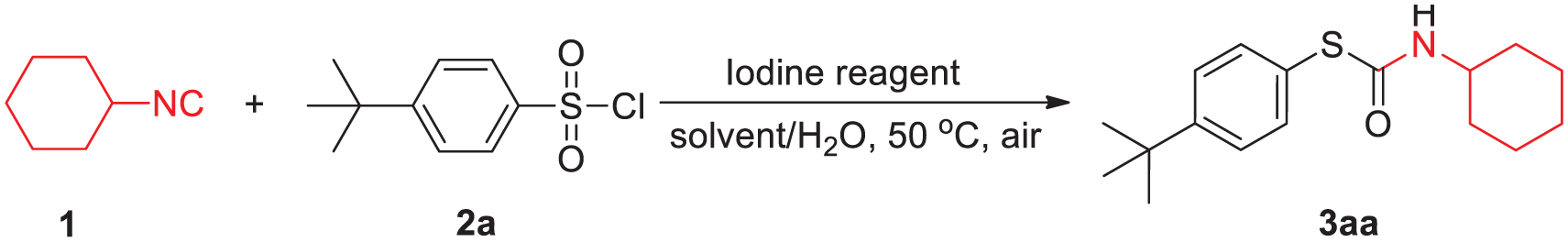Abstract
A simple and efficient process for direct generation of various S-thiocarbamates is developed by cross-coupling of readily available sulfonyl chlorides with cyclohexyl isocyanide. The yields are excellent and the structures of the generated S-thiocarbamates are characterized by nuclear magnetic resonance spectroscopy, infrared spectroscopy, and high-resolution mass spectrometry together with X-ray crystallographic analysis. The protocol has the advantages of using easily available reagents, employs inexpensive KI as the reagent, demonstrates good functional group tolerance, and utilizes mild reaction conditions.
Introduction
In the past few decades, the synthesis and study of organic sulfur chemicals has become a popular research topic.1–6 S-Thiocarbamates represent key building blocks that are common in many synthetic pharmaceuticals and natural biologically active compounds. 7 They have numerous biological applications in the manufacture of pharmaceuticals and agrochemicals including the HIV-1 nucleocapsid protein NCp7 inhibitor 8 and bactericidal, 9 anesthetic, 10 fungicidal, 11 pesticidal, 12 and herbicidal 13 substances. Therefore, the development of new methods for the synthesis of S-thiocarbamates is of widespread interest. Classic methods for the synthesis of S-Thiocarbamates include (1) reacting toxic phosgene/triphosgene or carbonyldiimidazole with an amine and thiophenol (Scheme 1(a)),14,15 (2) visible-light/rose bengal reactions (Scheme 1(b)),7,16 and (3) molecular iodine–catalyzed reactions of thiosulfonates (prepared from thiols or sulfonyl chlorides) with isocyanides (Scheme 1(c)). 17 However, these methods have shortcomings such as poor atom economy, harsh reaction conditions, and the use of difficult-to-obtain precursors as well as unstable, toxic, and/or hazardous chemical reagents. 7 Thus, it is necessary to develop an efficient and general route for the construction and enrichment of S-Thiocarbamate compounds.

Different synthetic routes for the synthesis of S-Thiocarbamates.
Sulfonyl chlorides are readily available and have been widely used in the fields of materials science as well as in organic and medicinal syntheses.18–21 Nonetheless, there is no example of the direct generation of S-Thiocarbamates from sulfonyl chlorides. On the contrary, methods based on metal iodide catalysis have gained significant attention in recent years because these processes are environmentally friendly and economical. In this paper, we report a simple and flexible synthetic route for the synthesis of various S-Thiocarbamates under mild reaction conditions using sulfonyl chlorides as the sulfur source (Scheme 1(d)).
Results and discussion
As a model reaction, the iodide-promoted reaction of cyclohexyl isocyanide (
Optimization of the reaction conditions a .

