Abstract
Silver nanowires were synthesized by a polyol method with a detailed control of synthesis conditions such as injection rate, concentration of copper (II) chloride, and silver nitrate to poly(vinylpyrrolidone) ratio. The silver nanowire films coated on poly(ethylene terephthalate) film/glass cover slips and lower density of longer silver nanowires and their contact junctions were observed lower. Silver nanowire films with a low sheet resistance (approximately 0.02 Ω/sq) and a high transmittance (approximately 90%) were resulted from longer silver nanowires. Further, sheet resistance and transmittance without any agitation in a synthesis process are demonstrated and compared. These results offer to design specifications of silver nanowires for highly transparent and metal conducting films.
Introduction
As we can see many applications in wearable electronics in day-to-day life, future electronic industry is expected to be stretchable, flexible, and friendly to human in the form of electronic devices. A new class of electronics is fascinating, which can be stretched, 1,2 twisted, 3 –5 and bent into many complex forms such as nonlinear rubber-like shape, 6 epidermal electronics, 7 three-dimensional photovoltaics, 8 artificial skin, 9 strain sensors, 10 and solar cells 11 while maintaining better conductivity 12 performance. In electronic industry, transparent electrodes are essential elements for solar cells, liquid crystal displays, light emitting diodes, and touch screens that have been considered as an extraordinary growth recently.
Indium tin oxide (ITO) has been used for many years for most of the transparent electrode applications. 13 However, it is not used in printable electronic applications due to toughness and high cost. To resolve these issues, it is required to replace ITO with carbon nanotubes, 14 –16 graphene, 17 –19 high conductive nanowires films, 20,21 nanorods, 22 nanowires transparent flexible films, 23 and metal nanowires 24 for next-generation wearable 25 electronic materials. Among these materials, silver nanowires (AgNWs) are the most promising materials due to their high electrical conductivity, 26,27 transparency, mechanical flexibility, 28 –31 and excellent optical properties. 32,33 In transparent and flexible electronic applications, AgNWs have shown very limited applications because their short length leads to higher contact resistance with lower electrical conductivity. 34 –36 The performance of AgNWs depends on many factors such as length, diameter, dispersion, surface roughness, and morphology. Recent research has suggested that the length and diameter are the primary factors to achieve excellent electrical conductivity and higher flexibility with higher transmission. 35,37,38 The AgNWs with longer length and smaller diameter can perform a good electrical active network by providing shorter contact resistance between nanowire junctions. 39 Hence, the longer length AgNWs with smaller diameter are good for better performance using a simple solution process method, and it is of great interest and challenge in the transparent and flexible electronic industry. 39
In recent years, many chemical synthesis methods have been reported for the synthesis of AgNWs. 40,41 For the past few years of research, the polyol process is the most significant way to synthesize nanowires using poly(vinylpyrrolidone) (PVP) as capping agent and ethylene glycol (EG) as solvent-reducing agent. 42 During the synthesis of longer length AgNWs, the main factors such as reaction temperature, stirring rate, concentration of chemicals, dropping rate, and ratio of chemicals have affected the high yield of AgNWs. 34,37,38,43,44 Recently, there are few other studies reported that they fabricated longer length AgNWs at a low temperature of 110°C with low stirring speeds, and the observed length was in the range of 20–100 µm with a very wide diameter distribution. 35 Hence, there is a need for suitable synthesis methods of controlling the diameter and length of AgNWs.
The focus of this study was to synthesize longer length AgNWs through a copper (II) chloride (CuCl2)-mediated thermal method by reducing silver nitrate (AgNO3) with EG and using a PVP capping agent. It is also observed that the formation of AgCl colloids in the initial stage plays an important role in the final product of AgNWs. The longer AgNWs were successfully synthesized by controlling the drop rate, temperature, and chemical ratio of AgNO3 to PVP. In this process, uniform AgNWs are grown with a length of about 70 ± 30 µm and diameter of 230 ± 50 nm. The AgNW films were successfully fabricated with approximately 90% transmittance and approximately 0.02 Ω/sq sheet resistance at room temperature, which shows higher performance compared to reported values, to the best our knowledge.
Experimental section
Materials and methods
PVP-25K, EG, cupric chloride, AgNO3 (99.9%), acetone, and ethanol were purchased from Sigma-Aldrich Co. All these chemicals were of analytical grade and were used without any further purification.
