Abstract
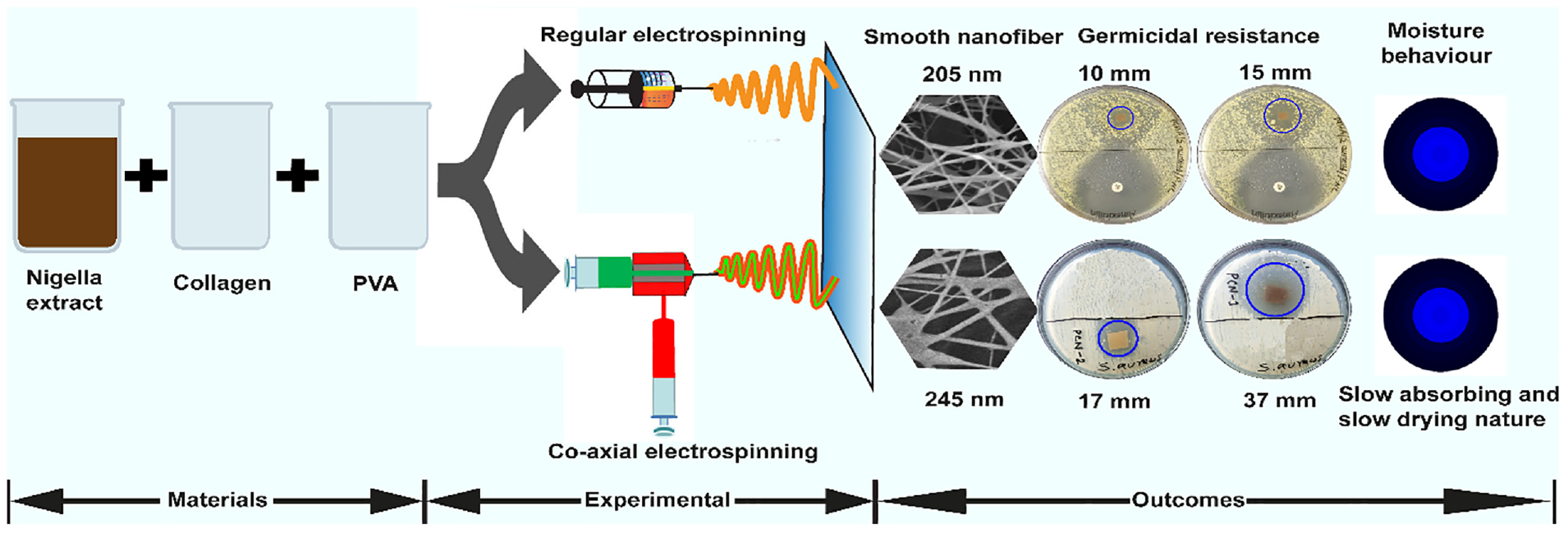
Mitigating health issues utilizing medicinal plants is an ancient practice that has surged in recent times due to the advent of sophisticated technology. Plant extracts incorporated in electrospun nanofibers having biocompatibility and germicidal activity are, therefore, become a competitive choice for biomedical applications. In this study, a novel co-axial electrospun nanofibrous mat with enhanced antimicrobial performance was successfully developed using poly (vinyl alcohol) in the core and collagen–Nigella sativa in the sheath. The structural analysis of the developed nanofibrous mat through a scanning electron microscope revealed the formation of nanofibers with diameters varying from 205 to 250 nm randomly oriented in the membrane. The FT-IR spectroscopy confirmed the existence of poly(vinyl alcohol), collagen, and nigella extract in the nanofibrous mat from their respective characteristic peaks. The bactericidal assay against Gram-positive Staphylococcus aureus bacteria through the agar diffusion method demonstrated an improved antibacterial performance with a higher zone of inhibition (17 and 37 mm) of the coaxial electrospun nanofibers with the increased amount of nigella extract. The moisture management profile indicated an adequate interaction between nanofibers and moisture/liquid, transferring the fluids through the membrane satisfactorily. The formation of such electrospun nanofibers will pave the way for selecting electrospinning techniques for appropriate designing and fabricating nanofibrous materials with enhanced functional properties for biomedical applications.
Introduction
Electrospinning is a prominent technique to produce continuous ultrafine fibers or nanofibers from tens of nanometers to a few micrometers with high surface-to-mass ratios. 1 Considerable interest in this technique has been sparked in recent times because of its simplicity, versatility, and potential applications in various fields. 2 Nanofibers made of natural or modified natural polymers using electrospinning exhibit unique physicochemical characteristics such as biocompatibility, light weight, low toxicity, and, most significantly, a large surface covering.3,4 Over the last decades, the techniques forming nanofibers through electrospinning have evolved into a variety of modified patterns, including coaxial, multiaxial, triaxial, bicomponent, conjugate, bubble edge, and so on, 5 to create complex nanostructures with superior functionalities for diverse application areas. Coaxial electrospinning, a modified form of regular electrospinning, produces nanofibers with a core–shell structure.6,7 A core–shell nozzle in this technique is employed in a double-chamber with a spinneret designed using concentric inner and outer needles connected to the core and shell solution supply, respectively.8,9 Due to forming nanofibers with enhanced performance, this technology has superseded regular electrospinning to develop functional nanofibrous materials for diverse fields of applications, especially in biomedical (drug delivery, antibacterial),10–13 electrochemical (supercapacitors, batteries, energy harvesting),14–17 and environmental (molecular filters, oil filters) fields.18,19 It also nullifies the problem of using biocidal compounds (metal nanoparticles, plant extract, antibiotics) in the electrospinning solution with elevated concentrations.
