Abstract
To investigate the effect of linear, rod and granular nano-silver structures on the electrical conductivity and wear resistance of cotton fabrics, silver nanowires (AgNWs), silver nano-rods (AgNRs) and silver nanoparticles (AgNPs) were separately obtained by solvothermal method, and then adsorbed on the cotton fabrics. Compared with AgNRs and AgNPs that silver nanowires could form network structure on the cotton fabrics. As the ratio of nano-sliver length to diameter increases, the effective adsorption mass of nano-sliver on the cotton increases, improving the conductivity of the textile electrode. No matter how many times of rubbing, it had no effect on the square resistance of the AgNWs/cotton. The cotton fabrics treated with silver nanowires had the best performances and wear resistance.
Introduction
In recent years, the emergence of wearable electronic equipment has expanded the application of textiles. Conductive textile fabrics [1–4] have attracted widespread attention and applied in capacitors [5,6], electromagnetic shielding [7], sensors [8]. Textile fabrics have better flexibility, air permeability and wear resistance. They could perform bending, torsional rubbing and other mechanical movements to meet the movement requirements of people's daily life [9,10]. Therefore, conductive fabrics based on fabrics are more suitable for the development of wearable equipment and are widely used in many fields [11,12]. Among many kinds of fabrics, cotton fabric has been widely used as the base material of conductive textiles because of its low price, wide range of uses, extra absorbent, soft and comfortable characteristics [13,14].
Conductive cotton fabric [15] is composed of cotton fabrics, electronic components, and conductive materials and so on. Conductive material is an indispensable part of conductive cotton fabrics, which is mainly used to transmit electric energy and signals [16], providing good conductivity for cotton fabrics. Silver nanoparticles have high specific surface area, thermal conductivity, conductivity, transparency, ductility, mechanical strength and other properties [17,18]. It is widely used in flexible conductive films, solar cells, touch screens, display screens and sensors. And also is one of the ideal conductive materials for conductive textiles. Silver nanoparticles can not only give the cotton fabrics excellent conductivity, but also endow the cotton fabrics good anti-bacterial [19] and anti-ultraviolet [20] and other additional properties. Preparation methods of nano-silver can be divided into chemical reduction method [21], photo reduction method [22] and solvothermal method. Solvothermal method [23] is one of the effective methods to synthesize silver nanostructures such as nano-films, nano-wires, nano-rods and so on. It has the characteristics of relatively simple preparation process, easy control and effective prevention of volatilization of toxic substances in closed system. Kwak et al. [24] formed a uniform and continuous layer of nano-silver prepared solvothermal method on the surface of cotton fabrics by low temperature thermal reduction process, which made cotton fabrics have conductive properties (3.92 + 0.18Ω/sq) and good antimicrobial.
Researchers tend to use different methods to prepare conductive nano-silver/cotton fabrics with excellent properties, but few reports mention their wear resistance. Wear resistance is an important factor to determine the service life of conductive nano-silver/cotton fabrics. Therefore, it is difficult for nano-silver particles to be fixed on the surface of fabrics for a long time, which results in the wear resistance of most nano-silver conductive fabrics are poor. The high wear resistance of silver-plated cotton fabrics is an important problem affecting its use, because in complex working environment, the silver particles on the surface of cotton fabrics are easily destroyed and even worn out. Therefore, the preparation of cotton fabrics with high conductivity and wear resistance poses a great challenge to the preparation technology of conductive fabrics.
Generally, the conductivity of nano-silver depends on its shape and size [25,26]. But whether different morphologies of nano-silver can also affect the wear resistance capacity of conductive cotton fabrics. With these in mind, we herein designed three morphologies of nano-silver (wire-like, rod-like, granular) by hydrothermal method, which was easy to hydrogen bond with hydroxyl groups of cotton fabrics. Comparing the electrical conductivity and wear resistance of cotton fabrics treated with three morphologies of nano-silver. Moreover detail mechanism is discussed and calls for contribution to understand the wear resistance properties of nano-silver in order to facilitate the development of more efficient electrical conductivity and long used cotton fabrics.