AgNWs were synthesized by reducing 0.1 M AgNO3 with EG in the presence of a PVP capping agent. Five milliliter EG solution of 300 µM CuCl2 was prepared followed by stirring and a 5 mL of EG solution of PVP with twice the quantity of 0.1 M AgNO3 was prepared followed by vigorous stirring. The PVP solution was added to CuCl2 solution and then the mixed solution was injected drop by drop (drop rate of 0.03 mL/min) using a syringe into 10 mL of EG solution of AgNO3 (0.1 M), which was set at 150°C in an oil bath. The CuCl2 acted as a mediating agent to facilitate the growth of AgNWs. We believe Cu(II) and Cl− ions are necessary for AgNWs production. Finally, the solution color became milky white and then the solution was cooled to room temperature. AgNWs were easily separated from EG with the addition of a large amount of acetone followed by centrifugation three times at 2000 r/min for 20 min. To separate particle and wires, centrifugation has been done twice by adding ethanol and then the final nanowires were dispersed in ethanol for further characterization. The schematic representation of the synthesis procedure is shown in Figure 1

Schematic of the synthesis of AgNWs. AgNW: silver nanowire.
Characterization
Ultraviolet–visible spectrophotometer (Perkin Elmer, Waltham, Massachusetts, USA) was used to record the absorbance at room temperature with quartz cuvettes and documented transmission of AgNW films on a poly(ethylene terephthalate) (PET) film as well as glass cover slips. Sheet resistance was measured using a four probe method. Transmission electron microscopic (TEM) images were obtained using the Tecnai G2 F30 (Hillsboro, Oregon, USA) at 300 kV. Microstructure of AgNWs was assessed using a field emission scanning electron microscopy (FE-SEM). The evolution of morphology, dimension analysis of AgNWs was captured using atomic force microscopy (AFM, NT-MDT, Russia) at room temperature employing semi-contact mode with a cantilever of force constant from 0.03 N/m to 0.2 N/m and radius of curvature 10 nm.
Results and discussion
The morphology of AgNWs is shown in Figure 2(a). The length and diameter distribution of AgNWs was measured using SEM images. The structure and morphology analyses have also been measured using AFM and optical microscope images (Online Supplementary Figure S1). The average length and diameter of the AgNWs, which were found to be 70 ± 30 μm and 230 ± 50 nm, respectively, were measured by statistical analysis of the normal distribution (Figure 2(b)). The SEM images showed that the diameter distribution is not uniform in the absence of stirring conditions and observed a broader range of diameter (100–300 nm). The lower stirring rate improves the uniform diameter (Online Supplementary Figure S2), but the length of wires becomes shorter. In this case, the AgNWs have successfully synthesized AgNWs with an average length of about 70 ± 30 µm without any stirring process, which is larger than the reported value, to the best of my knowledge, in a single-step process using a small molecular weight PVP-25K capping agent. A temperature of 150°C has been chosen during the synthesis process to synthesize longer length nanowires, and this leads to a wider diameter distribution of AgNWs. The main difference between our synthesis and previous synthesis method 35,37,38 was the use of PVP capping agent and the rate of injection of PVP + CuCl2 solution. Here, a very low-molecular weight PVP-25K capping agent and ultraslow injection rate (0.03 mL/min) of PVP + CuCl2 to mix with AgNO3 solution were chosen. The ultraslow rate process will lead to synthesize even longer length wires with lower molecular weight capping agent PVP and a ratio of AgNO3 to PVP.

(a) SEM images of AgNWs with mediated CuCl2 (a′ to c′). (b) The average distribution of length (a′) and diameter (b′) of AgNWs. SEM: scanning electron microscope; AgNW: silver nanowire; CuCl2: copper (II) chloride.
In general, in the previous AgNWs synthesis process, AgNO3 solution was added drop wise to the PVP + CuCl2 solution, whereas here it is used in reverse process with ultraslower injection rate. The ultraslow injection process will lead to uniform crystal growth of longer nanowires. To synthesize longer length nanowires using polyol process, the main possible factors would be temperature, stirring rate, and a ratio of AgNO3 to PVP.
The HR-TEM image of AgNWs (Figure 3(b)) shows that lattice fringes are related to {111} planes of a face-centered cubic (fcc) phase of Ag. The AgNWs have grown along {011} direction 8 and the lattice spacing 2.11 Å could be indexed as the fcc {111} planes. The corresponding selected area electron diffraction pattern of AgNWs is shown in Figure 3(a), indicating that the AgNWs are crystallized as a single crystal in the fcc five-fold twinned structure. The diffraction spots can be indexed as (111) and the fast Fourier transformation process on the AgNWs would avoid misfit problem caused by the machine and directly reflect the crystalline planes.

TEM images of silver nanostructures with a corresponding electron diffraction pattern (a) and crystalline pattern (b). TEM: transmission electron microscope.