The development of co-axial electrospun nanofibers with desired structures using various polymeric materials from different origins, including natural, modified natural, and synthetic, has been enriched by many scholars.20–22 A typical carrier polymer utilized in nanofiber formation is poly(vinyl alcohol) (PVA), which is a water-soluble and, most importantly, a biocompatible synthetic polymer, being suitable for various applications, for example, tissue regeneration, bone regeneration, surgical gowns, biomedical hydrogels, drug delivery systems, film formation, and packaging.23–25 Another biocompatible polymer, collagen, is commonly used in biomedical applications owing to its inherent medicinal properties in terms of cell growth, bone creation, and structural care of human conjunctive tissues and organs.26–28 Due to excellent biocompatibility, biodegradability, and compatibility with synthetic polymers, collagen scaffold materials made via electrospinning, are suitable for tissue engineering.29,30 However, the key problem associated with the low mechanical strength of collagen scaffolds confines its large-scale clinical applications. 31 This limitation is, therefore, overcome by cross-linking or blending with synthetic or natural polymers or inorganic compounds complying with the biological function. 27
The desired functionalities of PVA–collagen nanofibers are typically enhanced by incorporating several biocidal compounds, including metal oxides, metal nanoparticles, and antibiotics.14,15,32,33 But, the negative impact of these nanoparticles on ecological and public health issues has prompted the utilization of natural germicidal compounds, which are expected to be nonhazardous, ecologically sustainable, and resistant to the growth of bacteria. 34 In light of this, the development of functional electrospun nanofibers by incorporating novel biomaterials natural resources such as chitosan, curcumin longa, henna, keratin, cellulose, collagen, gelatin, aloe-vera, honey, and ginger for biomedical applications has been investigated in some contemporary research studies.34–42 Nigella sativa, generally known as black seed, is one of the most commonly used antediluvian medicinal herbs holding therapeutic potential for biomedical applications. 43 The presence of numerous active medicinal constituents including thymoquinone (TQ), dithymoquinone, thymohydroquinone (THQ), thymol, nigellicine, nigellimine-N-oxide, carvacrol, nigellidine, and alpha-hederin as well as elevated germicidal, wound healing, tissue regeneration, anticancer, antidiabetic, and anti-inflammatory performances has collectively made it a prospective choice for biomedical research, particularly wound dressing applications.44–46 The development of antimicrobial nanofibers via regular electrospinning incorporating this particular herb has been investigated by several scholars8,47 but achieving optimum germicidal performance with maximum nigella concentrations is still challenging due to its oily nature that requires further investigation. In this regard, this study delineates a comparative study based on germicidal activity between nanofibers fabricated by regular and coaxial electrospinning from Nigella sativa extract as a natural antimicrobial compound to assist in designing nanostructures with desired functionalities for biomedical applications.
Experimental
Materials
Nigella sativa seeds of the finest quality were purchased from the local market and cleaned thoroughly with water to remove foreign materials. The seeds were then dried under sunlight before grinding to a fine powder for proper extraction. Water soluble PVA (molecular weight: 115,000 g/mol) in the particle form was sourced from Loba Chemie Ltd, India. Distilled water was used as a solvent to prepare the PVA solution. Pure methanol (99.0%) was obtained from Merck, Germany. Pharmaceutical-grade fish collagen peptide powder (molecular weight: 250,000 g/mol) (Type-I) was procured from Baoding Faithful Industry Ltd, China.
Methods
Material Preparation
PVA solution (10.0% w/v) was prepared by dissolving polymer granules in distilled water with a constant stirring at 70° C for 3 h. Collagen solution of 40.0% (w/v) was prepared by dissolving the powder in distilled water at room temperature. Nigella seeds in granules were impregnated into methanol at a ratio of 1:2 and stored at room temperature for 24 h in a sealed bottle for extraction. The extract was then filtered twice through a very fine nylon mesh cloth with a quadruple layer. Solvents were then evaporated at 70° C and stored in the refrigerator at 4° C for further use.
Preparation of Electrospinning Solution
The experimental investigation was carried out by fabricating nanofibrous membranes employing two distinctive but widely used techniques, regular and coaxial electrospinning. The nanofibers developed by coaxial electrospinning consist of two individual layers called core and sheath. In the core layer of nanofibers, pure PVA solution, due to its high film-forming properties was used to provide strength to nanofibers, and the shell layer was developed using nigella extract and collagen with the proportions of 50:50 and 60:40 (v/v) owing to their medicinal properties. The schematic diagram of the development of nanofibers through the coaxial electrospinning technique is presented in Figure 1.
