Abstract
Owing to its high specific surface area, high thermal stability and excellent thermal insulation, silica aerogel is used as an insulating material in protective clothing. In this study, the hydrophobicity and thermal insulation properties of the electrospun polyacrylonitrile (PAN) nanofibers were enhanced by integrating silica aerogel (SA), which was synthesized using silicic acid as silica precursor instead of silicon alkoxides or sodium silicate and modified with phenyltrimethoxysilane. The electrospun PAN-SA composite nanofibers showed higher surface roughness and hydrophobicity (water contact angle of 142°) compared to the smooth and hydrophilic PAN (control) nanofiber. Further, the thermal stability of the PAN nanofibers was increased by 17 °C while their thermal conductivity was decreased from 15.8 W/mK to 0.4 W/mK (reduction of 97.5%). These features make the PAN-SA composite nanofibers particularly suitable for use in thermal insulation protective clothing.
Introduction
Protective clothing that protects the human body from external threats such as bullets, chemical and biological agents, fire, cold and heat has been widely used in military, sports and industrial applications [1]. Aerogels with unique properties such as low density, low thermal conductivity, high specific surface area, and excellent thermal insulation have been used in many applications such as construction, aerospace, and defense, as well as protective clothing [2,3]. Due to its superior insulation, thermal stability and flameproof properties, silica-based aerogels are a potential candidate as insulation material, especially for thermal insulation protective clothing [4].
Silica aerogel (SA) production includes three general steps: gel preparation, aging and drying. Gel formation is based on the sol-gel reaction consisting of a silica source solution (precursor) and a catalyst. The silicon alkoxides derivatives (tetraethoxysilane, TEOS; tetramethoxysilane, TMOS; polyethoxydisiloxane, PEDS; methyltriethoxysilane, MTES; methyltrimethoxysilane, MTMS; etc.) and water glass (sodium silicate) solution have been used by many researchers as silica precursors [5–9]. Hydrolysis of the silica precursor is performed with an acid or a base catalyst. The aging strengthens the gel; hence shrinkage is minimized during the drying step. Drying can be carried out under ambient pressure (APD) or critical pressure (supercritical drying, SCD) conditions. Freeze drying method (FD) is also used to dry the gel [10,11].
Nanofibers are fibrous materials with very small diameters compared to microfibers and can be produced by various types of techniques like drawing, template synthesis, phase separation, self-assembly and electrospinning. Among these, electrospinning is the simplest and most effective way using a high voltage source, pump, needle and collector [12,13]. Because of their high surface area, high tensile strength and flexibility, electrospun nanofibers are suitable materials for protective clothing applications [14,15].
Polyacrylonitrile (PAN) is one of the popular polymers owing to its high dielectric constant that helps to produce ultrafine nanofibers [16]. PAN nanofibers has high chemical resistance, high thermal stability and high mechanical features due to the strong intermolecular interactions of adjacent –C≡N groups, which also allows PAN to have a high melting point (317 °C) [17–19]. Recently, several studies have been reported on PAN-SA composite nanofibers used in various application areas. In one of these studies, Lee et al. prepared PAN-SA composites for thermal insulation. They used single-needle and co-axial electrospinning to increase the fraction of aerogel particles in the composite sheets, and therefore determined the weight ratio of PAN:SA as 1:0.5 and 1:1 for single-needle and co-axial electrospinning, respectively. They observed that when the aerogel content doubled, the average thermal conductivity of the composite decreased by about 12.5% [15]. Yanilmaz et al. fabricated the silica/PAN hybrid nanofiber membranes by using sol-gel and electrospinning methods to utilize as separators in lithium-ion batteries [20]. In another work, Wang et al. presented the fabrication of multilevel structured filtration membrane by stacking up bimodal sized and silica nanoparticles incorporated electrospun PAN nanofibers [21]. Zhao et al. also produced the electrospun PAN/silica-based fibrous composite membranes and investigated the electrochemical properties of the membranes for use as dye-sensitized solar cells [22]. Besides, Pirzada et al. synthesized PAN-silica hybrid fibers through sol-gel electrospinning by using TEOS as a silica precursor. A single-needle electrospinning was applied to the solutions containing the silica/PAN weight ratio in the range of 3.5:5, 9.3:5, and 14:5. They found that the intermolecular interaction between nitrile group of PAN and surface silanols of silica caused disappearance of glass transition and a broad thermal degradation zone for the hybrids with larger silica content [23].
