Abstract
A series of polystyrene (PS)/unmodified Na-montmorillonite (Na-MMT) composites were prepared via in situ radical polymerization. The prepared composites were characterized using various techniques. The presence of various functional groups in the unmodified Na-MMT and PS/unmodified Na-MMT composite was confirmed by Fourier transform infrared spectroscopy. Morphology and particle size of prepared composites was characterized by X-ray diffraction (XRD), scanning electron microscopy (SEM), and transmission electron microscopy (TEM). According to the XRD and TEM results, the interlayer spacing of MMT layers was expanded. SEM images showed a spongy and porous-shaped morphology of composites. TEM revealed the Na-MMT intercalated in PS matrix. The thermal stability of PS/unmodified Na-MMT composites was significantly improved as compared to PS, which is confirmed using thermogravimetric analysis (TGA). The TGA curves indicated that the decomposition temperature of composites is higher at 24–51°C depending on the composition of the mixture than that of pure PS. The differential scanning calorimetry (DSC) results showed that the glass transition temperature of composites was higher as compared to PS. The moisture retention, water uptake, Brunauer–Emmett–Teller specific surface area, and specific pore volume of composites were also investigated. Water resistance of the composites can be greatly improved.
Introduction
During the past decade, polymer/clay composites have attracted a great deal of attention due to a significant improvement in mechanical, thermal stability, gas-barrier properties, and biodegrading compared to pure polymer. 1 –4 These polymer composites show significant improvements in low clay loadings, especially in mechanical and reduced flammability. 5,6 Clay is the most important material for improving the performance of polymers against fire and is used in synergistic combinations with other traditional flame retardants. 7 They also exhibit a remarkable improvement in mechanical properties such as high tensile strength and modulus due to the incorporation and dispersion of high-order anisotropic nanoscale reinforcements. 8 –10
The inorganic-layered material clays are relatively low cost, abundant and have excellent chemical and thermal resistance for the synthesis of polymer/clay composite. Among the many layered materials, the Na-montmorillonite (Na-MMT) is one of the widely used inorganic materials and consists of layers of two tetrahedral silica sheets sandwiching one octahedral alumina sheet. 8,11 It has intercalation/exfoliation characteristics.
Polystyrene (PS), a hard, stiff, and brilliantly transparent synthetic resin, is produced by the polymerization of styrene. It is widely employed in the food-service industry as rigid trays and containers; disposable eating utensils; and foamed cups, plates, and bowls. PS is also copolymerized or blended with other polymers, lending hardness and rigidity to a number of important plastic and rubber products. As a commercialized and mass-productive polymer, PS has wide applications in industries and it is easily polymerized. 12,13 It is widely used for many applications such as construction and food packaging. Also, PS is an inexpensive and hard plastic. However, the especially poor thermal, mechanical properties, especially thermal stability, and also antibacterial properties need to be improved. For this purpose, unmodified Na-MMT clay was used for the in situ preparation of PS composites.
A considerable number of studies on polymer/clay composites exist in the literature, but there are very few research work available on preparation and characterization of PS/unmodified Na-MMT composites. Also, the present study describes the morphology, the moisture retention, water uptake, and thermal properties of the newly synthesized PS composites.
Experimental
Materials
The Na-MMT (silicon dioxide: 57.76%; aluminium oxide (Al2O3): 13.56%; iron(III) oxide: 4.27%; calcium oxide: 0.69%; magnesium oxide: 1.74%; sodium oxide: 1.37; potassium oxide: 0.33; Loss on Ignition (LOl): 19.58) was obtained from Reşadiye (Tokat/Turkey) and was used after further purification as previously described in the literature by the methods of dispersion and sedimentation from its aqueous suspension. 14 Its specific surface is 67.56 m2 g−1, and its X-ray diffraction (XRD) analysis shows that the interlayer spacing of Na-MMT is 1.18 nm. Benzoyl peroxide (Bz2O2; Merck), a free radical initiator, was purified by recrystallization of methanol and chloroform mixture. Styrene (Merck) monomer was purified by distillation at reduced pressure before use. All other chemicals were of analytical reagent grade and used as received.
Synthesis of PS/Na-MMT composites
In situ polymerization of PS/unmodified Na-MMT composites was carried out with different amounts of styrene monomer in aqueous medium. 3,15,16 The nomenclature of composite samples and polymerization conditions is summarized in Table 1. In this synthesis procedure, 1 g of Na-MMT was dispersed in 24 mL of distilled water in a 100-mL Pyrex glass tube and the obtained dispersion was stirred at a rate of 100 r min−1 in overnight at the room temperature for each polymerization. The styrene and the aqueous Na-MMT suspension were mixed with different weight ratios. By the addition of the solution of Bz2O2 in 1-mL acetone (3.59 × 10−3 mol L−1), the mixture was polymerized in an ultrasonic bath (Bandelin Sonarex RK 100H, Germany) at 85°C for 2 h to synthesize the PS/unmodified Na-MMT composites. At the end of polymerization, the mixture was precipitated into methanol and filtered. Finally, the obtained composites were left dry in a vacuum oven at 30°C and weighed. Pure PS sample was also polymerized under the same conditions, except Na-MMT clay, for a comparative study.
Composite samples and polymerization conditions.a

PS: polystyrene; Na-MMT: Na-montmorillonite.
a Bz2O2: 3.59 × 10−3 mol/L, temperature: 85°C, time: 2 h.
Characterization of PS/Na-MMT composites
Fourier transform infrared (FTIR) spectra were recorded in KBr pellets by a Bruker IFS 66/S Model FTIR Spectrometer in the range 400–4000 cm−1.
The XRD patterns of Na-MMT, pure PS, and PS/Na-MMT composites in powder form were recorded using a Rigaku Ultima IV X-ray Diffractometer. Composites were analyzed in a 2θ range of 2–40° at the scanning speed of 0.15° min−1 using CuKα radiation (λ = 1.5418 Å).
