Abstract
To reduce the environmental pollution caused by medical protective materials, such as masks and protective clothing, biodegradable antibacterial materials have received more and more attention in recent years. In this study, poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) and poly(butylene-adipate-co-terephthalate) (PBAT) were electrospun together and then treated with nano-TiO2 to develop and evaluate a biodegradable, antibacterial nanofiber membrane for medical protective fabric. The SEM images displayed that the nanofiber membrane with a mass fraction of 13 and a mass ratio of 50:50 PHBV/PBAT had the smallest diameter and the best morphology of all samples. In addition, the mechanical properties test and water contact angle test results demonstrated that the PBAT/PHBV composite nanofiber membrane had better mechanical properties and hydrophobicity without compromising its fundamental structure than pure PHBV. The addition of TiO2 nanoparticles decreased the fiber diameter of this nanofiber membrane. When the TiO2 concentration was 1.0 wt%, the average fiber diameter was 367 nm, which might approach the sub-micron level. Meanwhile, the presence of TiO2 reduced adhesion between fibers of the PBAT/PHBV membrane, resulting in a more uniform fiber distribution. Additionally, the elongation at the break of the PHBV/PBAT/TiO2 nanofiber membrane with 1.0 wt% TiO2 was raised from (135 ± 5)% to (203 ± 2)%. The PHBV/PBAT/TiO2 nanofiber membrane containing 1.0 wt% TiO2 inhibited Escherichia coli and Staphylococcus aureus, and its antibacterial rate was over 98%. In this research, we successfully prepared composite materials that were both biodegradable and antibacterial, which can be applied in the field of medical protection. It can promote the development of protective textile materials in the direction of functionalization and degradation.
Introduction
The current worldwide pandemic environment has made medical protective equipment, such as surgical masks, a part of our everyday lives. 1 Healthcare personnel and the general public are in danger of infection if such personal protective equipment is not used. Generally, most masks are made of non-renewable petroleum polymers such as polypropylene, polystyrene, polycarbonate, polyethylene, and polyester, which are not biodegradable. 2 Therefore, these polyethylene-based plastics accumulate in the environment at an alarming pace of around 25 million tonnes per year, whereas such materials generally take 200 years to decompose.3,4 In this context, the development of antibacterial biodegradable composite materials will increase their application in textiles and apparel, biomedicine, and other sectors. Furthermore, this strategy can lower the danger of bacterial infection when using medical textiles while also addressing waste fiber combustion and disposal environmental issues.
Many microorganisms often generate poly-hydroxy-alkanoates (PHAs), a polyester family member, and develop unchecked in cells as carbon and energy storage. 5 The well-known PHA bioplastic is the poly(3-hydroxybutyrate-co-3-hydroxy valerate) (PHBV). The advantages of PHBV include a wide range of biological sources, low cytotoxicity, piezoelectricity, thermoplasticity, biocompatibility, biodegradation, and high crystallinity. However, its extensive applicability is restricted due to its expensive cost, material brittleness, and low impact resistance. 6 These drawbacks can be solved by combining PHBV with other biodegradable polymers. 7 Poly(butylene adipate-co-terephthalate) (PBAT) is a biodegradable aliphatic-aromatic copolyester formed by polycondensing 1,4-butanediol with adipic and terephthalic acids. 8 It has a tensile strength of 32–36 MPa and an elongation at a break of about 700%. 9 Given the sufficient toughness of PBAT, mixing it with PHBV can increase the overall qualities of composites with the same degree of biodegradability.