This study covers the production of electrospun PAN-SA composite nanofibers and their morphological, wetting and thermal properties. The silica aerogel was synthesized by sol-gel reaction using a commercial silicic acid as silica precursor instead of silicon alkoxides or sodium silicate, which is commonly used in the literature. Thus, hydrolysis of the silica precursor with a catalyst in the gel-formation step was eliminated. The aerogel was modified with phenyltrimethoxysilane (silylating agent) to increase the hydrophobicity of surface, and was characterized by FTIR, BET and thermal conductivity analyses. Single-needle electrospinning was performed with solutions containing different amounts of the aerogel. The weight ratio of PAN:SA in the electrospinning solutions was adjusted as 1:0.09, 1:0.21, 1:0.36, 1:0.56, and 1:0.83. The structural, morphological, wetting and thermal properties of the composite nanofibers were examined, and the results were presented in detail.
Experimental
Materials
Commercial silicic acid (TANA®FINISH 40119) used as the silica precursor was supplied by Spot Kimya, Turkey. Phenyltrimethoxysilane (commercially called Dow Corning® Z-6124 Silane, referred to as Z-6124) used as the silylating agent was purchased from Dow Corning Co., USA. Other chemicals including ethanol (EtOH, absolute), n-hexane, ammonium hydroxide (NH4OH, 25%), N,N-Dimethylformamid (DMF), polyacrylonitrile (PAN, molecular weight 150 kg/mole) were purchased from Sigma-Aldrich, USA.
Silica aerogel synthesis
Hydrophobic silica aerogels were synthesized by the sol-gel process, followed by solvent exchange, surface modification with phenyltrimethoxysilane, and ambient pressure drying (APD), as shown in Figure 1(a). Briefly, 50 mL of silicic acid was added dropwise to 150 mL of 1 M NH4OH until the solution had a pH of ∼10. Silicic acid polymerization, which means gel formation, began with the contact of the droplets with the alkaline ammonia solution. After gently stirring for 30 minutes, silica gels were aged with ethanol (200 mL) for 2 h at 40 °C. Solvent exchange with n-hexane was carried out with vigorous stirring for 5 h at room temperature. The silica gels were then collected by centrifugation and again vigorously stirred in n-hexane (200 mL) for 5 h at room temperature. Surface modification was performed in a mixture of n-hexane (100 mL) and Z-6124 (10 mL); where the silica gels were gently stirred for 2 hours at room temperature. The modification here is based on the alcohol condensation reaction between the hydroxyl groups

(a) Schematic diagram of experimental procedures for the synthesis of silica aerogel. (b) Chemical reaction between silica gels and silylating agent (Z-6124).
(–OH) of silica gels and the methoxy groups (–OCH3) of the silylating agent, as given Figure 1(b). The modified gels were then washed with n-hexane excess to remove unreacted silylating agent and by-product methanol, and dried at 150 °C for 30 minutes to obtain aerogels, then allowed to dry at room temperature for characterization [24].
Nanofiber production
Single-needle electrospinning technique was used for the nanofiber production. 6 wt.% of PAN solution and 5 wt.% of silica aerogel dispersion (SA) were prepared by stirring in DMF for 24 h at room temperature. The electrospinning solutions containing PAN-SA mixtures in the volume ratios of 90/10, 80/20, 70/30, 60/40 and 50/50 were prepared by ultra-sonication for 4 h at room temperature, the solutions contents are given in Table 1. Electrospinning was performed for 1 h at room temperature and 60–75% relative humidity, using a voltage in the range of 6 – 12 kV, a tip-to-collector distance of 15 cm, and a flow rate of 1.5 mL/h. The electrospun nanofibers were expressed as PAN (control), PAN-SA10, PAN-SA20, PAN-SA30, PAN-SA40 and PAN-SA50 according to the volume of SA in the electrospinning solutions. Since the aerogel affected the physical properties of solutions such as viscosity, conductivity and surface tension, the electrospinning ability of the solutions changed; therefore different voltages were applied to obtain nanofiber. The morphological properties of nanofibers produced at different voltages were examined by microscopic analysis; the details are given in the relevant section (see the Morphological properties of the nanofibers section).
Content of electrospinning solutions, applied voltages and diameters of electrospun nanofibers.