The thermal stability of Na-MMT, pure PS, and the composites was determined using a Shimadzu simultaneous DTA-TG apparatus (DTG-60 H Model) under a nitrogen atmosphere at a heating rate of 10°C min−1 and 100 mL min−1 from 25°C to 800°C and α-Al2O3 was used as an inert material. The Shimadzu differential scanning calorimetry (DSC)-60 was performed to estimate the glass transition temperatures (T gs) of pure PS and the composites.
Scanning electron microscopy (SEM) was applied to observe surface morphology of pure Na-MMT and composites using a Quanta 400F Field Emission SEM instrument. The surface of all samples was coated by thin-layer gold prior to analysis.
The microstructure of composites was observed using an FEI Tecnai G2 Spirit BioTwin CTEM model transmission electron microscope (TEM) at an accelerating voltage of 120 kV. The cured epoxy resin samples were microtomed with a Leica ultracut-R. Ultrathin sections were cut with a glass knife and deposited on one layer of carbon 300 mesh copper grids prior to analysis.
The Brunauer–Emmett–Teller (BET) surface area and micro-mesopore size of Na-MMT and composites were determined using the Quantachrome Nova 2200 surface area and pore size analyzer.
The weighed dry samples in the form of pellet were conditioned at 25°C in a medium having 100% humidity for 24 h for the moisture retention measurements. The percentage of moisture retention was calculated from the difference between the weights of the conditioned and unconditioned samples (i.e. moisture retention % = (W 2 − W 1) / W 1 × 100, where W 1 and W 2 are the weights of the conditioned and unconditioned samples, respectively). 15,16
The dry samples in the form of pellet were weighed, W1 , and then kept in distilled water for 2 h at 25°C. After immersion, the wet samples were wiped using a filter paper and reweighed immediately, W2 . The percentage of water uptake of samples was determined using the formula (W 2 − W 1) / W 1 × 100. 15,16
Results and discussion
FTIR spectroscopy
The formation of PS and its incorporation in the composite were indicated by the FTIR spectroscopy. Figure 1(a) to (c) shows the FTIR spectra of Na-MMT, pure PS, and PS/Na-MMT7 composites. For PS, the characteristic peaks observed at 3060–2920 (aromatic and aliphatic C–H stretching) and 1492–1451 cm−1 (C=C stretching for CH2 bending); the characteristic peaks of Na-MMT at 1040 cm−1 (Si–O), 515 cm−1 (Al–O), 470 cm−1 (Mg–O), and 3630 cm−1 (O–H stretching) as broad single peak are observed in the composite spectra (Figure 1(c)). The peaks have been identified consistent with previously published data. 17,18
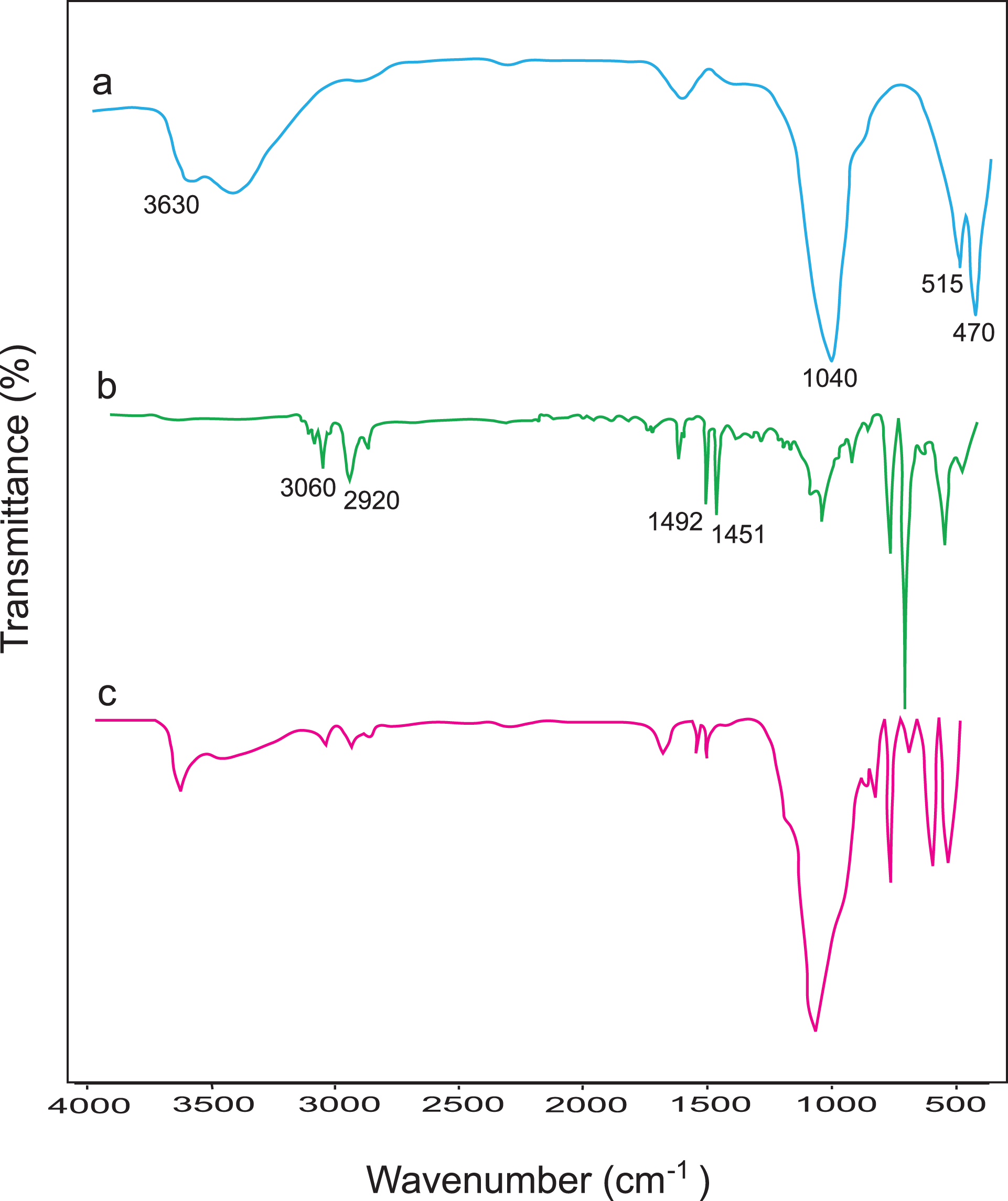
FTIR spectra of (a) Na-MMT, (b) pure PS, and (c) PS/Na-MMT7. FTIR: Fourier transform infrared; Na-MMT: Na-montmorillonite; PS: polystyrene.