Experimental
Materials
Sliver nitrate (A.R.≥99.8%) was purchased from Tianjin Kermel Co., PVP was purchased from Hai Lanji Biological Co., FeCl3·6H2O (A.R.≥99.8%) was purchased from Tianjin Kermel Co., ethylene glycol was purchased from Tianli chemical Co., as based materials. Commercially available cotton fiber was used, and all chemicals were used without further purification.
Preparation of nano-silver structures
In this paper, silver nanoparticles with different morphologies and length-diameter ratios were prepared by hydrothermal method using PVP as growth guiding agent and ethylene glycol as reducing agent. At the first, we prepared two solutions. Solution 1: 30 mL of PVP (MW = K30), Glycol(EG) (99.8%) solution with FeCl3·6H2O (control the mole of silver nitrate) were added and completely dissolved in the room temperature. Solution 2: a certain mass of AgNO3 (0.09 M, 99.8%) solution was added in 30 mL of EG to dissolve. After that, two solutions were poured into the reactor and synthesized at high temperature. In the process of synthesis, based on the single factor of temperature, time and FeCl3·6H2O concentration, the reaction conditions were controlled to explore the best process for the synthesis of silver nanowires. After synthesis, cooled to room temperature. Finally, separated silver nanoparticles with acetone (1:1 ratio) and centrifuged two times for 30 minutes at 3,000 rpm. The Ag nanoparticles then dispersed in ethanol andcentrifuged at 3,000 rpm for 30 minutes. By controlling the reaction temperature, reaction time and the amount of FeCl3·6H2O, a series of nano-silver solution with different colors were obtained. Solutions was directly used for characterization of ultraviolet-visible spectra, scanning electron microscopy and so on.
Preparation of nano-silver/cotton fabrics
We prepared three kinds of cotton fibers with different nano-silver structure by simple post-finishing method. Firstly, dipping process was carried out at room temperature for 60 seconds on the surface of cotton fabrics (5 × 5 cm size). Preparation of silver nanoparticles with three different morphologies: AgNPs, AgNRs, AgNWs. Then, washed cotton fibers were impregnated in three nano-silver solutions. Cotton fabrics with silver nanoparticles was dried at 105 °C for 10 minutes. Finally, three different morphologies of nano-silver cotton fabrics were obtained: AgNPs/cotton fabric, AgNRs/cotton fabric, AgNWs/cotton fabric. A schematic representation of the fabrication method for the conductive cotton fabrics process was shown in Figure 1.

Schematic of fabrication of nano-Ag/cotton fabrics.
Characterization
The evenly dispersed silver nanowire solution was dripped onto the tin paper with a needle tube, the electric heating wind was gently blown dry, and the morphologies of nano sliver was detected by scanning electron microscope (SEM) (Rigaku Co Ltd, Japan). After drying the silver nanowire solution with diluted concentration of 1 wt%, the morphology of silver nanoparticles was observed by transmission electron microscope (TEM) (FEI Inch, America). The structure and composition of nano sliver were measured by X-ray diffraction (XRD) (Bruker Co Ltd, Germany) and ultraviolet visible absorption spectrum (UV) (Agilent Co Ltd, America). The sliver contents of the cotton fibers were measured by using inductively coupled plasma atomic emission spectrometry (ICP) (Waters Co Ltd, American). The sensitivity of the cotton fibers were measured by a cyclic voltammetry test using electrochemical workstation (Chen Hua Co Ltd, Shanghai, China) and the potentia l (E) and electric current (I) were recorded. The resistance (R) of the conductive cotton sheets (1 × 1 cm) was measured by a four probe tester. By using rubbing color fastness tester, the treated cotton fabrics were placed at the end of the tester, and rubbed back and forth for different number of rubbing on the dry-rubbed sample along the track straight line of (104 ± 3) mm until the surface of the treated cotton fabrics ruptured. The number of rubbing was recorded in the meanwhile. The resistance of the rubbing fabrics was measured and the photos of the rubbing fabrics were taken. E. coli was cultured by bacteriostasis method to detect the antibacterial properties of cotton fabrics and calculate the antibacterial rate.
