Abstract
Solid polymer electrolytes (SPEs) are the best choice to replace liquid electrolytes in supercapacitors, fuel cells, solar cells and batteries. The main challenge in this filed is the ionic conductivity and thermal stability of SPEs which is still not up to mark, therefore more investigations are needed to address these issues. In this study, PVA/salt based SPEs was fabricated using both solution cast and electro-spinning methods to probe the effect of different salts such as (NaCl, KCl and KI) and their concentrations on the ionic conductivity. Scanning electron microscopy (SEM) x and Fourier Transform Infra-Red (FTIR) have been employed to study the morphology as well as the different functional groups of SPEs, respectively. It was noted that small addition of NaCl, KCl and KI salts in SPEs dramatically increased the ionic conductivity to 5.95×10−6, 5.31×10−6 and 4.83×10−6 S/cm, respectively. Importantly, the SPEs obtained with NaCl via electro-spinning have higher ionic conductivity (5.95×10−6 S/cm) than their casted SPEs (1.87×10−6 S/cm). Thermal stability was also studied at two different temperatures i.e. 80 °C and 100 °C. The weight loss percentage of electrospun SPEs have zero percent weight loss than the solution based SPEs. The combined results clearly indicated that the nature of salt, concentration and fabrication process play a vital role in the ionic conductivity. Also, the NaCl salt with low molecular weight at low concentrations shows an enhanced ionic conductivity.
Keywords
Introduction
In recent years, solid polymer electrolytes (SPEs) are markedly investigated owing to their peculiar properties and promising practical and industrial applications, particularly in energy storage devices and fuel cell developments [1–3]. Among the various methods employed to fabricate SPEs, electro-spinning technique is highly favored due to ease of procedures, production of very thin fibers with large surface areas, ease of functionalization, and superior mechanical properties for improved efficiency [4,5]. Given that polymer nanofibers membrane has the capability for ion exchange, it can be used as electrolyte as well as separator [6]. Polyvinyl alcohol (PVA) is vital for the design of SPE and plays a leading role in the PVA-based SPE using different salt concentrations. In this respect, PVA offers excellent mechanical resistance, flexibility, high ion solvation capability and prominent ionic flow [7,8]. Furthermore, different salts such as NaCl, KI, or KCl can used, given that every salt has its own property and imparts different ionic conductivity, electrical conductivity, together with optical, thermal and mechanical properties [9–11]. SPEs have proactive application because of its advantage of low-cost production, durability, light weight and resistance to deteriorate. Importantly, SPEs are considered as conducting polymers having metallic properties due to their extended π-electron (polarons) along the chain [12,13]. Numerous research work has been conducted on the development of smart materials for rechargeable batteries and energy applications [14–16]. SPEs have several applications which is depend upon the nature of work [17,18].
Currently, researchers are developing many conducting polymers for different applications such as electromagnetic shielding, ant statistic coating, compact capacitors and smart windows [19–21]. The dry SPE
Experimental
Fabrication of SPEs
Polyvinyl alcohol (PVA), sodium chloride (NaCl), potassium chloride (KCl) and potassium iodide (KI) were of analytical grade and purchased from Sigma Aldrich Karachi Pakistan. And they were used without further any treatment. The synthesis of SPEs werefollowed by two methods i.e solution cast and electrospinning method. In solution cast method, PVA (2 g) together with NaCl, KCl and KI (0.1 g, 0.2 g and 1 g) were dissolved in 18 ml of deionized water. Same process was repeated without the use of salt. The solution was poured in the petri dish and left for drying till complete solidification takes place. After solidification the samples were peeled from the petri dish. The sample was then symbolized as PVA-C, PVA-NaCl-C (20:1), PVA-NaCl-C (10:1), PVA-NaCl-C (2:1), PVA-KCl-C (20:1), PVA-KCl-C (10:1), PVA-KCl-C (2:1), PVA-KI-C (20:1), PVA-KI-C (10:1) and PVA-KI-C (2:1).While, in the electrospinning method, 2 g of PVA was dissolved in 18 ml of deionized water with and without addition of salt having concentration of 0.1 g and 1 g. The solution was filled into 20 ml syringe and positioned horizontally in syringe pump. In electro-spinning setup, two terminals power supply unit is utilized to deliver the electrostatic force for the formation of nanofibers. One terminal is attached with the tip of needle and other is connected with aluminum foil (fiber collector) in order to complete electric circuit. The distance between the syringe and collector was maintained at 7 cm and the flow rate of polymer solution and voltage were setup according to sound fabrication of nanofibers. The electro-spun samples were then air dried for 48 hours. After drying, the samples were peeled from the aluminum foil. These samples were labelled as PVA-E, PVA-NaCl-E (20:1), PVA-NaCl-E (2:1), PVA-KCl-E (20:1), PVA-KCl-E (2:1), PVA-KI-E (20:1) and PVA-KI-E (2:1).