Confirmation of intercalation by XRD analysis
XRD analysis was utilized to confirm the existence and formation of Na-MMT in PS. The patterns of the Na-MMT, pure PS, and PS/Na-MMT composites are shown in Figure 2(a) to (f). The d 001 interlayer spacing values of Na-MMT and composites are also demonstrated in Figure 2. The important diffraction peak of Na-MMT appears at 2θ =7.46°. The d 001 interlayer spacing value of Na-MMT is found to be 1.18 nm using Bragg’s law (d = nλ/2sinθ, with λ = 1.5418 Å). The XRD pattern of PS/Na-MMT1 shows a characteristic (001) diffraction peak around 7.55°. Two (001) diffraction peaks were observed around lower diffraction angle (Figure 2(a) and (b)). However, the XRD pattern of the PS/Na-MMT2 revealed a broad peak at composite, which showed that the characteristic crystalline peak was shifted toward smaller angles, consequently, increasing the interlayer spacing. This confirmed the intercalation of Na-MMT layers in PS matrix (Figure 2(c)). The interlayer spacing was calculated to be 1.30 nm for PS/Na-MMT2. An increase of approximately 0.12 nm expansion in the d-spacing value demonstrated that the intercalation. However, no more expansion in the interlayer spacing was observed at higher styrene content (Figure 2(d) and (e)). These results indicate and support that the PS molecules penetrate through the galleries of clay and expand the interlayer spacing. This is a direct evidence that intercalated PS/Na-MMT composites have been synthesized. Similar behavior has been reported in the literature by many other researchers. 12,19
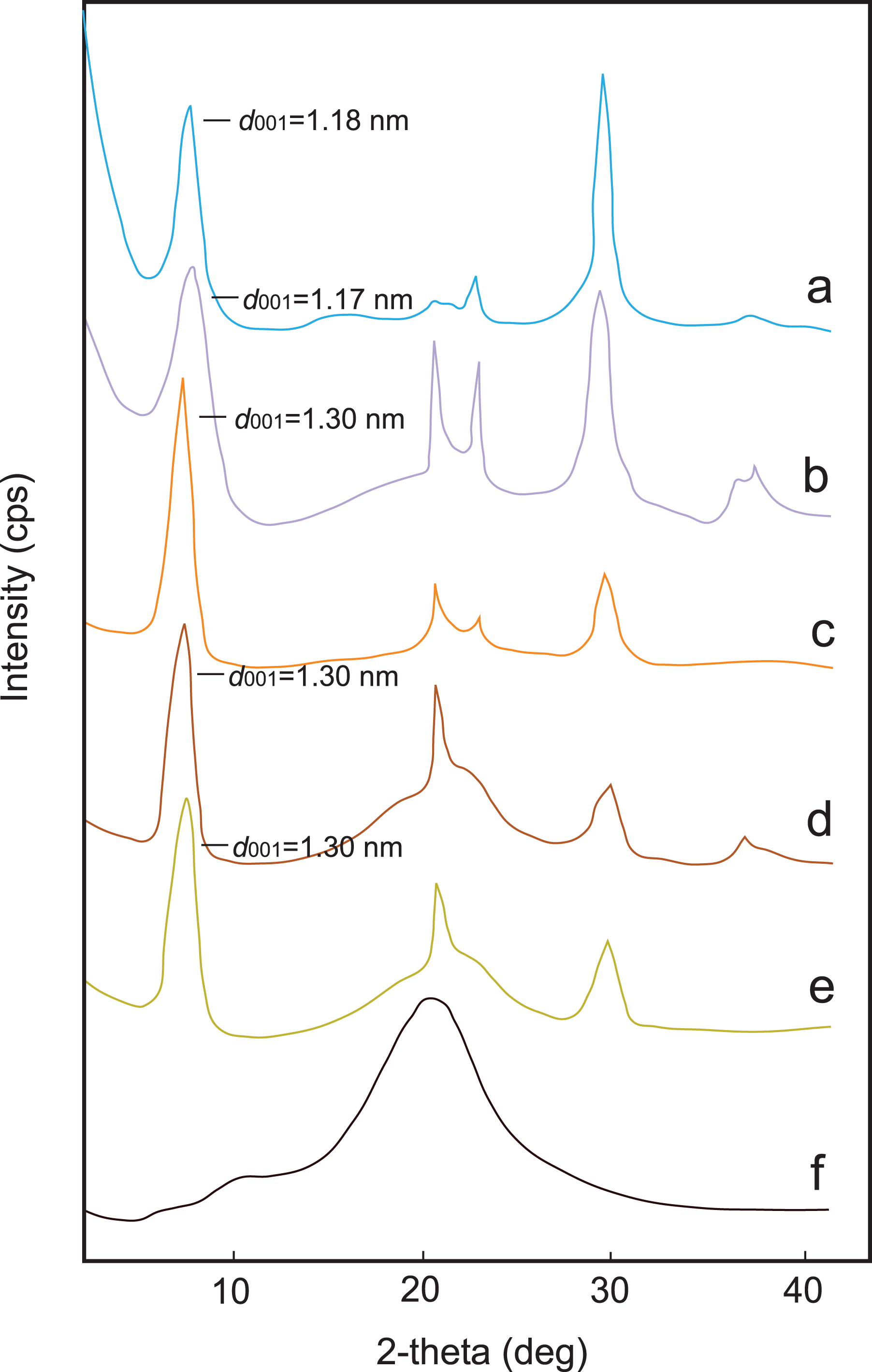
XRD patterns of (a) Na-MMT, (b) PS/Na-MMT1, (c) PS/Na-MMT2, (d) PS/Na-MMT4, (e) PS/Na-MMT6, and (f) pure PS. XRD: X-ray diffraction; Na-MMT: Na-montmorillonite; PS: polystyrene.
