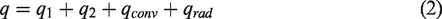Abstract
Flexible and durable electrically driven heaters (EDHs) was prepared by using silver nanowire/polydopamine modified cotton fabric (AgNW/PDA-C) as Joule heat generator and polydimethylsiloxane (PDMS) as stretchable matrix.The electric property of the EDHs was investigated, and it can show excellent electrical property and stability. The EDHs can be seen from the SEM morphology that cotton fabric was well protected by the PDMS and has good conductive network. The EDHs also exhibit excellent electrothermal performance under applied voltage. At low-voltages, it can quickly reach satisfactory steady-state temperatures that meet the needs of wearables. By improving the electrothermal model, it can be verified that there is a quadratic function relationship between electric power and steady-state temperature. Under cyclic voltage and bending tests, the EDHs can well maintain steady-state temperature and exhibit excellent dynamic and static electrothermal performance.
Keywords
Introduction
Electrically driven heater is an active heating product that converts electrical energy into heat through Joule heat [1,2]. EDHs have received much attention because of the potential applications in defogger/deicer, optoelectronics and protective devices [3–6]. Metallic materials are the most commercially used EDHs, but exist disadvantages such as high density, high-temperature process, easy corrosion and weak flexibility [7–9]. Recently, flexibility and stretchability have become the development requirements of the next generation of flexible wearable devices [10–13].
With the development of electronic textiles, textile-based EDHs gradually become enter people's attention [14–16]. The traditional textile-based EDHs that combine metal materials with fabrics are produced by compound technology [17]. Though it has certain flexibility and stretchability, it still exists weakness like high density, complicated process and uneven heating [18]. Currently, a common strategy to prepare EDHs is to disperse conductive fillers such as metal nanomaterials, carbon nanotubes and graphene nanosheets on textiles [19–22]. The novel textile-based EDHs process sufficient flexibility and stretchable to perform sufficient electrothermal properties in personal heating systems and healthcare management [23–25]. However, the textile originally has a three-dimensional structure and mainly combined with the conductive fillers by electrostatic adsorption and entanglement [26,27]. During practical use, the conductive fillers are prone to oxidation, fall off and abrasion [28,29]. These will cause increase in the resistivity and even the destruction of the conductive loop.This means that it is necessary to obtain a satisfactory effect by increasing the applied voltage or adding more conductive filler. In general, wearable devices need to take into account the unpredictable effects on the human body and need to avoid high voltages as much as possible. Therefore, it is still challenging to develop low-voltage EDHs that have excellent durable and long-term usability.
In order to solve the above problems, polydopamine modified cotton fabric with AgNW/flexible polymer having high conductivity may be a potential candidate for EDH. AgNW has excellent conductivity and is currently the hottest conductive filler [30,31]. Researches have shown that AgNW can exhibit excellent electrothermal performance at low-voltage as it compounds with polyethylene terephthalate (PET), polyimide film (PI), polyvinyl acetate (PVA) and other substrates [32–35]. Cotton fabrics are the most widely used for electronic textiles and easily functionalized to exhibit various properties [36,37]. Polydopamine (PDA) can form a functional coating to strengthen better adhesion between fillers and matrix [38–41]. The Young's modulus of PET, PVA or PI is too excessive to restrain the stretchability of the material. In contrast, polydimethylsiloxane is a more promising candidate due to its high flexibility and stretchability [42–44].
In this paper, flexible and durable EDHs were prepared by using PDA-modified cotton fabric with AgNW network and polydimethylsiloxane (PDMS). It is expected to obtain excellent electrothermal performance at low voltage. The PDA-modified cotton fabric with AgNW is sandwiched with stretchable PDMS film, which combines the high conductivity of AgNW with the flexibility of PDMS. As expected, the EDH could reach 59.4°C in a short time at 1.8 V and exhibit excellent electrothermal performance. Under cyclic voltage and bending tests, the EDH can still maintain relatively stable electrothermal performance due to the flexibility of the matrix.
Experimental
Materials
Silver nitrate (AgNO3), sodium chloride (NaCl), polyvinylpyrrolidone (PVP, K30) and hydrochloric acid (HCl) was obtained from Sinopharm Chemical Reagent Co., Ltd (China). Tris(hydroxymethyl)aminomethane and dopamine hydrochloride were suppliedby Shanghai Aladdin Chemical Reagent Co., Ltd. (China). Ethylene glycol (EG) was purchased from Shanghai Lingfeng Chemical Reagent Co., Ltd (China). Acetone and anhydrous alcohol were obtained from Changshu Hongsheng Fine Chemical Co., Ltd (China). Silicone elastomer kit (SYLGARD 184TM) was purchased from the Dow chemical company (China). The plain cotton fabric (2.5 cm × 2.5 cm, 50 weft cm−1, 50 warp cm−1, 100 g cm−2) was obtained from Shaoxing Shamanlou Textile Co., Ltd(China).