Characterization
The bulk density of the silica aerogel was calculated from its mass to volume ratio. The mass of the aerogel was measured by an electronic balance with 0.00001 mg accuracy (Precisa XR 125SM). The volume was calculated by filling a graduated cylinder with the synthesized silica aerogel. The bulk density calculation was repeated 5 times, where the average value was reported. The specific surface area, pore size distributions (PSD), average pore diameters, and N2 sorption isotherms of the aerogel were determined by the Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods, based on the amount of N2 adsorbed at several different relative pressures (0.05 – 0.3) were taken at 77 K. (ASAP 2020, Micromeritics, USA). The pore size distribution and pore volume were calculated from the desorption isotherm [25].
The structural analysis of the silica aerogel was carried out by the FTIR measurement with Perkin Elmer Spectrum 100 FTIR spectrometer in attenuated total reflection (ATR) accessory. Infrared spectra were collected between 4000 and 650 cm−1 at a resolution of 4 cm−1. FTIR analysis was also used to characterize the electrospun PAN-SA composite nanofibers.
Morphologic analysis of the obtained composite nanofibers was investigated by scanning electron microscopy (SEM) (VEGA3 TESCAN). Before the SEM analysis, the samples were covered with Au nano-powder under a current of 20 mA for a minute. Thermal stability of PAN-SA nanofibers were tested using TG/DTA analyzer (SEIKO EXSTAR 6200) with a heating rate of 10 °C/min from room temperature to 750 °C in nitrogen atmosphere.
Wetting behavior of the nanofibers was evaluated by measurement of the contact angle between a water droplet and surface using Theta Lite Optical Tensiometer TL 101 according to BS EN 828:2013 standard. For each sample, five droplets of water with a volume of 6 µl were left at different positions on the sample surface, and both sides of the droplets were measured using the Sessile Drop method within five seconds. The average values of the measured contact angles were reported with standard deviations.
Thermal conductivity analyzer (TCi) was used to measure thermal conductivity of produced silica aerogel powder since it allows the thermal conductivity measurement in powder form. Alambeta instrument (Sensora, Czech Republic) was used to determine the thermal conductivity of the electrospun PAN-SA composite nanofibers. The averages of five measurements for each sample were calculated, and the mean values of the thermal conductivities were reported.