Thermal properties
The thermogravimetric analysis (TGA) can determine the enhancement of thermal properties of the composites. Figure 3(a) to (d) shows the curves for TGA measurements used to determine weight loss due to thermal degradation of Na-MMT, pure PS, PS/Na-MMT3, and PS/Na-MMT7. The TGA curve of Na-MMT indicates that there is two-step decomposition (Figure 3(a)). First, a slight weight loss (approximately 6.13%) just below 125°C is attributed to the removal of moisture and physisorbed water in the structure. At higher temperatures, weight losses occurred due to dehydroxylation of Na-MMT. The total mass loss is only 18.85% up to 800°C. As could be expected, the Na-MMT shows a high thermal stability. 3,16,20
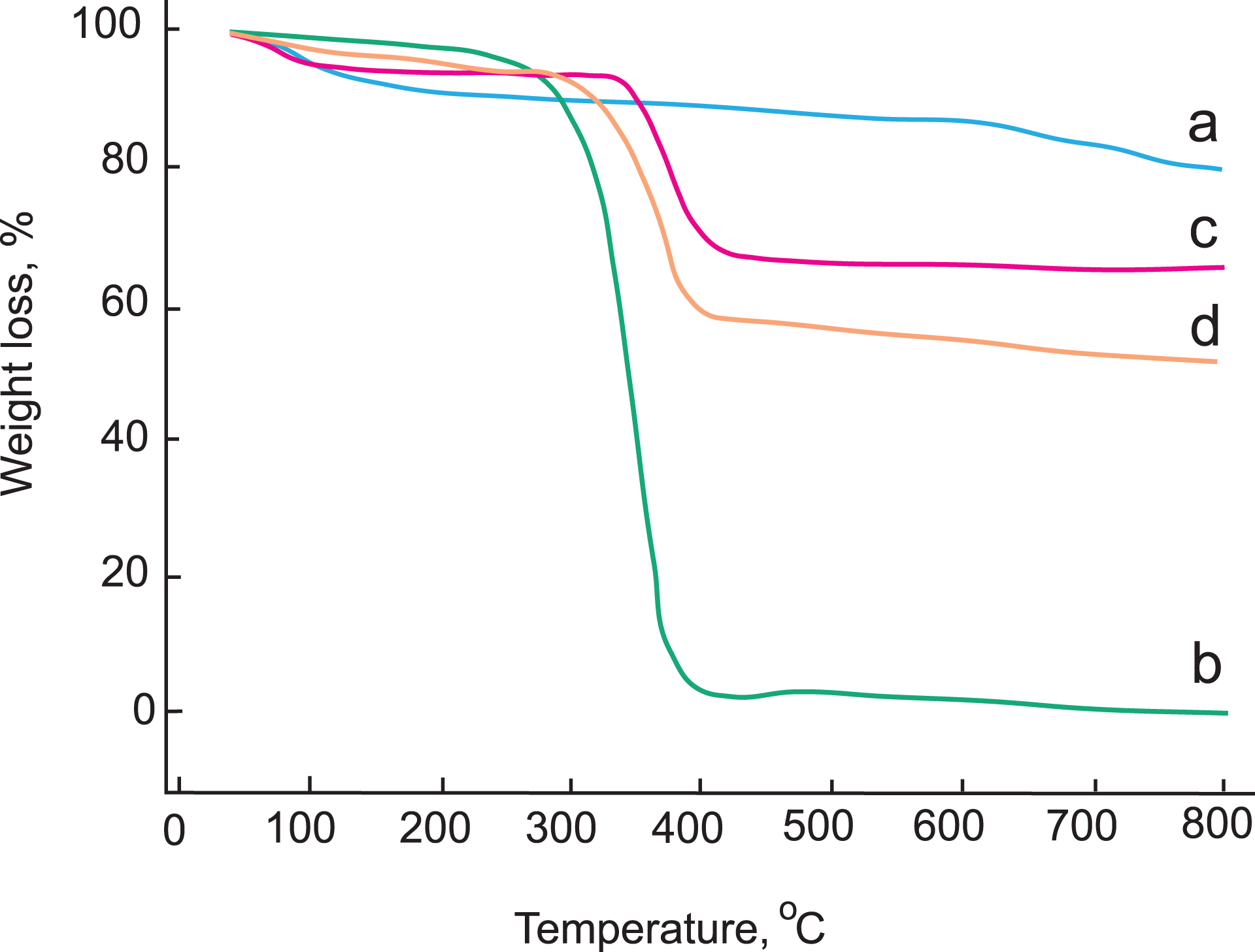
TGA curves of (a) Na-MMT, (b) pure PS, (c) PS/Na-MMT3, and (d) PS/Na-MMT7. TGA: thermogravimetric analysis; Na-MMT: Na-montmorillonite; PS: polystyrene.
