Abstract
Functional electrically conductive fabric with acceptable mechanical properties, which could be applied in electromagnetic shielding, was developed. Conductive cotton fabrics (cotton/PANI, cotton/Mn, cotton/Cu, and cotton/Co) were prepared by in situ chemical oxidative polymerization for (cotton/PANI) and pad dry curing method was used for nanometals application. The Nano size of the metals and polyaniline inclusion were proven through both Dynamic Liquid Scattering (DLS) and X-ray diffraction (XRD) which showed an increase in crystallite density in unit space and the nanoparticles ranged from 100–200 nm. The effect of gamma irradiation on different treated cotton fabrics was investigated. The mechanical properties against irradiation dose showed an improvement up to 40 kGy, for all treated fabrics. On the other hand, Young’s modulus for untreated cotton recorded the lowest value, while cotton/Co recorded the highest one. Moreover, both AC (Alternating Current) and DC (Direct current) conductivities values can be calculated. In DC conductivity cotton/PANI was found to be more conducive than the remainder of the treated fabric by surface metallization with transition metals; while in AC conductivity cotton/Mn was found to be more conducive than the rest of the treated samples. The conductivity value increases by increasing the gamma irradiation dose for cotton/PANI fabric. Also, g-factor values can be estimated from ESR signals and vary from 0.009 up to 0.059 for conductive cotton fabrics; whilst cotton/Mn fabric has six hyperfine splittings, indicating that it is a paramagnetic element.
Keywords
Introduction
Textiles with a rare blend of properties are fundamental for developing technologies continuously. To achieve nanomaterials with determined functionality, numerous efforts have been made in the past using innovative nanotechnology and nanoscience knowledge.
Cotton as a natural fiber has an extensive difference in its properties [1].In the dry state, cotton fabric possesses modest mechanical strength whereas in the wet state it has the highest strength properties. The cotton fabric is comfortable as summer be dressed in because of the high moisture absorption of the fabric. The cotton fabrics have tremendous adhesion to coat or plastic-coated polymeric film [2]. Well-designed terminating fetches an alteration on the mechanical and surface properties of textile materials [3].
In recent times, nanoparticles are progressively used to acquire smart textiles. The vital mechanisms of smart textiles are conductive fabrics. In order to obtain conductive textiles with electrical conductivity, there are various approaches such as surface metallization, coating of carbon materials, coating of conductive polymers, etc. [4,5]. The wires and inflexible plastic cases are prickly to wear against bodies. Therefore, the electronic circuits are being manufactured individually out of textiles to deliver data and power and perform touch sensing, the protection has given the comfort of the wearer [6]. Stainless steel, ferrous alloys, nickel, aluminum, copper, and titanium are used as electrically conductive metals. Among them, textile surface metallization is a unique method that can provide multifunctional properties such as electrical conductivity, EMI shielding, anti-static and anti-bacterial properties, UV radiation screen, and radar reflectivity [7–9].
Electromagnetic shielding generally implies the absorption or reflection of electromagnetic waves by shielding materials. The shielding materials can be furnished on the wall surface and covered around the radiation source. Also, the protector or shielding materials protect the environment or a circuit from harmful electromagnetic radiation and also used to isolate a space (a room, an apparatus, a circuit, etc.) from outside sources of electromagnetic radiation, or to prevent the unwanted emission of electromagnetic energy radiated by internal sources. Moreover, there are many types of EM protection products such as shielding tents and shielding curtains. Furthermore, a safety strategy for radioactive waste containment and isolation for the proposed storage and transportation focuses on two objectives: (i) to provide stabilization of the radioactive waste within the including package; (ii) to limit the radiation exposure dose of the public during the transportation or other handling processes [10]. It is difficult to use concrete or lead during source transportation from one place to another due to the densities of heavyweight concrete aggregates. The new generation of shielding materials is conductive textiles which are lighter materials that may be more effective in this case. Electromagnetic shielding properties vary and they depend on the conductivity of shielding material [11].
Polyaniline (PANI) is the main and easy of the conductive polymers. It has excessive thermal and chemical stability, small price, effortless polymerization steps, luxurious synthesis, ecofriendly stability, dissimilar oxidation steps. Including, it has extra property such as color variation, high capacitance, and high absorption in shielding [12–15]. PANI’s green protonated emeraldine type has high electron conductivity as high as (1.1–1.2) × 103 S/cm on a semiconductor level. Polyaniline is familiar to be a true metal; however, it is similar to nanometal. Polyaniline is found to be a p-type carrier semiconductor and its conductivity occurs due to electron movement at gaps [16]. Also, conductive polymers such as (PANI) are compared with other materials and the main usage for electromagnetic shielding.
Metallic fabric produced by traditional manners consists of defects, such as stiffness, poor air permeability, and heavyweight [17]. The sputter coating, vacuum deposition, flame, and arc spraying, electroless plating, and pad dry curing methods are some of the novel methods of surface metallization which can overcome the limitations of traditional methods [17,18]. These effective modifications for the cotton fabric surface with controllable conductivity and high durability in safe conditions. Suitable materials for the EMI shielding like copper, silver, and stainless steel, cobalt were produced and must have very specific properties, including magnetic properties. On the other hand, composite fabrics not only reflect but also absorb the incident wave [19]. This property, along with advantages of low density, good flexibility, and good shielding effect, convenient processing, ensures conductive fabrics to get more widely used [20].
Mechanical properties may be reflected in the most important of the mutually physical and chemical properties of textile materials for most applications. The mechanical properties at an inexpensive standard have very important for the end-user to classify between the different textile materials. Instead, any chemical treatment to textile material to impart required properties will affect the mechanical properties to some extent [21].
Herein our study was motivated by the improvement of electrical conductivity for treated fabrics with refining mechanical properties. This paper presents a novel method for the accumulation of nanometals particles on the surface of cotton fabrics by successive dipping in metal salts and then sodium borohydride solutions, while polyaniline was carried out by oxidative polymerization of aniline. The mechanical and electrical properties of treated cotton fabrics were examined. The depositions of nano metal particles were confirmed by Dynamic Liquid Scattering (DLS), X-ray diffraction (XRD), scanning electron microscope (SEM) analysis, and Electron Spin Resonance Spectra (ESR).
Materials and methods
The cotton fabric was taken as a substrate in this work, which was delivered by El-Mahalla Textiles Company, Egypt. The cotton fabric was raw materials and not subjected to any auxiliary finishing processes. The raw cotton fabrics were washed in a bath containing 0.2 gm/l, Na2CO3, and 0.1g/l Nonionic detergent for one hour at boiling temperature, and then soaked in 1M NaOH for 1h, finally washed several times by deionized water. All chemicals were of methodical must be analytical used without supplementary purification Aniline, Hydrochloric Acid (HCl, 37%), Ammonium Peroxydisulfate (NH4)2S2O8, sodium borohydride (NaBH4), Manganese Chloride, copper sulfate, and cobalt sulfate were obtained from (LoBA- Chemie), and other chemicals were used as customary.
Preparation of PANI by chemical oxidative polymerization
We started by investigating the cotton sample was allowed to soak in aniline solution (0.1 mol) afterward that aqueous solution of APS (0.1 mol) was inserted dropwise. The temperature was maintained at ice-bath and kept under stirring for 6 hr. While the polymerization continues, the fabric sample revolutions green color which is the color of emeraldine salt which is a conducting PANI. After the success of the period of polymerization, the in situ polymerized polyaniline cotton fabric was washed in dilute HCl to eliminate untreated chemicals and oligomers, then the fabric sample washed afterward by deionized water. Finally, it was dried and weighed.
Deposition of nanometal particles
To verify the validity of the Pad Dry Curing method, we carried out by Copper Sulfate, Cobalt Sulfate, and Manganese Chloride were used as the improper material along with sodium borohydride (NaBH4), as a reducing agent for in-situ deposition of different nanometals on cotton fabrics. In the beginning, 0.1 M of different metal salts was dissipated in distilled water. Then, the cotton fabrics were immersed in the solution and dried at 100οC for 5 min. This technique of dipping and drying was repeated for 20 cycles. The dwell time of fabric in solution was about 30 s against each dip. Subsequently, the treated fabrics were transferred to the 0.2 M sodium borohydride solution. The concentration of sodium borohydride was determined based on the concentration of metal salts. The reduction was continued for 20 min. The time of reduction treatment was clarified based on the change in the color of cotton. The cotton fibers transformed their color from blue to blackish gray for copper, red to dark color for cobalt, and light pink to brown for manganese, after 15 min of reaction with the reducing agent, consequently, 20 min of reduction treatment was recognized to complete the reaction.
Characterization
Morphology and elementary analysis
The morphology of nanometals deposited on the fabric surface and polymerized PANI on the cotton fabrics was investigated by scanning electron microscope (SEM), the elementary analysis was characterized energy-dispersive X-ray spectroscopic (EDX) analysis (JEOL-JSM5400, Japan, and along with an acceleration voltage of 30 kV).