Characterization of the prepared SPEs materials
Scanning electron microscopy (SEM) images were recorded on scanning electron microscope JSM-6380L JEOL operated at an accelerating voltage of 20 kV. Fourier transform infrared (FTIR) spectroscopy was obtained on a (Thermo Scientific Nicolet iS10 in region of 3500-750 cm−1) at room temperature. Thermal stability of SPEs was determined by weight loss techniques. In this technique, the initial weight (Wi) of samples was determined and these samples were placed into oven at 80 °C and 100 °C for 2 hours. After that final weight (Wf) was determined. Percentage weight loss was determined by using the following formula:
Electrochemical impedance investigation
All of the electrochemical impedance measurements were carried out at room temperature on a (VERSASTAT 4-500 Princeton Applied Research) using a two-electrode system at frequency 1×10−6 to 4×105 Hz. These two terminals of electrode were connected with two stainless steel plates of 2.5×2.5 cm area dimension. The SPEs samples were taken in similar dimensions and hold in between two stainless steel plates then load is applied. The ionic conductivity (σ) in S/cm was determined by following formula:
Where
Result and discussion
Material characterization
The FTIR spectraof solution cast samples including pure PVA and PVA complexed with different salts such as KCl, KI and NaCl, respectively are shown in Figure 1(a). In pure PVA, the major peak of intermolecular hydrogen bond (O-H Stretching) observed at range of 3223–3422cm−1 [31], while in case of PVA with added KCl, KI and NaCl is shifted to 3250–3344 cm−1, 3237–3367 cm−1 and 3230–3376 cm−1, respectively [32]. Another peak occurs at 2924 cm−1 in pure PVA, assigned to C-H stretching of alkyl group is shifted to 2923 cm−12,921 cm−1 and 2918 cm−1 in the presence of KCl, KI and NaCl, respectively. Moreover, the C = O peak is detected in pure PVA at 1719 cm−1, which is almost similar in range but the size of peak may vary in PVA salt complex. In pure PVA, O-H bending peak is shifted from 1434 cm−1 to 1426 cm−11,424 cm−1 and 1420 cm−1 and whereas C-O stretching peak is shifted from 1054 cm−1 to1068 cm−11,060 cm−1 and 1065 cm−1 in case of added KCl, KI and NaCl, respectively [33]. The FTIR study has confirmed that the salts are chemically loaded on the PVA. The interaction between salts and PVA is mainly governed by electrostatic force.

FTIR spectrum (a) cast sample (b) electrospun sample.
The FTIR spectrum of electro-spun samples of pure PVA and complexed PVA with various salts like KCl, KI and NaCl, respectively are shown in Figure 1(b). The major changes that have been noted in spectrum included the O-H stretching peak appeared at 3174–3313 cm−1 range and shifted to 3234–3285 cm−1 and 3153–3263 cm−1 by the addition of KCl, KI and NaCl, respectively. Interestingly, the C = O stretching peak that observed at 1734 cm−1 in pure PVA is higher than the solution samples, possibly due to differences in the morphology of PVA in the two cases (solution vs. electro-spun). In case of complexed PVA, the C = O stretching band is shifted to 1701 cm1,1681 cm−1 and 1686 cm−1 whereas the O-H bending peak detected at 1406 cm−1 is shifted to 1413 cm−11,406 cm−1 and 1401 cm−1 in the presence of KCl, KI and NaCl, respectively. Moreover, the C-O stretching peak appeared in pure PVA at 1057 cm−1 is shifted in complexed PVA to 1063 cm−11,056 cm−1 and 1057 cm−1 for KCl, KI and NaCl, respectively [34,35]. Taken together, these IR data clearly indicate that addition of different salts may facilitate the electrostatic interaction between the opposite charged ions of the salts with PVA.
It is reported that when salt is added in PVA then O-H bending and stretching peak range decreases. It occurs due to decrease in decomposition temperature of PVA with addition of salt. Therefore, the backbone of PVA which is O-H group is complexed with cations of salt [36].