Results and discussions
Structural and textural properties of silica aerogel
Figure 2 presents the FTIR spectra of silylating agent (Z-6124), unmodified and modified silica aerogels. The unmodified silica aerogel displays a broad absorption band at 3422 cm−1 and a weak vibrational band at 1640 cm−1, corresponding to the –OH stretching of silanol groups [26,27]. The strong vibrational band at 1080 cm−1 corresponds to the characteristic asymmetric stretching of siloxane groups (Si–O–Si) [28–30].
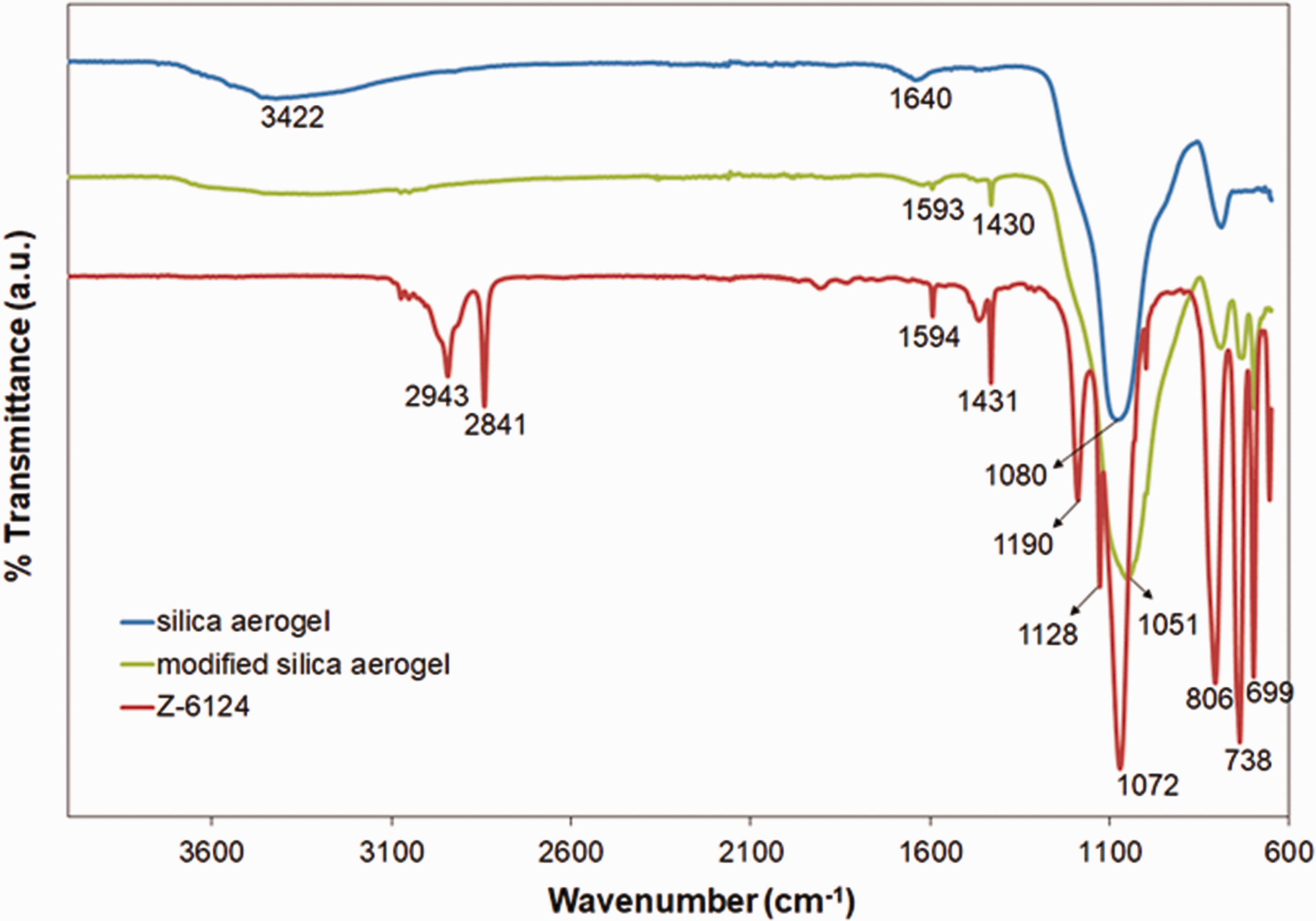
FTIR spectra of the Z-6124, unmodified and modified silica aerogels.
The silylating agent Z-6124 (phenyltrimetoxysilane, the chemical formula (CH3O)3–Si–C6H5) shows asymmetric and symmetric vibrational bands of the methyl group (–CH3) at 2943 and 2841 cm−1, respectively [31]. The vibrations detected at 1594 and 1431 cm−1 correspond to C = C stretching of aromatic benzene ring, while the in-plane and out of plane C–H deformation vibrations in the benzene ring are observed at 806, 738 and 699 cm−1, respectively [32,33]. Further, the vibrational bands at 1128 and 1072 cm−1 are assigned to the Si–O stretching of silane group [34].
The modified silica aerogel exhibits a weak –OH stretching at 3422 compared to the unmodified. In addition, the vibrations at 1593, 1430, 738 and 699 cm−1 are matching with the silylating agent. These results reveal that Z-6124 has been successfully grafted to silica aerogels surfaces and replaces –OH groups on the surfaces.
The N2 gas adsorption-desorption isotherms and PSD curve of the modified silica aerogel are shown in Figure 3. Bulk density, porosity, BET surface area, pore diameter, and pore volume of the aerogel are reported in Table 2. The N2-sorption isotherm of the aerogel exhibits type-IV isotherm with H3 hysteresis loop corresponding to the aggregates of plate-like particles forming slit-shaped pores according to IUPAC classification [35,36], which indicates that the aerogel possesses the typical features of mesoporous material. Further, the aerogel has a broad pore size distribution with an average diameter of 20.05 nm as listed in Table 2. In spite of having a high pore diameter, the BET surface area and the pore volume of the aerogel, 105.62 m2 g−1 and 0.47 cm3 g−1, respectively, remained low values.

N2-sorption isotherms and PSD curve of the modified silica aerogel.
Physical properties of the modified silica aerogel.