Structure analysis
The XRD analysis was performed to (XRD-6000 Shimadzu), X-ray diffractometer using Cu Kα radiation (λ = 1.54059Aο), and 2θ in the range between (4 and 900). The mitigate dispersion of nanometal particles was prepared in deionized water. It was ultrasonicated for 15 min with an ultrasonic bath before DLS (Dynamic Light Scattering) measurement of particle size distribution with count rate 415 kHz, index of ref. 1.333, and viscosity 0.933cp at room temperature.
Electrical conductivity and vibrational properties
The DC conductivity was computed by the two-point- probe method for surface and volume conductivities by using a Keithley model 6517A at room temperature, and the relative humidity RH = 54%. The free radicals created by Electron Spin Resonance spectra (ESR signals) were verified by an X-band EMX spectrometer (Bruker, Germany) using a standard rectangular cavity of ER 4102. The operating parameters applied during the ESR test are microwave power, mW; modulation amplitude, Gauss; modulation frequency, 100 kHz; the number of x-scans, resolution in x, sweep width, Gauss; microwave frequency, GHz; time constant, ms; conversion time, ms and sweep time, s. All the tests were carried out at room temperature
Mechanical properties measurements
The mechanical properties consist of tensile strength and elongation to break was determined at room temperature, according to the ASTM standard D5034 for textile fabrics. Quadrangular specimens at dimensions of 50 mm × 100 mm were tested. Each data point is the average of 3 tests, (Mecmesin tester, UK), fitted out with software was used and by employing a crosshead speed of 70mm/min. In this scheme, the various mechanical parameters were estimated directly.
Gamma radiation
Irradiation was approved in the Cobalt-60 gamma cell source (made in Russia) installed at the National Centre for Radiation Research and Technology, Cairo, Egypt (dose rate 1.33KGy/h). Direct radiation was used to studying the effect of different doses (10, 20, 40,50KGy) on the treated cotton fabrics.
Results and discussion
Electrical conductivity (DC conductivity) of untreated and treated fabrics
Volume resistance can be calculated by operating a voltage potential across the conflicting edges of the sample and evaluating the resultant current flowing through the sample. Volume resistivity, ρV [Ω.m] can be computed by the following equation (1)

The values of surface and volume conductivity of untreated and treated irradiated fabrics at 40 kGy.

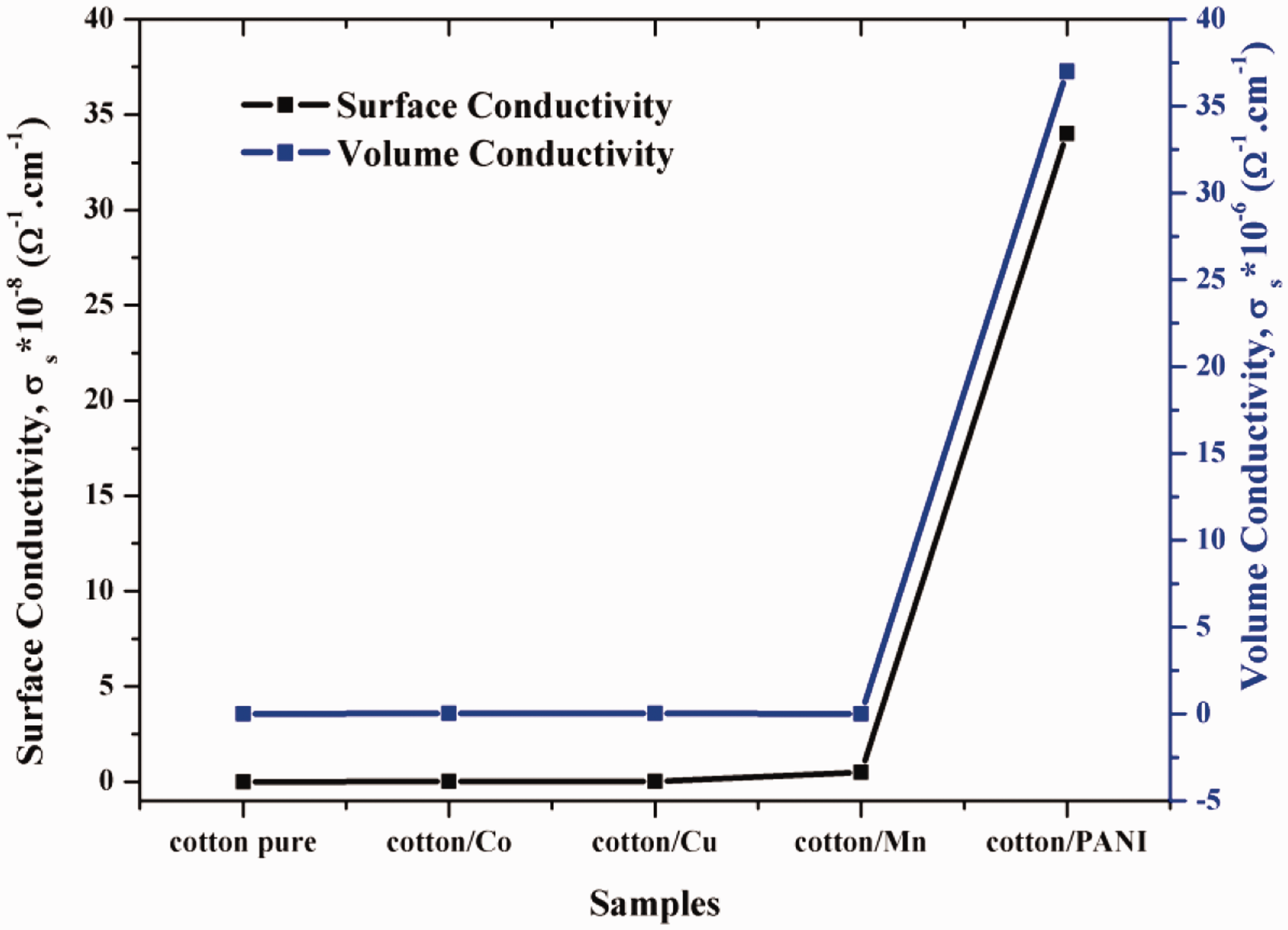
Surface and volume conductivities for untreated and treated fabrics with PANI and metals-dipped fabric samples.
The elemental composition of nonmetals deposited on cotton fabrics.

It was clear from Table 1 and Figure 1 that surprisingly, higher values of surface and volume conductivities resulted in treated with PANI than these metal-dipped fabric samples. This manner could be assigned to the formation of an ionic bond between the metals (II) and negative groups available on the cotton surface. Metal salts created more conductive fabrics due to the formation of the percolated network by the creation of continuous connectivity between the small-sized metal particles. Then, the cotton fabrics absorbent properties take place due to several reasons, including its specific molecular structure and structure of polyaniline. The cotton materials contained a negative charge which attracts the molecules easily and absorbed them quickly. The fiber in cotton has hydrophilic properties; this makes them absorb the polyaniline particles regularly and is placed over the surface of cotton fabrics. Later, cotton fabrics coated by polyaniline have low specific surface resistance and good conductivity. The enhancement in surface and volume conductivities of the polyaniline coated cotton can be concluded.
The effect of grafting ratios of polymerization for PANI on cotton fabrics on electrical conductivity was examined and showed in Figure 2. The conductivity of the fabrics was supposed to encourage increasing grafting ratios of the PANI. This suggestion was approved by the electrical conductivity experiments accomplished on the advanced conductive fabric samples. The effect of gamma irradiation doses on conductivity for cotton/PANI was showed in Figure 3. It shows that the dc conductivity of the cotton/PANI is increased by increasing the radiation dose and at 0 kGy the conductivity is 1.69 × 10−5 Ω/cm and at 40 kGy is 1.68 ×10−4 Ω/cm. the electrical conductivity increases with increasing the radiation dose due to the formation of polarons created at the site of the defect, which moves towards the polymer backbone. This implies that radiation-induced degradation becomes effective and hence increases the motion of free radicals, thereby increases the conductivity [22,23].

The effect of grafting ratios of polymerization for PANI on cotton fabrics on electrical conductivity.

The effect of gamma irradiation doses on conductivity for cotton/PANI.
SEM and EDX analysis
The SEM was employed to observe the deposition of nano-metals and PANI on the fabric surface. The scanning electron micrographs of blank cotton, cotton/PANI, and cotton/Cu Np are shown in Figure 4. The results suggested that the reaction product was comprised of abundantly pure metal nanoparticles. From Figure 4(b) after PANI polymerization, fibers appeared homogeneously coated with a film of PANI on the cotton fabrics; it can be envisioned from some areas of fabric surfaces that were covered wholly and some areas were painted by small and large granulated PANI. The inclusive observation of the SEM study shows that the cotton fabrics have been effectively treated and these aniline diffusions on the individual fibers deliver conductivity to the fabrics [24]. Figure 4(c) revealed the nanometer scale of copper particles deposited on the fabric surface; this indicated the formation of a percolated network of copper particles on the cotton fabric surface. EDX spectrum was obtained for samples analyzed shown in Figure 5 for cotton/Co Np and cotton/Cu Np. EDX analysis showed the elemental composition of copper and cobalt nanometals deposited fabrics. It was shown that, the increase in contents of copper and cobalt nanometals after 20 cycles of dips. Table 2 showed the elemental composition of nanometals deposited on cotton fabrics determined by EDX analysis.

