Abstract
In this study simple, facile and highly active silver coated ZnO-chitosan (Ag/ZnO-CH) textile cotton supported nanocomposites were developed. The nanocomposites were characterized by UV-visible spectroscopy, scanning electron microscopy (SEM), energy dispersive X-rays spectroscopy (EDX), fourier transform infrared spectroscopy (FTIR) and X-rays diffraction (XRD). The prepared nanocomposite fibers were used for the selective removal of seven model pollutants, including para-nitrophenol (p-NP), meta-nitrophenol (m-NP), ortho-nitrophenol (o-NP), 2,4,6-trinitrophenol (TNP), and dyes of methyl orange (MO), congo red (CR), and methyl red (MR). The apparent rate constant (Kapp) of pseudo first order kinetic for p-NP was 2.813 × 10−3 s−1 and 1.663 × 10−3 s−1 for MO dye. Among the different nitroarenes, the reaction rate matched the ordered of p-NP >TNP > m-NP > o-NP, while for the dyes it was MO > CR > MR. Ag/ZnO-CH nanocomposites were recycled multiple times without any significant loss of its catalytic activity. The higher stability of the Ag/ZnO-CH nanofibers also allows the catalyst to be separated easily by just pulling the catalyst from the reaction mixture and reused. The clean and facile, simple synthesis procedure, outstanding properties and low-cost supports allow these catalysts to be used in the reduction of the organic pollutants individually as well as collectively in the mixture of dyes in wastewater at room temperature.
Introduction
Metal nanoparticles due to their specificity, interaction and higher chemical reactivity as compared to their bulk state, receive more attention. Nanoparticles have a large surface to volume ratio that makes nanoscale materials attractive to a large number of applications. Due to their electron-donating tendency, zerovalent metal nanoparticles (MNPs) are considered to be a powerful reducing agent [1]. Nanoparticles provide extremely active centers, but due to their extra-large surface area and high energy; they are unstable at nano size and agglomerates or oxidize in air easily. This leads to inactivation of both reactive and adsorption sites of nanoparticles, leading to a reduction in contact between MNPs and target molecules [2]. To achieve this goal, a number of approaches have been adopted. Over the past decade immobilizations of nanoparticles on heterogeneous support have gained considerable attention. Recently the support materials used for the zerovalent MNPs include boron nitride [3], biochar [4], graphene [2], MgO and zeolites [5], chelating resin [6], plant powder [7,8], and chitosan [9].
Due to its broad accessibility, non-toxic, low cost, biodegradability and distinctive structural property, chitosan is mostly chosen as support polymer for pure MNPs. The presence of active amino and hydroxyl groups in the molecular structure of chitosan allows multiple metal ions to be attached. The sorption of metal ions includes electrostatic and chemical interaction with amine and hydroxyl groups of chitosan. But due to poor dissolution of chitosan in water and weak mechanical strength make it unable as a support to adsorb a large number of metal ions [10,11]. Thus, to enhance the chitosan efficiency for MNPs as a support material; alteration in their physical and chemical properties are essential. Different methodology has been used for the modification; however, the coating of chitosan is a new approach. Coating enhances mechanical and chemical properties of chitosan by mixing appropriate and readily available materials. ZnO nanoparticles are of best choice for coating as compared to other metal oxide due to their ability to improve mechanical properties of chitosan as well as to enhance its chemical stability, biocompatibility, antibacterial and photocatalytic properties. ZnO has been used extensively in photocatalysis [12], water splitting [13], environmental remediation [8], and biologically as an anticancer [14], antifungal, and antibacterial agent [15]. ZnO nanoparticles due to high surface area, possibly adsorb positive metal ions effectively in the wastewater. ZnO/chitosan nanocomposites possess good antimicrobial activities as well as high mechanical strength [16]. The problems with the use of ZnO/chitosan composites are their dispersion, reusability and recyclability. In many applications the catalysts are usually embedded in a solid matrix/support, which allows them multiple or even permanent use. To cover this issue, different type of materials such as cellulose microfibers [17], cellulose filter paper [18], or clay mineral montmorillonite [10] have been used to enhance the reusability of nanocomposites for different applications.
Common carrier materials are polymeric or mineral in nature. However, their production and charging with the catalysts are often complex and costly. Here in this study, we used textile cotton fibers as a support for the ZnO/chitosan nanocomposites due to their cost-effective and abundant availability.
Household and textile wastewater contain a number of pollutants and toxic substances, including dyes, nitroarenes and organic loads, which are non-biodegradable and represent a risk to ecosystems and human health. Such organic contaminants can be detected in both surface and groundwater wells. A number of aromatic organic compounds, especially synthetic dyes are highly toxic and carcinogenic, i.e., causing cancer, malformation, nephritis and a number of other diseases [19]. Phenol and its nitro compounds are listed as main concern contaminants by the United State Environmental Protection Agency (US-EPA) [20]. Nitroaromatic compounds are toxic to aquatic plants and animals. In addition to the issue of odor, nitrophenols also induce colour problems in water bodies to some extent and thus become sources of pollution by reducing the sunlight by causing a disturbance in aquatic life [21]. Tri-nitrophenol or picric acid is widely used in various industries as a reagent for the research and clinical chemistry and also for the military purpose. Tri-nitrophenol exposure is explosive because of its sensitivity to shock, heat, and friction [22]. However, it is difficult to degrade dinitroaromatic and particularly trinitroaromatic substances due to its electron withdrawing property. Mostly nitroaromatic compounds are non-biodegradable due to poor electron density, making the electrophilic attack difficult on the ring [23].