Results and discussion
Characterization of nano-silver structures
Effect of reaction temperature on the morphology of silver nanowires
In the process of preparing AgNWs by hydrothermal method, ethylene glycol must be converted to glycol aldehyde first, then Ag+ can be reduced to Ag, and the corresponding environment for the subsequent growth of AgNWs is provided. Only at high temperature, ethylene glycol could be converted to glycol aldehyde, so the reaction temperature has an important influence on the morphology and size of the final product. First of all, we controlled the reaction time and FeCl3·6H2O concentration as invariants and carried out single-factor optimization for the reaction temperature.
Figure 2 displays the SEM photographs of nano-Ag structure prepared at different reaction temperature with reaction time of 6 h, FeCl3·6H2O concentration of 1‰. During the synthesis process, it was found that the nano-silver structure could not be generated under the condition of less than 140 °C when the other conditions were the same. When reaction temperature was 140 °C, the solution was full of silver nanoparticles. The decomposition of silver atoms requires higher energy in the crystal growth process. But the temperature isn't high enough to provide energy for the silver nanoparticles to grow into rods. When the reaction temperature rose to 150 °C, nano rod-like products could be observed in the picture, which were irregular in length and shape. This is due to the low activity of the whole reaction system, the insufficient kinetic energy of the growth of silver nanowires. When the reaction temperature was 180 °C, we could see that the morphology of silver nanowires were better appearance, and the length is about 5–10 μm. This is because the reduction rate of silver nitrate is accelerated by high temperature, and the activity of the reaction system is enough to provide energy for the growth of AgNWs [27]. When the temperature continues to rise to 190 °C, the morphology of the silver nanowires is similar to that of 180 °C. Considering of energy conservation, the optimum reaction temperature of silver nanowires was 180 °C.

SEM images of nano-Ag with different reaction temperature.
Effect of reaction time on the morphology of silver nanowires
Figure 3 shows the scanning photographs of silver nano-Ag structure prepared at different reaction time in 180 °C, FeCl3·6H2O concentration of 1‰. As can be seen from Figure 3, the morphology and particle size of AgNWs in the SEM resultant are controlled by changing the reaction time. When the reaction time was 6 h or 7 h, the product was a mixture of irregular Ag nanoparticles and rod-like with uneven size. This is because at the beginning of the reaction, a large number of PVP could not play the role of structure-directing agent because of its poor solubility, resulting in a large number of irregular Ag nanoparticles in the product. When the reaction time was extended to 8 h, a large number of AgNWs were in the product. The PVP molecule interacts with Ag and selectively adsorbs on a crystal surface of Ag, resulting in anisotropic growth of Ag nanoparticles, which leads to the product transformation from rod-like to linear. When the reaction lasted for 10 hours, the number of nanowires in the product decreased but the number of nanoparticles increased. Because in the process of crystal growth, the synthesis reaction is excessive and a large number of silver monomers are formed, resulting in new twins and sums. Over reaction time is not conducive to the growth of silver nanowires, thus affecting the final reaction structure.

SEM images of nano-Ag with different reaction time.
Effect of FeCl3·6H2O concentration on the morphology of silver nanowires
In the process of synthesizing silver nanowires by hydrothermal method, Cl− in FeCl3·6H2O can promote the growth of silver nanowires to a certain extent. At the initial period of the reaction, chloride ions combine with silver ions to form silver chloride colloids. Then silver ions gradually agglomerate to form seeds, silver elements could grow orderly along the (111) crystal plane.
In order to investigate the effect of different FeCl3·6H2O dosages on the growth of AgNWs, concentration of FeCl3·6H2O varied from 0‰–5‰ when the reaction temperature was 180 °C, and the reaction time was 8 h. From the morphology comparison of Figure 4, under the lower content of FeCl3 ·6H2O the length of silver nanowires was relatively long and diameter was smaller. With the increase of chloride ions concentration, the length of silver nanowires decreased and the size of silver nanowires increased. This may be due to the fact that when the concentration of chloride ions in solution is too high, more twins are formed, while the total amount of silver ions is limited. Silver elements need to grow along the (111) surface of the twins and the number of silver nanowires reduce. When the content of FeCl3·6H2O was 1‰, the quantity and quality of silver nanowires were the best, and the length-diameter ratio of silver nanowires reached the maximum.

