPPS [54]. The FE remain unchanged or more than 95% benchmark for aerosols optical diameter greater than 1.0 µm for tested sterilization methods (dry heat for 30 and 60 min, gamma irradiation for 15 and 25 kGy dose and H2O2 sterilization).
Further, the certified Venus (NIOSH) facemasks are sterilized with moisturized heat (Autoclave) for 30 and 60 minutes and 10% H2O2 concentrated liquid soaking; after that, FE is evaluated for two flow rates and shown in Figure 5. The FE of Venus (NIOSH) masks found to be 94.61 ± 1.04% and 94.70 ± 1.12% for 20 L/min and 90 L/min flow rate respectively after 30 minutes of autoclave sterilization. The average FE did not get changed significantly after 30-minute autoclave sterilization. However, for 60 minutes autoclave sterilization, FE found to be 92.49 ± 1.89% for 20 L/min flow rate (not significantly changed) and 86.50 ± 1.99% for 90 L/min flow rate (significantly reduced p < 0.05) for the optical diameter of aerosols 0.3–0.5 µm. In the case of H2O2 sterilization, FE found to be increased from 93.44 ± 1.49 to 97.28 ± 0.89% for 20 L/min flow rate and 92.58 ± 1.59 to 97.68 ± 0.81 for 90 L/min flow rate for an optical diameter of aerosols 0.3–0.5 µm. The increase in FE (p < 0.05) after autoclave and H2O2 sterilization might be due to absorption of moisture by the fiber layers of the mask [55,56], and thickness of the overall filter layer might be increased (not measured in this communication), which is reflected in breathing resistance. The observation is equivalent to other published data [19,57]. The pressure drops for Venus (NIOSH) increased after H2O2 sterilization, which also reflected in FE (p < 0.05) of the face mask.

FE of certified (Venus NIOSH) facemasks after autoclave and H2O2 sterilization for 20 and 90 L/min flow rates.
The other three certified facemasks (Magnum cup shape, V-44+ and Lot No. 210307) are sterilized with an autoclave for 30 minutes only, and FE is presented in Figure 6. The FE of the mask decreased from control masks for both flow rates. The FE of Magnum cup shape, V-44+ and Lot No. 210307 facemasks reduced from 97.37 ± 0.69 to 96.24 ± 0.52% (p > 0.05), 99.56 ± 0.09 to 92.53 ± 0.69% (p < 0.05) and 98.38 ± 0.87 to 85.27 ± 1.29% (p < 0.05) respectively for 20 L/min flow rate. Similarly, for 90 L/min flow rate, there is a significant reduction in FE of Magnum cup shape, V-44+ and Lot No. 210307 facemasks from 93.39 ± 1.69 to 88.20 ± 1.39% (p < 0.05), 96.08 ± 0.78 to 89.34 ± 1.02% (p < 0.05) and 82.98 ± 1.5 to 79.14 ± 2.01% (p < 0.05) respectively. The reduction of FE after autoclave sterilization is more for Magnum cup shape and V-44+ facemasks in high flow conditions than the low flow rate condition.