Nanoparticles are commonly employed as antibacterial compounds,10,11 with TiO2 being one of the most common due to the photocatalysis of TiO2. 12 TiO2 is non-toxic, environmentally friendly, low-cost, and stable. 13 Reactive oxygen species produced by TiO2 photocatalysis would attack the weak places in the bacterial cell wall, allowing internal components to seep out and then destroy the injured cells, 14 resulting in an antibacterial action. Since then, several investigations on TiO2 photocatalytic antibacterial performance have been conducted. 15 For instance, Wu et al. 16 designed a novel TiO2/SiO2/carbon electrospun nanofiber mat (TSC NFM) with flexibility and porous hierarchy. Pant et al. 17 successfully prepared electrospun nylon-6spider-net-like nanofiber mats containing TiO2 nanoparticles (TiO2NPs). TiO2 has been found in studies to exhibit antibacterial action against Gram-positive bacteria, 18 Gram-negative bacteria, 19 fungi, 20 and viruses. 21 As a result, TiO2 may be injected into the PHBV/PBAT nanofiber membrane, conferring antibacterial activity. 22
Nanofibers are thinner and have a larger specific surface area than regular fibers. 23 Electrospinning can be used to prepare nanofiber mats from diverse polymers, polymer blends, or polymers doped with other materials. 24 Electrospinning provides various benefits in the manufacture of nanofibers, including convenience, cheap cost, and mature technology.20,21 Therefore, it is widely employed in biomedical, 25 textile and clothing, 26 food packaging, 27 and a variety of other sectors. Furthermore, the electrospun filter material has a smaller pore and greater porosity than standard filter materials, allowing for efficient filtering of micron or even nanoscale pollution particles. 28 Cui et al. 29 created a novel form of air filtration membrane for outdoor use, using green electrospinning technology and a thermal crosslinking process of polyvinyl alcohol (PVA) and sodium lignosulfonate (LS). The electrospinning technique is also frequently employed in the textile and clothing industries. For example, Joseph et al. 30 combined the substrate-free electrostatic spinning process with textile technology to create a new collector design that generates continuous high-strength yarns and can be developed as core-sheath structures to generate potential nanotextiles for a variety of biomedical applications. Buttaro et al. 31 used the electrospinning technique to turn a polylactic acid-polyethylene glycol block copolymer (PLA-B-PEG) into the biodegradable, hydrophilic water-insoluble fiber. This membrane was found to be used for sweat absorption and moisture removal in clothes.
Under many environmental circumstances, biodegradation can occur. Polymer biodegradation results from a synergistic interaction of biological and abiotic variables, and numerous studies have demonstrated that abiotic forces are more significant than biological factors. Due to the photocatalytic ability, TiO2 exhibits strong antibacterial activity under the suitable light wavelength. Films of PHVB and its nanocomposites, containing TiO2 with different mass fractions, were submitted to biodegradation tests by Natália et al. 32 After testing of aqueous medium and garden soil, all samples were found to be biodegradable. As a result, TiO2 addition into a biodegradable material does not affect its breakdown function. In this study, PBAT and PHBV were first physically mixed and PHBV/PBAT nanofiber membranes with varying mass ratios and mass fractions were effectively synthesized. Then, TiO2 nanoparticles as antibacterial agents were added into PHBV/PBAT/TiO2 nanofiber membranes via the blended spinning process. The mechanical characteristics, crystallization melting properties, and hydrophilicity of PHBV/PBAT/TiO2 nanofiber membranes would be investigated using SEM, FTIR, DSC, tensile test, and water contact angle goniometer. Finally, the antibacterial characteristics of the nanofiber membrane were investigated against E. coli and S. aureus.
Experimental section
Materials
Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) and poly(butylene-adipate-co-terephthalate) (PBAT) were purchased from Shanghai Shengquan Plasticization Co., LTD. Trichloromethane, dimethylformamide (DMF), potassium dihydrogen phosphate, dinar hydrogen phosphate, and sodium hydroxide were purchased from Tianjin Fengchuan Chemical Reagent Technology Co., LTD. TiO2 nanoparticles (with diameters of 40 nm) were provided by Shanghai Maclean Biochemical Technology Co., LTD. Beef immersion powder was made by Nanjing Quan Long Biotechnology Co., LTD. Peptone from Tianjin Damao chemical reagent factory. Phosphate Buffered Saline (PBS) was made by ourselves. Agar powder from Tianjin Zhiyuan Chemical Reagent Co., LTD. E. coli and S. aureus were purchased from Nantong Kai Hengsheng Science and Technology Development Co., LTD.
Preparation of PHBV/PBAT nanofiber membranes
The mass ratio and mass fraction range of the PHBV/PBAT spinning solution was investigated. The spinning state was found to be optimal in the pre-experiment at roughly the same temperature and humidity when the mass fraction of the solution was between 12 and 15%. Therefore, we choose the middle two mass fractions (13% and 14%) to experiment with. Furthermore, as shown in Table 1, six groups of exploratory experiments were set up. Based on the pre-experiment, the PHBV and PBAT polymer particles were then blended in different mass fractions and mass ratios using chloroform/DMF (7:3) as the solvent. For 40 min, an ultrasonic cleaning machine (KQ5200E) was used to dissolve the PHBV and PBAT polymer particles in solution, the ultrasonic frequency of the ultrasonic cleaner is 40 kHz. The blend solution was then magnetically stirred for about 7 h at a speed of 10 r/s at 58°C until the mixed solution was completely dissolved. The spinning solution was then set aside for a few minutes to allow the bubbles in the solution to settle. The prepared PHBV/PBAT precursor was loaded into a 20 mL disposable sterile syringe with a 0.6 mm flat head metal needle. The spinning voltage was set to 17 kV. The flow rate was adjusted to be 1.5 mL/h. The spinning distance was 15 cm. After covering the receiver with smooth A4 paper for collection, a PHBV/PBAT nanofiber membrane was obtained.