FTIR analysis of the nanofibers
The FTIR spectra of the electrospun PAN-SA composite nanofibers are given in Figure 4. As the control group, PAN nanofiber displays the typical vibrations at 2926, 2243, and 1452 cm−1 corresponding to –CH2 asymmetric stretching, –C≡N stretching, and –CH2 scissor vibration, respectively [37]. On the other hand, the PAN-SA composite nanofibers show strong peaks around 1109 cm−1 corresponding to the characteristic asymmetric stretching of siloxane groups, compared to the PAN (control) nanofiber. This result confirms the presence of the silica aerogel in the composite nanofibers.
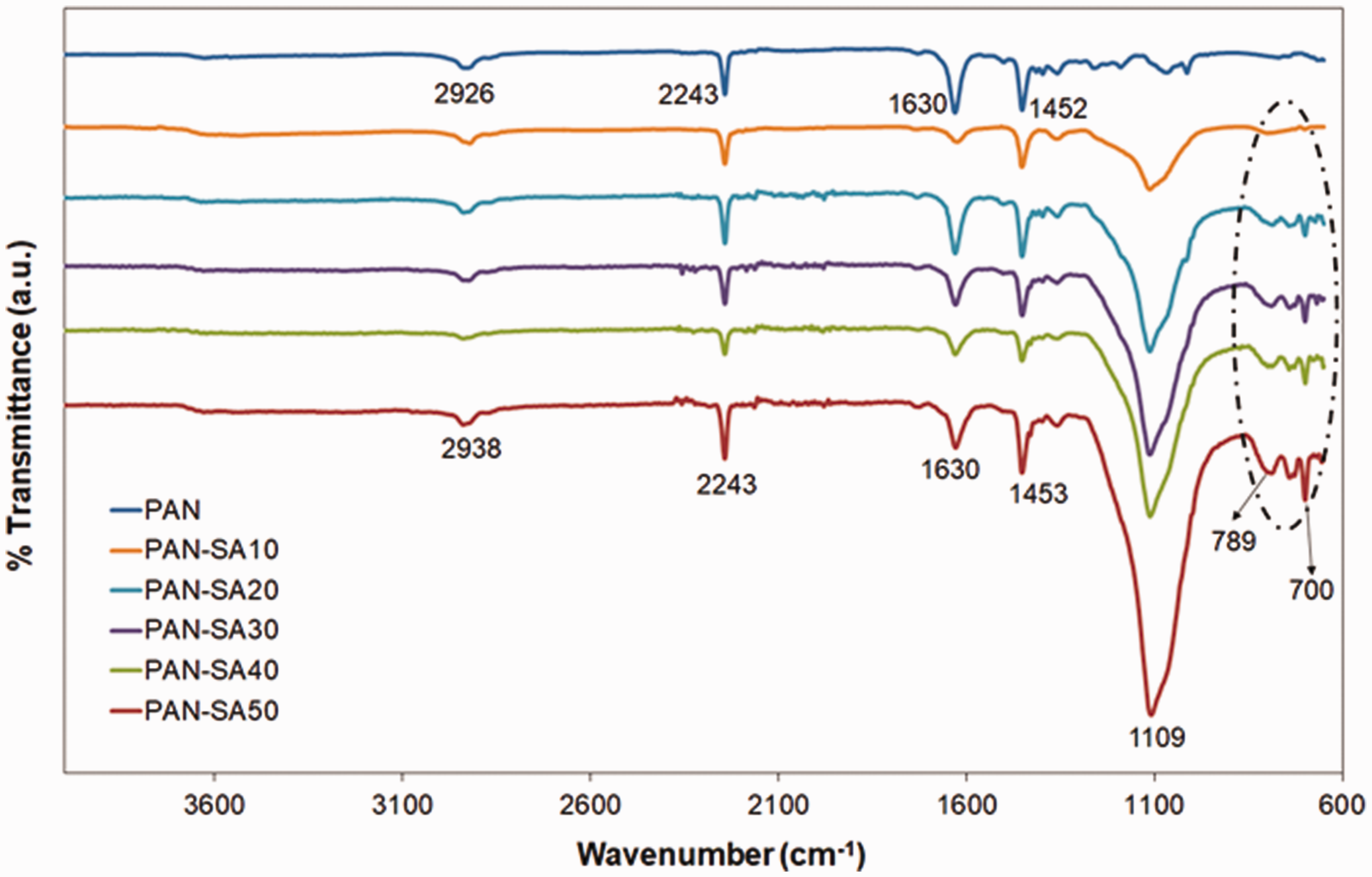
FTIR spectra of PAN (control) and PAN-SA composite nanofibers.