Besides nitroarenes, azo dyes due to chemical stability, versatility, color and non-biodegradability cause water pollution when released to the outdoor environment. Some azo dyes are extremely hazardous, mutagenic and carcinogenic. MO dye is a part of organic monoazo dyes that are hazardous by nature and are frequently used in the leather, paper and pulp, foodstuffs, and textile industries. MO dye is commonly used in research laboratories as a pH measure and are usually released to environment without further treatment. Due to the complicated aromatic chemical structure and xenobiotic properties, MO is not biodegradable [24,25]. Benzidine-based anionic diazo CR is very toxic due to the production of well-known carcinogen benzidine upon its metabolism. It has been reported that carcinogenic CR metabolite benzidine causes urinary cancer in humans. Due to severe health concerns, CR has now been banned by several countries, but it’s still commonly used in different countries [26,27]. Azo dye MR is a highly persistent for marine life. MR induces of the skin, hair and eye sensitization, and when breathed or ingested causes inflammation of the intestinal tract [28]. Pure metal NPs catalyze the reduction and degradation of organic pollutant molecules such as nitrophenols and non-biodegradable azo dyes in wastewater due to their high surface reactivity, high surface area, stability and comparatively low cost [29–34].
In this study a simple and efficient nanocomposites were developed for the support of silver nanoparticles. This composite composed of silver coated ZnO-chitosan embedded in the textile cotton fibers. The prepared silver loaded chitosan ZnO fiber composites were used for the selective removal of p-NP, m-NP, o-NP, TNP, MO, CR, and MR.
Experimental
Chemicals and reagents
All reagents and chemicals used were of analytical grade. Sodium borohydride (NaBH4), zinc nitrate hexahydrate (Zn (NO3)2.6H2O) and silver nitrate (AgNO3) were purchased from Daejung Korea. High molecular weight of 800–2000 cP, yellowish chitosan powder of Sigma Germany was used with deacetylation degree were >75%. The required organic reagents p-NP, m-NP, o-NP, TNP, MR, CR, and MO were purchased from the BDH Chemicals, England. Acetic acid was purchased from the NTN UK Ltd. Double distilled water (DDW) was used throughout the experiment.
Synthesis of ZnO nanoparticles
For the preparation of ZnO nanoparticles, 0.05 M zinc nitrate hexahydrate and 0.05 M sodium borohydride were prepared in 1000 mL of DDW. For the synthesis, 200 mL of zinc nitrate solution in 500 mL round bottom flask was taken and added 10 mL of the chitosan solution (2 weight% of chitosan solution in 2% v/v aqueous solution of acetic acid) as a capping agent and stirred on the magnetic stirrer at 50 °C. After 20 minutes, 200 mL of sodium borohydride solution was added into a reaction flask as reducing agent. With the addition of sodium borohydride, appearance of white precipitate started which indicates the formation of nano ZnO. The precipitate was collected by centrifugation at 4500 rpm for 12 min. After centrifugation, the collected pellets of ZnO washed twice with DDW and dried for 24 hours at 80 °C. The dried pellets were collected and used for analyses.
Synthesis of cotton chitosan-zinc oxide (ZnO-CH) composites
Chitosan solution was prepared by dissolving 2% of chitosan powder in 2% v/v aqueous solution of acetic acid and stir for 1 hour. For the synthesis of chitosan-ZnO composite, 1 gm of ZnO nanoparticles was dispersed in 50 mL of chitosan solution by stirring for 30 min. The composite of ZnO-CH cotton fibers was prepared by dipping 0.5 g of cotton fiber in 50 mL of ZnO-CH solution for 5 hours. After 5 hours the cotton fibers were taken out of the solution and dried at room temperature. The dried Zn-CH cotton fibers were treated with 0.2 M NaOH solution, and then washed three times with DDW and dried.
Synthesis of Ag coated ZnO-CH cotton fibres nanocomposites
The prepared ZnO-CH fibers composites were further used as a support material for the synthesis of silver supported nanocomposites. Firstly, 0.1 M of silver nitrate solution was prepared in DDW. For silver ion uptake, 0.1 g of dried ZnO-CH fibers was put in 100 ml of 0.1 M silver nitrate solution for six hours to saturate the adsorption site. The change in color was observed by putting the ZnO-CH fibers in silver nitrate solution. The color of ZnO-CH fibers becomes brownish due to the adsorption of Ag+ ions. The silver nitrate treated ZnO-CH fibers were collected, and then dried at room temperature.
For the synthesis of Ag loaded catalyst, 10 mg of Ag+ loaded ZnO-CH fibers were treated with 10 mL of freshly prepared 0.5 M sodium borohydride. The Ag+ loaded ZnO-CH composite fibers were reduced to the Ag/ZnO-CH nanocomposites in the presence of sodium borohydride as a reducing agent. These fibers were remaining in a solution of sodium borohydride for 1 hour, till the complete reduction of Ag+ into Ag(0) nanoparticles as shown in the equation (1)
Thus, zerovalent AgNPs (M0) templates on ZnO-CH cotton fibers were prepared by treatment with sodium borohydride, where clusters of silver ions were reduced to Ag nanoparticles.
Due to electrostatic interaction of Ag+ ions with amine and hydroxyl groups of chitosan chains, the silver ions are adsorbed on the composite surface of ZnO-CH and as already stated, that ZnO nanoparticles in ZnO-CH increased the surface area for adsorption of silver ions. The standard reduction potential of sodium borohydride in aqueous system is −1.33 V and capable to easily reduce Ag+ (0.799 V) to Ag(0).
Catalytic reduction study
The synthesized Ag/ZnO-CH composite fibers were investigated for the catalytic reduction of different organic pollutants in the presence of reducing agent sodium borohydride. For the catalytic reduction of NP, 1 mM aqueous solution was used, while for the TNP, 0.1 mM solution was used.
For the reaction experiments, 3 mL of nitroarenes solution (o-NP, m-NP, p-NP and TNP) were added into a cuvette to record its UV-visible spectra. Then, 0.5 mL of NaBH4 (0.5 M) solution was added to the previous samples with recording the change in their spectra. The catalytic reduction of nitrophenols follows pseudo first order reaction and independent on NaBH4 concentration. For the catalytic reduction, 15 mg of Ag/ZnO-CH nanofibers were added to the UV cuvettes containing the aforementioned samples. The variation and change in the absorption peaks at λmax in UV-visible spectra (417 nm for o-NP, 391 nm for m-NP, 403 nm for p-NP, and 396 nm for TNP) were noticed and then compared with the earlier calibration curve.
