Abstract
Benzoyl peroxide is a widely used active agent for acne vulgaris treatment that has antibacterial, anti-inflammatory, keratolytic and wound-healing properties. The common complaints for benzoyl peroxide are skin irritation and dryness. To reduce these side effects, microsponge formulations are used as an effective tool in pharmaceutical and cosmetic industries. In this study, a medical plaster was developed containing benzoyl peroxide microsponges which gives an opportunity to explore the potential of the microsponge systems in textile industry. Benzoyl peroxide microsponge was obtained by using quasi-emulsion solvent diffusion method and characterized by SEM, FT-IR, particle size analysis and porosity studies. The microsponges were applied onto 100% cotton woven fabric by using dip-coating technique. In the treatment solution, different binding agents as acrylic binder, and cross-linking agents as poly(ethylene glycol) diglycidyl ether and 1,2,3,4-butanetetracarboxylic acid were used. Microsponges were around 78.4 µm in diameter and had a spherical porous surface. SEM analyses of the treated fabrics proved that the microsponges were succesfully transferred on cotton fabrics. The drug contents of the fabrics containing Benzoyl peroxide loaded microsponge were found to be within the range of 68.512–102.873%. The in vitro drug release results show that the release from both samples was more than 40% within 6 h. The water vapor permeability of plaster prepared with acrylic binder (S1) was significantly higher than the other plasters. This study presents a novel approach for acne treatment based on textiles containing microsponges. The results revealed that microsponges had a promising potential in textile field.
Introduction
Acne vulgaris is one of the most common skin disease which dermatologists have to treat. It mainly affect teenagers, though may present at any age [1,2]. Acne vulgaris is generally characterized by formation of seborrhea, comedone and inflammatory lesions in the face, back and chest and presence of bacteria Propionibacterium acnes, Staphylococcus epidermidis and Staphylococcus aureus in the follicular canal and sebum production. Acne can be classified as mild, moderate or severe. Topical treatment is the first choice in mild and moderate acne lesions [3,4].
Benzoyl peroxide (BPO) has an anti-inflammatory, keratolytic and comedolytic activities, and is commonly used in topical different formulations for the treatment of acne vulgaris [1,5]. The use of BPO has advantages in comparison to the use of antibiotics because potential bacterial resistance is avoided, and it is preferred over keratolytic agents due to its bactericidal effect. However, its use can cause mild skin irritation and skin dryness. The degree of irritation is believed to be related to the amount of BPO present in the formulation. It has been shown that encapsulation of BPO in vesicular and particulate delivery systems can reduce the side effects to a great extent [3,6,7].
Microsponges are uniform polymeric delivery systems composed of porous microspheres having spherical particle size range from 5 to 300 µm [3,8,9]. These effective, stable, non-toxic and non-irritant delivery system when applied to the skin, the release of drug can be controlled through diffusion or other variety of triggers, including rubbing, moisture, pH, friction or ambient skin temperature. Controlled release of active substance from a delivery system to the skin could reduce the side effect while reducing percutaneous absorption [3,10].
Microsponges are capable of absorbing skin secretions, therefore reducing oiliness and shine from the skin. Microsponge polymers possess the ability to load a wide range of actives providing the benefits of enhanced product efficacy, mildness, tolerability and extended wear to a wide range of skin therapies [3,11]. The quasi-emulsion solvent diffusion method is a popular method to develop microsponges. The solvent diffusion process has the advantage of avoiding solvent toxicity. It is easier to conduct and is less prone to agglomeration of the microsponges and also is more economic. This method is a process by which microencapsulation can be readily performed in the laboratory without the need of specialized equipments [12,13].
The aim of this study was to develop a novel carrier system for BPO based on textiles containing microsponges for acne vulgaris treatment. BPO-loaded microsponges were prepared by quasi-emulsion solvent diffusion method using ethyl cellulose (EC) and polyvinyl alcohol (PVA). The microsponges were characterized by surface morphology, particle size distribution, pore structure and fourier transform infrared (FT-IR) spectroscopy. In our previous study, BPO microsponges were applied onto cotton fabric by spraying technique [13]. In this study, BPO microsponges were applied onto cotton fabric by using dip-coating technique. Furthermore, the effects of different binders used in the application were investigated [14].
Experimental
Materials
EC (polymer) and PVA (emulsifying agent) were both purchased from Sigma (USA). BPO (active agent) was supplied from Merck (Germany). 100% cotton fabric (plain weave, 115 g/m2) was used for textile applications during the experiments. Hydroxypropyl methylcellulose (HPMC) polymer was purchased from Dow Chemicals (USA). The acrylic based commercial linking agent was supplied from BASF (Germany). The cross-linking agents, poly(ethylene glycol) diglycidyl ether (PEGDGE) was supplied from Polyscineces (Germany) and 1,2,3,4-butanetetracarboxylic acid (BTCA) was purchased from Alfa Aesar (USA). The transparent adhesive plaster tape was purchased from a local commercial supplier. All other chemicals were used in analytical grade.
Methods
Preparation of microsponge
Microsponge formulation containing BPO were prepared by using quasi-emulsion solvent diffusion method. To prepare the internal phase, 0.25 g EC was dissolved in 5 mL dichloromethane (DCM). Then 0.5 g BPO was gradually added under agitation. The resulting internal phase was then gradually poured into the solution of 0.075 g PVA: 30 mL water. The solution containing the emulsion droplets was continuously stirred for 3 hours at 500 rpm. Then the mixture was filtered, washed with distilled water and dried at room temperature [13,14].
Particle surface morphology
The particle surface morphology of the BPO microsponge and the fabrics that were treated with BPO microsponge were visualized by Scanning electron microscope (SEM) (Carl Zeiss 300VP, Germany). The samples were coated with a thin layer of gold prior to the SEM analysis by using QUORUM Q150 RES device.
Particle size and distribution
The mean particle size and distribution were measured by laser diffraction measurements with the Mastersizer (Malvern Instruments, Worcestershire, UK) using the wet dispersion technique. Dv(50) value was expressed as the mean particle size by volume and the size distributions (Span) were calculated by using following equation:
Dv(90): the maximum particle diameter below which 90% of the sample volume exists.
Dv(10): the maximum particle diameter below which 10% of the sample volume exists.
Dv(50): the maximum particle diameter below which 50% of the sample volume exists - the median particle size by volume.
The D50 has been described as the diameter where half of the particles’ size below the given value. 90% of the particles’ size below the D90, and 10% of the particles’ size below the D10.
Fourier transform infrared spectroscopy
FT-IR spectrums were obtained by Perkin Elmer Frontier, USA. The measurements took place at the room temperature in the wavelength range of 4000–450 cm−1. Origin Pro 2016 software was employed to smooth the spectral data.
Porosity studies
Porosity studies were performed by using mercury porosimetry (Micrometrics Autopore 4, USA). The samples were placed in the device. Then the pressure was gradually increased from 0.1 psia to 60000 psia (∼0.69 kPa to 413685,44 kPA), thus allowing mercury to enter the smallest pores of the microsponge (from 0.003 µm to 1100 µm). As the mercury (mercury intrusion and extrusion volume 0,1 µL) penetrated through the pores the volume of mercury in the volume-calibrated cell of the device gradually decreased and this change was measured and analyzed by the software of the device.
Application of BPO microsponges onto textile materials
BPO microsponges were applied onto cotton fabric by using dip-coating technique. The solution of which content is given in Table 1 was prepared and applied onto textile material.
Content of treatment solutions.