Figure 4 shows the absorption spectra of AgNWs with CuCl2-mediated agent. The peak at 405 nm usually indicates the absorption peak for silver nanoparticles (AgNPs) 45 and the AgNPs slowly grow into larger ones via Ostwald ripening because the corresponding peak becomes broader for AgNWs. The appearance of a peak position at approximately 390 nm could be considered as the optical signature of long AgNWs. 34,38,40 The absorption peak position range of 385–400 nm could be considered as the optical indication for the longer AgNWs. 46,47
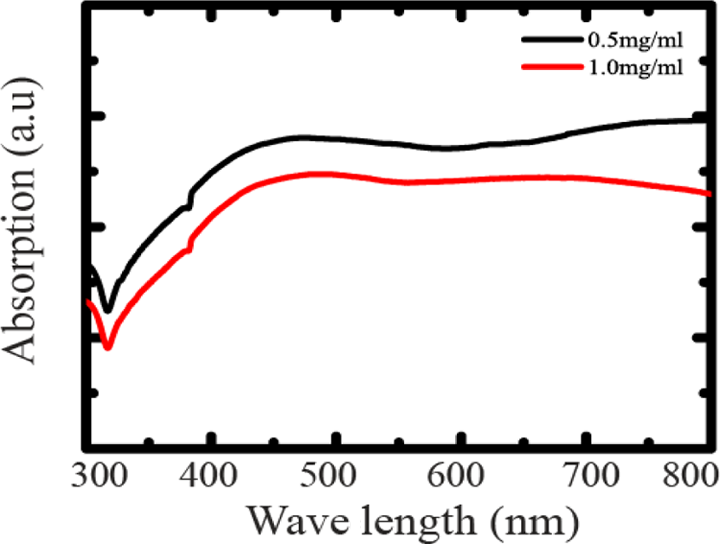
Absorption spectra of AgNWs with CuCl2-mediated agent. AgNW: silver nanowire; CuCl2: copper (II) chloride.
The transparent film was fabricated on a cover slip by drop casting method and followed by annealing at 80°C for 20 min. The AgNWs were deposited on 2 × 2 cm2 cover slip/PET film and the sheet resistance of cover slip/PET was measured by the four probe method. The transmittance of AgNWs films is shown to be 60–90% at 500 nm wavelength for different thicknesses from 150 nm to 800 nm (Figure 5(a)). Figure 5(b) shows the relation between transmittance and sheet resistance of AgNWs after annealing treatment at 80°C for about 15 min. The sheet resistance decreased by 10% after annealing at 80°C. The sheet resistance and transmittance depends on AgNWs distribution (concentration) on the substrate and higher transmittance (approximately 90%) observed at AgNWs concentration (1 mg/mL) with a higher sheet resistance (approximately 0.02 Ω/sq). The film with a sheet resistance of approximately 0.022 Ω/sq observed about 90% optical transmittance. The optical transmittance is about 70% when the sheet resistance increased significantly from 0.008 Ω/sq to 0.02 Ω/sq. A very low sheet resistance of 0.02 Ω/sq was achieved at approximately 90% transmittance for longer length nanowires compared to reported values, to the best my knowledge. We believe that the possible reason is the longer length nanowires with only less number of contact points and form a random network that reduces a contact resistance significantly in the film per unit area compared to the shorter AgNWs.

(a) The transmittance spectra of AgNWs network as the function of wavelength for different thicknesses (150–800nm) and (b) the transmittance versus sheet resistance at 500 nm wavelength of AgNWs network. AgNW: silver nanowire.
Conclusion
In summary, longer AgNWs have been synthesized successfully with a low molecular length PVP-25K capping agent, ultraslow injection rate of 0.03 mL/min, and without any stirring using a novel polyol method. The sheet resistance achieved approxiamtely 0.02 Ω/sq with approxiamtely 90% transmittance after annealing treatment. The future work should include fabrication of various thicknesses of flexible thin films and testing for electrode activity. The highly transparent and metal conducting films can be more useful for future transparent and flexible electronics industrial applications.
Footnotes
Acknowledgments
The author would like to acknowledge Dr Jaydeep Kumar Basu, Department of Physics, Indian Institute of Science, Bangalore, Karnataka, India, for his fruitful discussion and use of his laboratory facilities. The author would also acknowledge Mr S R K Chaitanya Indukuri for helping in atomic force microscopy imaging and Dr Aveek Bid for using his laboratory facilities for electrical conductivity measurements. The author thank the Department of Science and Technology (Nanomission), India, for financial support and the Advanced Facility for Microscopy and Microanalysis, Indian Institute of Science, Bangalore, India, for providing access to transmission electron microscopic and scanning electron microscopic measurements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