For the catalytic reduction of azo dyes, 0.03 mM concentration of CR, MO and MR were used. For the reduction, 3 mL of the dye solution were inserted into a UV cuvette with the addition of aqueous 0.5 mL NaBH4 solution then recorded the UV-visible spectra of each sample. In order to reduce the dye molecules, 15 mg of Ag/ZnO-CH nanocomposite fibers were added as a catalyst. Dyes reduction initiated after the addition of Ag/ZnO-CH composites to the cuvette.
Besides dyes and nitrophenols, Ag/ZnO-CH nanocomposites were used in the reduction of mix solution of dyes and nitrophenols. For this reaction, equal volumes of dye and nitrophenol were used (1.5 mL each) and then 0.5 mL of the sodium borohydride solution were added to make the final volume of 3.5 mL. Then, 15 mg of Ag/ZnO-CH fibers were introduced into a reaction cuvette and the rate of reductions were recorded by UV-visible spectrophotometer.
Characterization
For the confirmation of ZnO nanoparticles and catalytic reduction studies of dyes and nitroarenes, UV-visible spectrophotometer Shimadzu-1800 Japan was used. For FTIR spectra of pure and Ag/ZnO-CH nanofibers, ATR-FTIR Perkin Elmer spectrometer was used in the spectral range of 400–4000 cm−1. FE-SEM instrument JEOL (JSM-7600F, Japan) was used for structural characterization of ZnO NPs and Ag/ZnO-CH. Elemental analysis was investigated by using an EDX system from JEOL (JSM-7600F, Japan).
Crystalline structure of nanomaterials was assessed using powder XRD PAN-analytical diffractometer with a radiation source Cu kα of λ = 1.54 A° wavelengths. Scherrer Equation was used for the calculation of the average crystalline size of nanomaterials using equation (2).
Results and discussion
Synthesis
Successful synthesis of ZnO nanoparticles was confirmed using UV-visible spectrophotometer. UV-visible spectroscopy is a vital and powerful tool to determine the optical properties of nanoparticles. ZnO nanoparticles give an absorption peak at 362 nm as shown in the Figure 1. The formation of a sharp absorption peak near 360 nm showed the successful formation of ZnO NPs [35]. On the spectrum, there was no other peak observed, which showed that the prepared ZnO nanoparticles were of high purity.
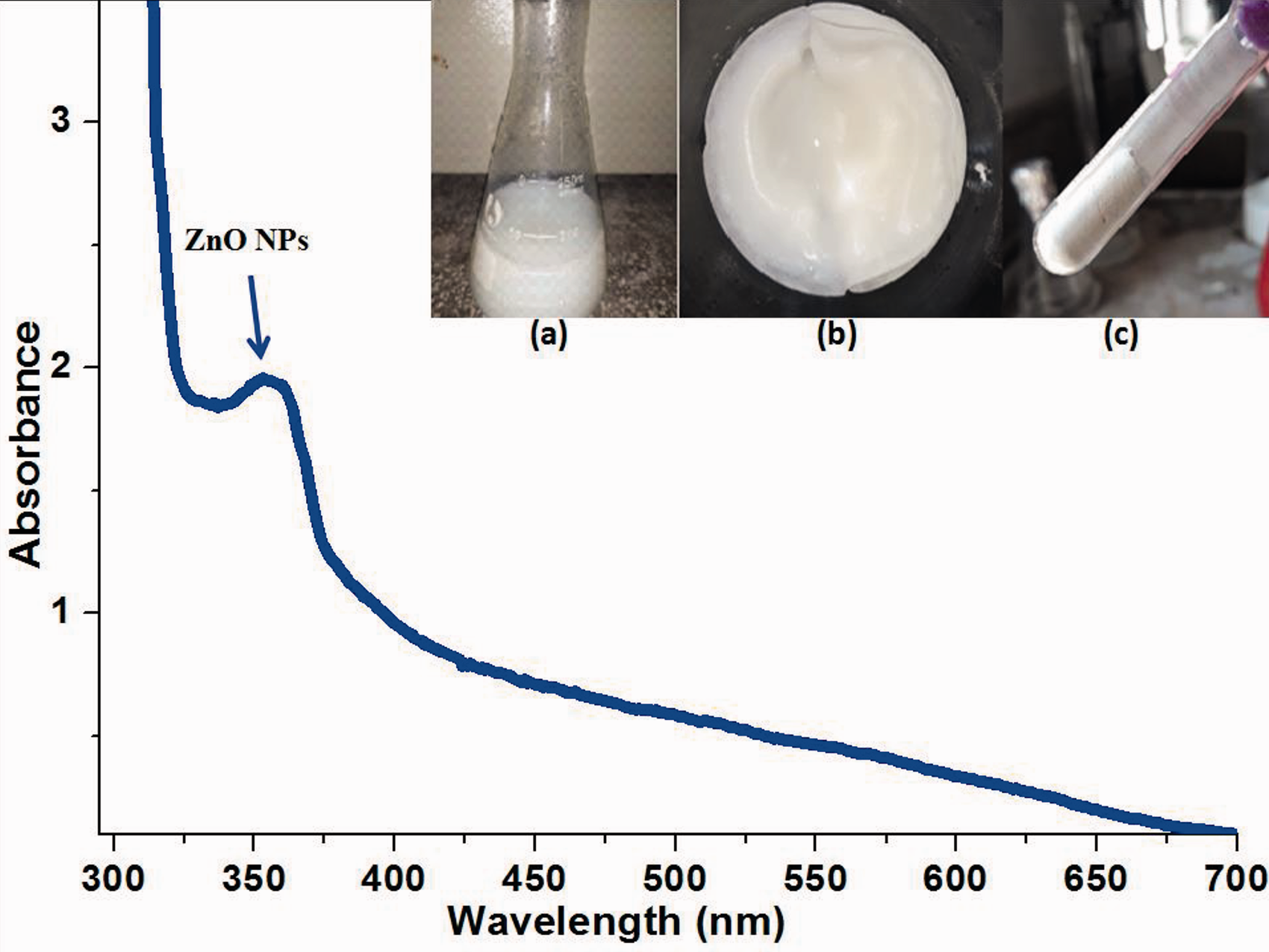
UV-visible absorption spectra of ZnO NPs, which clearly show a strong peak at 362 nm (inset shows the synthesis of (a) ZnO synthesis (b) white paste of zinc hydroxide (c) ZnO NPs).