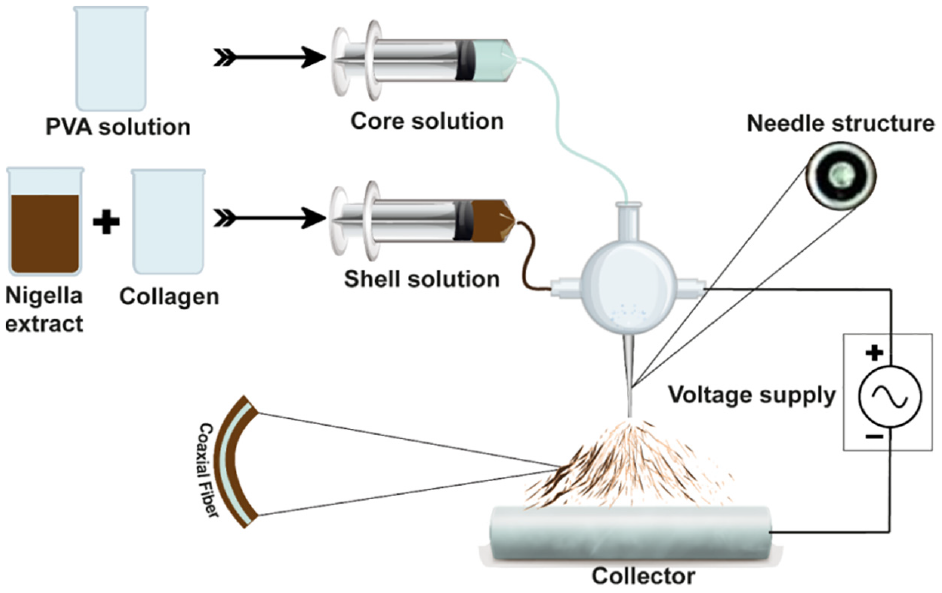
Schematic diagram of the development of nanofibers through coaxial electrospinning.
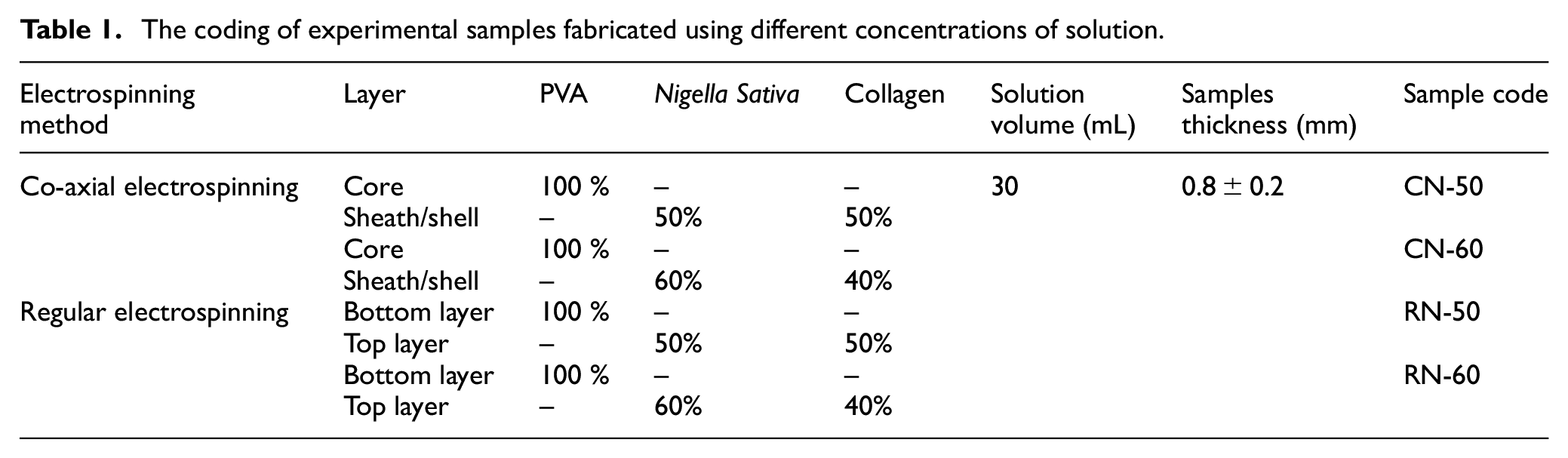
In the case of regular electrospinning, the nanofibrous membrane was developed with two layers, the bottom and top layers. The bottom layer was fabricated using pure PVA solution, and then nanofibers of collagen and Nigella sativa extract of different proportions were overlaid as a top layer. A constant collector distance (15 cm), solution feeding rate (2.45 mL/h), needle gauge (22), drum speed (400 rpm), positive voltage (24 kV), and negative voltage (11 kV) were maintained for all samples during the experiment. All the solutions were mixed in liquid form based on their amount (mL), For instance, 30 mL of solution was prepared at a 50/50 mixing ratio of nigella extract and collagen using 15 mL of nigella extract and 15 mL of collagen solution. The identification of test samples with different concentrations of PVA, Nigella sativa, and collagen extract are presented in Table 1.