Preparation of the EDH
0.2 g of AgNO3, 0.4 g of PVP and 20 ml of EG were used to prepare AgNW solution according to our previous research [45]. 0.12 g of Tris was dissolved in 100 ml of deionized water and titrated to pH = 8.5 with HCl to obtain a Tris-HCl buffer solution. Then, 10 ml of buffer solution was taken, and 20 mg of dopamine hydrochloride was added to form a dopamine solution. Simultaneously, the cotton fabric was immersed in the dopamine solution for 24 hours, then washed and dried. PDA-C was dipped in a certain concentration of AgNW ethanol solution for 1 minute and dried in an oven, then repeated 10 times.
The Sylgard 184 silicone elastomer kit solution was ultrasonicated for 30 minutes to remove the bubbles. Then, a layer of solution was coated on a polytetrafluoroethylene plate, put into an oven and heated at 50°C for 30 minutes to prepare a PDMS preform. AgNW/PDA-C was then placed on the above of PDMS preform and the surface was coated with another layer of mixed solution. Finally, put it into an oven and heated at 100°C for 2 hours to be cured totally to form EDH. The schematic diagram of the preparation process of the EDH is shown in Figure 1. Three different EDH4, EDH6, EDH8 were prepared according to the concentration (4, 6 and 8 g/mL) of AgNW in ethanol solution.
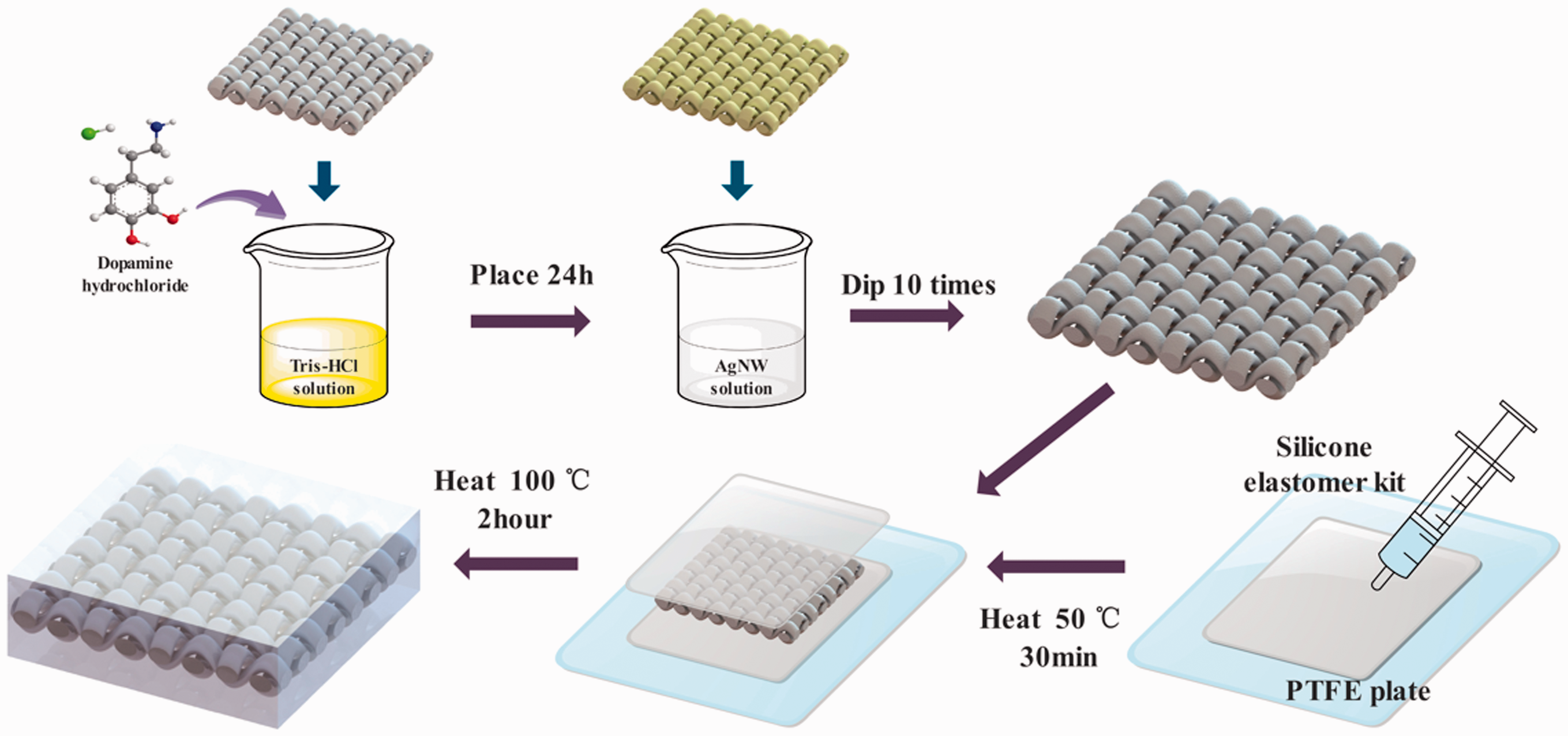
Schematic illustration of the formation mechanism of the EDH.
