Abstract
We prepared a solid polymer electrolyte (SPE) composed of a lithium borate salt and a polymer matrix, which can be employed for all-solid-state lithium-ion batteries. The lithium borate salt was made from lithium cations and bis (maleic acid) borate anions, and exhibits an excellent thermal stability as well as high ionic conductivity. The polymer matrix is an amorphous polymeric material having no crystalline regions, which is beneficial for the movement of lithium ions in the SPE. The polymer matrix also has good mechanical performance and thermal stability. Moreover, the SPE also has a relatively high ionic conductivity.
Keywords
Introduction
The requirement for sustainable energy has become a crucial and big challenge of the entire world due to our large population and the shortcomings of fossil energy such as pollution to environment and lack of renewability. 1,2 To address this issue, lithium-ion batteries (LIBs) were chosen by the scientific–technological community as one of the solutions because of their extraordinary advantages, such as high specific power and energy density, long cycle life, low self-discharge rate, and long-term stability, 3 -6 and have been widely used for cell phones, laptops, digital cameras as well as hybrid energy and pure electric vehicles. 7 -10
Electrolytes are critical components of LIBs, in which lithium ions move between cathodes and anodes during the charge/discharge process. 11,12 The current commercialized LIBs use liquid electrolytes, which have many virtues like high ionic conductivity. 13 Nevertheless, there are many drawbacks of liquid electrolytes that severely restrict the further application and development of LIBs, for instance, the presence of corrosive organic solvents, which may react with the seals and containers of the batteries, leakage of electrolytes, poor thermal stability, and high flammability. 14 -16 In recent years, solid electrolytes were proposed as the alternatives of liquid electrolytes for LIBs, which exhibit many merits, such as absence of organic solvents, no leakage, durable thermal stability, low flammability, and high energy density. 17 -19 Solid electrolytes have two types: inorganic solid electrolytes and solid polymer electrolytes (SPEs). SPEs have been intensively studied due to their prominent advantages over inorganic solid electrolytes, such as flexible geometry, excellent processability, and superior mechanical properties. 20 -22
SPEs of LIBs consist of polymer matrices and lithium salts. 23 Polyethylene oxide (PEO) is most commonly used as the polymer matrix for SPEs because of its strong solvating ability for lithium ions and excellent compatibility with lithium salts. 24 -26 Unfortunately, PEO-based SPEs have a low ionic conductivity because the crystalline regions of PEO restrict the movement of lithium ions. 27 -29 While amorphous polymers are much more beneficial than crystalline or semicrystalline polymers when used as polymer matrices of SPEs. With respect to lithium salts, lithium hexafluorophosphate (LiPF6) is the most popular one due to its excellent performance including high ionic conductivity. 30,31 But LiPF6 suffers from thermal instability and decomposition, which strongly impede its employment in LIBs. 32 -34 Researchers are paying much attention to the study of lithium salts looking for the ones with good comprehensive performance.
In this study, we prepared an SPE that is composed of a novel lithium borate salt and a polymer matrix. The novel lithium borate salt is made from lithium cations and bis (maleic acid) borate anions (abbreviated as LiBMB), which has excellent thermal stability and high ionic conductivity meeting the practical requirement of lithium salts of LIBs. The polymer matrix is an amorphous polymer material with extraordinary thermal stability according to the characterization data. Moreover, the SPE also shows good ionic conductivity.
Experiment
Materials
Lithium hydroxide monohydrate (LiOH·H2O), maleic anhydride (MAH), boric acid (H3BO3), methyl cyanide (MC), ethylene carbonate (EC), and diethyl carbonate (DEC) were all provided by Aladin Reagents Co. Ltd (Shanghai, China). Dimethyl acetamide (DMAc), hydroxyethyl acrylate (HEA), butyl acrylate (BA), and benzoyl peroxide (BPO) were purchased from Changzheng Chemical Company (Chengdu, China). All the chemicals above were analytical reagents. LiOH·H2O was dried in a vacuum oven at 100°C for 4 h before use, removing its crystal water. DMAc was distilled twice under reduced pressure before use. All the other chemicals were used as received.