The Scanning electron micrographs (SEM) of SPEs based on PVA with different salts like KCl, KI and NaCl at different concentrations, prepared by solution cast method are shown in Figure 2. Initial inspection of the obtained SEM images revealed that the particles appear as a surface structure with uniform distribution [37,38]. The size of PVA particles is around 3–5 microns. It is evident from the Figure 2(a) that both pristine PVA and PVA-based SPE samples have homogeneous phase with the later containing low amount of salts (20:1). This is most likely due to complete dissolution of added salts at low concentration in PVA and hence formed homogeneous phase. However, the PVA samples having higher percentage of salts with either (10:1) or (2:1) ratio, showed a precipitation phase. It is worthy to mention that if the amount of added salt exceeded the solubility limit, then salt is precipitated out from the PVA. Therefore, the precipitation phase is obtained at higher concentration of salt in PVA and it affects the ionic conductivity [39]. Virtually, the precipitation of salts in PVA caused a bigger size aggregates, which could reach tens of microns and the particles are not uniform.

SEM images of various samples (a) PVA-C, (b) PVA-NaCl-C (20:1), (c) PVA-NaCl-C (10:1) (d) PVA-NaCl-C (2:1), (e) PVA-KCl-C (20:1), (f) PVA- KCl-C (10:1), (g) PVA-KCl-C (2:1), (h) PVA-KI-C (20:1), (i) PVA-KI-C (10:1), (j) PVA-KI-C (2:1).
SEM images obtained forSPEs fabricated by electrospinning method are shown in Figure 3. Pristine PVA exhibited smooth and thin fibers [40] as shown in Figure 3(a), however addition of salt to PVA increases the diameter of fibers and results in agglomeration [41,42] as illustrated in Figure 3(b) to (g). Thus, it can be noted that electro-spun method has provided fiber like morphology with a 10 to 50 microns in length. This means that afterloading the salts, the morphology was markedly altered due to the swelling as the salt ions were carried by the nanofibers, thereby causing expansion of the generated fibers.

SEM images of various samples (a) PVA-E, (b) PVA-NaCl-E (20:1), (c) PVA-NaCl-E (2:1), d) PVA-KCl-E (20:1), (e) PVA-KCl-E (2:1), (f) PVA-KI-E (20:1), (g) PVA-KI-E (2:1).
The thermal stability of SPEs based on PVA-Salt is determined by weight loss method at 80 °C and 100 °C. The cast samples represent the weight loss in which pure PVA has high weight loss percentage while the samples having salt at different concentrations showed less percentage of weight loss as shown in Figure 4. It is evident that as the salt percentage increases the weight loss decreases. The weight loss decreases as the series of NaCl<KCl<KI. It is anticipated that decrease in weight loss is dependent on the melting point of the salt. Zero weight loss is seen in case of electro-spun samples with and without salt at the two used temperatures as shown in Figure 5. It is predicted that these samples would show good thermal stability at the working condition as SPEs. The electro-spun samples are much drier and free of moisture thus they have shown superior thermal stability than the solution based samples.

Weight loss of cast samples at (a) 80 °C, 9b) 100 °C.

Weight loss of electrospun samples at (a) 80 °C, (b) 100 °C.
Electrochemical impedance investigations
The electrochemical impedance spectroscopy (Bode Plot) of SPEs based on PVA-Salt fabricated via solution cast method are shown in Figure 6. The obtained data implied that pure PVA has low ionic conductivity of about 5.59x10−7 S/cm at 106 Hz frequency. As illustrated in Figure 6(a) small addition of NaCl caused the ionic conductivity to increase to 1.87×10−6 S/cm, however, further increase in the salt concentration decreases the ionic conductivity to 1.58×10−6 S/cm and 1.4×10−6 S/cm in case of PVA-NaCl (10:1) and PVA-NaCl (2:1), respectively.