Morphological properties of the nanofibers
The SEM images of the PAN nanofibers with fiber diameter distributions are given in Figure 5. The fiber diameters were calculated by measuring 50 nanofibers randomly selected in SEM images using Image J software for each sample, and the average values are given in Table 1. Nanofiber production could not be carried out at a constant voltage because the electrospinning ability of the solutions changed due to the increased silica aerogel content as well as the reduced amount of polymer. For this reason, the voltages ranging from 6 to 12 kV were applied, as seen in Table 1. On the other hand, since nanofibers with different diameters are produced depending on the applied voltage, the effect of the silica content on the diameters has not been fully investigated. For instance, when a voltage of 12 kV was applied to the 6% by weight of PAN solution, uniform fibers with an average diameter of 538 nm could be produced. Based on the decrease in their viscosities owing to reducing of polymer contents, the electrospinning solutions containing 10% and 20% the aerogel dispersion by volume could be fibered with average diameters of 623 and 506 nm, respectively, by applying a voltage of 7 kV. Similarly, the electrospinning solutions containing 30, 40 and 50% silica aerogel dispersion by volume could be fibered by applying a voltage of 6 kV, and the average fiber diameters were determined as 597, 407 and 538 nm, respectively. Considering fiber sizes, it can be assumed that the diameter of nanofibers produced by applying the same voltage decreases as the silica aerogel content increases. This trend has been observed in the PAN-SA10/PAN-SA20 and PAN-SA30/PAN-SA50 nanofibers (as an exception, the PAN-SA40 has the lowest fiber diameter). Here, reducing the fiber diameters due to silica aerogels can be attributed to the repulsive forces between the aerogels and polymer chains, which change the physical properties of solution and diminish the entanglement of polymer chains during electrospinning [20]. In addition, the SEM images show that the surface roughness and bead formation rise with increasing the silica aerogel content compared to the PAN (control) nanofiber.
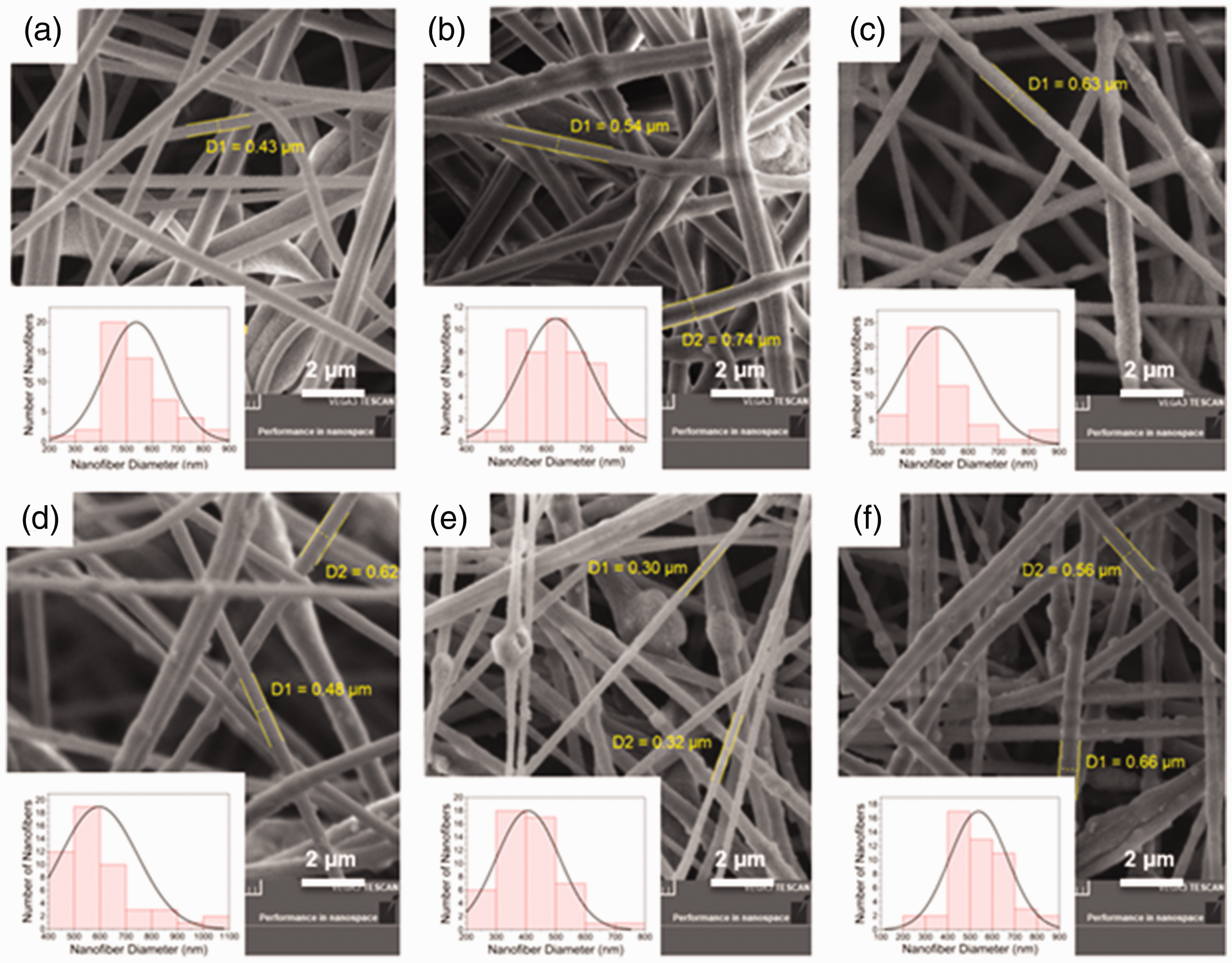
SEM images of electrospun nanofibers of; (a) PAN(control), (b) PAN-SA10, (c) PAN-SA20, (d) PAN-SA30, (e) PAN-SA40, and (f) PAN-SA50.
Wetting properties of the nanofibers