SEM images of nano-Ag with different FeCl3·6H2O concentration.
Generally speaking, at the same reaction temperature (180 °C) and time (8 h), the optimum process for preparing AgNWs are that the FeCl3·6H2O concentration is 1‰. AgNPs can be prepared without adding FeCl3·6H2O. When preparing AgNRs, 2‰ FeCl3·6H2O is added.
Morphology and surface property
Figure 5(a) presents the SEM and TEM photographs of AgNWs (a-a'), AgNRs (b-b') and AgNPs (c-c') by hydrothermal method. Their reaction temperatures were 180°C, reaction time is 8 h. The concentration of FeCl3·6H2O is 1‰, 0‰ and 2‰. AgNWs with average diameter of 40 nm (a') and length of 60 μm are prepared. Its length-diameter ratio is about 1000. AgNRs with a length of 10 μm and a diameter of about 83 nm (b'), its length-diameter ratio is about 100, as well as AgNPs (96–118 nm in diameter, c') are prepared.

(A) SEM and TEM images of AgNWs (a-a'), AgNRs (b-b') and AgNPs (c-c'); (B) Ultra-violet absorption spectra of a, b, c; (C) XRD pattern of a, b, c. (a-(AgNWs), b-(AgNRs), c-(AgNPs)).
At the same time, AgNWs, AgNRs and AgNPs are characterized by UV (Figure 5(b)). There are obvious absorption peaks (AgNPs) at 352 nm in all three samples, so a small amount of AgNPs are produced in all three samples, which is consistent with the results of SEM. There is a distinct characteristic peak (390 nm) in the spectrum in curve a. 390 nm is the characteristic peak of AgNWs, which represents the longitudinal and transverse resonance peaks of one-dimensional nanowires of AgNPs, indicating that AgNPs begin to grow into one-dimensional nanowires in an anisotropic direction. The result shows that AgNWs have been successfully prepared. For curve b, the characteristic peak at 425 nm is caused by the dipole resonance on the outer surface of AgNRs, which indicates that the final products should be AgNRs. However, compare with curve c, the absorption intensity at 425 nm is much lower than that in curve b, indicating that the content of AgNRs is very few in curve c. For curve c, there is another obvious peak at 352 nm, which proves the final product is AgNPs, which is consistent with the SEM results.
Figure 5(c) exhibits the XRD pattern in which the diffraction peaks of curves a, b and c at 2Ɵ degree was 38.2°, 44.5°, 63.8°, 77.4°, 82.6° can be seen respectively, consistent with those of (111), (200), (220), (311), (222) crystal plane diffraction peaks of silver in standard contrast cards, indicating that a, b and c are monocrystalline silver with plane-centered cubic structure. In addition, it can be found clearly the intensity of diffraction peak increased and FWHM reduced evidently, with the increase of length-diameter ratios. The highest diffraction peak of AgNWs indicates that its crystallinity is the best.
Performance of cotton fabrics loaded with nano-silver
Dipping treated fabrics
There are many hydroxyl groups in the surface of the cotton fibers, which can combine with silver nanoparticles to form hydrogen bonds. Cotton fabrics are mainly treated by dipping method in this experiment, so that the surface of cotton fibers is adsorption to silver nanoparticles to endow the fabrics with electrical conductivity. In the integral experiment, the content of nano-silver on the fabrics could be controlled by changing the impregnation times. The properties and applications of nano-silver materials are greatly influenced by their structures and morphologies.
The conductivity of fabrics is investigated by measuring the square resistance of fabrics with four probe method. The larger the square resistance of the fabric is, the worse the conductivity. The smaller the square resistance of the fabric is, the better the conductivity by contraries. Table 1 shows the square resistance of cotton fabrics dipped different times with different morphologies of nano-silver. Compared with the data in the table, it can be found that the square resistance of cotton fabrics can be significantly reduced when the three different solution are applied to the finishing of cotton fabrics respectively. When the AgNWs solution is applied to cotton fabric finishing, the square resistance of cotton fabric decreased significantly with the increase of impregnation times. It can be seen that the AgNWs has excellent conductivity. When impregnation times are more than 3 times, the square resistance of cotton fabric can reach 0.239 Ω/sq and tend to be stable, which gives cotton fabric excellent electrical conductivity. With the increase of dipping times, the square resistance of cotton fabrics tends to stability. This is because too many AgNWs would destroy the interpenetrating network.