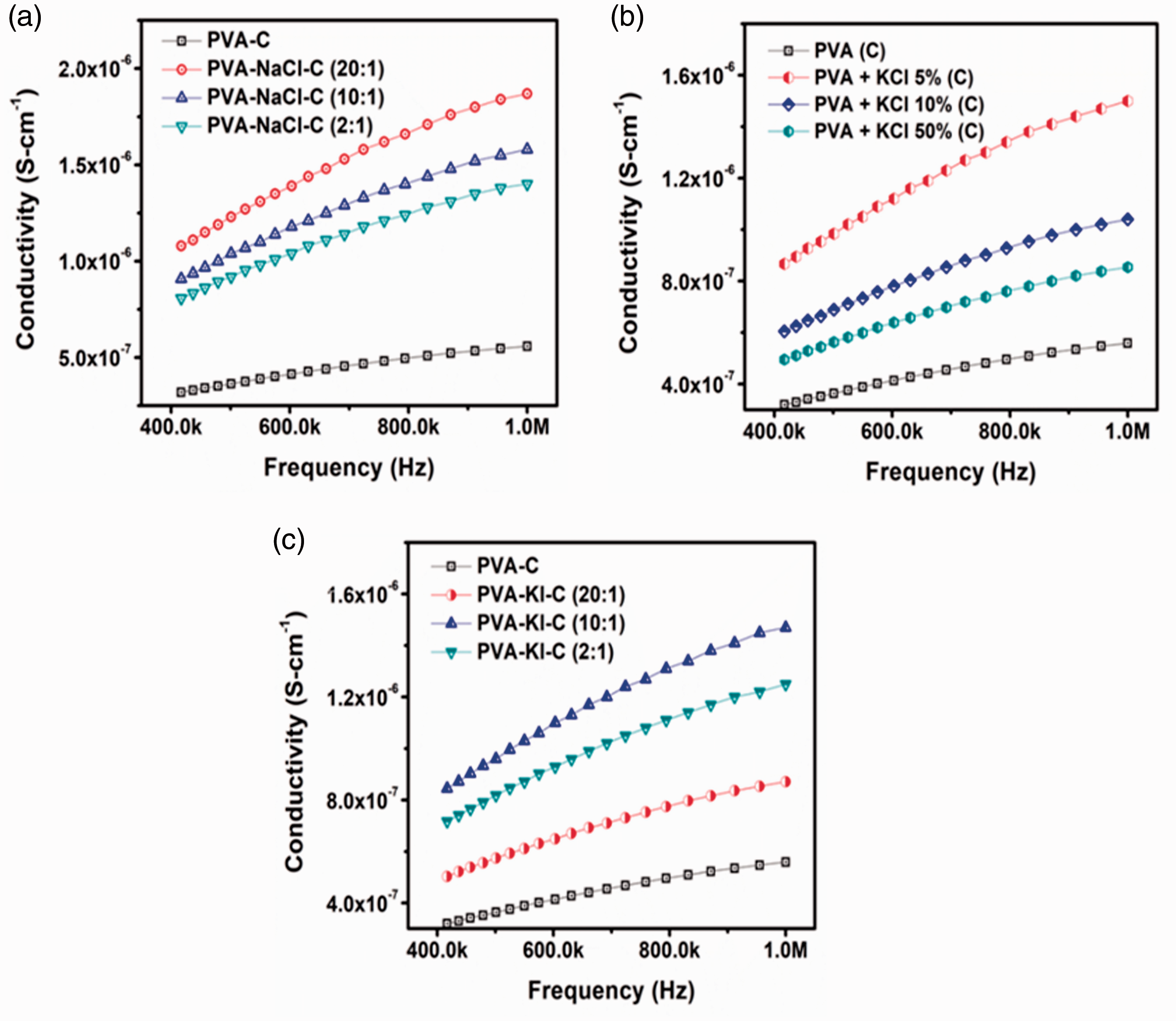
EIS Bode Plot of cast samples at various concentration with salts (a) NaCl (b) KCl (c) KI.
Similar trend is evident in case of KCl and KI where their addition to a certain limit enhances the ionic conductivity, while further increase in salt concentration caused decrease in the ionic conductivity. It is clearly established from the SEM images that higher concentrations of all salts caused precipitation on the polymer surface, which is responsible for the decrease in the ionic conductivity after certain concentration.
The PVA-NaCl (20:1) SPE shows high ionic conductivity as compare to SPEs fabricated using KCl and KI. This is because salts having lower molecular weight can contribute faster in ionic conductivity [43].