Synthesis of Ag/ZnO-CH nanocomposite fibers is represented in the Figure 2. The absorption of metal ions on nonoporous ZnO has recently been investigated and it was found that ZnO NPs shows good activity for metal ions adsorption from aqueous medium due to high surface area, narrow and wide pore size distribution [36]. Besides, it has been reported that ZnO NPs in ZnO/carbon black cellulose acetate sheets act as nanofiller that modify the molecular network of nanocomposites and increase the surface area for the adsorption of metal ions [37].

A visual representation of (a) pure cotton fibers, (b) chitosan and ZnO treated cotton fibers, (c) aqueous silver nitrate solution treated ZnO-CH fibers and formation of Ag/ZnO-CH cotton fibers supported catalyst by treating with NaBH4 solution and then the synthesized catalyst was applied for the reduction of o-NP, m-NP, p-NP, TNP, CR, MO, and MR.
Metal uptake study
The adsorption capacity of Ag ions onto supporting matrix of ZnO-CH was studied by use of ICP-OES with adsorbent dosage of 0.04 g in 15 ml of 0.1 M initial salt solution of AgNO3 at room temperature. The metal uptake capacity of the ZnO-CH was determined using equation (3):
Where qe is the amount of adsorbed metal ions by ZnO-CH (mgg−1), Ci and Ce are initial and final concentrations of the metal ions in aqueous solution (mgL−1), V is the volume (L) of solution and mads is the mass (g) of the adsorbent.
The silver ions adsorbed on ZnO-CH were then treated with NaBH4 aqueous solution to reduce the adsorbed Ag+ into zero-valent metal NPs, as shown in equation (4):
Characterization of the prepared materials
XRD analysis
XRD is an important tool for the determination of crystalline structure and average size of nanomaterials. The XRD patterns of the synthesized ZnO NPs and Ag/ZnO-CH nanocomposites were taken at 2-theta values of ranging from 20° to 80°. The major peaks observed for ZnO NPs at a 2Ɵ value of 31.11°, 34.38°, 36.07°, 47.21°, 56.67° and 68.52° (as show in Figure 3(a)), which corresponds to the Bragg’s peaks of (100), (002), (101), (102), (110) and (112). The Bragg’s peaks of ZnO are close in agreement with the Data Card of JCPDS No: 36-1451. XRD peaks of our synthesized ZnO NPs show similar lattice planes with the previously reported Cinnamomum tamala synthesized ZnO NPs [38]. Thus, it was concluded that synthesized ZnO NPs have hexagonal wurtzite structure, and the sharpness of peaks showed a high degree of crystallinity, ably supported by FE-SEM images. The average size of ZnO NPs calculated from the (100) Brags peak using Scherrer Equation was 69.32 nm (±10 nm).

XRD pattern of prepared Ag/ZnO-CH nanocomposites and ZnO nanoparticles.
In the XRD pattern of Ag/ZnO-CH nanocomposites (Figure 3), the major peak at a 2Ɵ value of 38.12 and a small peak at 43.74 were observed. The peaks at 38.12° and 43.74° correspond to the Bragg indices of (111) and (200), of the face centered cubic (fcc) structure of AgNPs [39,40]. The average crystallite size of AgNP calculated from the 111 Brags peak was 32.17 nm (±4 nm). Broadening of peak is a common trend in small sized crystallite. Noticeably, no peak for ZnO was observed in the XRD pattern of Ag/ZnO-CH nanocomposites, because the amount of ZnO is very low in the composite, so ZnO NPs peaks are suppressed by the polymer and AgNPs which exist in high amount as evident in the EDX profile which shows only 3.7% of zinc in Ag/ZnO-CH.
FTIR analysis
The FTIR spectra of the ZnO NPs, Ag/ZnO-CH nanofibers and pure chitosan (CH) are shown in Figure 4. In the FTIR spectrum of chitosan, the broadband from 3200 – 3400 cm−1 could be attributed to the O-H and N-H groups of the chitosan chain [41,42]. While the sharp peak at 1651 cm−1 corresponding to amide group N-H bending of the chitosan [43]. Besides some characteristic small bands are observed from 1020 to 1350 cm−1, which may be due to the O-H bending and C-N stretching and the asymmetrical C-O-C stretching vibration of the chitosan [44].

FTIR spectrum of chitosan, ZnO NPs and Ag/ZnO-CH nanocomposites.
Similarly, FTIR spectra of ZnO NPs and Ag/ZnO-CH nanofibers exhibit absorption bands at 3290, 2950, 1580, 1410, 1030, and 660 cm−1, which corresponds to the stretching of an O-H group of support, C-H asymmetrical stretching, N-H bending, C-N stretching vibrations, and C-O-C stretching. The special band at 660 cm−1 is attributed to the metal oxygen (Ag-O and Zn-O) stretching vibration modes [45], which confirm the presence of Zn and Ag in the chitosan-cotton matrix. Generally, the slight shift in the peaks of both ZnO and Ag/ZnO-CH nanofibers from chitosan suggests that loading of nanoparticles (AgNP) on chitosan-cotton matrix didn’t alter the chitosan chemical structure. Thus, FTIR study suggested that nanoparticles were present in the fibers of chitosan without altering its chemical composition.