FE of various certified facemasks after autoclave sterilization for 20 and 90 L/min flow rates.
Evaluation of FE of non-woven fabric masks after different sterilization
The non-woven facemasks are sterilized with the help of steam (autoclave) for 30 and 60 minutes, gamma irradiation for 15 and 25 kGy dose, 10% H2O2 concentrated liquid soaking, hot water soaking with and without detergent; after that, FE is calculated and shown in Figure 7. As expected from filtration principles, the minimum FE is found to be in between 0.3–0.5 µm particles for all untreated and processed non-woven masks and increases with an increase of aerosol optical diameter. The mean FE of untreated non-woven masks is 24.72 ± 2.54% and 20.78 ± 3.26% for low and high face velocity respectively, for 0.3–0.5 µm aerosol. The average FE is found to be comparable after gamma, H2O2 and hot water sterilization and found to vary from 21.66 ± 2.33–29.86 ± 1.49% and 16.52 ± 2.44–22.29 ± 1.59% for 20 and 90 L/min flow rates respectively for ambient aerosols size 0.3–0.5 µm.
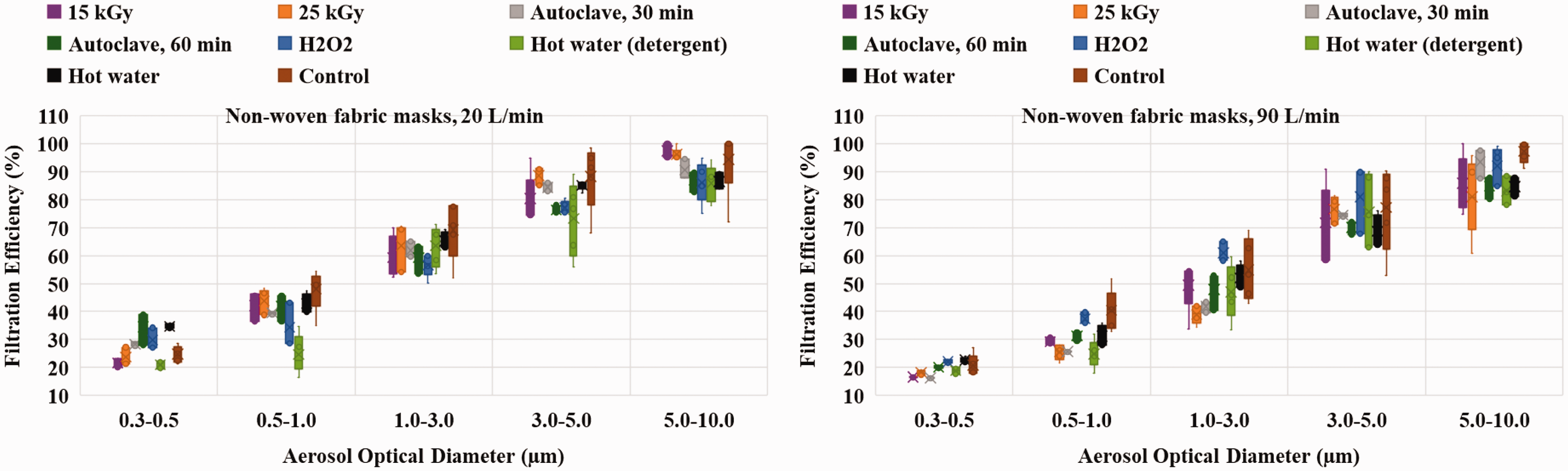
FE of non-woven fabric masks after gamma, autoclave, H2O2 sterilization and hot water with and without detergent sterilization for 20 and 90 L/min flow rates.
The average FE after autoclave sterilization increases from 24.72 ± 2.54 to 30.34 ± 1.025% (p < 0.05) for low face velocity while for high face velocity decreases from 20.78 ± 3.26 to 16.28 ± 3.52%. The FE increased more for 60-minute autoclave sterilization for both flow rates than 30-minute autoclave. In the case of hot water with detergent sterilization, the FE found to be in the range of 24.72 ± 2.54 to 21.01 ± 2.62% for 20 L/min and 20.78 ± 3.26 to 18.79 ± 2.98% for 90 L/min flow rate. The FE of the non-woven face mask is around 20–25% for MPPS. The spread in FE is less at MPPS and increases with larger aerosols for both face velocity. The lower efficiency attributed to the density of fibers in the non-woven type masks. However, these masks are adequate for the particles in a size range of 3.0 µm and above (about 60% efficiency).
Evaluation of FE of textile/fabric masks after different sterilization
The home-made double layer fabric facemasks (both layers have the same fabric) are sterilized with the help of autoclave for 30 and 60 minutes, gamma irradiation for 15 and 25 kGy doses, hot water soaking with and without detergent; after that FE, is calculated for two flow rates and shown in Figure 8.