HPMC is a non-irritant and non-toxic polymer, widely used in oral and topical pharmaceutical formulations. HPMC can be used in textiles as binders. In this study, HPMC was used to keep the particles in large size on the fabric surface which the cross-linking agent does not suffice to hold. Cyanamid was added to solution as a catalyst.
In order to bond the microsponges to the fabric, different non-toxic binding and cross-linking agents were used. As binder acrylic binder, and as cross-linking agents poly(ethylene glycol) diglycidyl ether (PEGDGE) and 1,2,3,4-butanetetracarboxylic acid (BTCA) are employed in the treatment solution.
In the dip coating process, the solution prepared was placed in a container of suitable size. The samples were immersed in the solution and kept for 5 minutes and removed from the solution and kept for 2 minutes vertically. After the treatment, the fabric samples were dried at 30°C and exposed to fixation process 20 minutes at 65˚C. Then, the treated cotton fabric samples were combined with adhesive tapes.
Quantification of BPO by UPLC
BPO content of the fabrics containing BPO loaded microsponge was determined by UPLC (Thermo Scientific Accela, USA). Separation was achieved on GL Sciences Inertsil ODS-3 HP (3 µm, 150 × 2.1 mm) C18 column at 25°C by using an isocratic elution method with water: Acetonitrile (15:85, v/v) at a flow rate of 200 µL/min. PDA detection was made at 225 nm. This method was validated according to the ICH guidelines including determination of linearity, specificity, accuracy, precision (repeatability and reproducibility), stability, limit of detection, and limit of quantification [15].
Drug content of textile materials treated with BPO microsponge
To determine the drug content, fabric samples (4 cm x 4 cm) was extracted in 50 mL acetone at 200 rpm during 48 h. In order to determine the amount of active substance loaded in the fabric samples, the solution was filtered through a 0.22 µm syringe filter and filtrate was analyzed by validated UPLC method. Experiments were carried out at least five times for reproducibility.
In vitro drug release of textile materials treated with BPO microsponge
To determine in vitro drug release profiles of the textile materials treated with BPO microsponge, fabric samples (4 cm x 4 cm) were placed in glass vessels containing 100 mL of PBS + acetone (6:4) solution. The release study was determined in a horizontal incubation shaker (Thermo Fisher Scientific Max Q™ 6000, USA) according to the sink conditions at 37˚C, 70 rpm. At appropriate time intervals, samples were withdrawn and released BPO amount was determined by validated UPLC method. All the experiments were repeated five times, and the data were expressed.
Water vapor permeability test
Water vapor permeability test was carried out with SDL ATLAS M261 Water Vapor Permeability Tester in accordance with BS 7209:1990. The fabrics to be tested were cut to the size of the sample cup and 3 replicate samples were prepared for each test. 46 ml of distilled water was placed in the sample container and the samples were placed on the sample container in such a way that they would not come into contact with water. The edges of the fabrics were fixed to the edges of the container with adhesive. In order to prevent water vapor leakage from the sides, an aluminium ring was placed on the side and the side surface was covered with a soft tape. The samples were weighed again after 1 hour and placed back into the device and re-weighed after 24 hours. Water vapor permeability was calculated according to the following formulation:
WVP = Water vapor permeability (g/m2/day)
M = Weight loss
A = 0.0054113 m2 (sample area)
t = time (hours)
Results and discussion
Particle surface morphology
For morphology and surface topography investigation, prepared microsponges were subjected to SEM analysis. Particle surface morphology of microsponges is important because it affects drug diffusion and particle degradation [16]. As seen in the Figure 1, the microsponge formulation is uniform and has a spherical shape. The presence of surface pores can be clearly observed. These morphological characteristics point out that BPO is dispersed all over the microsponges.