FE-SEM and EDX analyses
FE-SEM was used for the analysis of surface morphology and crystalline nature of nanomaterials. Pure ZnO and Ag/ZnO-CH nanocomposites fibers were checked with various magnifications of FE-SEM. The pure ZnO nanoparticles show hexagonal shape as presented in Figure 5(b). In the Figure 5(b), the crystalline structures of ZnO nanoparticles are clearly observing and support the hexagonal crystalline structure of the XRD pattern. The low to high magnification FESEM images of Ag/ZnO-CH nanocomposites are represented from Figure 6(a) to (d). In the FE-SEM images of Ag/ZnO-CH microfibrous structure formation was clearly observed from the low and high magnifications of Ag/ZnO-CH composite fibers (Figure 6(a)). Ag nanoparticles are distributed on the composites and also we can clearly observe that Ag nanoparticles accumulate on the ZnO-CH (as appeared in the Figure 6(b) and (c)), which demonstrate that Ag is secured successfully on the surface of ZnO as appeared in Figure 6.

FESEM images of ZnO nanoparticles.
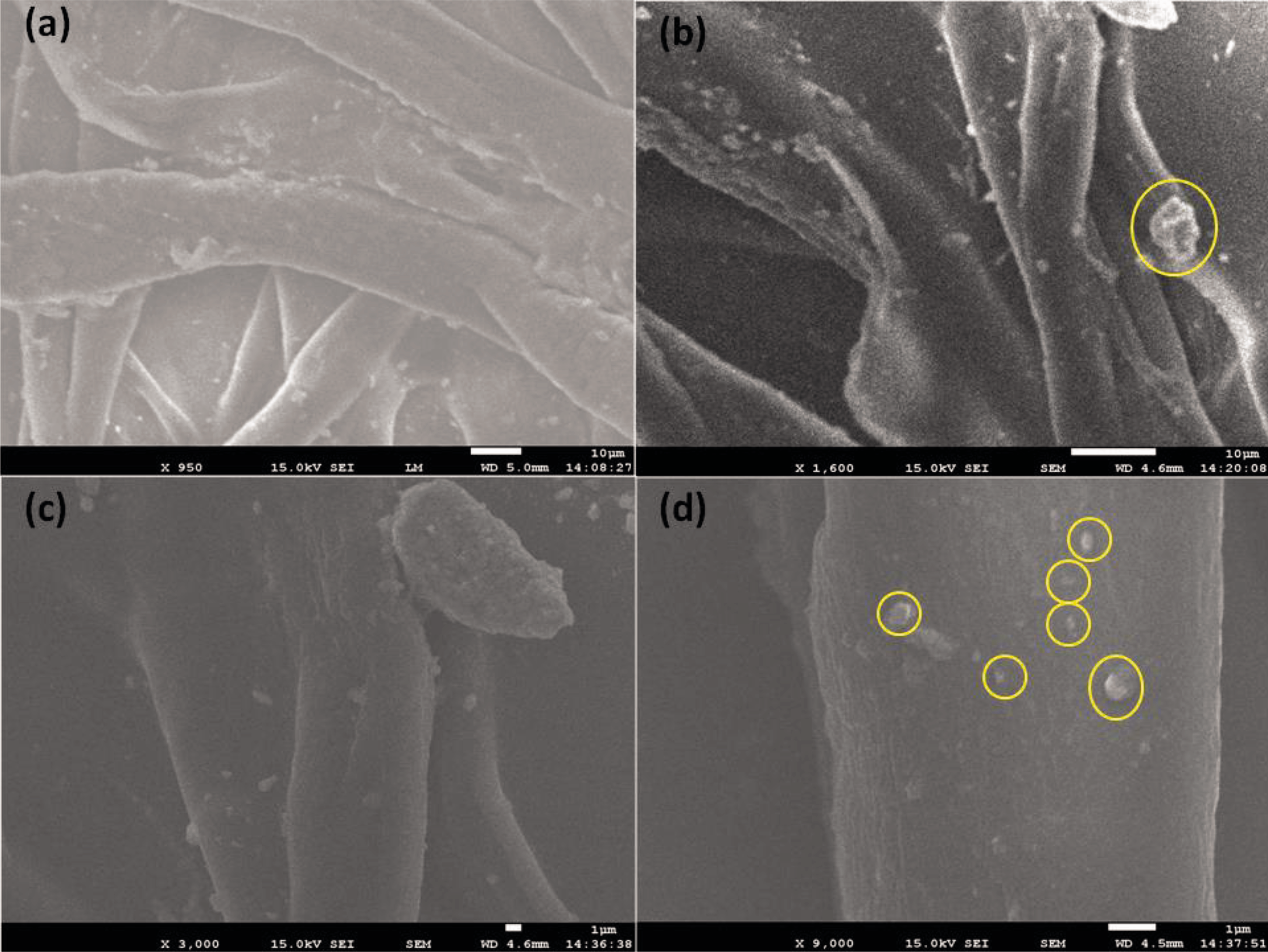
FESEM images of Ag/ZnO-CH nanocomposite fibers.