The wetting properties of the PAN-SA composite nanofibers were examined by measuring the contact angles of the water drop on the fibers. Figures 6 and 7 present the average contact angles and contact angle images of the nanofibers, respectively. The PAN (control) nanofiber exhibits a completely hydrophilic character with a contact angle of 0° due to absorbing the water drop within 3 seconds. As seen in the figures, the hydrophobicity of the composite nanofibers improves with increasing the silica aerogel contents. The average water contact angle of the PAN-SA10, which has the lowest silica aerogel content, is 65°, while the PAN-SA50 with the highest silica aerogel content has the highest water contact angle (142°) among the nanofibers. Further, this result confirms that the hydroxyl groups on the silica aerogel surface have been successfully silylated with Z-6124 thanks to the modification.

The average water contact angles of the electrospun PAN-SA nanofibers.

The water contact angle images of; (a) PAN-SA10, (b) PAN-SA20, (c) PAN-SA30, (d) PAN-SA40, and (e) PAN-SA50 nanofibers.
Thermal properties of the nanofibers
The thermal stability of the electrospun PAN (control) and PAN-SA composite nanofibers was examined by thermogravimetric analysis. Figure 8(a) and (b) show the TG (weight loss) and DTG (derivative weight loss) curves of the nanofibers, respectively.

(a) TG and b) DTG curves of electrospun PAN (control) and PAN-SA composite nanofibers.
The PAN (control) nanofibers undergo thermal decomposition in two stages. The first stage occurs in the temperature range of 60 – 130 °C, which corresponds to 6.6% weight loss due to moisture evaporation [38]. In the second decomposition stage, the main weight loss due to pyrolysis of acrylonitrile polymer chains begins at 290 °C, which corresponds to the DTG peak at 304 °C.
A similar decomposition process is observed for the PAN-SA composite nanofibers. However, with the increase in the amount of silica aerogel, the weight loss due to the evaporation of moisture in the first stage decreases up to 3.2%, while the DTG peak shifts from 90 °C to 122 °C. In addition, the silica aerogel retards the initial degradation temperature of the composite nanofibers to 302 °C in the second decomposition stage, while the DTG peak rises from 304 °C to 321 °C compared to the PAN (control) nanofiber. The char residues at 750 °C increase randomly with the addition of silica aerogel regardless of quantity. As a result, the modified silica aerogel removes the moisture among nanofibers due to its hydrophobic property and increase the thermal stability of PAN nanofibers by 17 °C.