SEM micrographs of BPO microsponges at 500× and 1000× magnifications.
Particle size and distribution
The particle size histograms were generated according to the data obtained by laser diffraction measurements (Figure 2). While histogram gives average of microparticles, green line shows the undersized particles. Microparticles in the range size of 0.1–50 µm, 50–100 µm and 100 µm are 31.74%, 28.29% and 39.97% in volume, respectively. Dv50 refers the particle diameter of 50% of the sample volume. It is known that higher span value indicates a wide size distribution and higher polydispersity. It is seen that the particle size (Dv50) of the microsponge formulation is 78.4 µm and the polydispersity index is identified as 3.013.
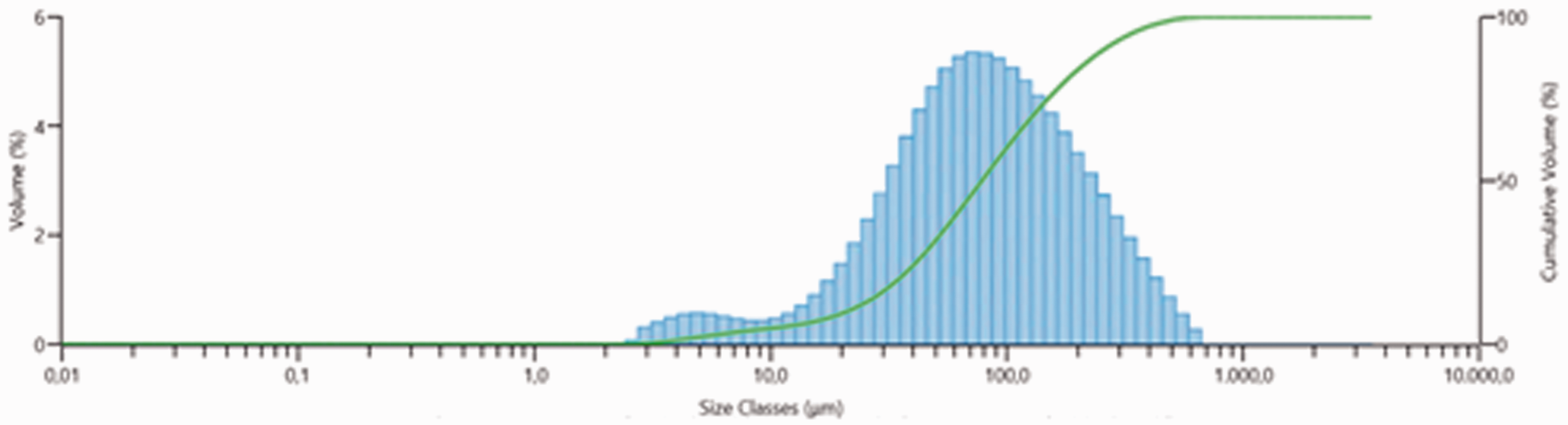
Particle size distribution of microsponges.
Fourier transform infrared spectroscopy
The FT-IR patterns of microsponges were evaluated to understand if there were any interactions between drug and polymer (Figure 3). FT-IR spectrum of BPO shows their characteristic peaks at 1757 cm−11,221 cm−1 and 996 cm−1 [17]. Spectrum of ethyl cellulose demonstrates the C-H bending at 1375 cm−1, C-O-C band at 1054 cm−1, and characteristic tensile vibrations of the C-H band at 2870 cm−1 and 2972 cm−1 [18]. The characteristic peaks of the BPO and EC were seen on the spectrum of the microsponge formulation. The results indicated that BPO and EC were compatible with each other and BPO preserve its structure after the microsponge formation. It was an expected result and in accordance with the literatures [6,19].