Characterization
Scanning electron microscopy (SEM, FlexSEM 1000, Japan) was used to investigate the surface morphology.The samples were attached to the microscope stage with conductive tape and apply a thin layer of vapor gold to the surface. Digital universal analyzer (VICTOR VC890D, China) was used to measure the surface-specific resistivity according to the AATCC 76-2005 test standard. A regulated DC power supply (APS3005S, China) was used to measure the electrical property of the samples. An infrared thermal imaging camera (FLTR T420, USA) was used to record the temperature distribution of the material under applied voltages.
Results and discussion
Electric property analysis
For electrical resistance of sample, two electrical conductive wires are adhered to the opposite two sides of rectangle sample respectively; then Keithley tester is used to measure the electrical resistance of sample by clamping the two electrical conductive wires. The curve of the resistance value of cotton fabric under different concentrations of AgNW solution is shown in Figure 2(a). The resistance value of cotton fabrics decreases with the increase of impregnation times. The resistance of the cotton fabric itself is very large, reaching 1015 Ω. With the continuous deposition of AgNW, the surface begins to form a conductive loop and the resistance declines significantly. Finally, as the AgNW approach saturation, the conductive loops become complete, and the resistance drop gradually begins to slow down.

Electrical property analysis: (a) resistances change of cotton fabric during impregnation; (b) resistance of cotton fabric and PDA-C after impregnation.
Figure 2(b) is the resistance comparison chart of cotton fabric and PDA after 10 impregnation times. It can be seen that the resistance decreases as the concentration of AgNW increases. In a relatively higher concentration solution, the resistance value is lower than that in a low concentration solution due to the cotton fabric has more opportunities to adsorb more AgNW. At the same concentration, the resistance value of PDA-C is lower than that of pure cotton fabric. This is because PDA has strong adhesion and absorb more AgNW during the dipping process. The conductive loop can form quickly, and the resistance value is relatively low.
The current-voltage curve of the EDHs at low voltage is shown in Figure 3. It can be seen that the current increases with the increase of the voltage, and there has a good linear tendency. Using polynomials for fitting, it can be found that the current-voltage curve of the EDHs can well fit the linear relationship (R2>0.99). This shows that the EDHs can well comply with Ohm's law, and maintain the resistance stability under low voltage.

Current-voltage curve.
SEM morphology
Figure 4(a) is the SEM of the internal heating element, and it has a characteristic interlaced woven structure. Moreover, it can be observed in Figure 4(b) that there exists a well-formed conductive cross-linking network among fibers. The surface and cross-section of EDH are shown in Figure 4(c) and (d). The surface is covered with a layer of PDMS film, which completely covers the interlaced structure ofcotton fabric. It also can be seen from the cross-section that the internal heating element itself still maintains a buckled structure, and combined with PDMS well without gaps. The above analysis shows that PDMS can wrap the internal heating elements well without changing its original buckling structure.

SEM photography of the heating element: (a) 100x, (b) 5000x; SEM photography of the EDH: (c) the surface (the upper right corner is digital photo), (d) the cross-section.
Electrothermal performance of EDHs
The electrothermal behavior of EDHs was studied by applying voltages. The temperature/time curves of the EDHs at different voltages are shown in Figure 5. The electrothermal behavior of EDHs under different applied voltages still conforms to the classic electrothermal curve trend, which can also be divided into three regions: heating, equilibrium, and cooling. In the first stage, the internal heating element generates Joule heat, which is transmitted to the air through PDMS, and the surface temperature gradually increases with time. In the second stage, the temperature no longer changes, and the Joule heat generated by the input voltage is equal to heat dissipation, the system reaches equilibrium. In the third stage, after the power is turned off, the EDHs slowly dissipate heat and returns to room temperature. The EDHs can reach steady-state temperature in a short time (∼75 s) and have good temperature response rate.

The temperature versus time curve of the EDHs under applied voltage: (a) EDH8, (b) EDH6, (c) EDH4; (d) Voltage vs. steady-state temperature curve; (e)
Further, the electrothermal performance of the EDH is analyzed. According to energy conservation, the input power (q) from the power supply is equal to the output power, which includes the energy absorbed by the heat generator(q1), PDMS (q2), thermal convection (qconv) and thermal radiation (qrad) [46,47].