DMF: dimethylformamide; TBAI: tetrabutylammonium iodide; NMP: N-methyl-2-pyrrolidone; THF: tetrahydrofuran; DMSO: dimethyl sulfoxide.
Reaction conditions:
Isolated product yields after column chromatography.
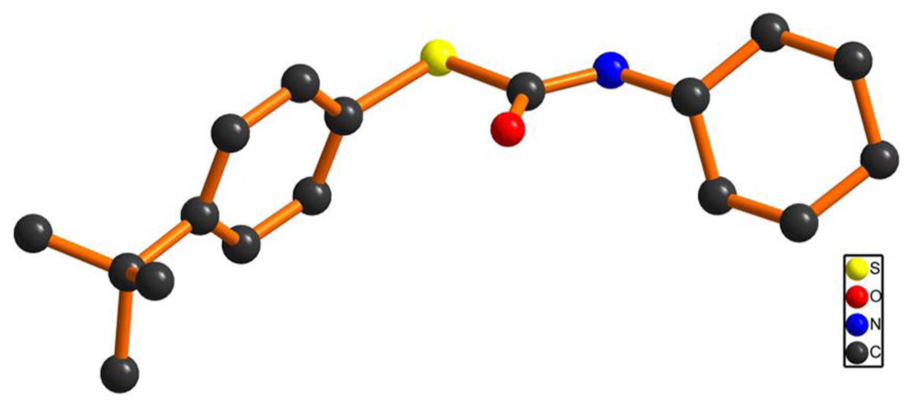
Ball-and-stick representation of the structure of
Utilizing the optimized reaction conditions, we extended the reaction scope (Scheme 2). Various substituted benzenesulfonyl chlorides were examined. It was confirmed that the reaction has good applicability for various substituent groups, giving the corresponding thiocarbamate derivatives in excellent yields. When 10 mmol of cyclohexyl isocyanide (

Substrate scope for the synthesis of

Gram-scale synthesis of
Based on the report of Zhao and Zhou,
22
a possible reaction mechanism has been proposed (Scheme 4). First, KI reduces sulfonyl chloride

A plausible reaction mechanism.
Conclusion
In summary, we have established a simple and convenient synthetic route for the preparation of S-Thiocarbamates. Using cheap and readily available sulfonyl chlorides as the sulfurizing reagents, S-Thiocarbamates with different substituents on the benzene ring can be constructed under mild reaction conditions. The protocol avoids the use of hazardous and unstable thiols. The reaction can be enlarged to gram scale, showing potential in industrial syntheses of S-thiocarbamates.
Experimental
All chemicals were commercially available and used without further purification. Analytical thin-layer chromatography (TLC) was performed on Merck Millipore silica gel 60 F254 plates. Column chromatography was carried out using 300–400 mesh silica gel (Qingdao Haiyang Chemical Co., Ltd).
The nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AV-II 500 MHz NMR spectrometer, operating at 500 MHz for 1H NMR and 125 MHz for 13C NMR. Mass spectrometric investigations were conducted with a Finnigan LCQ Advantage MAX mass spectrometer. Infrared (IR) spectra were recorded on a PerkinElmer spectrometer (Spectrum One). Melting points were measured with a Yanaco MP500 melting point apparatus and are uncorrected. Crystallographic determination of
General procedure for the synthesis of 3
Cyclohexyl isocyanide
S-4-tert-butylphenyl cyclohexylcarbamothioate (
S-phenyl cyclohexylcarbamothioate (
S-p-tolyl cyclohexylcarbamothioate (
S-mesityl cyclohexylcarbamothioate (
S-4-methoxyphenyl cyclohexylcarbamothioate (
S-4-(trifluoromethoxy)phenyl cyclohexylcarbamothioate (
S-4-fluorophenyl cyclohexylcarbamothioate (
S-4-chlorophenyl cyclohexylcarbamothioate (
S-4-bromophenyl cyclohexylcarbamothioate (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (No. 21772035), the Natural Science Foundation of Hunan Province, China (No. 2016JJ3050), the Scientific Research Fund of Hunan Provincial Education Department (No. 18A344), the Scientific Research Foundation of Hunan Institute of Engineering (XJ1707), and the Open Foundation of Key Laboratory of Environmental Catalysis & Waste Recycling, Hunan Institute of Engineering (2018KF10).