The heat transfer process through porous media consists of thermal conduction along the solid matrix, thermal convection of gases filling the pores, and thermal radiation across internal pores [39,40]. Since it has more than porosity of 90%, silica aerogel shows excellent insulation performance. Thermal convection can be completely ignored because the pores in silica aerogel and its composites are in micrometer or nanometer size. On the other hand, while the thermal radiation heat transfer becomes an important heat transfer mechanism for the porous matrices, it can also be considered as the main heat transfer mechanism for fibrous insulation structures such as fiber/aerogel composites. The fiber/aerogel composite materials act as an effective insulator due to the synergistic effect of fibers and aerogels, since the aerogels suppress the heat transfer through conduction and convection, while the fibers reduce the thermal radiation. The optical and physical (size, shape, density etc.) properties of the fibers affect the scattering and absorption parameters of thermal radiation. Some previous studies have emphasized that submicron fibers have a reducing effect on the thermal radiation conductivity [15,39,41].
In this study, although the electrospun PAN nanofibers have diameters in submicron; the thermal conductivities of the fibers vary significantly owing to their silica aerogel contents. The thermal conductivities of the PAN (control) nanofiber and the modified silica aerogel were determined as 15.8 and 0.037 W/mK, respectively, as presented in Figure 9. The PAN-SA10 nanofiber with the lowest silica aerogel content (PAN:SA weight ratio of 1:0.09) has a thermal conductivity of 10.6 W/mK, which is approximately 33% lower than the PAN (control) nanofiber. With the increase of silica aerogel content, the thermal conductivities of PAN-SA20, PAN-SA30 and PAN-SA40 nanofibers decrease to 3.5, 3.3, and 1.5 W/mK, respectively (78–90% reduction in thermal conductivity). Further, the PAN-SA50 nanofiber with the highest silica aerogel content (PAN:SA weight ratio of 1:0.83) have a thermal conductivity of 0.4 W/mK, reduced by nearly 97.5% compared to the PAN (control) nanofiber. Thanks to its high porosity and surface area, the modified silica aerogels reduce the density of the electrospun PAN-SA composite nanofibers while preventing free air flow through the fibers. Therefore, although the sizes of the electrospun PAN-SA composite nanofibers are almost similar; the modified silica aerogels show exceptional insulation performance with increasing amounts in the polymer matrix.

The thermal conductivities of PAN-SA nanofibers and modified silica aerogel.
Conclusions
The current study reports the morphological, wetting and thermal properties of the electrospun PAN-SA composite nanofibers. The silica aerogel was synthesized by sol-gel method using a commercial silicic acid precursor and then modified with phenyltrimethoxysilane (silylating agent, Z-6124) through alcohol condensation reaction. The modified aerogel was characterized by FTIR, N2 adsorption-desorption isotherms and thermal conductivity. The FTIR analysis confirmed that the aerogel surfaces were successfully silylated, while the N2-sorption isotherms showed that the aerogels were a typical mesoporous material. The average pore diameter, BET surface area, pore volume, and thermal conductivity of the modified silica aerogel were determined as 20.05 nm, 105.62 m2/g, 0.47 cm3/g, and 0.037 W/mK, respectively.
The single-needle electrospinning technique was used to produce the PAN-SA composite nanofibers. The electrospinning solutions were prepared by mixing 6 wt.% PAN solution (in DMF) and 5 wt.% of SA dispersion (in DMF) in the volume ratios of 90:10, 80:20, 70:30, 60:40 and 50:50. The PAN:SA weight ratios of these mixtures were 1:0.09, 1:0.21, 1:0.36, 1:0.56, and 1:0.83, respectively. The electrospun nanofibers were produced in diameters between 500 – 600 nm, and the surface roughness of the nanofibers increased as the aerogel content increased. The wettability properties were examined by measuring the water contact angle, and it was clearly seen that the hydrophobicity increased with the increased silica aerogel content in the nanofiber structures. The highest contact angle (θ = 142°) was observed for the PAN-SA50 nanofiber, which has the highest silica aerogel content (PAN:SA weight ratio of 1:0.83) among the samples. Moreover, the thermal stability of the PAN-SA50 nanofiber enhanced by 17 °C, while the thermal conductivity reduced to 0.4 W/mK, which was about 97.5% lower than the PAN (control) nanofiber.
Thanks to their hydrophobicity, high thermal stability and high thermal insulation efficiency, the electrospun PAN-SA composite nanofibers produced with this new approach can be promising candidates for thermal insulation protective clothing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