Synthesis of LiBMB
In a three-necked flask, 9.8 g of MAH reacted with 2.1 g of LiOH·H2O in 100 mL of DMAc at 80°C for 2 h. H3BO3 (3.1 g) was added and reacted at 80°C for 2 h and then further reacted at 120°C for 4 h. The crude product was obtained by vacuum filtration of reaction mixture, removing solvent of filtrate and subsequent drying at 120°C under vacuum. The pure product was collected after recrystallization twice in MC. The schematic synthesis process of LiBMB is shown in Figure 1(a).

(a) Schematic synthesis process and (b) FTIR spectrum of LiBMB.
Preparation of the polymer matrix solution
In a three-necked flask, 0.8 g of HEA, 0.2 g of BA, 0.02 g of BPO (the initiator), and 20 mL of DMAc were added and reacted at 75°C for 1 h. A monomer solution (7.2 of HEA, 1.8 g of BA, and 0.08 g of BPO dissolved in 30 mL of DMAc) was added dropwise during a course of 2 h and proceeded at 75°C for another 6 h and further reacted at 80°C for 2 h, resulting in the final polymer matrix solution.
The polymer matrix for testing was prepared by casting the polymer matrix solution into a Teflon baffle plate, drying at room temperature for 10 days, and further drying under vacuum at 60°C for 10 h.
Preparation of the SPE
In a three-necked flask, 10 g of the as-prepared LiBMB was dissolved in 60 g of the polymer matrix solution and homogenized at room temperature for 10 min, leading to the polymer electrolyte solution. The SPE was prepared by casting this polymer electrolyte solution into a Teflon baffle plate and drying at 50°C for 7 days.
Characterizations
Fourier transform infrared (FTIR) spectra of LiBMB and the polymer matrix were characterized by a Magna-IR-560 FTIR spectrometer (Nicolet Company, Madison, Wisconsin, USA), in the frequency range between 400 cm−1 and 4000 cm−1.
Proton nuclear magnetic resonance (1H-NMR) and carbon-13 nuclear magnetic resonance (13C-NMR) of LiBMB were confirmed by an AV-II 400-MHz NMR spectrometer (Bruker Corporation, Germany). Samples were dissolved in deuterated dimethyl sulfoxide for testing.
Thermal stability of LiBMB and the polymer matrix were tested by an SDT-Q 600 analyzer (TA Company, New Castle, Delaware, USA). About 5 mg of the samples was tested from 40°C to 600°C with a heating rate of 10°C min−1 under nitrogen atmosphere.
Molecular weight and polydispersity of the polymer matrix were tested by an HLC-8320 gel permeation chromatography (GPC, Tosoh, Japan). Dimethylformamide (DMF) was employed to dissolve the polymer matrix for GPC test.
Mechanical property of the polymer matrix was tested by a Force Gauge Tester (Changping Electronic Instrument Company, China) according to the standard of GB/T 528-1998 at 25°C and 60°C, respectively.
Thermal property of the polymer matrix was characterized by a differential scanning calorimeter (DSC 204; Netzsch Company, Germany). Samples of about 3 mg were tested in the temperature range between 10°C and 110°C with a heating rate of 10°C min−1 under nitrogen atmosphere.
Ionic conductivity of LiBMB in the mixture of EC and DEC was measured by a DDSJ-307 conductivity tester (Shanghai INESA Instrument Co. Ltd, China).
Ionic conductivity of the SPE was analyzed based on the electrochemical impedance spectroscopy (EIS) method using a CHI-660E electrochemical workstation (Shanghai Chenhua Company, China). 35
Results and discussion
Chemical structure of LiBMB
FTIR spectrum of LiBMB is shown in Figure 1(b). As can be seen in Figure 1(b), ester groups have an absorption peak at 1717 cm−1, which is assigned to C=O stretching vibration. The absorption peaks at 1624 and 1364 cm−1 are ascribed to C=C and B–O stretching vibration, respectively. C–O stretching vibration shows an absorption peak at 1111 cm−1. The absorption peak at 984 cm−1 belongs to O–B–O stretching vibration. Additionally, the residual hydroxyl groups from the raw materials show an absorption at 3458 cm−1.