Square resistance of cotton fabrics dipped different times with AgNWs, AgNRs, AgNPs.

When AgNRs and AgNPs are applied to cotton fabrics finishing separately, the square resistance of cotton fabrics with different dipping times do not change significantly. This is due to the shorter length of rod-like and granular nano-silver, which could not form conductive pathways between them, it has contributed to the square resistance changed slightly. When the times of dipping was five times, the square resistance of cotton fabric dipped with AgNWs solution was the smallest, followed by solution AgNRs and AgNPs. Therefore, the conductivity of AgNWs is better than that of rod-like and granular nano-silver, and the higher the length-diameter ratios, the better the conductivity of nano-silver. Therefore, by controlling the dipping times of fabrics, the optimal results of conductivity of AgNWs can be obtained, that give good electrical conductivity of fabrics, which provid possibilities for the application of fabrics in electronic textiles.
Table 2 below shows the size and morphology of silver nanoparticles in this paper and the properties of finished cotton fabrics compared with those in the literature. As can be seen from Table 2, the conductive effect of cotton fabrics is closely related to the morphology, diameter and size of silver nanoparticles [24,28–31]. The AgNWs/cotton fabrics prepared in this paper had excellent square resistance compared with the conductive cotton fabrics reported in the literature. This is due to the large length-diameter ratio (1000) of AgNWs, which is conducive to the formation of conductive networks and further increases the possibility of conductive network connectivity.
Comparison of the conductivity of the size and morphology of silver nanoparticles and the properties of finished cotton between the literature and the present work.

Morphology and content of nano-silver
Figure 6(a) and (d) is the scanning photographs of cotton fabric after five times immersion in nano-silver solution with different morphologies. Silver nanoparticles were loaded on the surface of cotton fabrics. It can be seen intuitively that the AgNWs in the surface of cotton fabrics treated with AgNWs intertwine with each other, forming a coherent conductive path, which is consistent with the square resistance test results of cotton fabrics. The rod-like and granular silver nanoparticles were loaded on the surface of cotton fibers, but couldn’t form a conductive network between the cotton fibers which makes the conductivity of cotton fabrics poor.

SEM images of cotton fibers dipped five times (a-untreated cotton, b-AgNWs/cotton, c-AgNRs/cotton, d-AgNPs/cotton); e-content of Ag of cotton fabric by ICP.
The Figure 6(e) presents ICP method to detect content of Ag of cotton fabrics after five times impregnation with nano-silver solution with different morphologies. Cotton fabrics treated with AgNWs, AgNRs and AgNPs are 1#, 2# and 3# respectively. It can be found from the figure that the maximum content silver loading of AgNWs/cotton fabric can reach 30.39 mg/g, indicating that cotton fabric has considerable adsorption capacity for AgNWs in solution, and that the lowest of AgNPs/cotton fabric is 3.086 mg/g. AgNWs/cotton with the maximum content silver loading has the best conductivity and AgNPs/cotton has the worst conductivity. In the process of adsorbing cotton fabrics, AgNWs not only deposit on the fibers, but also entangle and wrap the fibers, and the fibers also intertwine with each other. Silver nanowires have the highest adsorption capacity on fabrics, while AgNRs and AgNPs can only be deposited on fibers, which make the adsorption capacity lower. There are many nano pores in the sliver-coated fabrics, which are conducive to increase the factual contact area between the conductive material and the cotton fabrics. As the ratio of nano sliver length to diameter increases, the effective mass of nano sliver on the cotton increases, improving the conductivity of the textile electrode. Therefore, the biggest length-diameter ratios of AgNWs deposited the most on cotton fabrics, then AgNRs, and finally AgNPs.