As shown in Figure 3(b), it is a small mass loss (approximately 1.41%) just below 160°C which is attributed to the loss of absorbed water of pure PS. The decomposition temperature of the pure PS is 346°C, and the second mass loss is nearly 100.00% at 800°C, which was attributed to thermal decomposition of the PS chains. This stage is the thermal decomposition of polymer. Consequently, pure PS should exhibit lower thermal stability relative to Na-MMT. 17,19
For the PS composites, the PS/Na-MMT3 has a higher decomposition temperature (397°C), which is 51°C more than that of pure PS, and exhibited a mass loss of 34.65% at 800°C. It is a small mass loss (approximately 3.03%) just below 90°C, which is attributed to the loss of absorbed water (Figure 3(c)). But, with the styrene content further increased to 77.78 wt% (PS/Na-MMT7) has a lower decomposition temperature (370°C) and exhibited a mass loss of 46.97% at 800°C than 60 wt% styrene-containing composite (PS/Na-MMT3), because the clay content is low in this sample. Similarly, it is a small mass loss (approximately 7.47%) just below 120°C, which is attributed to the loss of absorbed water (Figure 3(d)). Evidently, the thermal stability of PS composite is enhanced by 24–51°C relative to that of pure PS. This enhanced thermal stability of the PS/Na-MMT composites is due to Na-MMT acting as a heat barrier to heat flow. These views are consistent with the observed trend. 12,21
DSC curves of both PS and PS/unmodified Na-MMT composites are shown in Figure 4(a) to (c). T g in case of pure PS was found to be 390°C, whereas it gradually increases in case of PS/unmodified Na-MMT composites with an increase in the amount of Na-MMT in resultant composites.
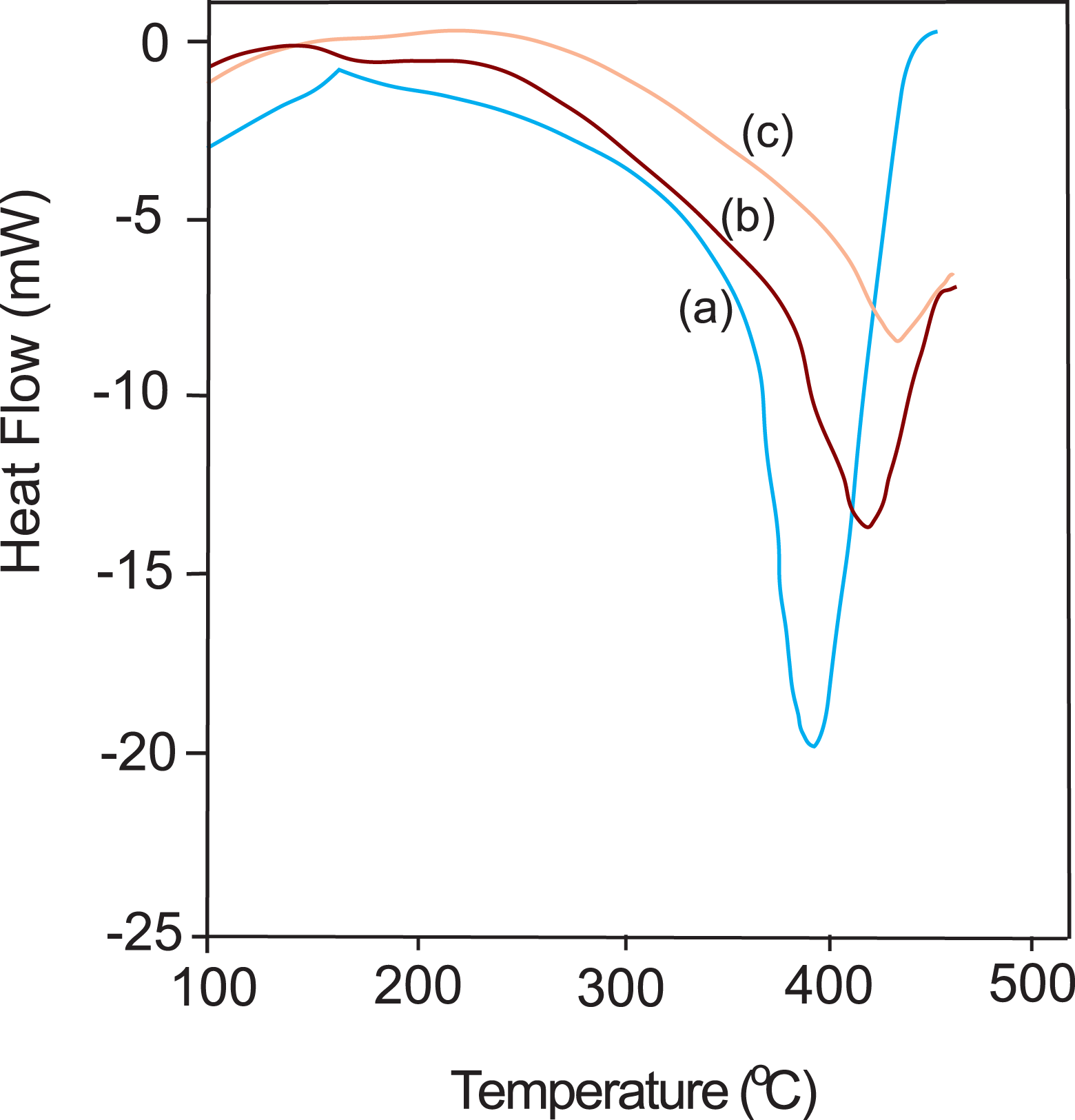
DSC curves of (a) pure PS, (b) PS/Na-MMT3, and (c) PS/Na-MMT7. DSC: differential scanning calorimetry; Na-MMT: Na-montmorillonite; PS: polystyrene.