1H-NMR and 13C-NMR spectra of LiBMB are shown in Figure 2(a) and (b), respectively. As shown in Figure 2(a), LiBMB exhibits a single absorption peak in 1H-NMR because of its symmetrical structure, which has a chemical shift at 6.5 ppm ascribed to unsaturated hydrogen atoms of –CH=CH–. All the chemical shifts below 3.5 ppm belong to the solvent and the impurities. It can be seen from Figure 2(b) that the absorption peak at 135 ppm is assigned to the chemical shift of carbon atoms of –CH=CH–, and the absorption peak at 167 ppm belongs to the chemical shift of carbon atoms of ester groups. FTIR and NMR results confirm the successful preparation of LiBMB.
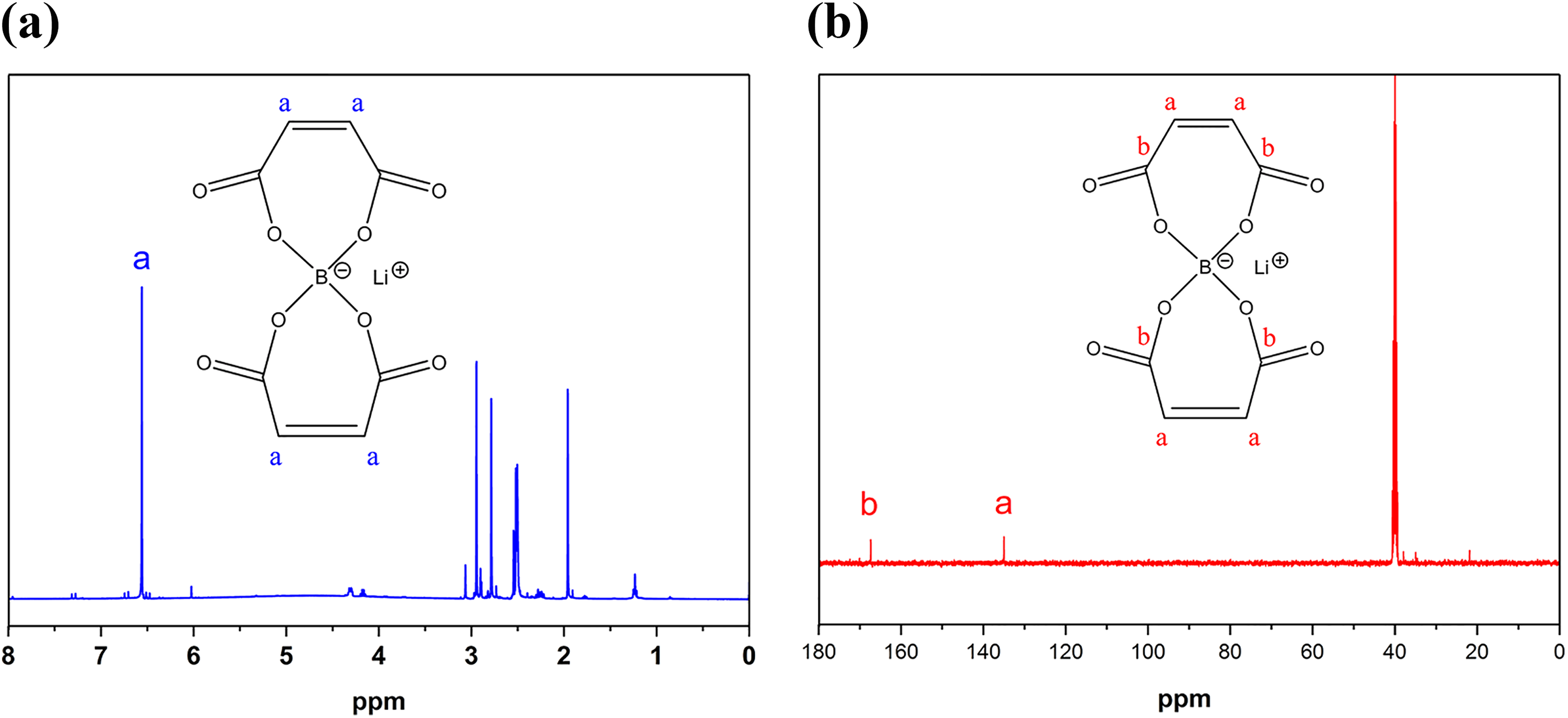
(a) 1H-NMR and (b) 13C-NMR spectra of LiBMB.