The coding of experimental samples fabricated using different concentrations of solution.
Material Characterization
The morphological examination of developed nanofibers was carried out using a scanning electron microscope (SEM) (SU 1510, Hitachi, Japan), and chemical characterization were accomplished by FT-IR (4700, JASCO, Japan) with the spectral range of 4000–500 cm−1. The bactericidal evaluation was performed against Gram-positive Staphylococcus aureus following the standard ATCC 6538 using the Kirby–Bauer agar disk diffusion technique through the measurement of inhibition diametric zone (mm). This qualitative analysis was conducted maintaining 1.5 × 105 CFU/mL bacterial concentration where amoxicillin (10 µg) was used as control. Moisture management performance with unidirectional moisture transport capability was evaluated using a Moisture management tester (MMT) (M 290, SDL Atlas, UK) following AATCC 195-2009 standard. All the specimens were conditioned in testing atmosphere before testing.
Results and Discussion
Structural Analysis
In the fabrication process of electrospun nanofibers, several factors, including solution viscosity, applied voltage, solution viscosity, collector distance, and ambient condition, typically influence the dimension and structure of nanofibers.48,49
In this regard, appropriate parameterization is a prerequisite for developing nanofibers with desired structures and functionalities. Therefore, a trial-and-error method was followed in the present experiment to find suitable parametric values. Observation reveals that nanofiber formation is not desirable until the applied voltage is more than 12 kV, indicating the conglomeration of enough charge density to distort the surface tension of electrospun solutions that assist positively in tailoring cone formation. Moreover, a constant collector distance of 15 cm and a flow rate of 2.45 mL/h were found appropriate in the current study for developing co-axial nanofibers with the desired diameter.
The SEM images in Figure 2 exhibit the formation of nanofibers possibly with three-dimensional structures, and multidirectional alignment in the nano mat. A higher diameter of coaxial nanofibers compared to regular electrospun nanofibers was also detected in the SEM micrographs of the nanofibrous membrane. The observed diameters of RN-50 and RN-60 were 207 and 205 nm, shifted to 250 and 245 nm for CN-50 and CN-60, as presented in Figure 2(f). This might be attributed to the formation of double-layered nanofibers with a core and shell in the structure. Because of forming nanofibers with larger diameters, less surface covering with a porous structure was observed for the specimens CN-50 and CN-60.

SEM micrographs of (a) top side of RN-50, (b) top side of RN-60, (c) bottom side of RN-50 and RN-60, (d) CN-50, (e) CN-60, and (f) fiber diameter distributions of all samples.
The existence of PVA, collagen, and nigella components in the prepared samples was affirmed by the spectral wavenumber of FTIR of as represented in Figure 3. The functional group region is located between 4000 and 1450 cm−1, while the fingerprint region spans between 1450 and 500 cm−1. The main phenolic compounds of nigella are TQ, dithymoquinone, thymol, and THQ, and FTIR spectra have revealed the presence of various sharp, strong, and weak peaks, along with prime chemical groups corresponding to -CH3, -CH2, C-H, C=O, C-O, and C=C as well. 50 The attributable peaks for the used nigella were located at the wave number of 2928 and 2869 cm−1 for C-H of the aliphatic group, indicating the existence of methyl and isopropyl substituent. Again, a significant peak was observed at a wavenumber of 1635 cm−1 belonging to the C=O stretching of TQ. The wave peaks at 1449 cm−1 related to C-H absorption scissoring, and 1040 cm−1 owing to -C-O stretch, were also observed. Besides, the characteristic peaks of PVA were observed at 3239 cm−1 (O–H stretching bonded), 2931 cm−1 (C–H stretching), and 1089 cm-1 (C–O–C stretching).
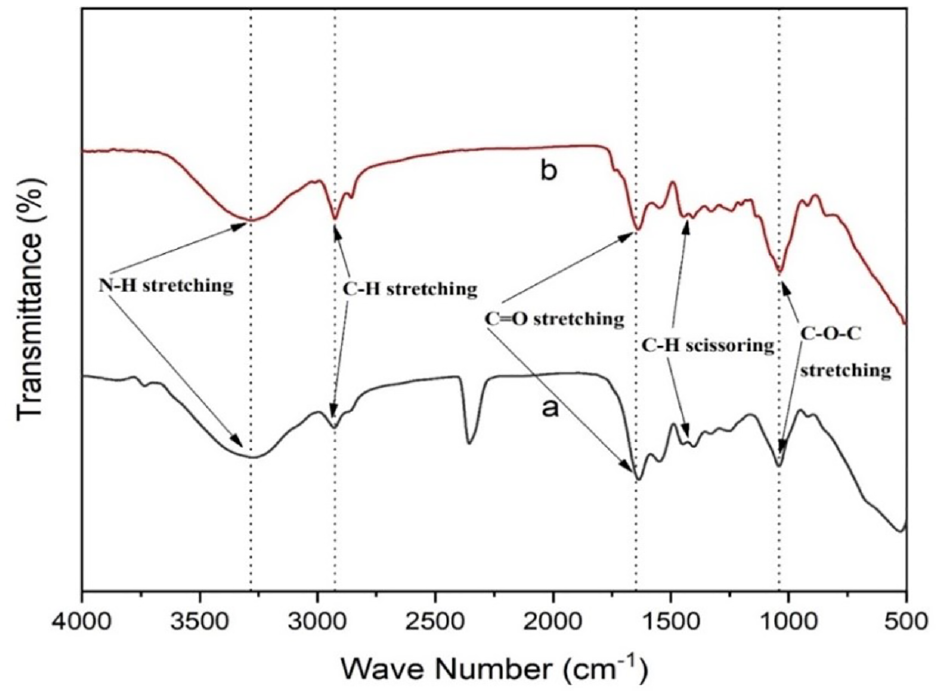
FT-IR spectra of (a) coaxial electrospun and (b) regular electrospun nanofibrous mat.