Sample numbers and blending parameters.

Preparation of PHBV/PBAT/TiO2 nanofiber membranes
Spinning was difficult in the preliminary experiment when the mass fraction of pelleted TiO2 (40 nm) exceeds 1.0 wt%. The maximum mass fraction of TiO2 was only 1.0 wt%. TiO2 with different mass fractions (0, 0.5, and 1.0 wt%, respectively) were added to the PHBV/PBAT blend system with a concentration of 13% and a mass ratio of 50:50, ultrasonic treatment was performed for 40 min, and magnetic stirrer was used at 58°C at a speed of 10 r/s for approximately 7 h until the solution was dissolved entirely and formed. The solution was removed from the oven and set aside for a few minutes to defoam. The samples were labeled 13%PHBV50PBAT50TiO2(0%), 13%PHBV50PBAT50TiO2(0.5%), and 13%PHBV50PBAT50TiO2(1.0%) based on the different mass fractions of TiO2. The spinning procedure was identical to the preparation process of PHBV/PBAT nanofiber membranes.
Characterization of nanofiber membranes
The morphology of the PHBV/PBAT nanofiber membrane and the PHBV/PBAT/TiO2 antibacterial nanofiber membrane was examined by Scanning Electron Microscopy (SEM) (HitachiS4800). When the accelerating voltage was 10 kV, the diameter of the fiber distribution was tested and then analyzed by the image analysis software ImageJ (National Institutes of Health, USA). The Fourier Transform Infrared (FTIR) spectroscopy (PerkinElmer) was used to characterize the chemical properties of the two nanofiber membranes. It was performed using KBr pellets. and the scanning range of wavelength was 500–4000 cm1, the number of scans was 32, and the resolution was 4 cm−1. The crystallization melting performance of the prepared nanofiber membranes was tested using a Differential Scanning Calorimeter (DSC) (200F3, Netzsch, Germany). The scanning temperature range was 20°C–200°C, and the heating rate was 10 °C/min. To eliminate the thermal history, the temperature was raised from 20°C to 200°C for the first time and held for 2 min. It was then cooled to 20°C and heated to 200°C twice. The use procedure was carried out in a nitrogen-protected environment. An electronic Universal Testing Machine (INSTRON 5900, USA) was used to measure the tensile strength, elastic modulus, and elongation at the break of PHBV/PBAT nanofiber membranes and PHBV/PBAT/TiO2 antibacterial nanofiber membranes. First, the nanofiber membrane was cut into 20 × 50 mm strips for stretching. The initial distance between the two collets was set to be 50 mm, with a stretching speed of 20 mm/min. Each sample group was tested three times, and the average was calculated. Finally, water contact angles were examined to analyze the hydrophobicity of the samples using an Optical Contact Angle goniometer (OCA15pro, DataPhysics, Germany). The contact angle was estimated using the five-point fitting method.
Antibacterial tests
The antibacterial performance of the PHBV/PBAT/TiO2 nanofiber membrane was evaluated using the plate counting method. 33 Gram-positive bacteria S. aureus and Gram-negative bacteria E. coli were used in this study as the model strain. And, 0.35 g PHBV/PBAT/TiO2 nanofiber membranes with various TiO2 mass fractions (0, 0.5, and 1.0 wt%) were achieved. These samples were sterilized using an ultraviolet lamp for 2 h. The samples listed above were placed in a conical flask with 70 mL of PBS buffer which was inoculated with 5 mL of bacterial solution. The samples were sealed with eight layers of gauze and used as the experimental sample group, while a blank group was set up with no samples. For 18 h, the samples were cultured in a constant temperature shaker at 37°C and 130 r/min to obtain homogenous conditions. At the end of the culture, 100 µL drops of the diluted bacteria solution were taken and added to the sterile agar plate (the average number of colonies was between 30 and 300 CFU). The sterilized glass coating stick was used for uniform coating in the shape of Z. Three agar plates should be prepared as parallel samples for each dilution ratio, and then the coated agar plates should be placed upside down in a biochemical incubator at 37°C for 24 h (E. coli) or 48 h (S. aureus). The number of bacterial colonies in the culture dish was counted after the removal. The antibacterial effect was assessed using antibacterial rate XS. The bacteriostasis rate was calculated using the following equation.

