Abstract
Functional polymeric membranes with antibacterial properties have gained significant importance in many applications. Silver NPs offer advantage over other materials for their effective antibacterial properties and being safer for humans at low concentrations. The synthesis of silver NPs may not always be environmental friendly and their incorporation in the polymer membranes is usually a multistep process. In this study, PVDF/PVP/AgNPs electrospun membranes were developed in a single step process where silver NPs were synthesized using reducing and stabilizing properties of PVP. The UV-vis spectroscopy confirmed the synthesis of silver NPs in PVP solution by sharp absorption peak at 398 nm. The membranes were loaded with various concentrations of silver NPs (1, 1.5, 2 and 2.5 wt%). The scanning electron microscopy of the developed membranes showed nano fibers of uniform diameter at optimized electrospinning conditions. FTIR spectroscopy also confirmed the successful development of polymeric composite (PVDF/PVP/AgNPs) membranes. The composite membranes demonstrated effective antibacterial properties against
Keywords
Introduction
Electrospinning is considered one of the methods for preparation of membranes [1,2]. It is more simple, versatile, reliable, and cost-effective compared with other techniques [3]. This method has been used widely for the manufacturing of polymeric membranes such as ultrafiltration, nanofiltration and microfiltration membranes [4,5]. These polymeric membranes are used for filtration of various pollutants such as sand particles, dyes, metals, effluents and pathogens from water [5–7]. For development of membranes, polymers [8] and ceramics [9] are widely used materials [8,9]. Polymeric membrane [10] offers desirable properties such as, ease of fabrication, robustness, cost effective, flexibility, high processability and reliability as well as readily availability [10]. Various type of polymeric membranes has been reported for water purification such as polysulfone, polypropylene, chitosan/polyamide, polyvinyl alcohol polyether sulfone, cellulose based and PVDF for water treatment and bacterial disinfection [11–14]. PVDF based membranes are preferred due to high chemical stability, excellent functionality, high permeability, cost effectiveness thermal resistance and higher efficiency [10]. PVDF membranes are used for nanofiltration through different modifications. This is carried out by physical blending, surface modification and chemical grafting [15]. Generally, the performance and surface properties of PVDF membranes have been improved by adding nanofiller, chemical additive and nanoparticles [16–18]. Addition of nano materials enhanced hydrophilicity, performance, functionality, mechanical strength, and permeability of polymeric membrane [19]. Metal nanoparticles such as silver, silica, gold, have been used to impart functionality in polymeric membranes [20–24]. Among various metal nano particles, silver nano particles are most widely used for water disinfection and wound healing [25]. Ibrahim et al. [26] reported that silver nanoparticles synthesized using banana peel extract showed good antibacterial capabilities. Silver is known for excellent antibacterial activity in its various chemical forms. Among them silver nanoparticles are widely studied in recent years due their possible usage as a strong bactericidal material with an application in water disinfection. The outstanding antibacterial activity is because of well-developed surface and large surface area to volume ratio providing better contact with microorganism. Moreover, even at low concentration silver shows excellent antibacterial activities [27,28]. Mostly, these nano particles are prepared by bottom up and top-down approaches. Formation of nano particle from colloidal dispersion and quantum dots are an example of bottom up technique [29].
Various techniques have been presented in literature for development of functional membranes [30]. Techniques including physical and chemical means were developed to prepare nano particles such as, electrochemical reduction, sol-gel method, photochemical reduction, heat evaporation and chemical reduction [31,32]. The physical ways require higher temperature and require expensive equipment’s while chemical reduction cause formation of by-products, environmental issues, multistep preparation, stability of nano particle is difficult to maintain, time consuming, require higher cost and require reducing agents or chemicals which are normally hazardous [10]. In this study, reducing properties of PVP were exploited to synthesize silver nano particles and develop PVDF/PVP/Ag NPs composites membranes with antibacterial properties. The development of such PVDF composite membranes loaded with silver NPs synthesized using PVP has not been reported in literature to the best of our knowledge. The developed membranes were characterized for their surface morphology, fiber diameter, pore size distribution, synthesis of Ag NPs, composition and antibacterial performance.
Materials and methods
Polyvinylidene fluoride (PVDF, Mw = 320000 g/mol−1), and Polyvinyl pyrrolidone (PVP, Mw = 13,00,000 g/mol−1) were purchased from Sigma Aldrich. Analytical grade Silver nitrate (AgNO3) and dimethylformamide (DMF) were also procured from Sigma Aldrich. All chemicals were used with no further purifications. Double needle electrospinning machine from Linari was used for the development of membranes.
Preparation of electrospun membranes
First, a 7.5 wt.% of PVP was dissolved in DMF with magnetic stirring at room temperature for 5 to 6 hours to prepare a homogeneous polymer solution. After that, different concentrations of AgNO3 (1, 1.5, 2 and 2.5 wt.%) were added to the PVP solution and stirred for 3 to 4 hours. Gradually, the silver ions were reduced, and the colorless polymer solution turned pale yellow and finally dark brown. Figure 1 represents the synthesis of silver nanoparticles in PVP solution.