FE of textile/fabric (same fabric) facemasks after gamma, autoclave, and hot water with and without detergent sterilization for 20 and 90 L/min flow rates.
The average FE of fabric masks found to be same after gamma, autoclave, and hot water (with and without detergent) sterilization as that of control mask and found to vary from 19.69 ± 1.44–25.86 ± 2.49% and 17.52 ± 2.84–20.29 ± 3.29% for 20 and 90 L/min flow rates respectively for ambient aerosols of size 0.3–0.5 µm. The average FE increases with an increase of aerosols optical diameter for both face velocity before and after sterilization. The home-made double layer fabric facemasks (different fabric) sterilized with an autoclave for 30 and 60 minutes and hot water (with and without detergent); after that, FE is calculated for two flow rates and shown in Figure 9. The average FE of masks found to be unchanged after sterilization and ranges from 18.28 ± 1.56–23.52 ± 2.79% for 20 L/min and 14.98 ± 1.87–19.83 ± 2.09% for 90 L/min for the aerosol size 0.3–0.5 µm. The mean FE increases as aerosol optical diameter increases for higher sized aerosols for both sterilized and non-sterilized conditions. The FE is relatively less for different fabric layer masks than with that of the same fabric double-layered facemask. The reason is attributed to the texture (lesser thread counts) of the second layer (inner side) in the different fabric mask. The FE of different layer fabric masks increased significantly after decontamination (p < 0.05). The uncertainty in FE found to be more for the large optical diameter for both fabric masks.

FE of textile/fabric (different fabric) facemasks after autoclave, hot water with and without detergent sterilization for 20 and 90 L/min flow rates.
Comparison of FE of various type of masks after different sterilization for ambient and laboratory-generated aerosols
The performance of multiple types of facemasks after diverse sterilization methods is evaluated using laboratory-generated aerosols and compared with that of ambient aerosols. The efficiency of the facemask also depends on the aerosol concentration apart from other parameters like aerosols particle characteristics, filter fiber characteristics (solid fraction, porosity and fiber diameter and charge on filter layer etc.), environmental conditions and flow rate of the air stream [24]. This type of analysis may be beneficial for some specific context where a person exposed to the higher aerosol’s concentration. The average number of bio-aerosols generated per cough by Influenza patient is 7.5 * 104 and the count median diameter (CMD) is between 0.6 and 0.9 µm with Geometric Standard Deviation (GSD) 1.53 to 2.28 [58]. The aerosols particle emission rate ranges from 102 to 103/sec during various activities (breathing, talking, and singing) with aerosol size 0.5–5 µm [59]. Similarly, another study mentions that the average number of droplet/aerosols expelled per cough by a person with a respiratory infection is 4.9 * 106. Most of the aerosols are in sub-micron [60]. Another study shows that 80% of droplets centered in the range of 0.74–2.12 µm during coughing and sneezing [61]. In this context, the mask's filtering efficiency needs to be investigated for high aerosols concentration (105–106 #/L). Table 7 shows that the FE of various types of facemasks for ambient and laboratory-generated aerosols (two order more aerosols concentration) after different sterilization methods for the two flow rate conditions and MPPS (0.3–0.5) aerosol optical diameter. The certified mask shows slightly higher FE for laboratory-generated aerosols (high aerosols concentration) than ambient aerosols even after sterilization for both flow rate conditions. The measured upstream aerosols count, for ambient aerosols < 1.0 µm size, ranges from 97.45–97.75% and > 1.0 µm, ranges from 2.55–2.25% for 20 L/min while for 90 L/min 98.03–98.22% and 1.97–1.78% respectively. Similarly, measured upstream aerosols count for laboratory-generated aerosol for < 1.0 µm is 95.04–95.37% and for > 1.0 µm is 4.96–4.63% for 20 L/min: while for 90 L/min is 88.90–89.52% and 11.10–10.48% respectively. The % of laboratory-generated aerosol concentration is higher for > 1.0 µm aerosols than ambient aerosol. This improves the overall FE efficiency for particles greater than > 0.3 µm, which is reflected in the higher aerosol’s concentration case. The increase of particle number concentrations enhances aerosol removal efficiency, and higher the ratio of large to small particle concentration further enhances this effect [62]. The FE of the non-woven mask found to be reduced for high aerosols concentration. However, the FE of both fabric masks has been increased for laboratory-generated aerosols. The FE of all three types of the mask is less for 90 L/min flow rate when compared to the 20 L/min, due to higher filtration capability for sub-micron sized aerosols by diffusion and interception at a low flow rate. The certified mask performed better in higher aerosols concentration; however, performance is slightly reduced for non-certified facemasks.
Comparison of FE of various types of facemasks for ambient and laboratory-generated aerosols after different sterilization.