Electrochemical detection
The Figure 7 shows the cyclic voltammetry of cotton fabrics dipped with nano-silver with different morphologies and times. With the increase of impregnation times, two sensitive and reversible redox peaks are obtained in the cyclic voltammetry of cotton fabrics with AgNWs/cotton when the times of impregnation is 5 times. −0.3 V is the reduction peak of Ag/Ag+ and 0.7 V is the oxidation peak of Ag+/Ag, which correspond to the transformation between Ag and Ag+. The above experimental results further proved that cotton fabrics are successfully modified with nano-silver and have high sensitivity. In the test results of AgNRs/cotton, with the increase of impregnation times, the oxidation and reduction characteristic peaks became more and more obvious. It indicate that the stronger the electrochemical signal is, and also proves that nano-silver has been successfully modified on cotton fabrics. Seen from Figure 7(c), the non-oxidation and reduction peaks of cotton fabrics treated with silver nanoparticles. The results show that the electrochemical response signal of cotton fabric is weak and its sensitivity is low, which is not conducive to electrochemical detection. Therefore, the same conclusion can be drawn from cyclic voltammetry: silver nanowires can give cotton fabric the best conductivity.

The cyclic voltammetry of cotton fabrics dipped with nano-silver with different morphologies and times. (a-AgNWs/cotton; b-AgNRs cotton; c-AgNPs cotton).

SEM photograph of frictionless cotton fabric and cotton fabric after friction (1,1′-untreated cotton; 2,2′-AgNWs/cotton; 3,3′-AgNRs/cotton; 4,4′-AgNPs/cotton); photograph cotton fabrics after rubbing (1″-untreated cotton; 2″-AgNWs/cotton; 3″-AgNRs/cotton; 4″-AgNPs/cotton).
Wear resistance
By using rubbing color fastness tester, the treated cotton fabrics were placed at the end of the tester, and rubbed back and forth for different number of rubbing on the dry-rubbed sample along the track straight line of (104 ± 3) mm until the surface of the treated cotton fabrics ruptured. The number of rubbing was recorded in the meanwhile. The resistance of the rubbing fabrics was measured and the photos of the rubbing fabrics were taken.
Table 3 records rubbing times and square resistance of cotton fabrics dipped 5 times with different morphology nano-silver. The untreated cotton fabrics are damaged when the rubbing times is 3000 times. After treated with nano-silver solution, the rubbing times of cotton fabrics are all increased, and the wear resistance of cotton fabrics is raised significantly. Among them, the rubbing times of cotton fabrics treated with AgNWs are as high as 11500 times. And the resistance after rubbing is still the highest, higher than that of unrobed AgNRs/cotton and AgNPs/cotton.
Rubbing times and square resistance of cotton fabrics dipped 5 times with different morphology nano-silver.

Note: 1#-untreated cotton; 2#-AgNWs/cotton; 3#-AgNRs/cotton; 4#-AgNPs/cotton.
Figure 8 is the scanning and optical photographs of the rubbing fabrics. Compared with Figure 8(1″), (3″) and (4″), the rubbing breakage of AgNWs/cotton fabrics (Figure 9(2″)) are not obvious, which indicate that the AgNWs/cotton fabrics have good wear resistance and improve the application scope of the fabrics. Based on the analysis of the results in Figure 8, it can be clearly found that after repeated rubbing, the silver on the surface of cotton fabrics dropped. However, a small amount of AgNWs can be interconnected to form a coherent conductive channel (Figure 8(2′)). The square resistance of AgNWs/cotton fabric can still reach 2.88 Ω/sq, and it still has good conductivity. Because AgNWs have a high aspect ratio and entered the fiber gap of cotton fabrics, improving the wear resistance of cotton fabric and making it difficult to fall off by rubbing. The AgNRs/cotton and AgNPs/cotton (Figure 8(3′) and (4′)) fabrics after rubbing have only a small amount of nano-silver deposited on the surface of the fibers to provide electrical conductivity to cotton fabrics. Therefore, when testing the square resistance of cotton fabric before and after friction in Table 3, it is found that the square resistance of 1#, 2#, 3# and 4# after friction decreases. And after friction the square resistance of 2# still the highest.