Furthermore, the sight of collagen ingredients is indicated through the peaks at 1635 cm−1 (amide I), which corresponds to C=O stretching vibrations coupled to N–H bending vibrations, and peaks at 1526 cm−1 (amide II), which correspond to N–H bending vibrations. The attainment of the attributable peaks of individual compounds in the developed samples resembles the findings of earlier studies. 47
Antibacterial Performance
The microbicidal nature of electrospun nanofibrous mat plays a decisive role in wound dressing material because it inhibits bacterial infection and eventually kills them during the wound-healing process. Otherwise, the wound area may become infected through bacterial colonization causing fatal disease. In this cornerstone, the antibacterial activity of the developed nanofibers was evaluated against the Gram-positive bacteria S. aureus, which generally causes skin infections and is likely to be resistant to treatment with some antibiotics. 51 The formation of zones of inhibition (ZOI) of the developed nanofibrous mats has been presented in Figure 4.
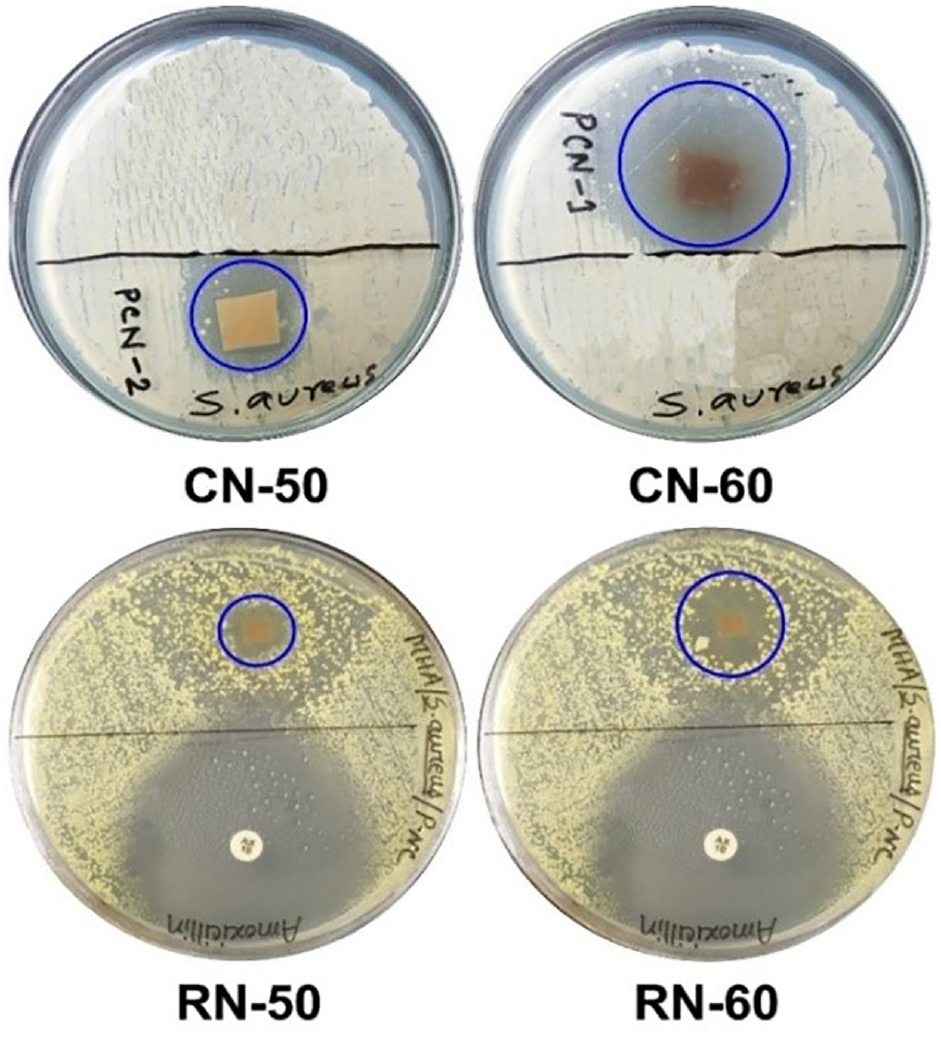
Forming ZOI against S. aureus bacteria of the developed nanofibrous mats through regular and co-axial electrospinning.