Because of the size of composite is small, the temperature gradients can be considered the same, therefore:
Where, C1, m1, C2, m2 are the specific heat capacity, mass of the heating element and PDMS, respectively; A is the surface area; T and T0 represent surface temperature and initial temperature, respectively; hc, Ɛ, and σ are convection heat transfer coefficient, emissivity (Ɛ = 0.95) and Stephen-Boltzmann constant (5.67 × 10−8 W·m−2·K−4); h is the total heat transfer coefficient.
In the equilibrium region, the temperature no longer changes (
According to the above formula, the value of h can be calculated and listed in Table 1. It can be found that there is a positive correlation between h and temperature, so it can be considered:
Parameters of the EDHs under different applied voltages.

Where, a, b are constant values;
Equation (4) can be rewritten as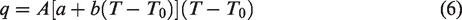
The equation (6) is a one-variable quadratic equation, and the solution is:
Where,
Substituting equation (5) into equation (3), it has.
In the heating region, the temperature material rises over time. According to 
Combining with equation (7), the above formula can be changed to equation (10).
Where,
In the cooling region, the temperature will gradually decrease to initial temperature due to the heat dissipation. Equation (8) can be changed to equation (11).
According to 
Where,
As shown in Figure 5(d), the electrothermal behavior depends on the applied voltage, and the steady-state temperature increases with the applied voltage. But there is no longer a quadratic relationship in the classic electrothermal model, it’s more tendency to a linear relationship.
According to 
The
In the modified electrothermal model, the important characteristic parameters of the electrothermal behavior areh,
The actual electrothermal curve and theoretical curve of the EDHs at 1.8 V voltage are shown in Figure 5(f). Comparing the two curves, it can be found that the electrothermal curve of the modified electrothermal model can better fit the actual electrothermal curve than the classic, and the steady-state temperature difference is smaller. The above analysis further shows that the EDHs can well match the modified electrothermal model under 1.8 V.
Static and dynamic electrothermal performance analysis
The static electrothermal stability was tested under constant voltage and gradient voltage cycles. The electrothermal behavior of the EDHs under voltage is shown in Figure 6(a) to (d). It can be found that the electrothermal curve keeps consistent and the steady-state temperature has not changed during 5 cycles. The gradient electrothermal behavior is consistent with that under single cycle and the steady-state temperature does not change.

The electrothermal behavior of the EDHs at a constant voltage: (a) EDH8, (b) EDH6, (c) EDH4; (d) electrothermal behavior under specific voltages; (e) Resistance change during 2000 repeated bending tests (f) The electrothermal behavior at 1.6 V after 2000 repeated bending tests.
The dynamic electrothermal stability was studied by comparing the electrothermal behavior after 2000 bends. It can be found in Figure 6(e) that the resistance change amount increases with the bending times and maximum change reaches about 0.12, which indicates that the EDHs have excellent bending resistance. The comparison of the electric heating curve before and after bending is shown in Figure 6(f). The electrothermal behavior of the two is almost the same, and the difference between the steady-state temperature is only about 4°C, which indicates that the EDHs have excellent dynamic electrothermal stability.
The above results show that EDHs have excellent static and dynamic electric heating performance and usability, which has the potential for wearable applications. This is mainly because the PDMS wraps the internal heating elements, making the cotton fabric fiber less prone to structural slack, filler oxidation and fall off. In addition, PDMS itself has superior anti-bending property, which can make the EDHs recover quickly after deformation and the internal structure not prone to damage.
Conclusion
In this paper, EDHs were successfully prepared by sandwiching the AgNW/PDA-C with stretchable PDMS. The electrical resistance of the composite decreases as the concentration of the AgNW solution increases and have excellent stability under low voltage. The PDA can strengthen the adhesion of the AgNW on the cotton fibers.The PDMS protects the internal Joule generator structure and maintains internal cross-linking conductive loop. At low-voltages, EDHs can reach a steady-state temperature higher than human body temperaturein a short time (75 s), which show fast response rate and excellent electrothermal property. The steady-state temperature is linear with the voltage and essentially quadratic relationship with electrical power, which can be well proven by the modified balance model.In periodic heating/cooling tests and anti-bending test, EDHs exhibit outstanding durability and can still achieve a satisfactory steady-state temperature, which means it has excellent flexibility and durability to meet the needs of wearable devices.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is jointly supported by Natural Science Foundation Project of Shanghai 2020 “science and technology innovation action plan” (20ZR1400200), Jiangxi Provincial Bureau for Quality and Technical Supervision (GZJKY201807), Jiangxi Provincial Administration for Market Regulation(GSJK201909).