Softness (a), whiteness and yellowness (b) of cotton fabrics dipped 5 times with different morphology nano-silver (1#-untreated cotton; 2#-AgNWs/cotton; 3#-AgNRs/cotton; 4#-AgNPs/cotton).
Mechanical properties
The test results in Figure 9(a) demonstrated the effect of nano-silver on the softness of treated cotton fabrics. The softness of cotton fabrics treated with silver nanowires increased slightly, and those treated with silver nano rods and silver nanoparticles changed little compared with untreated cotton fabrics. In a word, cotton fabrics treated with AgNWs were adequate for our needs for fabric softness.
The fabrics coated with nano-silver also showed different colors due to the local surface plasmon resonance (LSPR) [32] effect of nano-silver. LSPR is very sensitive to the size and shape of nanoparticles, which is due to the color difference caused by the plasma resonance effect of silver. Because silver nanoparticles show different colors depending on their size, silver nanoparticles show different colors according to their size, the larger the size, the darker the color. And the color of textiles gradually deepened (Figure 9(b)). However, the whiteness of AgNWs/cotton fabrics was higher than that of AgNRs and AgNPs. Therefore, AgNWs had little effect on the whiteness of cotton fabrics.
In order to further explore the mechanical properties of cotton fabrics, cotton fiber's breaking force and elongation at break were tested, as shown in Figure 10. The test results were plain to reflect that the tensile strength at break of the treated fabrics with different morphologies of nano-silver increased. Among them, the breaking strength of cotton fabrics treated with AgNWs was the highest, reaching 345 N, while the breaking elongation was the lowest, only 230%. Because nano-silver entered into the interior of fibers, forming force between fibers. When textiles were subjected to external force, the ability of the fibers to deform under external force increased and the breaking force increased. With the increase of the length-diameter ratio of nano-silver, the hydrogen bond crosslinking between nano-silver and cotton fabric became more sufficient, and the force of cross-linking between fibers increased. The mobility between fibers was restricted, and the external load was more uneven, which led to the decrease of elongation at break [33].