The formation of ZOI against S. aureus bacteria of the developed nanofiber specimens is shown in Figure 4. Co-axial electrospun nanofibers demonstrated a higher ZOI with 17 and 37 mm dimensions for CN-50 and CN-60, respectively. The maximum ZOI exhibited by CN-60 suggested its enhanced antibacterial efficacy due to incorporating increased proportions of nigella extract in the electrospinning solution. On the other hand, a considerably lower ZOI with the dimensions of 10 and 15 mm was observed for specimens RN-50 and RN-60, respectively, developed by regular electrospinning. For both cases, the nano mats electrospun through the co-axial technique outperformed the regular nanofibers in terms of germicidal activity with similar NS content.
Literally, the virtue of killing bacteria with nigella extract is attributed to its bioactive ingredients, such as carvacrol, thymol, terpenoids, TQ, and THQ. 47 However, TQ and THQ are among the two most significant constituents of NS responsible for germicidal activity owing to their free rotation bonds (Figure 5). TQ and other active NS biocomponents can alter their forms for easy binding to bacteria and ultimately kill them by penetrating or breaching their barrier. 36 The common strategies for inhibiting the growth of bacteria include the rupturing of the cell membrane, the reduction of the proton motive force, and the harming of the protein composition of the cell membrane. 52 Accordingly, nigella extract typically causes the bacterial cell membrane to rupture and expel the contents of the cell, resulting in the death of the bacterial cell, as illustrated in a previous study. 47
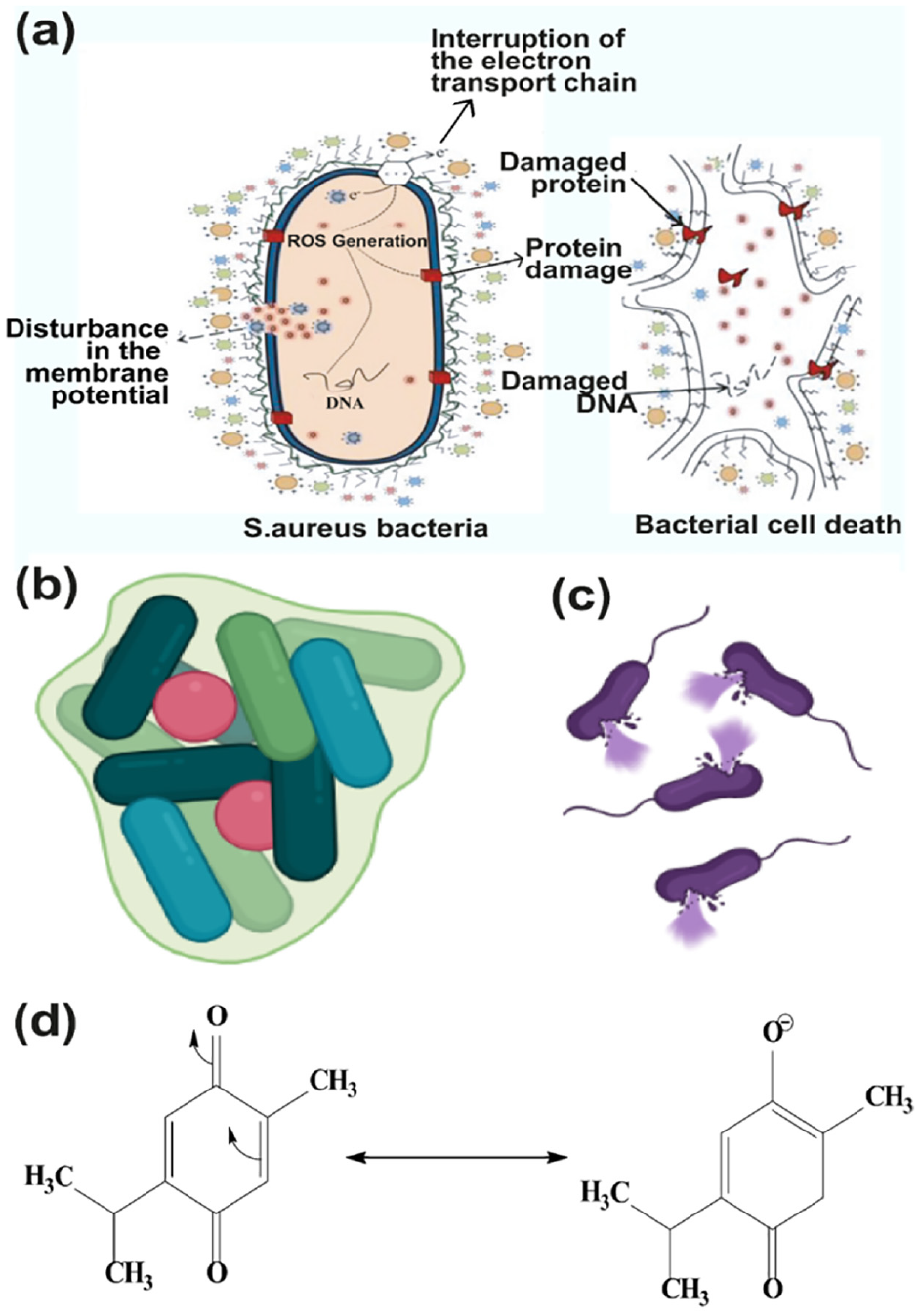
Mode of action for bacterial resistance of nigella extract in the nanofibrous membrane: (a) killing mechanism, (b) formation of biofilm, (c) cell lysing (created with BioRender.com), and (d) resonance structure of TQ. 53
In Gram-positive bacteria, this breakdown causes membrane-embedded proteins to malfunction, membrane fluidity and permeability to rise, respiration to be inhibited, and the ion transport pathway to change. Instead of this, several bacterial pathogens, including S. aureus, are thought to have key virulence factors that depend on the production of biofilm. Due to the microscopic gaps in the network structure of the produced nano mat, no bacteria can enter, effectively eliminating any illnesses in the area.