Where XS is the bacteriostasis rate (%); Wt is the mean number of colonies in the blank group (CFU/mL); Qt is the means the number of colonies in the samples group (CFU/mL).
Results and discussion
Analysis of PHBV/PBAT blend system
Surface morphology analysis
Figure 1(a) to (c) showed that the average diameter of the fiber was the smallest with a 50% fraction of PBAT, around 500 nm. The surface of the spun fiber became smoother, the distribution of fiber diameters was more uniform, and it could provide a more extensive specific surface area. Furthermore, theoretical predictions implied that nanofibers with less than 500 nm diameter would cause a noticeable slipping flow effect, reducing airflow resistance, and superior protective products.28,29 The average fiber diameter grew larger, and the fiber surface became rougher as the proportion of PBAT in the PHBV/PBAT system increased. The average diameters of the nanofiber membranes generated by PHBV/PBAT electrospinning solution with a 14% concentration at mass ratios of 50:50, 30:70, and 0:100 were 600 nm, 1.2 and 2.8 µm, respectively, as shown in Figure 1(d) to (f). As seen in the figure, the fiber diameter was much coarser at a 14% concentration than at a 13% concentration, and the fiber diameter likewise grew coarser as the PBAT content increased. It was probable that increasing the mass fraction of solution and the PBAT concentration would increase solution viscosity. 34 Higher viscosity electrospinning polymer solutions exhibited more excellent resistance to elongation during the electrospinning process, increasing the diameter of nanofibers. 35 In other words, as the solution viscosity decreased, the revolving jet may be stretched and extended more easily. As a result, more delicate fibers could be manufactured.32,33 The high viscosity spinning solutions were prone to plugging the needle and are difficult to spin.

(a–i) SEM micrographs and diameter distribution: (a) 13%PHBV50PBAT50, (b) 13%PHBV30PBAT70, (c) 13%PHBV0PBAT100, (d) 14%PHBV50PBAT50, (e) 14%PHBV30PBAT70, (f) 14%PHBV0PBAT100, (g) 13%PHBV50PBAT50TiO2(0%), (h) 13%PHBV50PBAT50TiO2(0.5%), and (i) 13%PHBV50PBAT50TiO2(1.0%).
FTIR analysis
FTIR was used to characterize the PHBV/PBAT blend system to investigate the reaction connection between the PHBV and PBAT components. Infrared spectra of various PHBV/PBAT mixes with mass fractions of 13 and 14% were given in Figure 2. The chemical structures of PHBV and PBAT were shown in Figure 3 and the infrared characteristic peaks of PHBV and PBAT were reported in Table 2. 1724 cm1 corresponded to the stretching vibration peak of the C=O in the PHBV molecule. 36 And, 3459 cm1 corresponded to the stretching vibration peak of the hydroxyl terminal. It suggested the presence of water molecules in the fibers. The peaks at 2931 and 2861 cm1 were assigned to the antisymmetric stretching vibration and symmetrical stretching vibration of methylene -CH2. 728 cm1 corresponded to the stretching vibration. The blend system comprises the typical absorption peaks of PHBV and PBAT, as determined by infrared spectrum analysis. By comparing the infrared spectra of pure PBAT and PHBV/PBAT nanofiber membranes, it was possible to determine that no additional characteristic peaks occur in the blend system. No variation in the stretching vibration band of any distinct group was observed. This showed that PHBV and PBAT did not form a chemical connection or that the force between them was extremely low and weak under the experimental conditions. The blending of PHBV with PBAT was a straightforward physical blending process.
Infrared characteristic peaks of PHBV and PBAT.


The Fourier transform infrared (FTIR) spectra of samples.