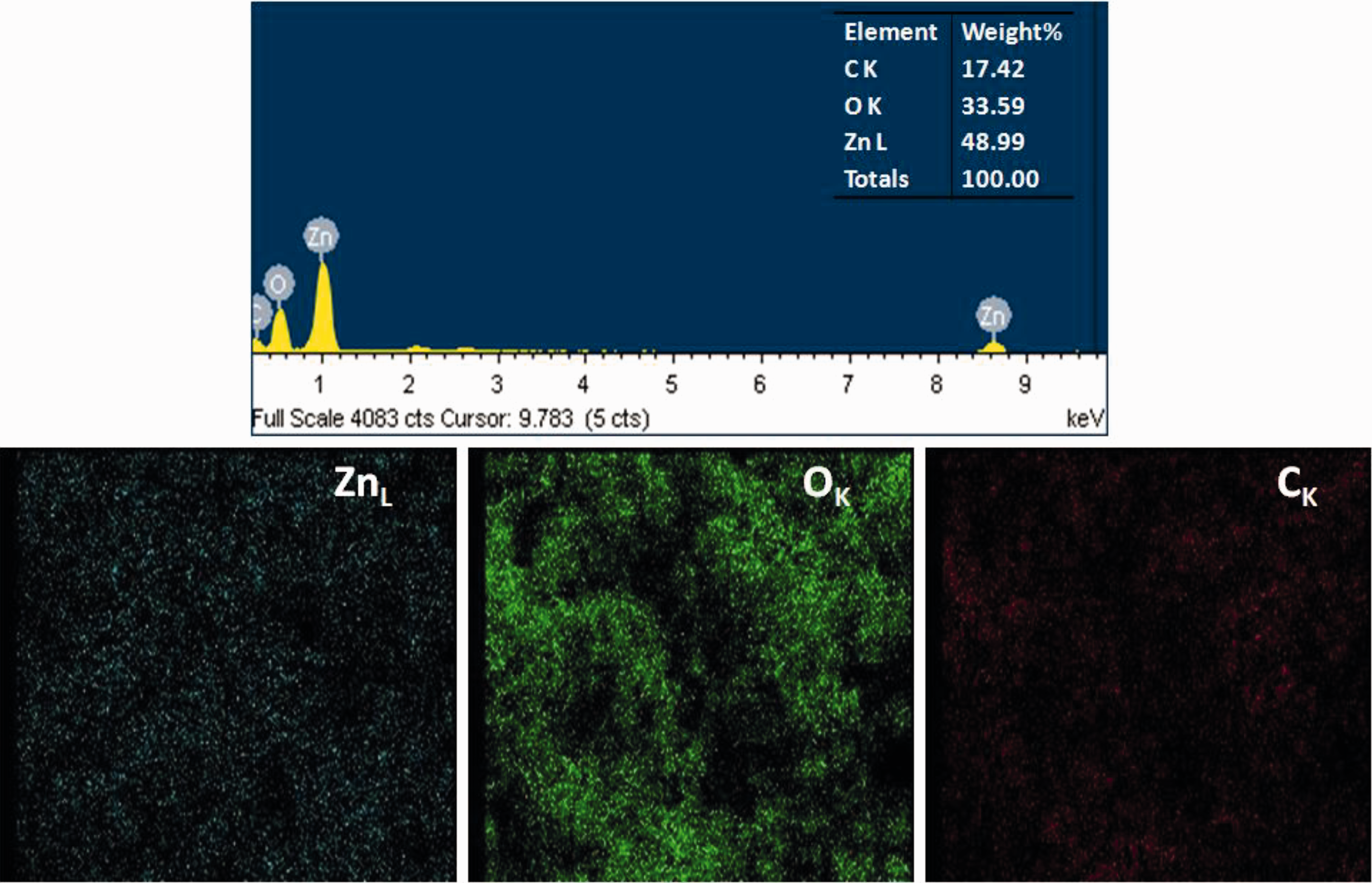
For the confirmation of AgNPs in the composite host, the prepared materials were subjected to EDX analysis. The successful synthesis of ZnO NPs and Ag/ZnO-CH nanofibers were confirmed by EDX. EDX spectra of ZnO NPs and Ag/ZnO-CH nanofibers are shown in Figure 7 and Figure 8 respectively. Due to surface plasmon resonance metallic Ag show absorption peak in the EDX spectrum at 3 keV, while Zn showed a peak at 1 keV. The elemental Ag can be clearly observed in the sample composition of Ag/ZnO-CH microfiber (Figure 8).
EDX profile of ZnO nanoparticles.

EDX profile of Ag/ZnO-CH nanocomposite fibers.
The elemental mapping result of the ZnO NPs showed (blue color in Figure 7) that Zn is distributed maximally (48.99%) and other elements were oxygen (33.59%), and carbon (17.42%). The elemental mapping result of the Ag/ZnO-CH nanofibers is shown in Figure 8, which suggests that silver (brown colour) was the predominant metal of the composite fibers. The elemental distribution of Ag/ZnO-CH nanofibers shows 24.87% of silver, 3.769% of zinc, 46.15% oxygen and 25.22% of carbon. The concentration of Zn is very small in the EDX spectrum of Ag/ZnO-CH nanofibers, which shows that the nanoparticles of ZnO are covered by AgNPs as indicated in the Figure 6(b). This is an evidence that Ag was successfully coated on the ZnO-CH to form the Ag/ZnO-CH nanocomposite fibers.
Catalytic reduction of nitroarenes
Catalytic reduction of nitrophenols
Organic aromatic pollutants have considerable adverse effects on the environment. Nitroaromatic compounds present a harmful effect on human health due to their toxic, mutagenic and carcinogenic potential. Therefore, the presence of even low concentration of nitrophenols poses a real danger to the ecosystem.
The prepared Ag/ZnO-CH nanocomposites were used for the selective hydrogenation of o-NP, m-NP and p-NP. Ag/ZnO-CH nanocomposites show outstanding activity in the reduction of nitrophenols as compared to ZnO-CH.