The scanning electron micrographs of (a) blank cotton, (b) cotton/PANI, and (c) cotton/Cu Np.
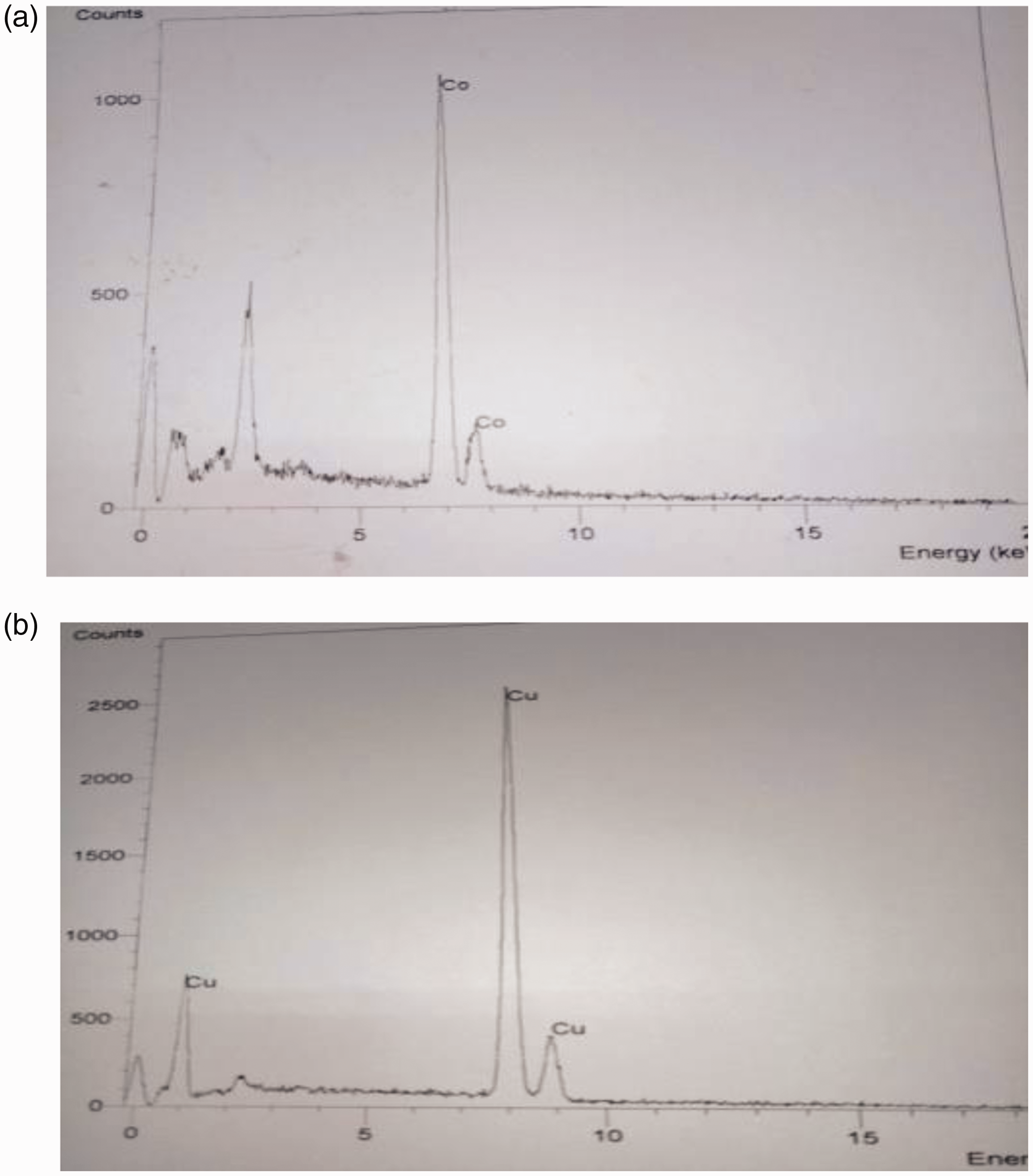
The EDX spectrum for cotton/Co Np and cotton/Cu Np.
XRD analysis
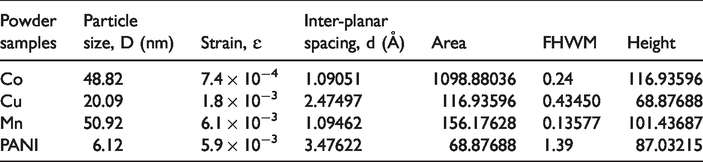
The X-ray diffraction (XRD) analysis was carried out to know the phase composition of deposited metal nanoparticles on cotton fabrics, and to study the structural effect of diffusion of PANI on the crystalline and amorphous regions of the fabric. The XRD pattern of the cotton blank, cotton/PANI, and cotton/Mn, cotton/Cu, and cotton/Co were given in Figure 6. From Figure 6 it can be seen that two peaks of cotton blank at 2θ about 15° and 23° correspondings to the planes of (100) and (002) respectively. The JCPDS file matching of the cotton blank was found to be 50-2241. Also, there were peaks of cotton/PANI detected at 2θ about 11°, 22°, 28°, 33° which indicates PANI deposited on cotton fabric. The realized values are in well-behaved agreement with the reported values [25]. This shows the diffusion of PANI and deposited metal nanoparticles into amorphous and para-crystalline regions of cotton fabric, without affecting its crystalline. Moreover, the peaks of cotton/Cu at 2θ 43.4, 50.5, and 74.2° corresponding to the (111), (200), and (220(respectively planes of copper [26]. Besides, the wide reflex diffraction peak of cotton/Co with a maximum at 79.6° can be attributed to the metallic Co, in which the alternating microdomains with cubic and hexagonal packings were observed. Furthermore, the peak reflections of cotton/Mn at 2θ about 28.33, 40.53, and 50.01ο matching to lattice planes which can be indexed to face-centered cubic manganese, which further proved the formation of crystal manganese nanoparticles [27]. These peaks matched with the Joint Committee on Powder Diffraction Standards (JCPDS No. 04-0326). Afterward surface metallization for nano-metals (Cu, Co, Mn) on cotton fabrics and polymerization of aniline on cotton fabric, the peaks of the cotton blank were slightly shifted to higher values of 2θ with a small shift in interplanar (d) spacing, then particle size of functionalized fabrics are about 5.2 to 7.5 nm. The values of particle size (D), strain (ɛ), inter-planar spacing (d), area, full half-width maximum (FHWM), the height of Cu, Co, Mn Nps, and PANI powders are tabulated in Table 3.
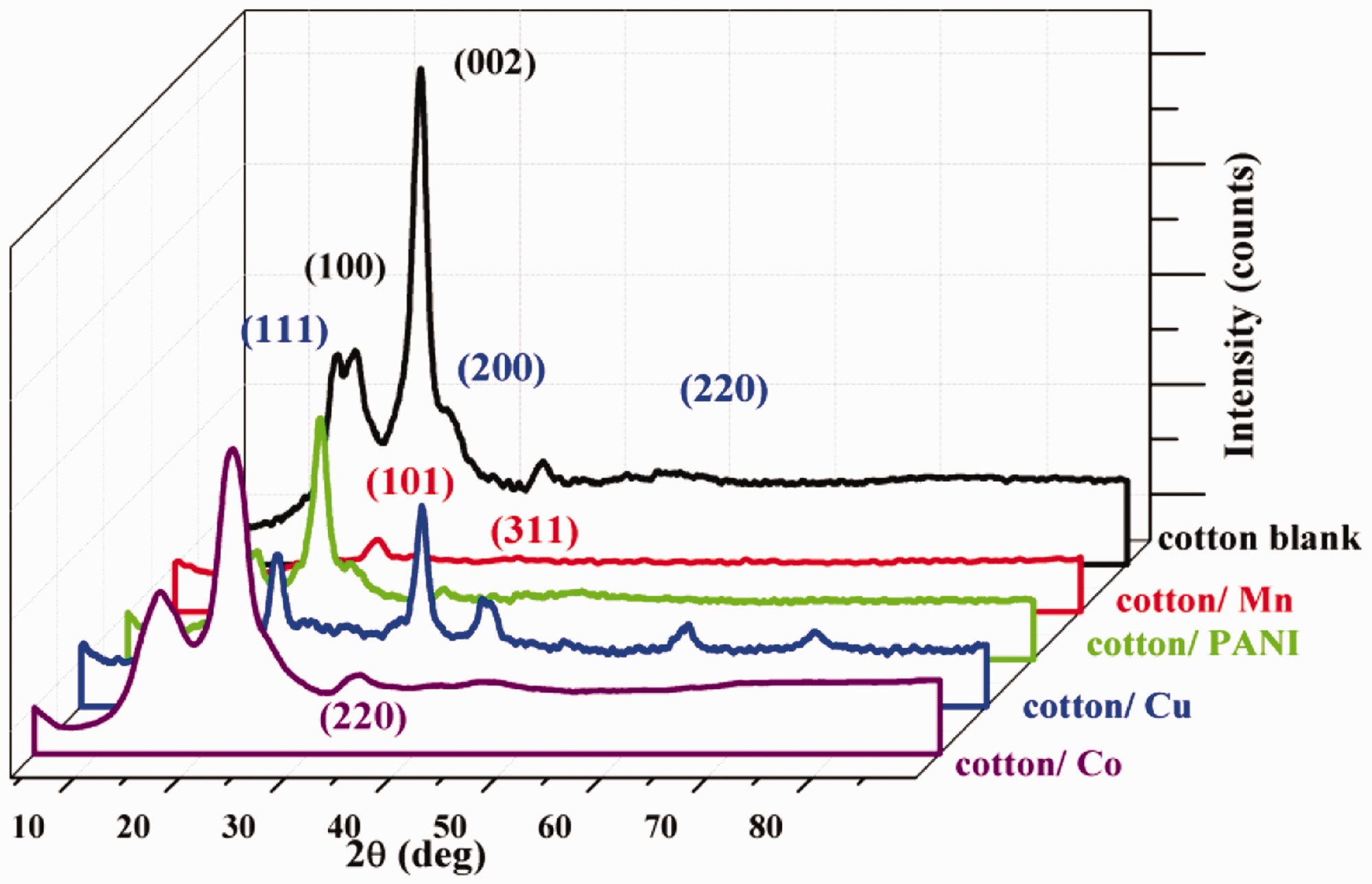
The XRD pattern of the cotton blank, cotton/PANI, and cotton/Mn, cotton/Cu, and cotton/Co.