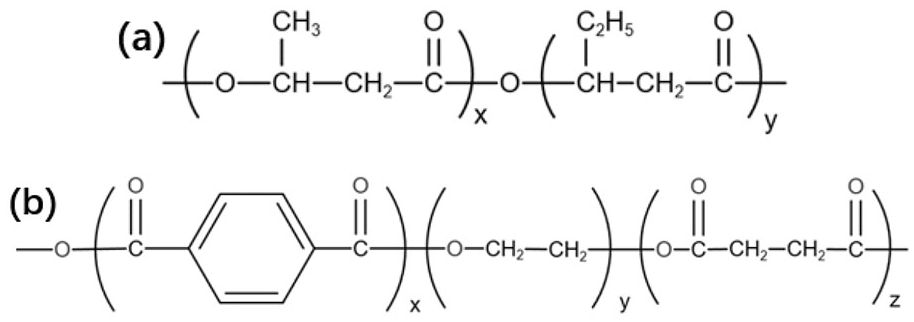
The chemical structure of: (a) PHBV and (b) PBAT.
DSC analysis
The Differential Scanning Calorimeter (DSC) was used to investigate the crystallization and melting characteristics of PHBV/PBAT nanofiber membranes with various mixes. Secondary heating eliminated thermal history, resulting in a more lucid graph. In Figure 4(a), the DSC heating curves for samples 13%PHBV50PBAT50 and 14%PHBV50PBAT50 revealed that the PHBV/PBAT nanofiber membranes had two melting peaks. The low-temperature melting point (Tm1) and the high-temperature melting point (Tm2) of PHBV/PBAT nanofiber membranes with a mass ratio of 50:50 was (152 ± 1)°C and (167 ± 1)°C, respectively, with the high-temperature melting point taking the lead. In the DSC test, double or multiple melting peaks may happen because partial melting and recrystallization may occur during the heating process due to the difference in crystal form or the different molar mass of the substance. 37 However, there was no discernible heat transfer in the heating curve of pure PBAT, and no melting point was visible between (152 ± 1)°C and (167 ± 1)°C. Thus, the two-fold melting peak seen in the PHBV/PBAT nanofiber membrane may result from PHBV melt-recrystallization-remelting process. The melting temperature remained constant when the fraction of PBAT in the PHBV/PBAT blend system was increased, but the area of the low temperature melting peak (Tm1) and the high temperature melting peak (Tm2) decreased, which may be owing to PBAT’s diluting impact on the crystallization of PHBV.

DSC thermograms of PHBV/PBAT samples of different blend systems and TiO2 samples: (a) (c) the second heating curve and (b) (d) the cooling curve.
The cooling curve of the PHBV/PBAT nanofiber membrane was shown in Figure 4(b). Pure PBAT crystallizes at (45 ± 1)°C, whereas PHBV/PBAT nanofiber membranes exhibited two exothermic peaks, one at around 100°C and the other at approximately (60 ± 1)°C, throughout the cooling process. The two exothermic peaks were likely crystallization peaks for PHBV and PBAT in the mix. With the rise of PBAT content, the crystallization temperature (Tc1) of PHBV in the mix did not change much, as shown in Table 3. Compared to pure PBAT, the crystallization temperature (Tc2) of PBAT in the blend rose from (45 ± 1)°C to around (60 ± 1)°C. In addition, it is possible to assess whether the mixture changes the crystallinity of the precursor based on the room temperature crystallinity (Xc) of the polymer and nanocomposites. 38 By calculating the room temperature crystallinity of different blending systems, we found that the incorporation of TiO2 does not affect the crystallization and melting behavior of the PBAT/PHBV. 39
DSC data on different samples obtained from second heating and cooling curves.

Mechanical property analysis
The stress-strain curves of PHBV/PBAT nanofiber membranes with various blending methods were shown in Figure 5(a). As Table 4 illustrated the specific mechanical properties of PHBV/PBAT nanofiber membranes. When PHBV/PBAT had a mass fraction of 13% and a mass ratio of 50:50, and the tensile strength was (0.7 ± 0.2) MPa and the elongation at break was (138 ± 2)%. When PHBV/PBAT had a mass ratio of 30:70 and a mass fraction of 13%, the tensile strength was (1.3 ± 0.6) MPa, and the elongation at break was (211 ± 2)%. The fiber membrane of PBAT exhibited an elongation at break of up to (407 ± 1)% and tensile strength of (2.3 ± 0.4) MPa. It may be inferred that increasing the PBAT concentration improved the tensile strength of the mix while increasing the elongation at break. 40 Because PBAT possessed strong toughness, the toughness of the PHBV/PBAT combination rose as the PBAT content increased, 41 and the tensile fracture behavior likewise changed from brittle fracture to ductile fracture, proving that PBAT increases the elasticity of PHBV. Similar phenomenon had been found in experiments by Jost and Miesbauer 42 exploring PHBV mixing with nine different biopolymers and polymers to improve the processing and mechanical properties of films. Tensile strength and elongation at break were both enhanced when the mass fraction of the PHBV/PBAT nanofiber membrane was raised to 14% compared to 13%. This might because when concentration and PBAT ratio rise, the fiber diameter thickens, increasing the breaking strength and elongation at the break of the nanofiber membrane. Furthermore, when the mass fraction and PBAT percentage rose, so did the elastic modulus.