FT-IR spectrum of microsponges.
Porosity studies
The results obtained by mercury porosimetry analysis confirm the porous structure of the microsponges as observed in the SEM images (Table 2). In the literature, it has been reported that the pore diameter can have a significant effect on the release rate of the ingredient, and can also affect the migration of the active ingredient from the microsponge particle into the vehicle in which the material is dispersed [7,20]. Jelvehgari et al. [6] calculated the total porosity of microsponges, total surface area and total volume of pores of microsponges to determine the effect of the number of pores on release rate of BPO from microsponges. The results showed that generally, the lower release rate was obtained for microsponges with a low porosity, low surface area and low volume.
Mercury porosimetry analysis results.

SEM micrographs of the treated fabrics
After fabric treatment with the solution containing microsponge and different type of binders and crosslinkers, SEM micrographs of the fabrics were taken (Figure 4). It was shown that microsponges were successfully transferred onto the fabric and retained their structure after the application process. It has been found out that acrylic binder and BTCA have showed better fixation properties on microsponges to textile fabrics. Obtaining more homogenous distribution of microspheres in textiles produced using acrylic binders may be explained by the effect of physical coating.

SEM images of treated fabrics with the solutions containing BPO microsponges and (a) acrylic (S1), (b) PEGDGE (S2), (c) BTCA (S3) binders.
Quantification of BPO by UPLC
The UPLC method was developed and validated following the ICH guidelines recommendation Q2 for validation of analytical procedures [15]. The method gave a peak of BPO in 3.67 min. BPO chromatogram is shown in Figure 5 and obtained chromatogram is compatible with the literature. It was successfully separated from other compounds. The calibration curve of CHD was linear in the concentration range of 0.5–10 µg/mL (r2 = 0.9996). The limit of detection (LOD) and the limit of quantification (LOQ) of BPO were found to be 0.242 and 0.403 µg/mL, respectively.