The values of particle size (D), strain (ɛ), inter-planar spacing (d), area, full half-width maximum (FHWM), the height of Cu, Co, Mn Nps, and PANI powders.
Particle size distribution
The dynamic light scattering (DLS) method was retained to determine the particle size based on the Brownian motion of particles. The zeta potential of the nanoparticles gives an idea about the stability and dispersed in the medium. The nanoparticles in this case are suspended in distilled Millipore water. If the potential exists in the +30 to −30 millivolts range the particle is said to be stable. We have all the three particles of Cu, Co, and Mn in this range which conclude that they are not aggregating and are stable. The particle size distribution of cobalt and copper particles were presented in Figure 7. The copper nanoparticles were obtained to have a multi-modal distribution with size varying from micrometer to the nanometer range. The average particle size was gauged around 200 nm and it has advantages over those particles which are below 100 nm. The reduced inflammatory response and toxicity can be predictable from the copper particles produced in this work as compared to smaller copper particles (i.e. below 100 nm) of the same chemical composition [28,29]. In addition, since the scattering intensity is directly proportional to the sixth power of the particle radius, this technique is extremely sensitive towards the presence of small aggregates. From the figure, the relation between diameter size (dam. nm) and size distribution by intensity (rel.) can deduce that the particle size of copper and cobalt nanoparticles around 85 and 312 nm respectively. The radius of curvature effect is the main contributing factor for the large difference observed on the averaged diameter from DLS and XRD. For a particle larger than 100 nm, there are some factors such as electro-viscous and surface roughness effects that should be taken into attention for the interpretation of DLS results [30]. Moreover, In DLS, the range of sample concentration is highly dependent on the sample size. On the other hand, if the sample is too concentrated, then multiple scattering can occur. At high concentration, the particle might not be freely mobile with its spatial displacement driven solely by Brownian motion but with the strong influences of particle interactions. In the case of Co NPs, it is too dilute and has inter-particle magnetic dipole-dipole interactions, so paramagnetic particle has no surface coating, dispersed in deionized water. So in the case of Co Nps, particle size can be determined with XRD analysis (Table 3).

The particle size distribution for (a) cobalt and (b) copper particles.
Mechanical properties of treated cotton fabrics
The mechanism of textile action under stress-strain test is described as a calculation of the material resistance on the activity of outward forces causing the change of the fabric shape. The reaction of the textile material changes the material properties, the method of load, and its tension. Several parameters affect the mechanical properties of woven fabrics. Firstly, there are the structure, fabric properties, and molecular properties, where the macromolecules can be arranged in crystalline (exclusive arrangement of molecules) or amorphous (spontaneous arrangement of molecules) structure [21]. The macromolecules are faced beside the fiber axis and are associated with intermolecular bonds. When a force is operated, the supermolecular structure leads to altering [31].
In this work, we study different effects on the mechanical properties of treated cotton fabrics. The mechanical properties of treated cotton fabrics were characterized by tensile strength at breakpoint and elongation (%) measurements. For a visual representation of the dependence the value of Dissipated Energy for untreated and treated irradiated fabrics at 40kGy the reader is referred to Table 4. The ultimate value of dissipated energy is for cotton fabric treated by polymerization PANI, while the lowermost value is for cotton fabric untreated.
The values of dissipated energy for untreated and treated irradiated fabrics at 40 kGy.

Effect of graft yield on the mechanical properties
The data show the tensile strength of different graft yields for cotton fabrics with polyaniline (from 1% up to 14%). Figure 8 represents the tensile strength of cotton fabrics increased by grafting. The untreated fabrics tensile strength at breakpoint was 10.8MPa and its aniline treated forms had a tensile strength of 16.5 MPa, 19.5 MPa, 22.4 MPa, 27.6, and 31.3MPa for polyaniline graft yields. The tensile strength at the breakpoint of the fabrics was encouraged after the treatment and was reliant on the polyaniline graft yield. On all occasions, this increase can be ascribed to the rigid nature of PANI. The tensile strength at breakpoint rise was associated with the modification of the surface of cotton fabrics by the deposition of polyaniline due to reinforcing the effect of the conductive polymer [32]. Also, Figure 8 illustrates the elongation (%) of treated cotton fabrics was affected as well by grafting. In which, the elongation (%) was increased slightly at small graft yield (1%) and then diminished over the increase in graft yields up to 14%. These trends may be attributed to the fact that the formation of coordination bonding and entanglement similar to cross-links. Also, this decrease in the elongation values of polyaniline treated fabric is that it is influenced by the rigid nature of PANI [22].
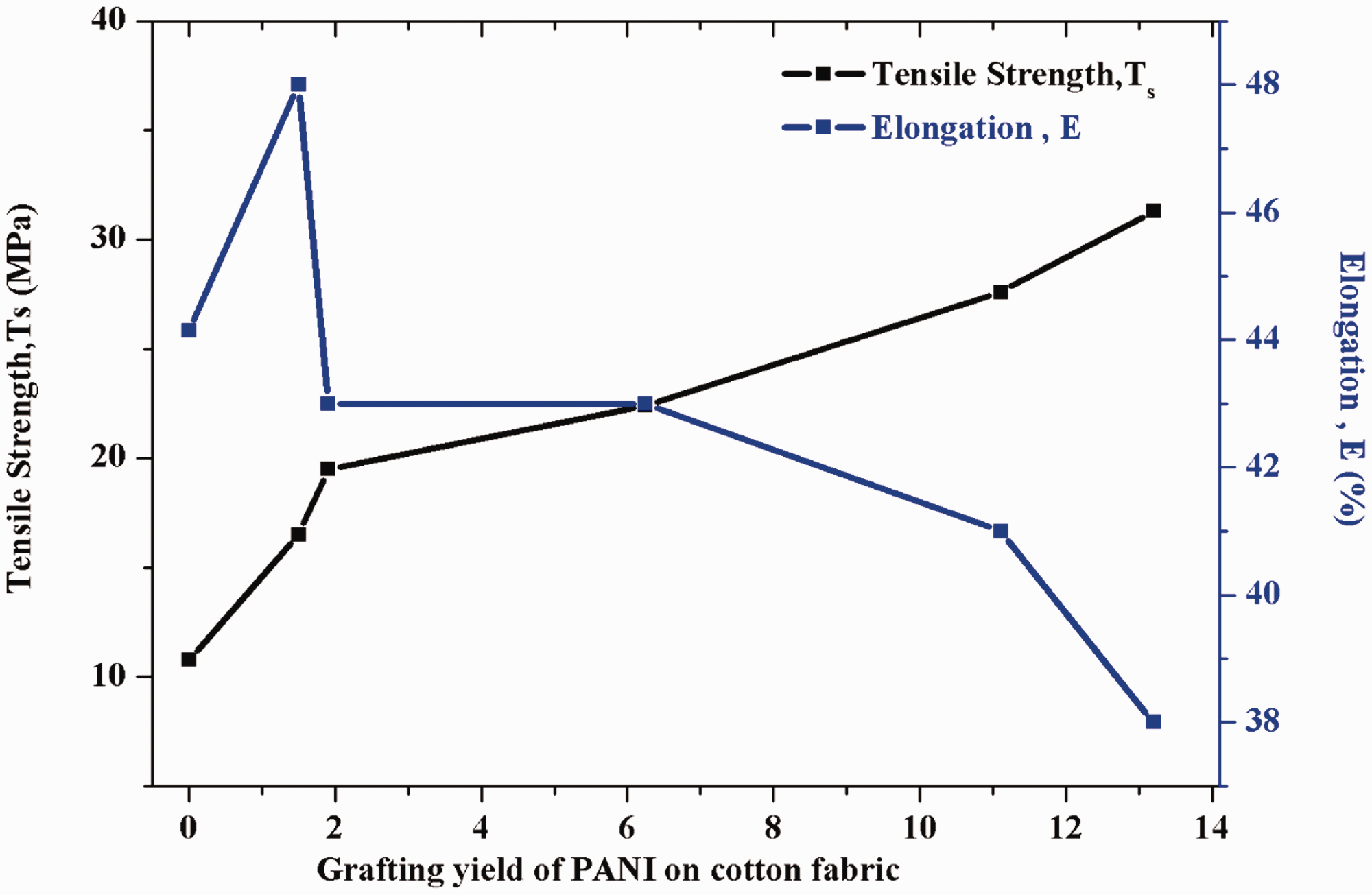
The relation between grafting yield of PANI on cotton fabrics with tensile strength, and elongation.