Tensile data of PHBV/PBAT samples of different blend systems and samples of TiO2 samples.

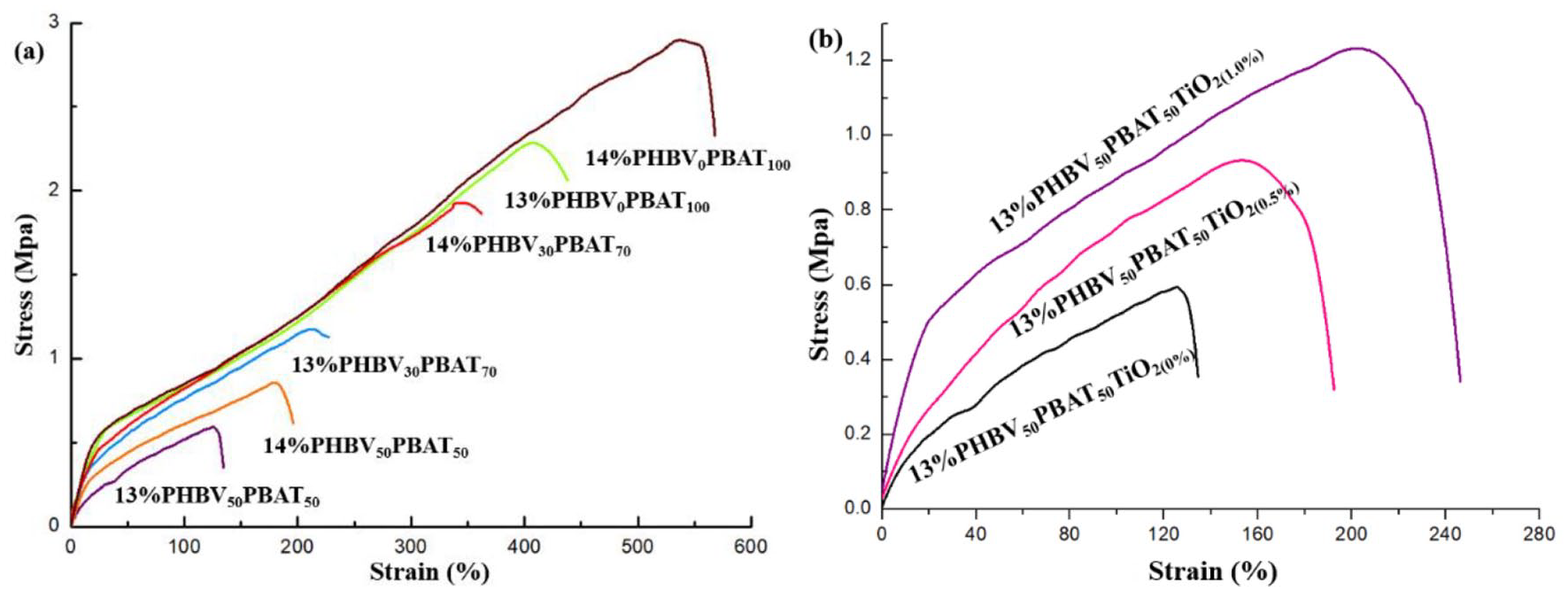
The stress-strain curves: (a) PHBV/PBAT samples and (b) TiO2 samples.
Contact angle analysis
The contact angle was employed to assess the wettability of the nanofiber membrane. And Figure 6(a) to (f) showed that both the PHBV/PBAT nanofiber membrane and the pure PBAT fiber membrane exhibited contact angles larger than 90°, indicating hydrophobicity. In Figure 6, the water contact angle was (109.61 ± 1.0)° with the PHBV/PBAT mass ratio of 50:50 and the mass fraction of 13%. The material hydrophobicity increased with the steady increase of the PBAT ratio material hydrophobicity. The water contact angle of pure PBAT may reach (137.1 ± 1.0)°. Besides, SEM micrographs of pure PBAT fiber membrane demonstrated that the fiber surface was rougher, indicating higher hydrophobicity. 43 However, when the mass fraction of the PHBV/PBAT mix system grew, the hydrophobicity of the materials diminished because the diameter of the spins caused various pore widths. If the substance was hydrophobic, the thinner the silk, the smaller the stacking pores, and the higher the hydrophobicity. The water contact angle of PHBV/PBAT nanofiber membrane findings revealed that samples with varying mass fractions and mass ratios were all hydrophobic.