The findings of the antibacterial performance in the current study align with the previous studies conducted in the development of electrospun nanofiber incorporated with nigella sativa extract47,54–56 (Table 2). The overall result suggests a rational correlation between the extent of nigella extract in nanofibers and the zone of inhibition, confirming the existence of TQ and THQ in the nano mat released from the nigella extract to kill the bacteria. This improved germicidal activity of the co-axial electrospun membrane affirms the presence of Nigella sativa in the shell of nanofibers that could exhibit resolute resistance against bacterial attack during their use as wound dressing materials.
Comparative outcomes based on antibacterial performance of the current and previous studies.

Moisture Management Properties
In addition to antimicrobial activity, nanofibrous mats intended for biomedical applications should also have comfortability at the dermis and epidermis and, therefore, have adequate moisture transport properties. Table 2 displays the absorbance properties of test nanofiber specimens developed through coaxial and regular electrospinning. Samples CN-50 and CN-60 exhibited wetting times at the top and bottom surfaces of fibrous mats of 3 and 14 s, respectively, indicating their fast- to medium-absorbing nature. In contrast, regular electrospun samples RN-50 and RN-60 showed values of 5 and 17 s, respectively. This may be attributed to the presence of the PVA outer layer of nanofibers. Overall, there was no significant difference in moisture management profile between the coaxial and regular nanofibers (Figure 6(a) and (b)). Both specimens exhibited slow absorption and slow-drying properties.
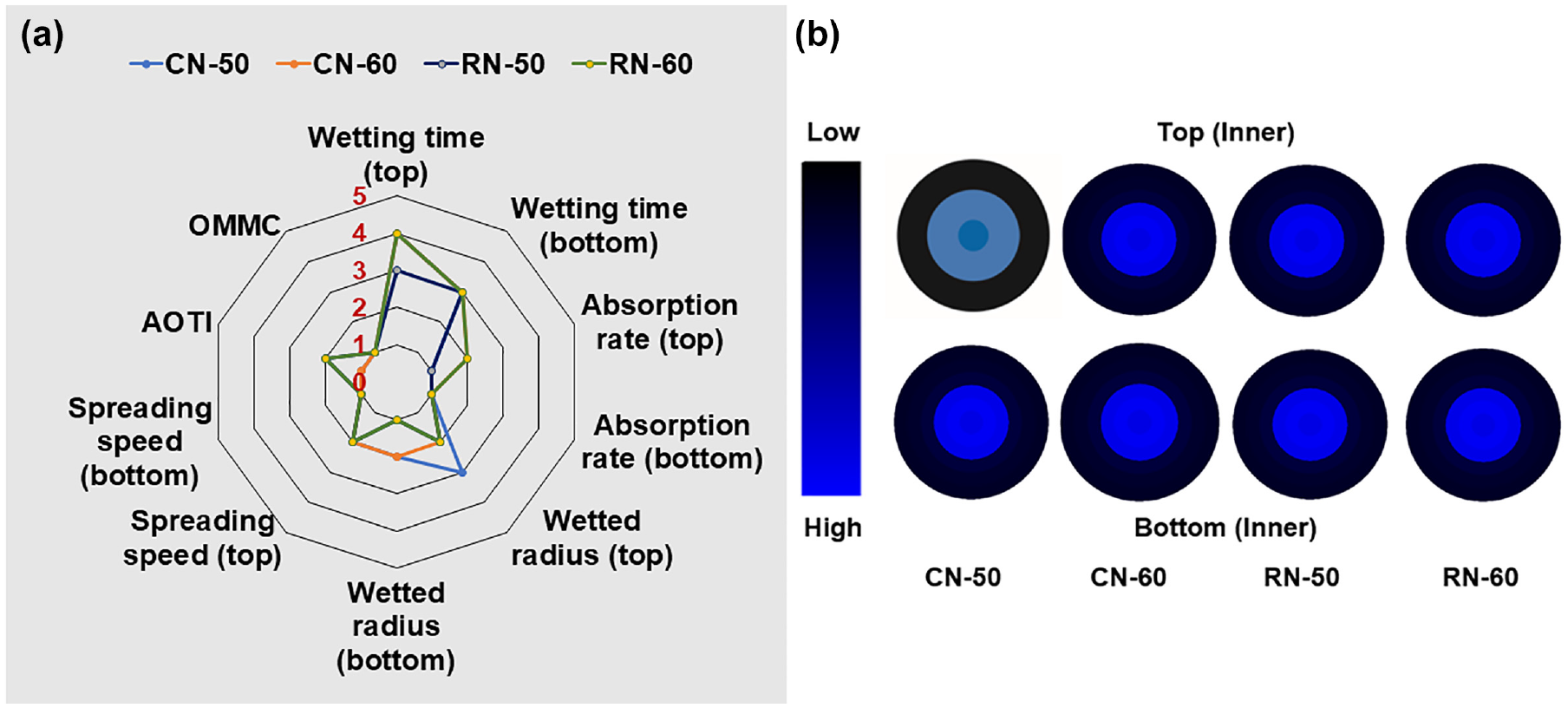
Moisture management profiles of test specimens: (a) grading of the test parameters and (b) time-dependent water location radius.