Effect of different treatments of cotton fabrics on the mechanical properties
The data show the tensile mechanical of untreated and different treated cotton fabrics with polyaniline, copper, cobalt, and manganese nanoparticles. As shown in Figure 9(a), the tensile strength at the breakpoint of cotton fabrics was affected by different treatments. Despite this, the tensile strength of grafted cotton fabrics with aniline was increased. The tensile strength of cotton fabrics treated with nano-metals was also increased by different values. It was spotted, the tensile strength of cotton fabrics treated with copper metal increased slightly compared to the tensile strength of other metals (Co and Mn). Consider Figure 9(b), which plots the elongation of cotton fabrics was not greatly marked by grafting with aniline and nano-metals deposition on cotton fabrics subject on the kind of metals. In which, the elongation (%) was increased slightly for cotton-PANI compared to the elongation of untreated cotton fabric. These trends may be attributed to the fact that creating more free radicals on fabric surfaces that are taken on the polymer chain molecule more effective way [33]. Oppositely, the elongation of cotton fabrics deposited with nano-metals was decreased slightly compared to untreated cotton fabrics. These trends may be attributed to the increase of the tough segment content on the surface of cotton fabrics. Upon tension, these semi-network structures withstand and resist tension.
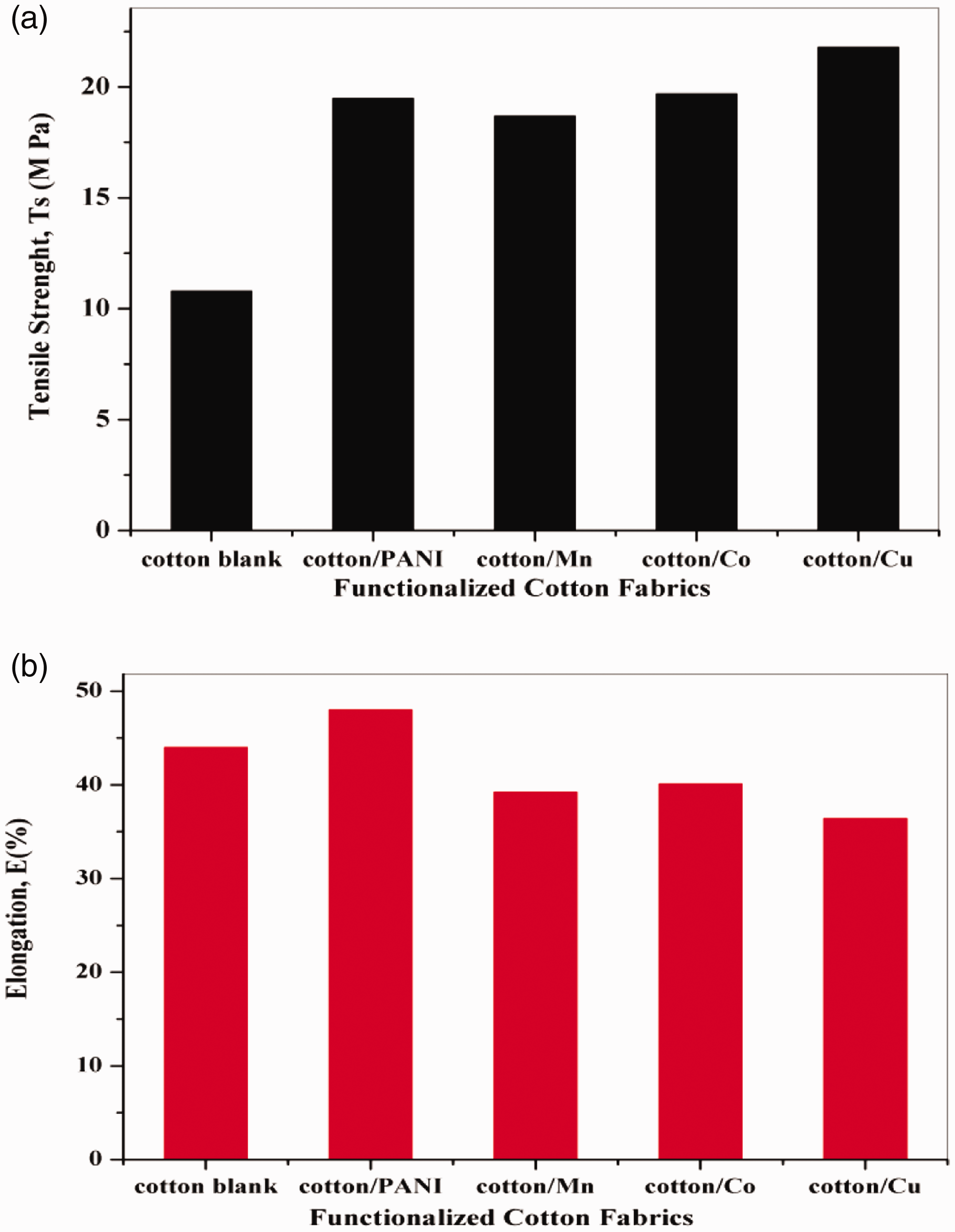
The relation between different treatment with (a) tensile strength Ts, and (b) elongation E%.
Effect of dose on the mechanical properties
The influence of the dose of radiation on the mechanical properties of treated cotton fabrics with (polyaniline, nano Cu, Co, Mn metals) was investigated. The irradiation process can cause two competing reactions in the polymer matrix; crosslinking or chain scission processes, depending on the total irradiation conditions [34]. Therefore, due to the important effect of the irradiation on the mechanical properties of treated cotton fabrics, the tensile strength at breakpoint and elongation (%) of the treated fabrics as a utility of irradiation dose can be found in Figure 10(a) and (b). It can be seen that the tensile strength of the treated fabrics was enlarged after different treatments. It can be realized that the values of the tensile strength for all treatments were escalated with increasing of the irradiation dose, accomplishment to their maximum values at about 40 kGy, and then it inclines to decrease faintly as it remarked with irradiation to 50 kGy, in which the cracking process begins as a result of the radiation chain scission route.
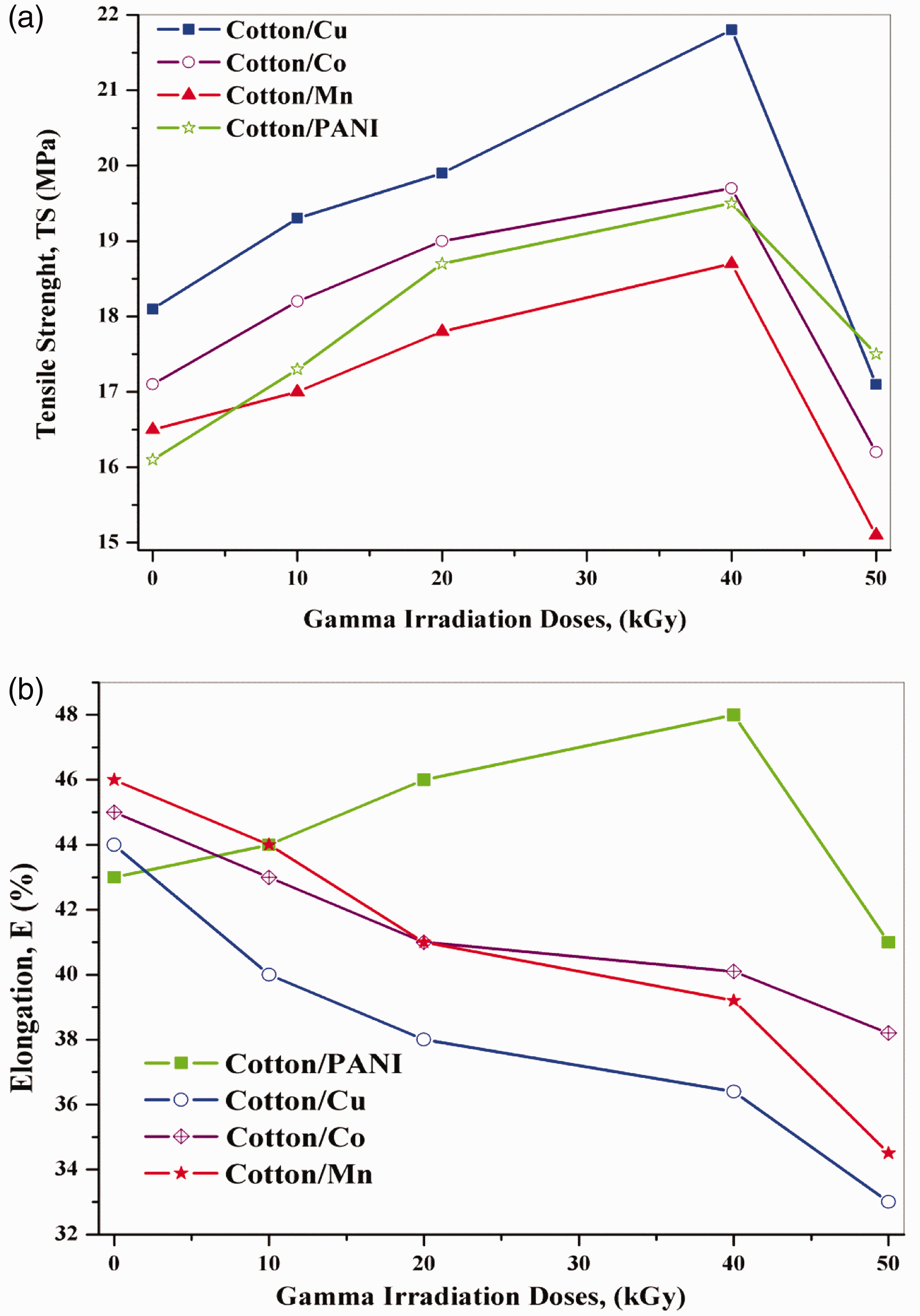
The relation between doses at different treatment with (a) tensile strength Ts, and (b) elongation E%.