For the reduction of p-NP solution, 3 mL of p-NP were added in a quartz cuvette with recording its UV-visible spectra. The maximum absorption for p-NP appeared at 317 nm in the UV-visible spectra as shown in Figure 9(a). With the addition of NaBH4 to the cuvette, a shift in peak was observed from 317 nm to 404 nm. Actually the addition of borohydride solution raises the pH of the p-NP and generates nitrophenolate ions which are indicated by deep yellow colour. The peak at 404 nm was due to para-nitrophenolate ions as shown in Figure 9, with the blue curve. The intensity of peak at 404 nm remains unchanged and stable for longer time without addition of the catalyst. This demonstrates that borohydride alone cannot reduce p-NP. Thus, in the absence of an appropriate catalyst, it is difficult to complete the reduction/hydrogenation of p-NP to aminophenol. This is due to the higher kinetic barrier of 4-nitrophenolate (C6H4NO3−) ions and borohydride (BH4−) ions. The decline in the peak intensity at 404 nm of p-NP initiated after the addition of 15 mg of catalyst (Ag/ZnO-CH or ZnO-CH). With the addition of catalyst, a new peak at 303 nm appeared. The decline in the peak intensity at 404 nm and the formation of new peak at 303 nm for p-aminophenolate was due of the catalytic hydrogenation of p-NP to aminophenol [46,47]. Catalytic reduction of p-NP to p-AP is thermodynamically favourable by reducing agent NaBH4 at ambient conditions (E for p-NP/p-AP is −0.76 V) because BH4− ions are very strongly reducing agent in aqueous media (H3BO3/BH4− = −1.33 V) but extremely slow (unfavourable) kinetically and the probability of the p-NP reduction is lower due to the of potential difference and kinetic barriers between acceptor p-nitrophenolate ions and donor borohydride ions. The catalytic reduction of p-NP by NaBH4 obeys pseudo-first order reaction with respect to substrate p-NP. The reduction of p-NP into p-AP takes place on the surface of Ag/ZnO-CH catalyst. The catalytic reduction of p-NP begins with the transfer of electrons from borohydride to para-nitrophenolate ions once borohydride and nitrophenolate ions adsorbed over the surface of catalyst. Thus, Ag/ZnO-CH facilitates the p-NP reduction by lowering the energy of activation and playing a catalyst role in this reaction.

UV–visible reduction spectra of p-NP (a) by Ag/ZnO-CH nanocomposites, (b) by ZnO-CH nanocomposites (c) percent reduction and (d), plot of the ln(At/Ao) vs time.
Besides p-NP, Ag/ZnO-CH catalyst was used for the reductive transformation of o-NP to o-AP. The UV–visible spectrum of o-NP gave strong peaks at 282 and 351 nm as shown in Figure 10(a) by the red curve. With the addition of sodium borohydride, the position of the peak at 351 nm shifted to 411 nm (Figure 10(a)). The red shift in position of peak from 351 nm to 411 nm was due to formation nitophenolate ions. The position and intensity of peak at 411 nm remains unchanged without catalyst. However, the decline in peak intensity at 411 nm begins with the addition of Ag/ZnO-CH catalyst. The decrease in absorption of nitrophenolate ions at 411 nm was monitored at constant intervals of time using UV-visible spectrophotometer (Figure 10(c)). At the same time the peak at 282 nm shifted slowly to 291 nm. The disappearance of peak at 411 nm for o-nitrophenolate ions and formation of new peak at 291 nm (aminophenolate ions) was due to the catalytic hydrogenation of o-NP to o-AP [29].

UV–visible reduction spectra of (a) o-NP by Ag/ZnO-CH nanocomposites, (b) m-NP by Ag/ZnO-CH nanocomposites (c) percent reduction and (d), plot of the ln(At/Ao) vs time.
Similarly, the synthesized Ag/ZnO-CH catalyst was used in the catalytic reduction of m-NP. The UV–visible spectrum of m-NP shows strong absorption peak at 333 nm as indicated in Figure 10(b). The shift in peak was observed from 333 nm to 391 nm with the addition of NaBH4. The shift in peak was due to the formation of phenolate ions. The peak intensity at 391 nm remains constant without catalyst for 30 min. However, the decline in peak intensity at 391 nm started after the insertion of Ag/ZnO-CH catalyst. The decline in the peak intensity of m-nitrophenolate ions at 391 nm was due to the reduction of m-NP to AP as reported previously [44]. Aminophenols are used the preparation of a number of analgesic and antipyretic drugs like acetanilide, paracetamol and phenacetin [48].
The percent reduction of p-NP, m-NP, and o-NP was calculated from the λmax of UV-visible absorption spectra by using the equation (5):
Herein, the Ag/ZnO-CH catalyst exhibit outstanding catalytic activity in the p-NP reduction and complete 93.2% reduction in 11 min, while for ZnO-CH nanocomposites took 34 min for 78.4% p-NP reduction as shown in Figure 9(c). Similarly, for o-NP and m-NP, the Ag/ZnO-CH catalyst reduces 89.3% o-NP in 33 minutes and 65.2% of m-NP in 29 minutes (Figure 10(c)).
It has been reported that the reduction of nitrophenols follows pseudo first order reaction. The pseudo first order equation was applied to determine the rate constant of the reaction.
In which r is the rate of reduction reaction of NP; t show the time of the reaction; C is the NP concentration; C0 show relative concentration of reactant at time zero (initial concentration); Ct is the concentration of the reactant NP at different interval.
From here the reactant NPs are coloured and show strong absorption peaks in the UV and visible region, therefore the rate of pseudo-first order reaction could be expressed and determined from the relative absorbance intensity at time t. So, the rate equation for pseudo first order kinetic could be express as equation (7),
The plot between ln(At/A0) versus time (t) and the kinetic data of p-NP reduction catalysed by Ag/ZnO-CH and ZnO-CH are shown in Figure 9(d). The calculated rate constant (kapp) for the pseudo first order reaction, was 2.813 × 10−3 s−1 and 4.621 × 10−4 s−1 for p-NP for Ag/ZnO-CH and ZnO-CH respectively. The reduction of p-NP by Ag/ZnO-CH is comparable with our previously reported Ag@CAF catalyst [49]. Similarly, the plot of ln(At/A0) vs t and the kinetic data of o-NP and m-NP hydrogenation catalysed by Ag/ZnO-CH nanocomposites is shown in Figure 10(d). The calculated rate constants were 8.155 × 10−4 s−1 and 4.245 × 10−4 s−1 for o-NP and m-NP, respectively for the Ag/ZnO-CH catalyst.