T g of PS/Na-MMT3 and PS/Na-MMT7 shifted the higher temperature than pure PS. This enhancement in the thermal stability is due to the fact that the introduction of a well-dispersed Na-MMT can prevent the heat to transmit quickly and then improve the thermal stability of the composite. This result is consistent with the reports by many other researchers. 12,17,22
SEM analysis
The surface morphologies of Na-MMT, PS/Na-MMT3, and PS/Na-MMT7 composites and also the distribution of the Na-MMT layers in the polymer matrix were examined by SEM. As shown in Figure 5(a), Na-MMT exhibited an aggregated and sheet-like plate structure before the intercalation. 20 After Na-MMT was intercalated with PS, the significant changes in morphology are observed, which is shown in Figure 5(b) and (c). It can be seen that PS polymerization occurred within the Na-MMT layers and a satisfactory dispersion of clay layers in the polymer matrix. It is particularly interesting to note that the composite morphology exhibits a compact and porous-shaped morphology at higher monomer contents (Figure 5(c)). These morphological images are also evidenced from XRD analysis of PS/Na-MMT composites, as shown in Figure 2.

SEM images of (a) Na-MMT, (b) PS/Na-MMT3, and (c) PS/Na-MMT7. SEM: scanning electron microscopy; Na-MMT: Na-montmorillonite; PS: polystyrene.
TEM analysis
TEM observations are necessary to determine the morphology of composite and it also provides additional information that will aid in the interpretation of XRD results. The TEM micrograph of PS/Na-MMT3 composite is shown in Figure 6. The black lines represent the individual MMT layers, while the light areas correspond to polymer matrix in the micrograph. TEM image of PS/Na-MMT3 composite show that individual layers of the Na-MMT are well dispersed uniformly and homogenously nanoscale in the PS matrix and separated one from the other. Some agglomeration of clay platelets was observed. As shown in Figure 6, the TEM micrograph of PS/Na-MMT3 composite had layered structure and the interlayer spacing of Na-MMT is expanded. The circle mark demonstrates the intercalated structure. The obtained intercalated structure in PS/Na-MMT3 composite is in good agreement with the XRD result. This feature was similar to the observations of other researchers. 13,22
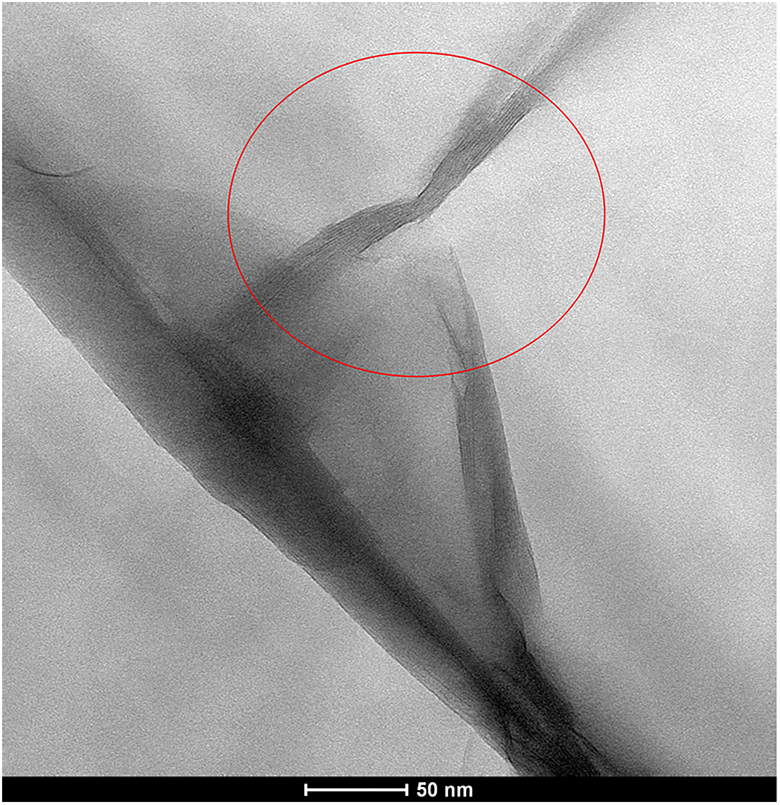
TEM image of PS/Na-MMT3. TEM: transmission electron microscopy; Na-MMT: Na-montmorillonite; PS: polystyrene.
The BET surface area measurements and micro-mesopore volumes
The determined BET-specific surface areas and micro-mesopore volumes of Na-MMT and PS/Na-MMT composites using both adsorption and desorption isotherms are listed in Table 2.
The BET specific surface areas (A) and specific micro-mesopore volumes (V) of Na-MMT and PS/Na-MMT composites.