Macro and microscopic analysis of the physical structure of masks
The macro and microscopic analysis of the physical structure of certified, non-woven and fabric/textile mask was carried out by using an optical microscope and gravimetric analysis. An optical microscope image of the fabric mask is shown in Figure 10. The thread count (yarn/inch2) is denser and thicker in the same fabric layer mask than in different fabric mask. The threads counts are more on the front side than on the backside of the different layer mask. The gap between threads decreased after autoclave and hot water sterilization for both cloth masks. Since, after sterilization by the above two methods, thread diameter enlarges (swell up) due to the hydrophilic nature of the fabric mask. This observation is qualitative, but it reflected in FE of the fabric mask, where FE increased after decontamination for aerosols larger than 1.0 µm [55,56]. The optical image of the same fabric layer and different fabric layer of all sterilized mask is shown in Figure 11.

An optical microscope image of fabric/textile mask (a) same fabric layer, (b) frontside of the different fabric layer and (c) back side of the different fabric layer mask. The scalebar represent 500 µm in length.

An optical image of fabric/textile masks after autoclave, hot water and gamma irradiation sterilization (a) same fabric mask, (b) frontside of different fabric and, (c) backside of different fabric. The scalebar represent 500 µm in length in each image.
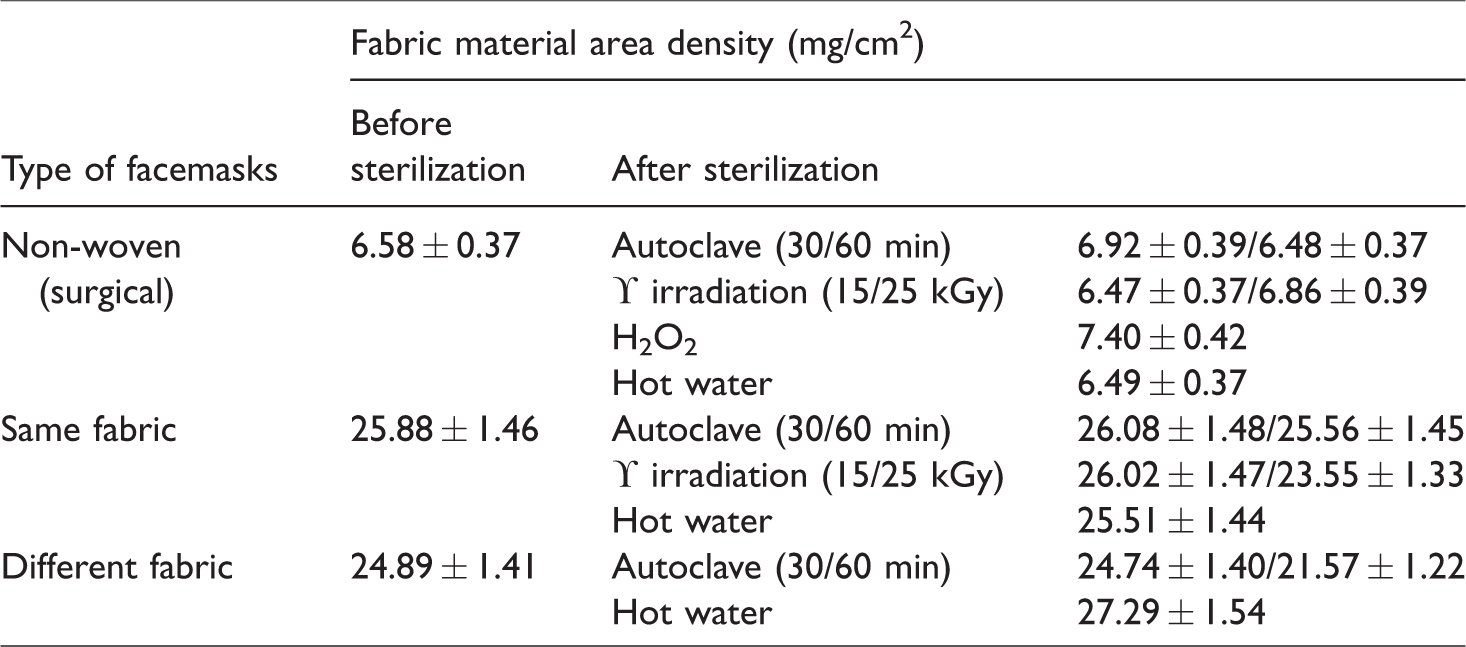
Further, material areal density of both fabric masks before and after various sterilization is measured and given in Table 8. The average material areal density of same and different fabric layer mask is 25.88 ± 1.46 and 24.89 ± 1.41 mg/cm2, respectively. The material areal density remained almost same before and after decontamination and the variation is not significant. The fabric material area density is more for the same fabric layer mask when compared to the different fabric layer mask; this may be the reason for the superior performance of the same fabric layer mask than different fabric layer mask.
The materials area density of the fabric and non-woven mask before and after various sterilization.
The non-woven mask consists of a three-layer viz. repellant (outer) layer, the filter (middle) layer and absorbent (inner) layer. A microscopic image of the outer, filter and inner layer of the control non-woven facemasks is shown in Figure 12. Each layer of non-woven mask made of spun-bond polypropylene fiber layers with fiber diameter range from 30–40 µm.