Thermal stability of LiBMB
Thermal stability is a vital property of lithium salts because LIBs always work under relatively high temperature. 36 In this article, we studied the thermal stability of LiBMB, and its thermogravimetry (TG) and derivative TG (DTG) data are shown in Figure 3. As shown in Figure 3, the thermal decomposition of LiBMB starts at 106°C with a weight loss of 5.0%. The peaks of DTG at 191°C and 291°C indicate the main decomposition of LiBMB. The decomposition ends at 600°C with a total weight loss of 74.1% (1–25.9%). The remaining weight of 25.9% is ascribed to the emergence of new inorganic lithium salts. It is certain that LiBMB has good thermal stability according to DG and DTG data, showing its competence as the lithium salt for LIBs.

TG data of LiBMB.
Chemical structure and mechanical performance of the polymer matrix
FTIR spectrum of the polymer matrix is shown in Figure 4(a). As can be seen in Figure 4(a), hydroxyl groups have a strong absorption peak at 3431 cm−1 that is assigned to O–H stretching vibration. The absorption peaks at both 2958 and 2875 cm−1 belong to C–H stretching vibration of methylene groups. The absorption peak at 1732 cm−1 is ascribed to C=O stretching vibration of ester groups. Methyl groups have an absorption peak at 1452 cm−1 attributed to C–H bending vibration. The absorption peak at 1169 cm−1 belongs to C–O–C stretching vibration of ester groups. Hydroxyl groups have an absorption peak at 1079 cm−1, which is assigned to C–O stretching vibration. All the characteristic absorption peaks of C=C double bond disappear.

(a) FTIR spectrum, (b) GPC data, and (c) mechanical performance of the polymer matrix.
The molecular weight and the polydispersity of the polymer matrix were tested by a GPC and shown in Figure 4(b). GPC data reveal that the number average molecular weight and the polydispersity index of the polymer matrix are 52335 and 1.93, respectively. The two-step method for the preparation of the polymer matrix presented in the section “Experiment” ensures that the polymerization takes place under a mild process, giving rise to the high molecular weight for the polymer matrix. As introduced before, excellent mechanical performance is one of the distinct advantages of SPEs over inorganic solid electrolytes for LIBs. Herein, we characterized the mechanical performance of the polymer matrix, and the results are disclosed in Figure 4(c). As exhibited in Figure 4(c), the polymer matrix has a tensile strength of 3.3 MPa and an elongation at break of 895% at 25°C, manifesting outstanding mechanical performance. As the temperature rises to 60°C, the polymer matrix still has a tensile strength of 2.1 MPa and an elongation at break of 542%. The reason why both the tensile strength and elongation at break reduce is that the intermolecular force of the polymer matrix is weakened at high temperature.
Thermal property and thermal stability of the polymer matrix
In this article, we studied the thermal property and thermal stability of the polymer matrix of the SPE by DSC and TG, respectively, and the results are shown in Figure 5. As shown in Figure 5(a), DSC curve of the polymer matrix has no melting peak in the temperature range between 10°C and 110°C, which means that the polymer matrix is in amorphous state and there are no crystalline regions to hinder the migration of lithium ions in the electrolyte. As we know, the homopolymer of each of the monomers chosen for preparing the polymer matrix in this study is in amorphous state, therefore, their copolymer, namely, the polymer matrix of the SPE, is obviously in amorphous state, too. As can be seen in Figure 5(b), the thermal decomposition of the polymer matrix begins at 259°C with a weight loss of 5.0%. There is only one peak in DTG curve at 421°C with a weight loss of 90.7%, which is assigned to the total decomposition of the polymer matrix. The remaining weight of 4.3% (1–5.0–90.7%) is the carbon residue of the polymer matrix. TG data indicate that the polymer matrix has an excellent thermal stability.