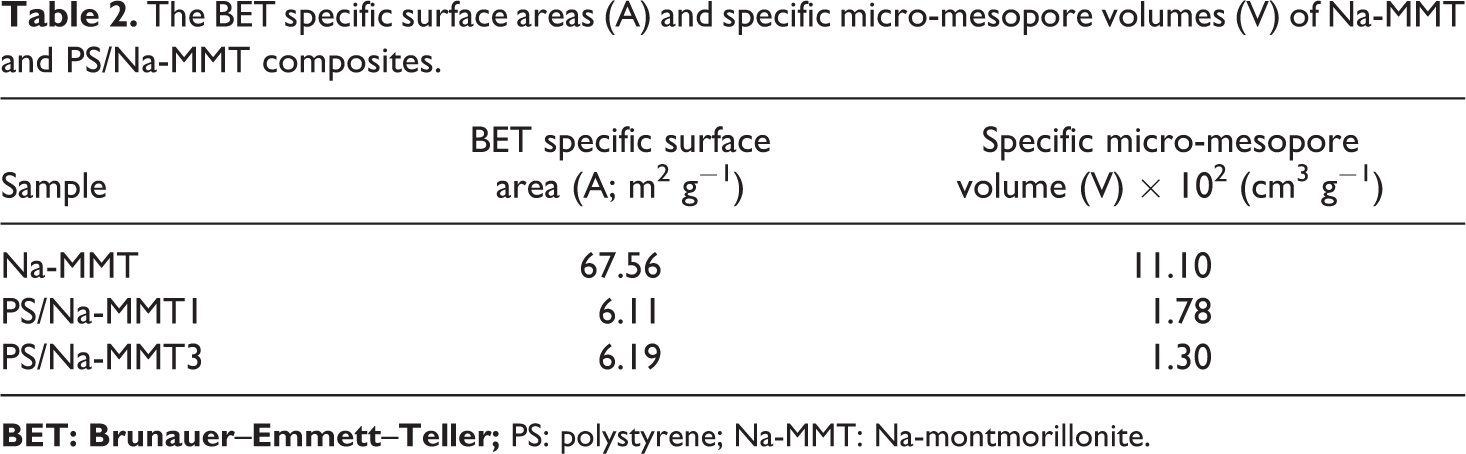
It could be as expected that the Na-MMT would exhibit higher BET surface area and micro-mesopore volume than PS/Na-MMT composites. The BET surface area and micro-mesopore volume of Na-MMT were 67.56 m2 g−1 and 11.10 × 10−2 cm3 g−1, respectively. Table 2 shows that these values decreased significantly to 6.11 m2 g−1 and 1.78 × 10−2 cm3 g−1 for PS/Na-MMT1 and 6.19 m2 g−1 and 1.30 × 10−2 cm3 g−1 for PS/Na-MMT3, respectively. Lower surface area of PS/Na-MMT composites than that of Na-MMT (Table 2) suggests that the composite samples may have considerable amount of covered or filled small- and medium-sized pores by PS. 23 –25
Measurements of moisture retention and water uptake
The moisture retention and water uptake values of Na-MMT and PS/Na-MMT composites decreased with PS content as shown in Figures 7 and 8. It was observed that moisture retention of composites decreased gradually, but water uptake decreased remarkably.
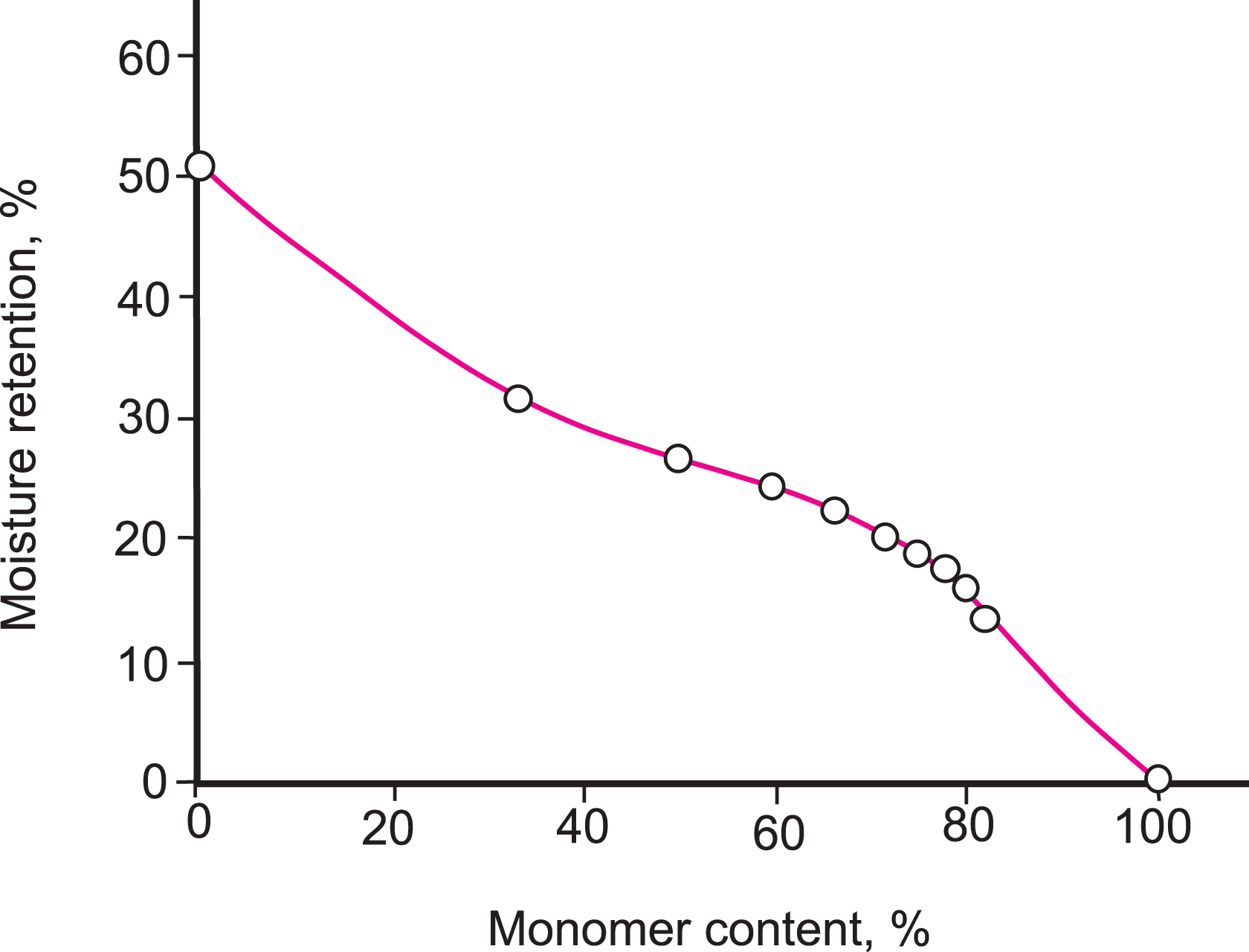
Percentage moisture retention values of Na-MMT and PS/Na-MMT composites obtained including different percentages of styrene. Na-MMT: Na-montmorillonite; PS: polystyrene.