In particular, the absorption rate, max wetted radius, spreading speed, and overall moisture management properties lie between the poor and fair grades (Table 3). A slight difference between the two samples may be due to the system followed by a coaxial method. However, moisture properties vary slightly due to the hydrophilic groups in the collagen, like hydroxyl, carboxyl, and amino groups.
Values of moisture management indexes of developed nanofibers specimens.
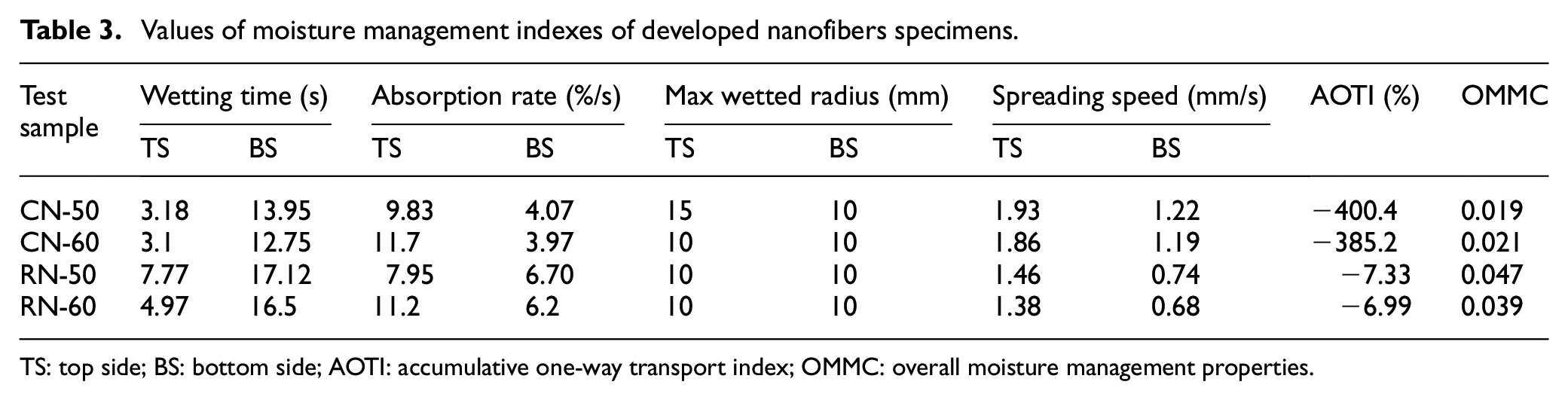
TS: top side; BS: bottom side; AOTI: accumulative one-way transport index; OMMC: overall moisture management properties.
Conclusion
Electrospun nanofibrous membrane with enhanced germicidal activity was successfully fabricated through the co-axial electrospinning technique using different amounts of PVA, collagen, and nigella extract. The nanofibrous mat developed through co-axial electrospinning demonstrated its antibacterial effectiveness against Gram-positive S. aureus bacteria. The SEM micrographs of developed nanofibers showed a regular and uniform architecture of fiber with their random arrangement in the membrane. The SEM images also revealed the increase in fiber diameter (245 ± 71 nm) developed through co-axial electrospinning compared to regular electrospinning (205 ± 70.9 nm). The chemical characterization through FTIR explored the existence of PVA, collagen, and nigella extract constituents by revealing their distinctive peaks. In view of germicidal activity, the developed co-axial nanofibers exhibited a higher bacterial resistance with a maximum ZOI of 37 mm against the Gram-positive S. aureus bacteria, suggesting its potential for use as a wound dressing biomaterial. In addition, the slow-absorbing and slow-drying moisture management properties indicated a soothing environment that could be provided by the developed nanofibrous membrane during its use in wound-healing treatment. All the outcomes suggest that the fabrication of a biodegradable nanofibrous membrane using collagen and nigella extract through co-axial electrospinning is a promising approach that could be utilized as a potential wound-healing biomaterial with enhanced germicidal activity for biomedical applications.
Footnotes
Acknowledgements
The authors thank the Department of Textile Engineering, DUET for providing required laboratory facilities to conduct the experiment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by UGC and DUET under the resolution of CASR 54.