Optical microscope image of the non-woven mask (a) outer, (b) filter and (c) inner layers. The scalebar represent 100 µm in length.
The optical image of the polypropylene fiber layer of non-woven mask after different sterilized is shown in Figure 13. There is no observable change in the polypropylene fiber structure of non-woven mask after different sterilization methods. The materials areal density of the non-woven fabric mask is included in Table 8. The areal density is 6.85 ± 0.37 mg/cm2 for the control facemask and remains almost same after various processing.

Optical image of the outer (a), filter (b) and inner (c) layers of non-woven mask after different sterilization (gamma radiation, autoclave, H2O2 and hot water). The scalebar represent 100 µm in length for the outer, inner, middle layers.
The certified masks were disassembled to understand their structural components before and after sterilization. An optical image of four-layer of certified control mask is shown in Figure 14. The spun-bond polypropylene fiber diameter (inner and outer layer) ranges from 18–22 µm, melt-blown polypropylene fiber diameter ranges from 1–5 µm and modacrylic fiber diameter range from 15–25 µm.

Optical microscope image of various layer of the certified mask (a) outer, (b) filter (c) middle and (d) inner layers. The scalebar represents 200 µm in length for the outer, inner, middle, and 100 µm for the filter layer.
The optical image of the certified mask fiber layer after different sterilization is shown in Figure 15 (three-layer mask) and Figure 16 (four-layer mask). There is no observable and significant change in morphology of the fiber found after various sterilization processes. The International Atomic Energy Agency has also specified that there is no significant change in the mask's texture concerning the fit factor of the facemask at 24 kGy radiation dose, which is needed to kill viruses and bacteria [63]. There is no observable visual change found after autoclave; however, the masks were shrunken, stiff and the plastic locking piece on the cord (black color) used in the facemasks for wearing is slightly melted. No visible change observed for 30–and 60 -minutes hot-air sterilization at a temperature 70–80 ± 3 °C. Soaking the facemasks in 10% H2O2 for 30 minutes produced no unaided visual change on the mask's layers. However, after H2O2 sterilization, the printed matter on the surface of facemasks is appeared to be faded.

Optical image of three-layer certified magnum mask (a) outer, (b) filter and (c) inner layers after gamma irradiation, dry heat and H2O2 soaking sterilization. The scalebar represent 200 µm in length for outer and inner layers and 100 µm for filter layers.