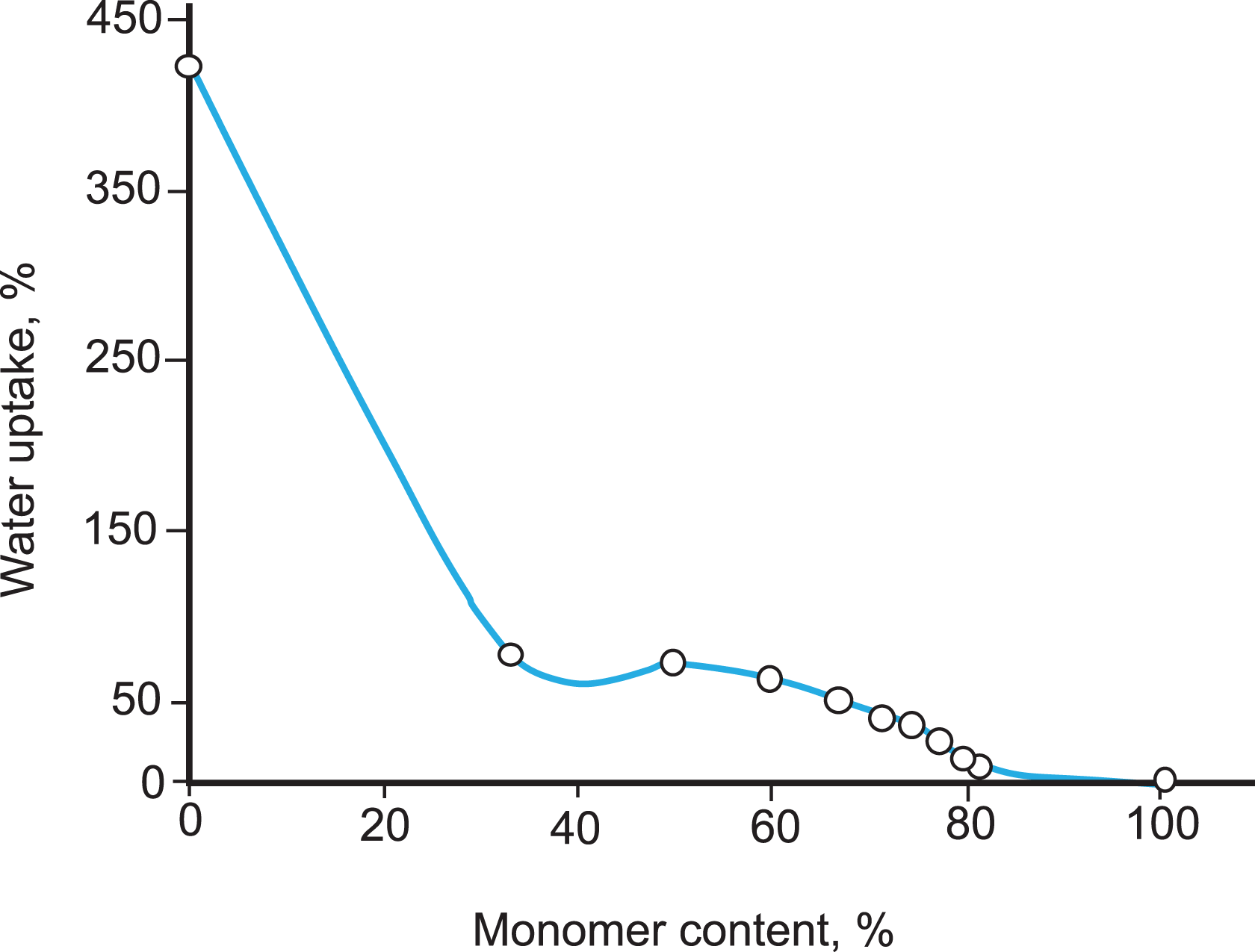
Percentage water uptake values of Na-MMT and PS/Na-MMT composites obtained including different percentages of styrene. Na-MMT: Na-montmorillonite; PS: polystyrene.
The decreases in moisture retention and water uptake can be attributed to the percentage of clay in the composites being limited, which reflects that the quantity of the polymer introduced in the layers reaches a limit and is enough to achieve maximum opening of the interlayers of clay and the formation of a cross-linked structure to a certain extent which prevents the addition of water molecules. Finally, water resistance of the composites can be greatly improved.
Conclusions
In this study, PS/Na-MMT composites were synthesized using in situ radical polymerization at different contents of styrene monomer. Intercalated morphology observed for composites and confirmed by XRD results. The thermal stability of PS/Na-MMT composites was improved by incorporating and dispersing of the Na-MMT in the PS matrix. The SEM revealed that, at higher content of styrene, the morphology of the composite is spongy and porous-shaped morphology and confirms the dispersion of silicate layers in the PS matrix. The TEM image showed the intercalation of PS between the clay layers in nanoscale. The resulting composites are more thermally stable than pure PS. The overall thermal stability trends as follows: Na-MMT > PS/Na-MMT3 > PS/Na-MMT7 > pure PS. BET surface area, micro-mesopore volume, moisture retention, and water uptake values of composites obviously decreased compared with that of Na-MMT.
Footnotes
Author’s note
Analyses for the characterization of composites were made at METU Central Laboratory.
Acknowledgment
The authors are grateful to Professor Müşerref Önal for her kindness of donation of clay sample.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Ankara University Scientific Research Projects Coordination Unit (project number 16L0430012, 2016).