(a) DSC and (b) TG data of the polymer matrix.
Ionic conductivity of LiBMB
Bulk LiBMB is not available for the testing of ionic conductivity. Consequently, we dissolved LiBMB in the mixture of EC and DEC with a volume ratio of 1/1 to characterize its ionic conductivity. The effect of temperature and concentration on the ionic conductivity of LiBMB is shown in Figure 6(a) and (b), respectively. As displayed in Figure 6(a), the highest ionic conductivity of LiBMB is 1.2 × 10−3 S cm−1 and 2.8 × 10−3 S cm−1 at 303 and 353 K, respectively, where the concentrations of LiBMB in the solvents are both 1.5 M. As for the lowest concentration of 0.2 M of LiBMB, the ionic conductivity increases gradually from 3.2 × 10−4 S cm−1 to 1.3 × 10−3 S cm−1 as the temperature rises from 303 K to 353 K. Notably, the ionic conductivity of LiBMB increases sharply when the temperature goes from 290 K to 303 K. This is because the solubility of LiBMB in the solvents is not high enough and the activity of lithium ions is inferior when the temperature is low (below 290 K). As the temperature is elevated, the solubility of LiBMB in the solvents and the activity of lithium ions both increase rapidly, leading to the fast improvement of the ionic conductivity of LiBMB.

Effect of (a) temperature and (b) concentration on the ionic conductivity of LiBMB in the mixture of EC and DEC (EC/DEC = 1/1 by volume).
As depicted in Figure 6(b), at 290 K, the ionic conductivity increases linearly from 3.2 × 10−4 S cm−1 to 8.0 × 10−4 S cm−1 as the concentration of LiBMB in the solvents increases from 0.2 M to 1.5 M. This is because the activity of lithium ions is relatively bad at low temperature. When the temperature is in the range between 303 K and 353 K, the ionic conductivity increases rapidly as the concentration increases from 0.2 M to 0.8 M and then increases slowly from 0.8 M to 1.5 M. It means that the ionic conductivity will not keep increasing with the concentration, because excessive lithium ions and their anions will hamper the migration of lithium ions in the solvents. More importantly, the results of Figure 6 imply that LiBMB meets the practical requirement of lithium salts for commercial batteries. 37
Ionic conductivity of the SPE containing LiBMB
EIS curves and the ionic conductivity of the SPE under different temperatures are disclosed in Figure 7(a) and (b), respectively. The value of horizontal ordinate of the intersection in each EIS curve is the bulk resistance (

(a) EIS curves and (b) the ionic conductivity of the SPE under different temperatures.

where
Conclusion
In this study, an SPE that consists of a novel lithium borate salt, namely, LiBMB, and a polymer matrix was fabricated. LiBMB has good thermal stability with an initial decomposition temperature of 106°C, as well as high ionic conductivity, for instance, which is 1.2 × 10−3 S cm−1 at 303 K in the solvents with a concentration of 1.5 M. The polymer matrix is in amorphous state and no crystalline regions would hamper the movement of lithium ions inside. Additionally, the polymer matrix exhibits good mechanical performance, for example, a tensile strength of 3.3 MPa and an elongation at break of 895% at 25°C. And the polymer matrix also has a distinct thermal stability with a starting temperature of thermal decomposition of 259°C. Finally, the SPE also has a good ionic conductivity. We are working on the improvement of the comprehensive performance of the SPE, especially the ionic conductivity at its service temperature.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Natural Science Foundation of China (21704089).