Figure 10(b) outlines the effect of irradiation dose on elongation (%) of treated fabrics. It can be seen that the elongation (%) of PANI-g-cotton was increased slightly with cumulative the irradiation dose up to 40KGy and then tends to decrease at 50KGy. This tendency may be attributed to the gamma radiation was in authority for the higher extension of fabric because upper radiation generates more free radical on fabric superficial that infected polymer chain molecule more efficient way [33] and then decreases at 50KGy because of the increasing of the crosslinking density, preceding to the hindrance of the mobility of the molecular chains, resulted in the decreasing of overall elongation.
Alternatively, the elongation of cotton fabrics treated with nano-metals not greatly affected by an increasing dose of gamma radiation. Conversely, the elongation of cotton fabrics treated with nano-metals decreased slightly with increasing dose, this may be attributed to the attenuation of gamma radiation by different metals which decrease the effect of radiation on treated cotton. Overall results showed that the tensile strength capable of a definite total dosage of gamma radiation and then diminished. The polymeric structure of the cotton fabrics may be affected by gamma irradiation, which may be produced by active sites. At low-down radiation dose, bond scission and cross-linking happened, but at greater dose scission was favored. Gamma irradiation may be removed moisture from the composite also, which in turn interjected to better fiber-matrix adhesion. This may well be the purpose behind the increased mechanical properties of the composites [35].
The stress-strain curves were achieved as revealed in Figure 11, the stress-strain curves with different directions of the fabric area close to each other, which can be explained by the particularities of the fabric used in sample preparation. From this relative between strain and stress for untreated and treated fabrics, the elastic modulus was governed to analyze the data with the maximum strain. Young’s modulus E (determined from the initial slope of the tensile curves), the value of Young’s modulus E for untreated and treated irradiated fabrics at 40kGy are summarized in Table 4.
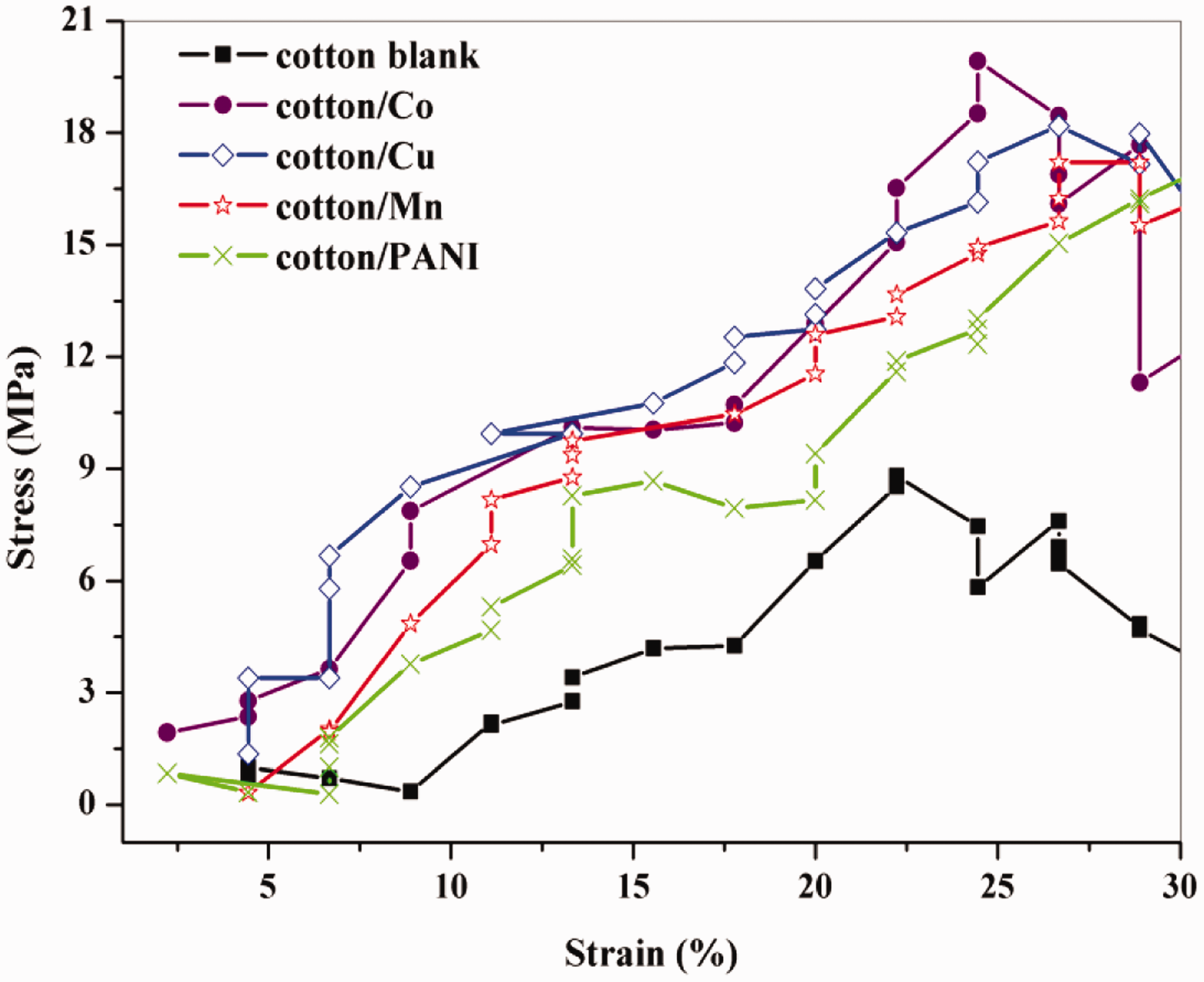
The stress-strain curves for untreated and treated cotton fabrics.
Dielectric properties (AC electrical conductivity) of functionalized cotton fabrics
The accountable of conduction in AC conductivities are surrounded by polarons and ions, equations (3) and (4) demonstrate that conductivity can be uttered according to [36]

AC Conductivity varies on materials electron conveying capacity. The conduction routes of conducting PANI can become supplementary incessant, nearby in the connection, and tinier the distance between the inter-chains and hopping of charges [37,38]. Figure 12 gives the AC conductivity with frequency for untreated and treated cotton fabrics. It can be seen that the AC conductivity increases with frequency increasing which a polymeric and metallic comeback; this was expected to the growth of the charge carrier mobility in PANI and nanometals-treated cotton composites. The AC conductivity showed individuality of frequency up to 3 × 106 Hz, which was due to unsystematic diffusion of the charge of a carrier. In the case of cotton fabric was treated by Mn Np AC conductivity was the highest values about other treated fabrics. After polymerization for PANI and dipping for (Co, Mn, and Cu Np), the increase in the AC electrical conductivity may be due to the uniform distribution of nanoparticles and an upturn in crystallite density in unit space which is evident from the XRD result. The mishmash of amorphous and crystalline structure in the composite material may similarly be the aim of perfected conductivity [39].