Water contact angles of PHBV/PBAT nanofiber membranes and PHBV/PBAT/TiO2 nanofiber membrane prepared of different mass fractions: (a) 13%PHBV50PBAT50, (b) 13%PHBV30PBAT70, (c) 13%PHBV0PBAT100, (d) 14%PHBV50PBAT50, (e) 14%PHBV30PBAT70, (f) 14%PHBV0PBAT100, (g) 13%PHBV50PBAT50TiO2(0%), (h) 13%PHBV50PBAT50TiO2(0.5%), and (i) 13%PHBV50PBAT50TiO2(1.0%).
According to FTIR and DSC results, PHBV and PBAT mixing did not affect the chemical structure of the two. On this basis, the mechanical properties test and water contact angle test demonstrated that the increase of PBAT could improve the mechanical properties and hydrophobicity of the material. And it was found from the comparison of the SEM results that when the mass fraction was 13%, the mass ratio of PHBV and PBAT was 50–50, the fiber diameter was the smallest, and the surface was the smoothest of all samples. Thus based on the findings of the prior studies, TiO2 was only added to the nanofiber membrane with a mass fraction of 13% and a mass ratio of 50:50 PHBV/PBAT for further assessment in the following sections.
Analysis of PHBV/PBAT/TiO2 blend system
Surface morphology analysis
Figure 1(g) depicted the fiber surface morphology of 13%PHBV50PBAT50TiO2(0%). The fiber had an average diameter of 500 nm, a smooth surface, and a generally uniform diameter distribution. The fiber diameter was lowered by 0.5 wt% TiO2, and the average diameter increased to 470 nm. The fiber was continuous and uniformly dispersed, with no beads between the fibers, and the shape of the fiber was improved. When the TiO2 concentration was 1.0 wt%, the average diameter of the fiber was 367 nm, which can approach the sub-micron level. The adhesion between the fibers was reduced, as was the fiber dispersion. The added TiO2 would enhance the solution’s conductivity. Consequently, under the influence of an electric field, it was simple to produce the fiber by massive jet elongation, 44 therefore lowering the diameter of the fiber.
Furthermore, no particle attachment was discovered for nanofiber membranes with TiO2. TiO2 was known to have a size of around 40 nm, much smaller than the diameter of the fiber. 45 TiO2 may be distributed evenly throughout the fiber. We found that 1.0 wt% TiO2 was shown to be the best.
FTIR analysis
Fourier Infrared Spectroscopy was utilized to characterize the effect of TiO2 addition on the PHBV/PBAT system. It showed that TiO2 does not influence the compatibility of the PHBV and PBAT systems. The absorption peak at wave number 1724 cm1 in Figure 3 was the typical absorption peak of the PHBV carbonyl group, while the stretching vibration peak at 728 cm1 in the PBAT molecule was the benzene substituted stretching vibration peak. Although the peak intensity increased dramatically in the TiO2 sample, the peak locations remained unchanged. Similar to the results of experimental observations made by Chenglin et al, 46 after the addition of TiO2 nanoparticles, the position and intensity of the eigenpeaks did not change much, so only a weak physical interaction between the nanoparticles and the PHBV/PBAT matrix can be found.
DSC analysis
Figure 4(c) depicted the second heating curve of the PHBV/PBAT nanofiber membrane and the PHBV/PBAT/TiO2 nanofiber membrane, whereas Figure 4(d) showed the cooling curve. According to the heating curve, PHBV/PBAT nanofiber membranes and PHBV/PBAT/TiO2 nanofiber membranes with varying TiO2 levels had two melting peaks with Tm1 and Tm2 as shown in Table 3. The additional TiO2 was found not to affect the melting capabilities of the PHBV/PBAT mixes. TiO2 did not modify the exothermic peak of the PHBV/PBAT mix system in the cooling curve, indicating that the addition of TiO2 did not change the crystallization property of the nanofiber membrane.