Optical image of four-layered certified magnum mask (a) outer, (b) filter, (c) middle and (d) inner layers after autoclave and H2O2 soaking sterilization. The scalebar represent 200 µm in length for outer, middle, and inner layers and 100 µm for filter layers.
Conclusions
Three types of facemasks viz. certified N-95, non-woven fabric and self-made double layer fabric/textile masks have been evaluated for particulate FE and pressure drop for two-flow rate condition viz. 20 and 90 L/min before and after various sterilization using ambient aerosols in a size range of 0.3–10 µm. Further, the FE has been compared with laboratory-generated aerosols (two order high aerosol concertation) of size 0.3–0.5 µm. The macro and microscopic evaluation of facemask fiber/fabric has been carried to characterize whether the decontamination processes caused any significant damage to the mask. The pressure drops increase with increasing face velocity; this reflects that it is harder to breathe through a facemask when the respiratory flow is large. The measured pressure drops during mask testing found to be the accepted range (the inhalation and exhalation resistance limit viz. 35 and 25 mm of H2O respectively) after sterilization. The performance of non-woven fiber masks and fabric/textile masks (double layer of the same fabric) for 0.3 to 10 µm optical diameters and for both flow rate is nearly same, but FE is slightly less for fabric masks than that of non-woven masks. The FE of fabric and non-woven masks increases with an increase of optical diameter from MPPS, and the efficiency decreases at a high flow rate than the low flow rate. The fabric masks sterilized using autoclave for 60 minutes have shown slightly better efficiency. The FE of non-woven masks remains same before and after decontamination for both flow conditions. The structure analysis shows that the non-woven and fabric mask remain intact after different sterilization. However, 60 minutes of autoclave sterilization damage elastic cords of the non-woven masks.
In the case of the certified mask, the FE of the magnum (NIOSH) mask found to be more than 95% after sterilization except for gamma irradiation. FE of certified facemask is found to be identical before and after dry heat sterilization for 30 and 60 minutes. The FE reduces to about 70% for most penetrating aerosols (0.3–0.5 µm) after gamma sterilization for 20 L/min flow rate and still lesser with a higher flow rate (90 L/min). The FE of other certified masks found to be reduced slightly after 30 and 60-minutes autoclave sterilization for both flow condition. FE of the certified facemasks remains same the control mask after the H2O2 sterilization, but the ink marks present in the outer surface is faded.
Face masks can provide additional protection when some measures, particularly social distancing, is challenging to maintain. The effectiveness of facemasks depends on consistent and proper use. Though the non-woven masks have slightly better efficiency than the double layer fabric masks, the efficiency can match if the fabric/textile masks have more dense thread counts preferably. We recommend using fabric/textile mask for the common public and ensuring that the fabric masks can be sterilized with any of the above-tested method (preferably hot water sterilization) before subsequent re-uses. The fabric/textile mask is quite good substitute of non-woven mask (but not for certified N-95 mask) as it prevents the inhalation of suspended droplets carrying virus in the environment and it also reduces the burden of micro plastic to the environment and slow down the spread of infection. The certified masks can be sterilized with dry heat and H2O2 vapors. Further, the autoclave sterilization methods are also recommended, but the stream temperature should be kept reasonable (∼ 100 °C) so that the elastic cords of the masks remain in good condition. The thermal decontamination can be performed at low cost in conventional ovens, which are widely available in commercial kitchens, laboratories, and sterilization facilities. Thermal disinfection can thereby provide a feasible solution at an affordable reach for all conditions (normal wear or during an emergency).
We highlight a few recommendations from the current studies: The breathability and FE test conveys that the fabric masks with a double layer, tight weaves, low porosity, and high thread count are a potential substitute for non-woven masks. We recommend that hot water washing (at 80–100 °C temperature) with or without detergent is quite suitable for sterilisation of fabric masks which is very much affordable for the common public. It is beneficial for the filtration of large respiratory droplets. Certified masks, which made of electret filtering media, are not recommended for sterilisation by ionising radiation; it will compromise the filtering efficiency. However, it suggested that masks can be sterilised with thermal decontamination and H2O2 vapours.
Supplemental Material
sj-pdf-1-jit-10.1177_15280837211028794 - Supplemental material for Evaluation of filtration effectiveness of various types of facemasks following with different sterilization methods
Supplemental material, sj-pdf-1-jit-10.1177_15280837211028794 for Evaluation of filtration effectiveness of various types of facemasks following with different sterilization methods by Amit Kumar, Basundhara Bhattacharjee, DN Sangeetha, V Subramanian and B Venkatraman in Journal of Industrial Textiles
Footnotes
Acknowledgement
The authors acknowledge Dr. S. Athamalingam, Associate Director, Health, Safety and Environmental Group (HSEG), Dr. R.Venkatesan, Head, Radiological and Environmental Safety Division (RESD) and Dr. C. V. Srinivas, Head, Radiological Impact Assessment Section (RIAS) for their encouragement and support to carry out this work. The Dr. Ch. Kishan Singh acknowledged for analysis of various layers of the mask by optical microscope. The authors would like to thank the reviewers for their valuable comments and suggestions, which significantly improved the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