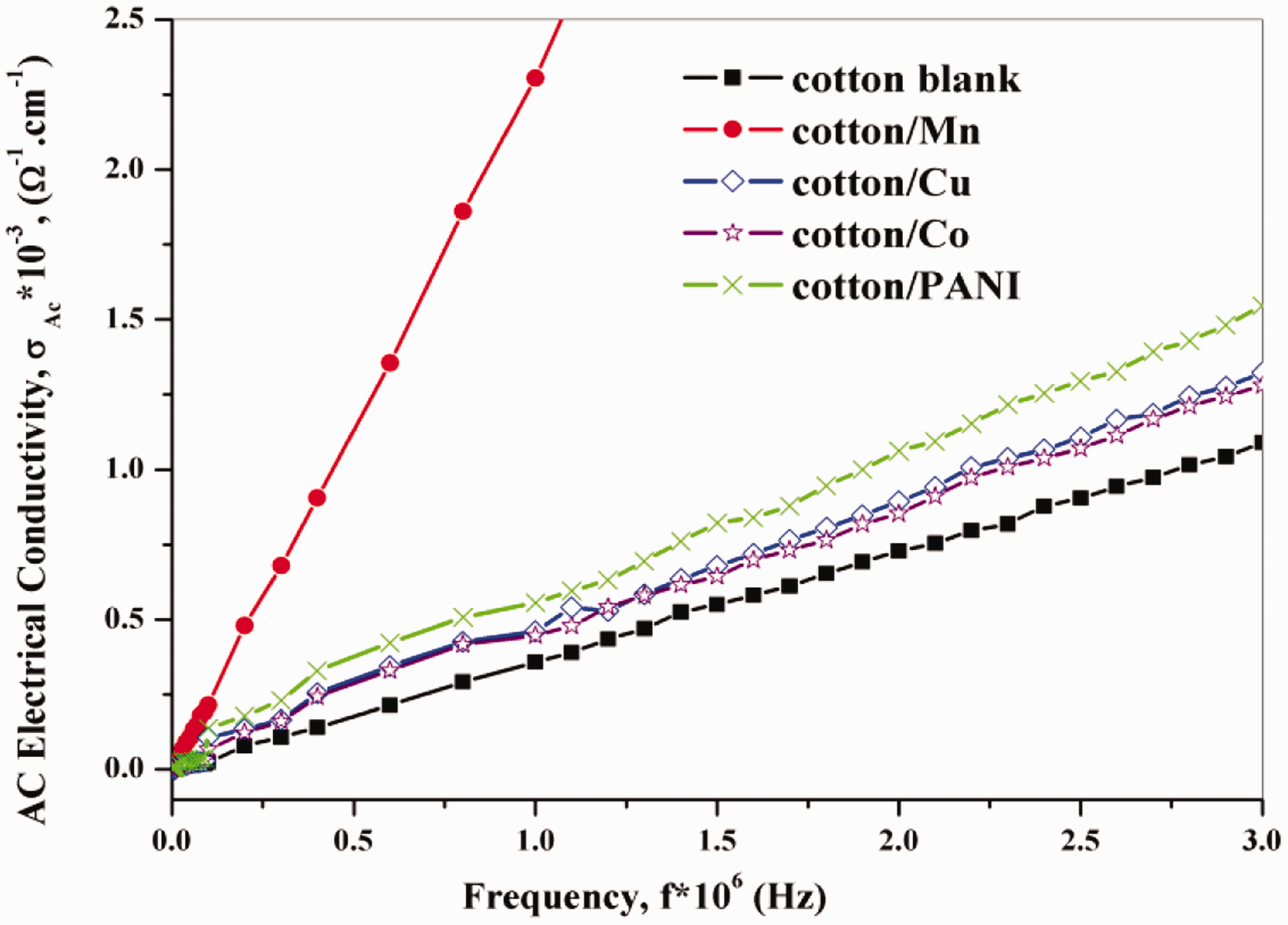
AC conductivity dependence of frequency for untreated and treated cotton fabrics.
When the material was subjected to an electromagnetic field, the electric field convinced two types of electrical currents inside the material, namely the conduction current and displacement current. The conduction current happened due to the occurrence of free electrons, which provided the real part of permittivity (ε1). The real and imaginary permittivity was called the ‘‘dielectric constant’’ and “dielectric loss” respectively. The dielectric loss was dependent on frequency for untreated and treated cotton Fabrics as shown in Figure 13. It can be seen that, the values of dielectric constant decrease with expanding frequency. The fundamental value of the dielectric constant of polar materials was elevated at lower frequencies.
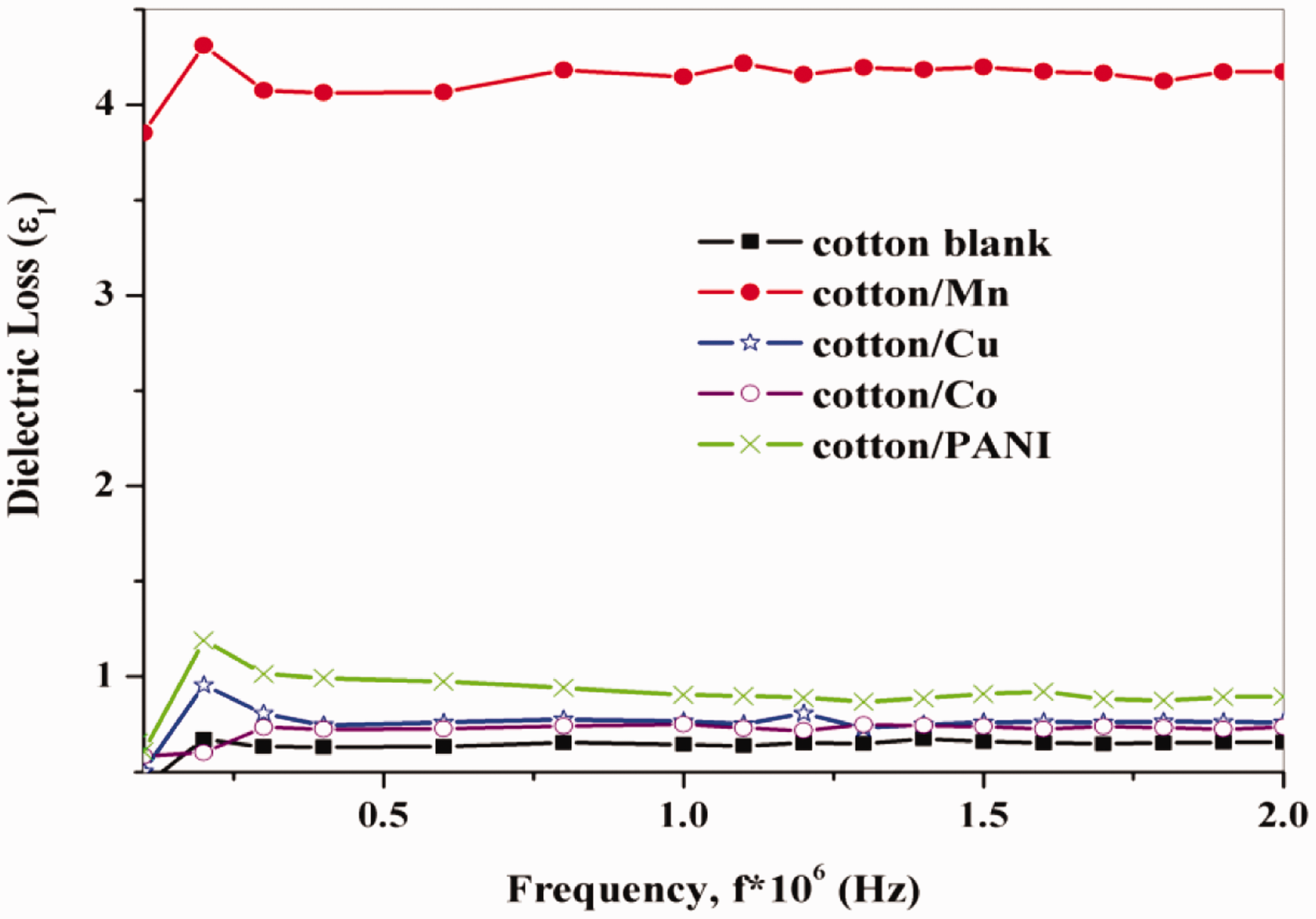
The dielectric loss dependent on frequency for untreated and treated cotton fabrics.
As follows from Figure 14 are shown the dependence of frequency with Impedance spectroscopy and parallel capacitance. The impedance decreases with increasing frequencies. This decrease occurred without bound until the LCR meter registers an overload reading and was due to a large amount of leakage current present in reverse bias. In the forward bias direction, we could see a small decrease in capacitance in most of the curves. This observation was consistent with the Inversion mode in curves made at a low frequency, but not quasi-static. At room temperature, we could be able to complete measurements over a much smaller voltage range on these devices. The capacitance seemed to be less dependent on the bias at positive voltages.