The electrochemical impedance spectroscopy (Bode Plot) of SPE
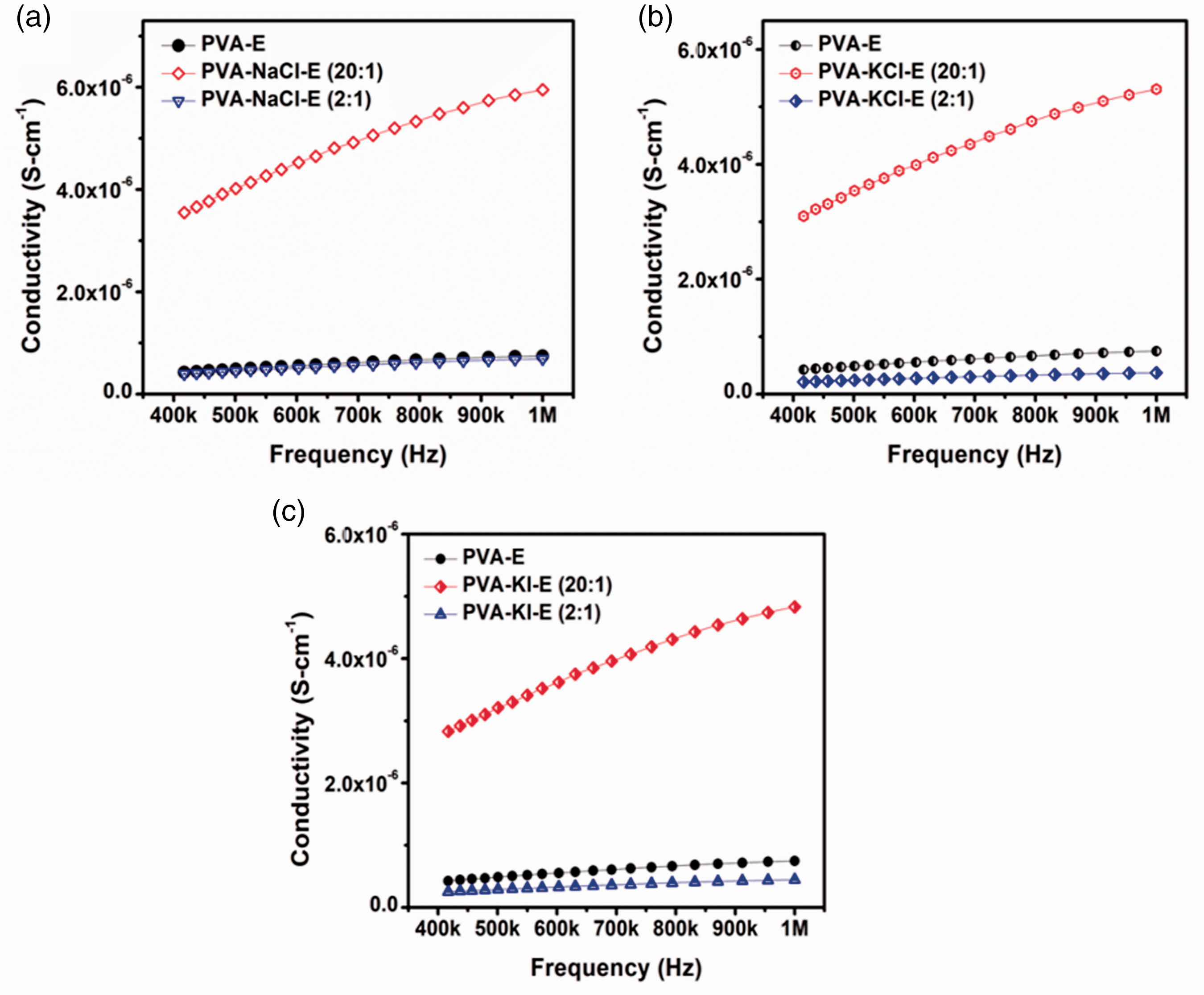
EIS Bode Plot of electrospun samples at various concentration with salts (a) NaCl (b) KCl (c) KI
The combined results clearly established that the trend of ionic conductivity of PVA-salt SPEs based nanofibers follows the order: NaCl>KCl>KI, which is highly dependent upon the molecular mass of the salt. Moreover, the decline in the ionic conductivities occurs when the concentration of added salt exceeds the solubility limit. This is likely due to the ionic pairing or ion association. Further it produced ion triplets which decreases the ionic conductivity [44,45]. Importantly, a high conductivity is found for the electrospun samples than the solution based samples due to their high surface area to volume ratio [46,47]. The electrospun samples exhibited a well-defined morphology which accelerated the charge transport.
Conclusions
In summary, SPEs based on PVA-salt, containing different salts such as NaCl, KI and KCl at various concentrations, were successfully fabricated using both solution cast and electrospinning methods. SEM micrographs of solution cast SPEs based on PVA-salt provide information that pure PVA has a homogenized phase. Small addition of desired salt does not disturb the homogeneity whereas addition of salt beyond the solubility limits induced fast precipitation on the surface. While in electro-spun sample, thin fibers of pure PVA are produced; as the percentage of salt increases then agglomerated fibers are obtained. The change in O-H stretching and bending in FTIR spectra confirmed the interaction-complexation of PVA with deliberately added salt. The electro-spun samples have high thermal stability, while the cast samples exhibited less thermal stability. Furthermore, The EIS (Bode Plot) of casted and electro-spun SPE
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was accomplished in Department of Metallurgy and Materials Engineering and Advanced Research Laboratory, MUET, Jamshoro, Sindh, Pakistan. We extend our sincere appreciation to the Researchers Supporting Project number (RSP-2020/79) at King Saud University, Riyadh, Saudi Arabia.