Scheme of synthesis of Ag NPs in PVP solution and development of composite membranes via electrospinning.
PVDF solution (24 wt.%) was prepared separately by dissolving PVDF in DMF and kept on stirring for 6 to 8 hours at 600 rpm at 60 °C. Finally, both PVDF and PVP/Ag NPs solutions were mixed together with magnetic stirring at room temperature for 3 to 4 hours to obtain PVDF/PVP/Ag NPs solution. This solution was loaded in a 10 ml syringe containing needle at its end. The syringe was inserted on an infusion pump. The needle of the syringe was connected to the positive terminal of power supply while the negative terminal was connected to the collector covered with aluminum foil. A high voltage (18KV) was applied between the needle tip and collector separated at16 cm. The flow rate of solution was kept at 1.5 ml/hr. These electrospinning parameters were selected after initial trials with the given solutions.
Characterization
The surface morphologies of nanofibrous membranes were studied by using the scanning electron microscopy (SEM Quanta 250) and the ImageJ software was used to check the diameter of these nanofibers. The diameter distribution and pore size distribution of the membranes were obtained by analyzing SEM images in ImageJ. The functional group and chemical structures of developed membranes were determined by Fourier transform infrared spectroscopy (FTIR-spectrum two, PerkinElmer) in the wavelength range from 4000-600 cm−1. UV-Vis spectrophotometer (Lamda 950 and manufacturer PerkinElmer) was used to assess formation of silver NPs in the solution.
The antibacterial activity of developed membranes was evaluated using disk diffusion test, a qualitative method to evaluate antibacterial activity. The samples were tested against
Results and discussions
For the development of antibacterial electrospun membranes, silver nitrate was dissolved in PVP solution for the synthesis of silver NPs. Figure 2(a) to (c) shows the change in color of the solution reduction as silver nitrate salt was reduced to silver NPs. Initially, the color of pure PVP solution was white and by adding silver nitrate salt (AgNO3) the polymer solution turned pale yellow as shown in Figure 2(b). With continuous stirring, the solution turned dark brown (Figure 2(c)) indicating the synthesis of silver NPs. Further, the synthesis of the silver NPs in PVP solution was evaluated using UV-vis spectroscopy.

(a) PVP solution (without silver nitrate), (b) PVP/Ag nano particles (pale yellow color), (c) PVP/Ag nanoparticles after continuous stirring (dark brown color).
The UV-Vis spectroscopy was carried out on pure PVP and PVP/AgNPs solution as shown in Figures 3 and 4 respectively. UV-vis spectra of PVP solution is shown in Figure 3. Pure PVP solution did not show any absorption in UV-vis spectra.
UV-Vis spectrum of pure PVP solution.

UV-Vis spectrum of PVP/AgNPs solution.
On the other hand, UV-vis spectra in Figure 4 clearly indicated the formation of silver nanoparticles by the appearance of strong absorption bands at 398 nm, which is a characteristic peak of silver nano particles [33]. Polyvinyl pyrrolidone (PVP) reduced metal ions (silver) to silver nanoparticles because of the oxidation of hydroxyl end group present in PVP that directs to the formation of an aldehyde functional group which later reduces the metal ions similar to the Tollens’s reagent.
The morphology of as spun nanofibrous PVDF/PVP/AgNPs membranes was analyzed using scanning electron microscope. By optimizing all process and solution parameters PVDF nanofibers were synthesized. Nano fibers with uniform diameter and smooth surface were prepared by dissolving PVDF polymer in DMF solution. The SEM images of nano fibers containing have been shown in Figure 5(a) and (b). Figure 5(a) shows that small particles have been deposietd because the concentration of polymer was very low, the distnace beetween needle to collector is lesser and no smooth fiber formation occured. With further increase in the polymer concentration beaded nanofibers were formed as obersved in Figure 5(b).