Mechanical property analysis
TiO2 was used as an antibacterial agent to prepare PHBV/PBAT/TiO2 nanofiber membranes with varying TiO2 levels. The stress-strain curve and tensile data were shown in Table 4. Figure 5(b) showed that the tensile strength and elongation at break of the nanofiber membrane improved dramatically after TiO2 was added to the PHBV/PBAT mix system. The tensile strength was (1.2 ± 0.4) MPa and the elongation at break was (203 ± 2)%, especially when the TiO2 concentration was 1.0 wt%. The inclusion of TiO2 enhanced the tensile strength and elongation at the break of the PHBV/PBAT nanofiber membrane. The reason for increased mechanical properties of the nanocomposites may be that when the composite material was subjected to a certain external force, the nanoparticles would absorb part of the energy to hinder the propagation of the crack. 47 As a result, TiO2 in the PHBV/PBAT blend system had high dispersion and created excellent surface adhesion, and the elastic modulus of the PHBV/PBAT/TiO2 nanofiber membrane was exceptionally modest, indicating that it was flexible.
Contact angle analysis
The Water Contact Angle test was used to assess the hydrophilic and hydrophobic characteristics of the nanofiber membranes. Figure 6(g) to (i) showed that the water contact angle of the PHBV/PBAT nanofiber membrane was (109.6 ± 1.0)°, whereas 0.5 wt% and 1.0 wt% TiO2 nanofiber membranes had water contact angles of (125.6 ± 2.5)° and (128.1 ± 3.3)°, respectively. It showed that the hydrophobicity of the nanofiber membrane rose. Highly hydrophobic materials can be utilized in the outer layer of medical protective fabrics to prevent the spread of foreign substances such as blood and droplets.
Analysis of antibacterial properties
The development and spread of residual colonies in E. coli and S. aureus liquid were examined by contact oscillation technique in the blank contrast method and all membrane samples were depicted in Figure 7(a). The agar medium touched by the nanofiber membrane (13%PHBV50PBAT50TiO2(0%)) without adding TiO2 was the same as the blank pair, with a significant number of E. coli and S. aureus growing on the surface. Colonies of E. coli and S. aureus on agar medium touched by a PHBV/PBAT/TiO2 nanofiber membrane were decreased in comparison. Furthermore, when TiO2 concentration increased, the quantity of E. coli and S. aureus decreased gradually. Figure 7(b) depicted the number of bacterial colonies remaining in the bacteria liquid of all membrane samples. However the bacteriostasis rate of all membrane samples against E. coli and S. aureus in Figure 7(c). The blank samples were used as controls. As a result, the nanofiber membrane lacking TiO2 had no antibacterial activity. When 0.5 wt% TiO2 was applied, the bactericidal rate of E. coli and S. aureus was around (92 ± 0.2)% and (91.5 ± 0.3)%, respectively, The bacteriostasis rate of E. coli and S. aureus increased to 99.7% and 98.5%, respectively, when the TiO2 concentration was raised to 1.0 wt%. When the bacteriostasis rate computed from the test findings was 70%, it showed that the test sample had an antibacterial action, according to GB/T20944.3-2008. In short, the PHBV/PBAT/TiO2 nanofiber membrane was antibacterial against E. coli and S. aureus. 48

(a) The antibacterial performances of the blank sample and the PHBV/PBAT/TiO2 nanofiber membranes against E. coli and S. aureus, (b) The average number of the bacterial colony-forming units, and (c) Bacteriostasis rate corresponding to the blank sample and the PHBV/PBAT/TiO2 nanofiber membranes.
Conclusion
In this work, we have prepared degradable PHBV/PBAT nanofiber membranes with varying mass fractions and proportions. The mass ratio of PHBV/PBAT was 50:50, with a mass fraction of 13% nanofiber membrane having good fiber shape and remarkable performance as determined by SEM, FTIR, DSC, tensile test, and water contact angle tests. Following that, different mass fractions of nano TiO2 were applied for characterization testing. In comparison to PHBV/PBAT nanofiber membrane, PHBV/PBAT/TiO2 nanofiber membrane with 1.0 wt% TiO2 had smoother surface morphology, better mechanical properties, and higher hydrophobicity. The antibacterial rate of the PHBV/PBAT/TiO2 nanofiber membrane was found to eradicate 98% of E. coli and S. aureus. The results of this study indicate that the nanofiber membrane has a potential future in the biomedical area.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