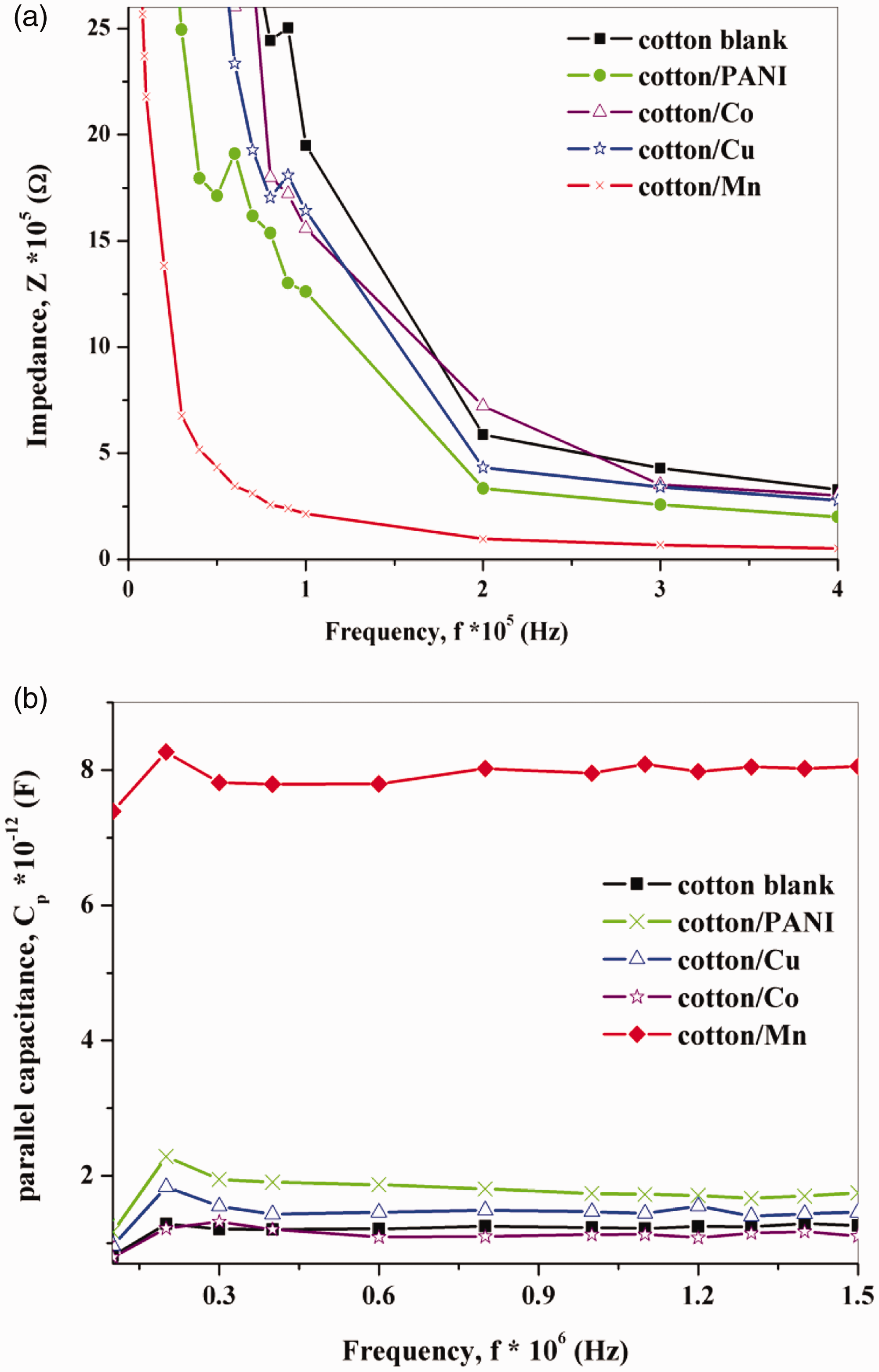
The relationship of the frequency with (a) impedance and (b) the parallel capacitance for untreated and treated cotton fabrics.
ESR spectra
The energy absorption spectra of many materials can be measured by Electron spin resonance (ESR). Also, the net amount of magnetic moments can be estimated by ESR spectroscopy, which is proportional to the sample amount. ESR- spectrometer permits the investigator to detect the molecules with unpaired electrons (free radicals) not including destroying these molecules in any sample. Also ESR –Spectroscopy is an essential tool to study the structure and dynamics of molecular systems inclosing one or more unpaired electrons, where these molecules are characterized by high oxidizing activity. Each molecular system contains unpaired electrons that have ESR spectra. Whilst, the molecular system containing paired electrons have not ESR spectra. Free radicals mind to react to gain one electron or lose its one electron to reach a stable state.
The electromagnetic radiation can be regarded as the coupled electric field (E) and magnetic field (H), where the two fields are perpendicular to each other and the direction of propagation and they oscillate by the same frequency ν within the range from 101 to 1011 spins, or from 1 to 100 GigaHertz .when a photon is captivated by an unpaired electron within the sample, we found that this unpaired electron transfer from the ground state to excited state, under the condition, ΔE = h ν (resonance condition), this implies, the absorbed energy h v must equal to the energy difference Δ E between to the ground state and excited state to occurring transition [40]. Where; h is Plank's constant, and υ is the frequency of the radiation. The number of spins (spin concentration) was calculated by using the formula
Figure 15 depicts the ESR spectrum for cotton/Co, Cu, and Mn nanoparticles and cotton/PANI. The hyperfine transitions in ESR spectra of cotton/Mn Np are contributed by the interaction of the Mn+2 ions that have 3d5 electronic configuration, electron spin, nuclear spin, and the electronic ground state of 6S5/2, and spectroscopic splitting factor of g ∼ 2.002. the characteristic sextet of hyperfine splitting (HFS) of Mn2+ ions, resolved six fine ESR signals which are interrelated to the “paramagnetic character” of Mn+2 ions are originate; because of the interchange between the thermal and radiofrequency energy population of the energy levels [41].

The ESR spectra for (a) surface metallization of nano-metals (Cu, Co, Mn) on cotton fabrics, and (b) polymerization of aniline on cotton fabrics (cotton/PANI).
The effective method for studies into the conduction mechanisms of conducting polymers is ESR. ESR has been often used to explore the nature of various quasi-particles [42–45] (such as excitons, polarons, and bipolarons) that can perform a range of roles in the electronic charge transport developments in the ground and excited states. ESR has also been surveyed the population and adjustable evolution of the polaron to the bipolaron in conducting polymers [44]. Conducting polymers classically exhibits a single broad resonance line located close to the g value of 2.0092 which has been ascribed to the existence of the non-spin paired (s = 1/2) polaron. At upper levels of oxidation, polarons have no ESR response as shown in Figure 15(b) for cotton/PANI, yet, this does not essentially denote the attendance of spinless, and bipolarons in this material. Cotton/PANI was shown to be redox immobile, incapable of supporting a delocalized polaron due to the break-in conjugation subsequent from these defects signifying that the reported phenazine-like defects [46,47]. The applied magnetic properties of the conducted form of PANI are due to polaron spins (radical cations). The narrowing of the ESR lines of polyaniline is probable due to the stationary delocalization of polarons mostly because of the linewidth of polyaniline. Table 5 summarizes g- values, peak-to-peak line width (ΔH), and spin concentration (N).
The values of (gǁ, and g⊥) g-factors, (H0) magnetic field at resonance peak, (Ns) number of the carrier, and (A) peak height for treated irradiated fabrics at 40kGy.

Cu+2 ion has 4s1 electronic configuration, the electronic ground state of 2D5/2, and a spectroscopic splitting factor with g ∼ 2.055 [48]. The g-value identical to the central resonance line of EPR spectra was established to be 2.063 where four resolved EPR resonance lines were observed.
Co+2 ion has 3d7 electronic configuration, electronic ground state of 4F9/2, and spectroscopic splitting factor with g//∼ 2.059 and g⊥ ∼ 2.0898 [48,49]. A discrete and unclear broad EPR resonance line was recorded.
Conclusion
From our research that can be prepared conductive fabrics by polymerization of aniline on cotton fabrics (cotton/PANI) and surface metallization for nano-metals (Cu, Co, Mn) on cotton fabrics by a novel method which named “pad dry curing method”. Based on the results, it can be concluded that the nano-metals prepared successfully and can be confirmed by DLS, XRD, and SEM analyses. The effect of (graft yield%, and the dose of radiation) for cotton/PANI fabric and different surface treatments cotton fabrics for nanometals on the mechanical properties was investigated. The tensile strength of treated fabrics was increased with increasing the graft yield of PANI. The findings of our research are quite convincing, and thus the following conclusions can be drawn: the tensile strength increase with an increasing dose of radiation from 10 to 40 kGy and then decreases at 50 KGy.While the elongation of treated fabrics decreased with the increase of graft yield of PANI and dose of radiation. Summing up the results, it can be concluded that the electrical conductivity values of different treated cotton fabrics were increased by two orders from untreated fabrics. The higher values of surface and volume conductivities resulted in treated with PANI than these metal-dipped fabric samples. Later, in this work, we develop multifunctional electrical conductive cotton fabrics with good mechanical properties. Afterward, The enhancement in the electrical conductivity of these conductive textiles is accepted to develop the Electromagnetic shielding application; Conductive fabric cotton/PANI is the best-functionalized fabric to be used in electromagnetic shield application. Shielding functionalized fabrics protect electronic devices, electrical equipment, and gadgets against electromagnetic energy; and can be furnished on the wall surface, and covered around the radiation source, and also used to isolate a space (a room, an apparatus, a circuit, etc.). EM protection products from conductive fabrics are such as shielding tents, and shielding curtain.
Footnotes
Acknowledgement
The authors gratefully acknowledge the generous financial support of the Egyptian Atomic Energy Authority (EAEA).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