The chromatogram of BPO.
Drug content
The effect of binders on drug content was examined. Table 3 shows the drug contents of the treated fabrics. The drug contents of the fabrics containing BPO loaded microsponge were found to be within the range of 68.512–102.873%.
Drug contents of the treated fabrics with the solutions containing microsponges and different types of binders.

In vitro drug release studies
Prepared BPO microsponge was applied onto cotton fabric and the drug release from the fabrics were studied PBS+acetone (6:4) (Figure 6). We added acetone to the receptor medium for increased solubility of BPO. There are some manuscript used same substance for similarly purpose [6]. As can be seen in Figure 6, no burst effect was observed, indicating that BPO was homogeneously dispersed in the fabrics. It was seen from the data obtained that after 6 h, the release rate of S1, S2 and S3 coded fabrics were 50.684%, 41.435% and 57.195%, respectively. Change in treatment solution significantly affects the release phenomena. The highest release rate was observed in the S3 coded fabric prepared with BTCA. Polycarboxylic acids have been confirmed as the most promising formaldehyde-free crosslinking agents for cotton cellulose among the various new reagents investigated. Among the various effective polycarboxylic acids, BTCA is the most effective cross-linking agent for cotton fabric [21,22]. However, the drug content of the S3 is low (68.512 ± 3.857). The binder can be a resin of acrylic, polyurethane, silicone type and its role is to fix the microsponges, microparticles on the fabric keeping them in place and thus preventing their loss during washing and wear. The efficiency of a binder to bind microcapsules, microsponges on the textile surface depends on the compatibility of the different interfaces of the products involved in the finishing process. The acrylics gained widespread market acceptance as coatings binders due to such outstanding properties as color stability, transparency, and resistance to weathering and aging. Acrylic based binders are cross-linkers that are widely used in printing and finishing applications of textile materials and can also be fixed at low temperatures. S1 coded fabric prepared with acrylic binder higher release rate than S2 coded fabric prepared with PEGDGE. It was concluded that acrylic binder appears to be the most optimal in both drug content and release profile.

In vitro drug release profiles of the treated fabrics with the solutions containing microsponges and different types of binders.
Water vapor permeability properties
In order to examine the comfort properties of the plaster design, water vapor permeability tests were performed. The results of the tests are given in the Table 4. As can be seen from the results, cotton alone had high vapor permeability, while the permeability value was considerably reduced when combined with plaster. This value was further reduced in the plaster combined with the treated cotton fabric. It was found that PEGDGE and BTCA were significantly reduced vapor permeability. It was seen that the water vapor permeability is significantly higher in the samples prepared with acrylic binder (S1).
Water vapor permeability test results.

Conclusions
Medical and cosmetic textiles are becoming increasingly important. Especially the rapid developments in the technological field provide a suitable environment for valuable studies. In this study, microsponge formulation was developed, characterized, and incorporated into medical plaster. The microsponges were developed by using quasi-emulsion solvent diffusion method. The particle morphology, FT-IR, particle size, porosity and drug release were studied. It was revealed that microsponge of the formulation were around 78.4 µm in diameter and had a spherical porous surface. Then, microsponge formulation was applied onto 100% cotton woven fabric by using dip-coating technique. The surface morphology, drug content and drug release of the fabrics containing microsponges were also studied. The results show that the drug release from both samples was more than 40% within 6 h. The treated fabrics were combined with a transparent adhesive plaster tape and their water vapor permeability properties were examined. It was concluded that plaster S1 appears to be the most optimal in both drug content and release profile. In addition, the water vapor permeability of plaster S1 is significantly higher than the other plasters. This study shows that microsponge drug delivery system is a versatile tool. It has a potential to convert a traditional or a modern plaster into a plaster containing BPO loaded microsponge. This study presents a novel approach for acne treatment based on plaster containing microsponge.
Footnotes
Acknowledgements
We would like to acknowledge E.U. Pharmaceutical Sciences Research Center for enabling us to use its laboratory instruments.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by the TUBITAK Scientific and Technological Research Council in frame of the Project 117M350.