SEM images of nanofibers at (a) 22% polymers concentration (PVDF/PVP/AgNs), distance 12 cm, flow rate 1.5 ml/h, voltage 20kv, (b) 24% polymer concentration, distance 14 cm, flow rate 1.3 ml/h and voltage 22 kv.
The electropsinning parameters were further optimized and morphology of resulting fibers was analyzed with SEM. The Figure 6(a) shows beaded nanofibers while Figure 6(b) shows uniform nanofibers. Beads in nanofibers occured due to high flow rate of polymer solution, low concentration of solution and lesser distance between needle tip to collector. Because there is no continous jet formation and lesser time avaialble for fiber to dry off. The needle to collector tip distance was kept 16 cm, voltage 24 kV and flow rate was 1.2 ml/h. These optimized parameters lead to formation of smooth nanofibers and reduced the beaded structures.

SEM images of (a) 22% polymer concentration (PVDF/PVP/AgNPs), distance 15 cm, voltage 24 kv, flow rate 1.3 ml/h, (b) 24% polymer concentration (PVDF/PVP/AgNPs), distance 16 cm, flow rate 1.3 ml/h and voltage 24 kv.
ImageJ software was used to determine the fiber diameter distribution as well as pore size distribution of electrospun PVDF/PVP/AgNP membranes as shown in Figure 7(a) and (b). A minimum number of 64 fibers were measured for each sample. The diameter of nanofibers gradually increased from 300 nm to nearly 760 nm. The average diameter of nano fibers was observed 513 nm and sharp peak was obtained in case of nano fiber diameter. This means that fiber diameter of most nano fibers is close to the average value. Based on the SEM image it was found that fiber diameter was highly influenced by the electrospinning parameters (both process and solution) [34]. The fiber diameter increases with increasing the polymer concentration [35]. Figure 7(b) represents the pore size distribution of electrospun samples. It is confirmed from the figure that the pore sizes of electrospun membranes are in nanometer range and most are within the 100 to 300 nm range. The pore size distribution varied with the fiber diameter. The blending of PVP with PVDF can enhance the pore size of membrane and increases the hydrophilicity [36].

(a) Diameter distribution of PVDF/PVP/AgNPs nanofibers, (b) Pore size distribution of nanofibrous membrane.
The FTIR of nanofibers and functional membranes was carried out at 4000–600 cm−1 range to analyze the chemical composition of samples. Figure 8 shows the FTIR spectrum of pure PVDF nanofibrous membranes. In the graph, the peaks at 2925.6 cm−1 and 2849.8 cm−1 represent CH2 symmetric and asymmetric vibrations [37]. The peak at 1739.3 cm−1 can be associated to the CH2 wagging vibration of PVDF and the peak at 870.41 cm−1 corresponds to the C–F stretch vibration [38].