The tensile strength and elongation at break of cotton fabrics dipped 5 times with different morphology nano-silver (1-untreated cotton; 2-AgNWs/cotton; 3-AgNRs/cotton; 4-AgNPs/cotton).
Anti microbial properties
In order to investigate the antibacterial properties of fabrics, the antibacterial properties were tested by the method of bacteriostasis circle, as show in Figure 11. The antibacterial rate of silver nanowire cotton fabric was 99.8%, which was higher than that of rod and granular cotton fabric. After washing five times, the antibacterial rate of the fabric decreased, but the antibacterial rate of the cotton fabric with silver nanowires could reach 96.1% still.
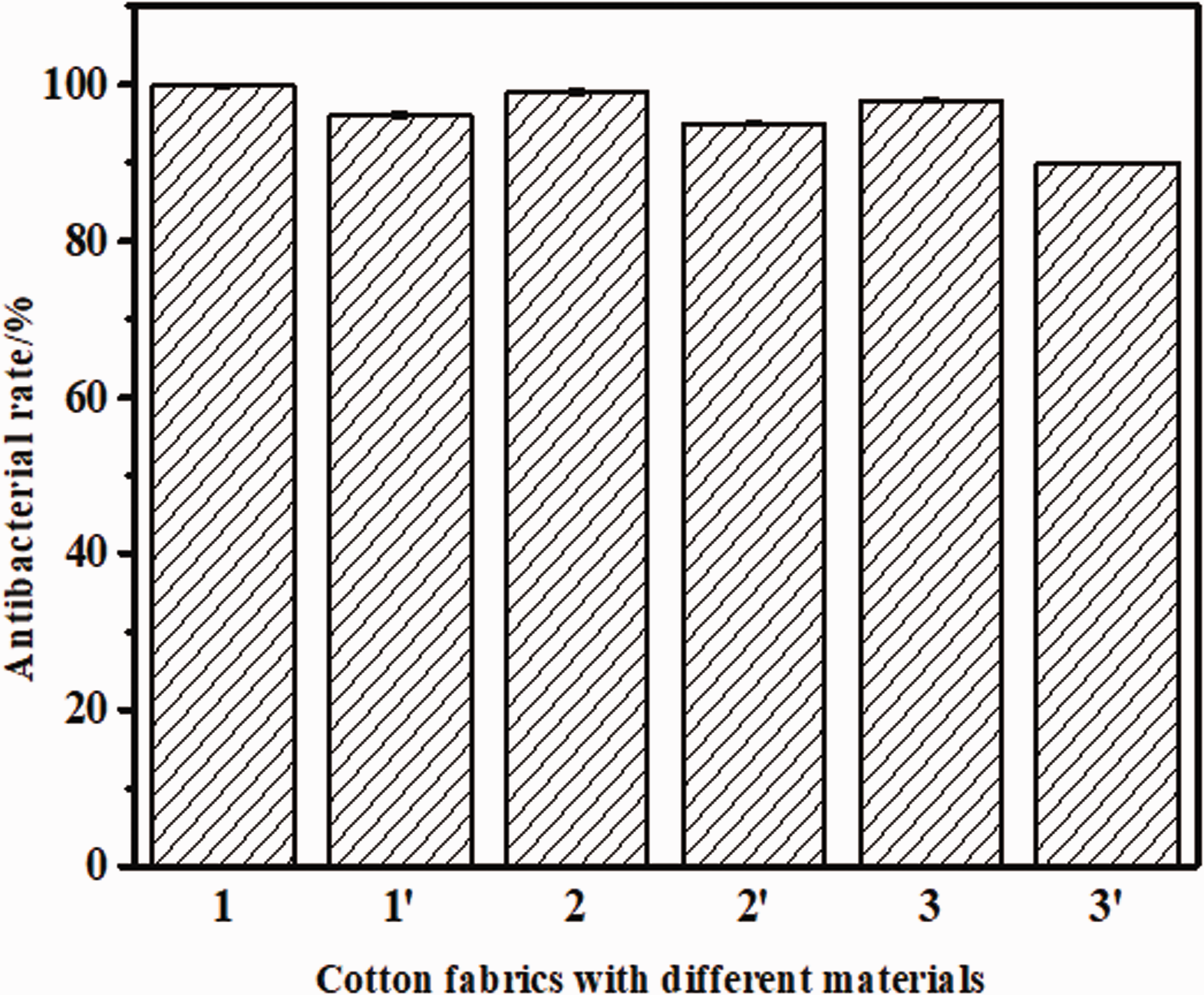
Detection of cotton fabrics finishing with different materials (1-AgNWs/cotton; 2-AgNRs/cotton; 3-AgNPs/cotton; 1′-wash five times of AgNWs/cotton; 2′-wash five times of AgNRs/cotton; 3′-wash five times of AgNRs/cotton).
Conclusions
AgNWs, AgNRs and AgNPs were prepared by simple hydrothermal method, and then used to make conductive cotton fabrics by impregnation method. Compared with cotton fabrics treated with silver nano rods and silver nanoparticles, the addition of AgNWs provides a fast charge transfer channel, which significantly improves the electrical conductivity of cotton fabrics. It shows that the square resistance of cotton fabrics can reach 0.22 Ω/sq when cotton fabric dipped 5 times. In addition, after 11500 times of rubbing, the resistance of cotton fabrics with silver nanowires could remain stable at 2.88 Ω/sq. It can effectively prevent the silver layer from falling off on the surface of cotton fabrics and ensure the stability of conductive cotton fabrics. Cotton fabrics treated with AgNWs nanowires have great potential in wearable flexible sensors. This work is conducive to the further development of flexible electronic textiles and is expected to be applied in flexible sensors, flexible supercapacitors, smart clothing and so on.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the support for this study from the Key Research Project of Shaanxi Province (grant numbers 2020GY-258); Shaanxi Provincial Education Department Serves Local Special Project (grant numbers 20JC006); Shaanxi Provincial Overseas Students Science and Technology Activities Merit Funding Project (Type of Outsdanding) (grant numbers 2019005); and Shaanxi Provincial “Special Supporting Plan for High-level Talents” Youth Top Talents Project.