FTIR spectrum of electrospun PVDF membranes.
The FTIR spectrum of functional membranes (PVDF/PVP/AgNPs) is shown in Figure 9. The broad peaks at 3,425 cm−1 is associated with O-H stretching and peak at 1648 cm−1 can be ascribed to C=O stretching vibrations of PVP [24]. The characteristic peaks at 870 cm−1, and 1180 cm−1 and 1400 cm−1 were attributed to PVDF [39]. FTIR peaks at 1402 cm−1 and 1073 cm−1 can be attributed to the deformation vibration of CH2 and stretching band of C–C respectively [40]. The peak present at 870.41 cm−1 corresponds to C–F stretch vibration due to presence of PVDF in composite membranes. With the deposition of AgNPs, the intense absorption peak at 1662.64 cm−1 was observed in the spectrum of the PVDF/PVP/AgNPs membrane, which might be due to the presence of AgNPs, suggesting the formation of AgNPs on the membrane. Peek at 1662.64 cm−1 could not be found in pure PVDF membrane.
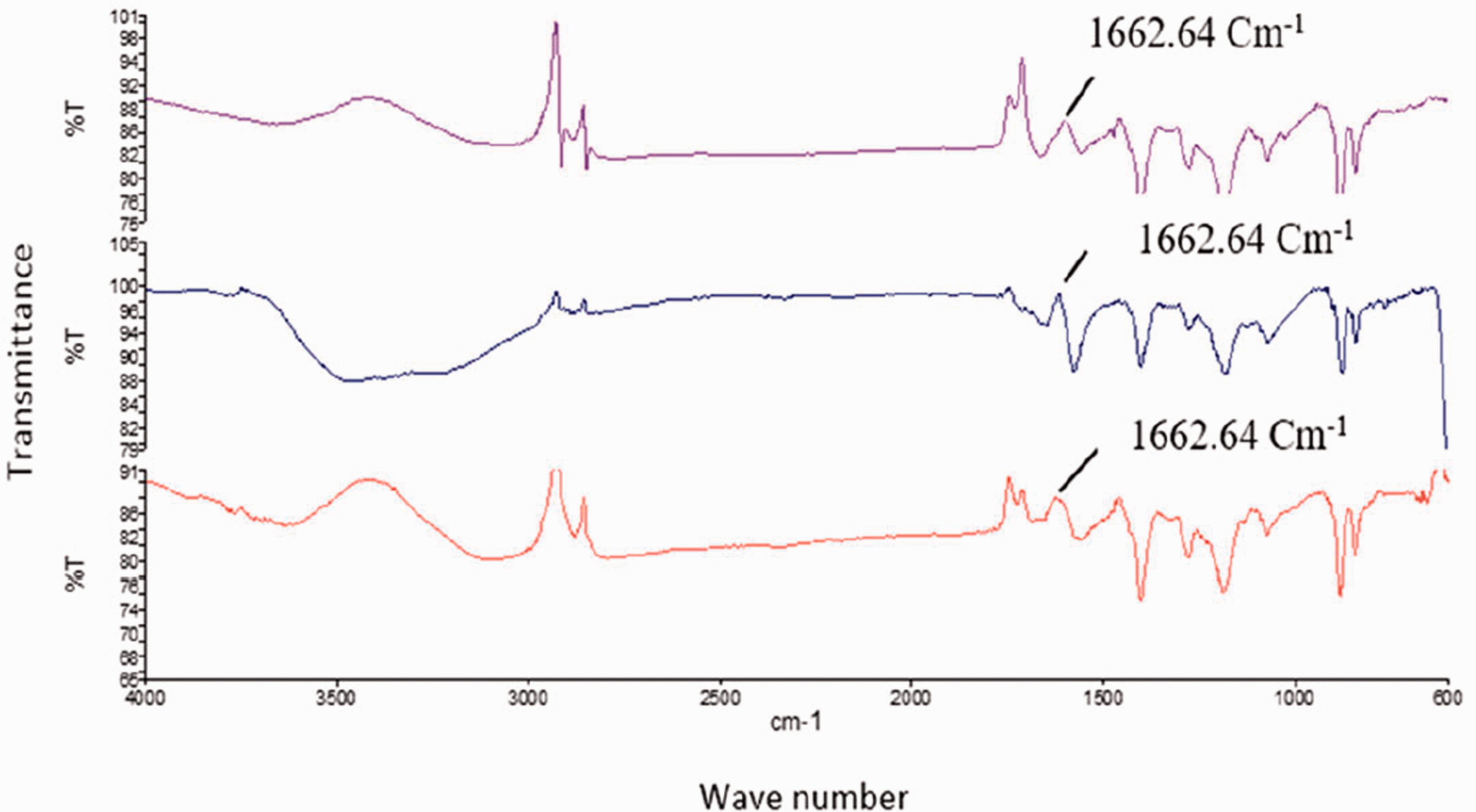
FTIR spectra of functional (PVDF/PVP/AgNPs) membrane.
The developed membranes were tested against
The membranes shown in Figure 10(a) to (d) contain silver nano particles in increasing concentration in the order: 1, 1.5, 2 and 2.5 wt% respectively. In Figure 10(a), the concentration of silver nano particle was the lowest (1%) and smaller zone of inhibition was detected. The size of zone of inhibition increased with increasing the concentration of the silver nanoparticles. Highest activity was achieved with functional membrane containing 2.5 wt% of silver nano particles. The membrane containing silver NPs did not allow the growth of bacteria on the sample surfaces and in the surrounding environment. During the test, silver NPs get oxidized and are released in the form of silver ions in the surrounding wet environment of the disk and form an inhibition halo. Silver ions penetrate in the cell membrane of bacteria and give antibacterial effect by disrupting the bacterial cell membrane. With increasing concentration of silver NPs, more silver ions will be released thus resulting in greater size of the inhibition zone in disk diffusion test. These results demonstrate that PVDF/PVP/AgNPs electrospun membranes exhibited good antibacterial properties.

Antibacterial activity of PVDF/PVP/AgNPs membrane against
Conclusion
Functional polymeric membranes containing silver nanoparticles were successfully prepared using a simple and environment friendly process. The composite membranes comprised PVDF, PVP and silver NPs. The silver NPs were synthesized exploiting reducing properties of PVP without using any additional reducing agent and composited with PVDF. The UV-vis spectra of PVP solution in DMF showed an absorption peak at about 398 nm, a characteristic peak of silver NPs showing their synthesis in the solution. FTIR results confirmed the development of composite membranes indicating peaks related to structural characteristics of PVDF/PVP/AgNPs membranes. Various concentrations of silver (1, 1.5, 2, 2.5 wt%) were added to the polymeric membranes. The developed membrane showed good antibacterial activity by forming a clear zone of inhibition around the edges of membrane samples against
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by Higher Education Commission, Islamabad, Pakistan for the completion of this research having research grant number 20-4179/NRPU/R&D/HEC/14.