Catalytic reduction of 2,4,6-trinitrophenol
Besides others nitroarenes, the reduction of trinitro-aromatic molecules (like 2,4,6-trinitrophenol) from the sewage is therefore the requirement of the day. The synthesized Ag/ZnO-CH catalyst was also used in the catalytic reduction/hydrogenation of TNP by sodium borohydride. TNP gave a strong absorption peak at 356 nm as shown in Figure 11(a) by the black arrow (TNP). With the addition of reducing agent sodium borohydride, the peak from 356 nm shifted to 396 nm with increasing intensity. The color of TNP also changed to dark yellow from light yellow after the insertion of borohydride. Phenolate ions were generated by sodium borohydride which leads to a shift from 356 to 396 nm. The position and intensity of peak at 396 nm remain constant without catalyst, which shows that sodium borohydride alone could not reduce TNP. However, with the addition of Ag/ZnO-CH catalyst, the decrease in intensity of the peak at 396 nm gets started. The decline in peak intensity at 396 nm and the formation of new peak at 304 nm was due to the catalytic hydrogenation of TNP (Figure 11(a)). The new peak formed at 304 nm is attributed to the reduction of 2,4,6-trinitrophenol to corresponding aminophenol [23,50–52]. It has been reported that the reduction of TNP is independent of sodium borohydride concentration [23]. Thus, the reduction of TNP by sodium borohydride follows the pseudo first order reaction.
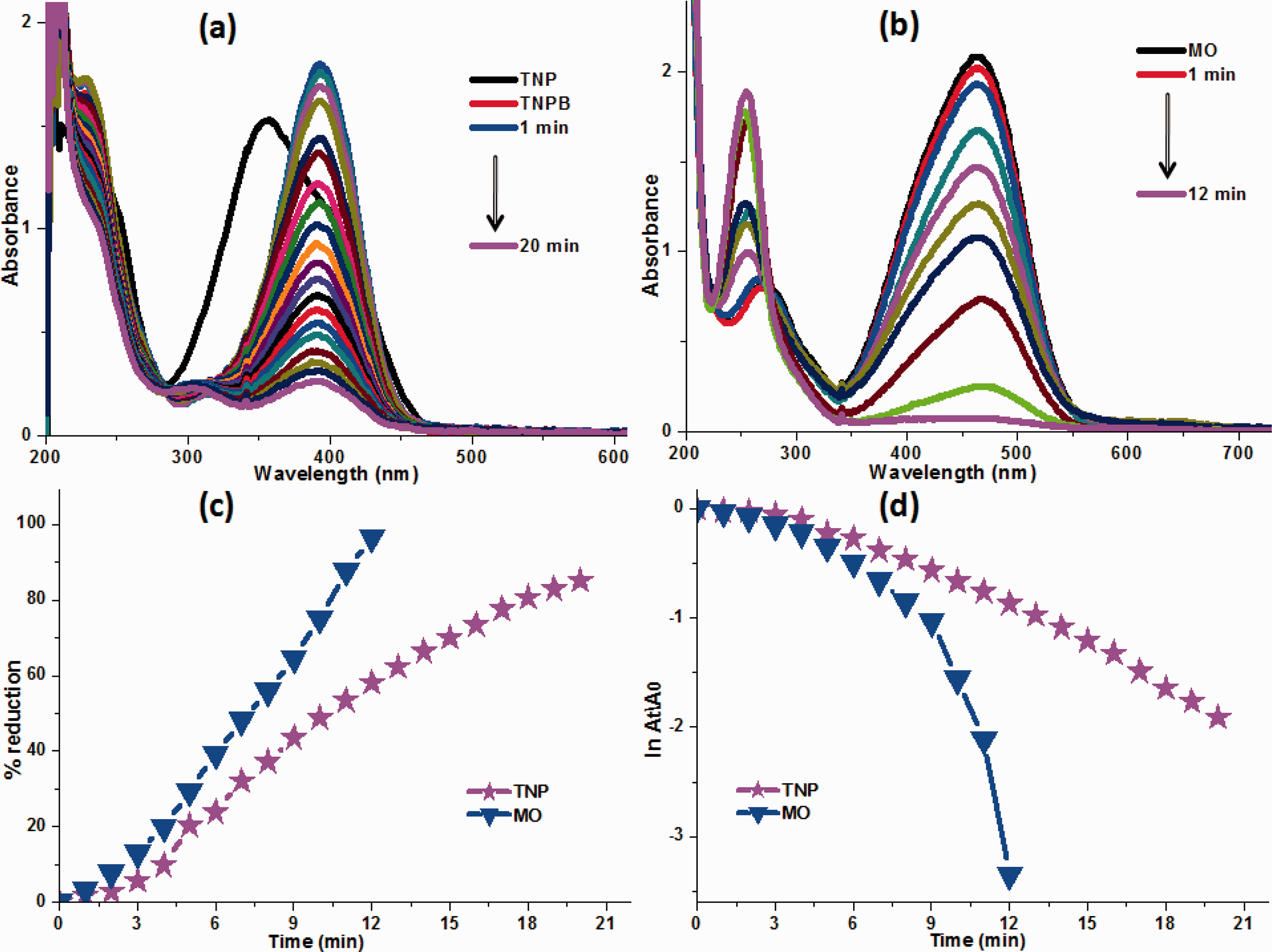
UV–visible reduction spectra of (a) TNP by Ag/ZnO-CH, (b) MO by Ag/ZnO-CH (c) percent reduction and (d), plot of the ln(At/Ao) vs time.
Ag/ZnO-CH catalyst reduced 85.18% TNP in 20 minutes, as shown in Figure 11(c). The calculated kapp for TNP at room temperature was 1.157 × 10−3 s−1 (Figure 11(d)). The kapp for our Ag/ZnO-CH catalyst is much higher than our recently reported catalyst Ag@CAF which was 8.97 × 10−4 s−1 for TNP [49]. Hence, the synthesized catalyst Ag/ZnO-CH shows better activity for the reduction of TNP in aqueous solution.
Dyes reduction
In addition to nitroarenes, the catalytic performance of Ag/ZnO-CH was evaluated for the reduction of azo dyes CR, MR and MO. Azo dyes are extensively used for textiles, inks, paper, leather, ceramics, cosmetics, food and plastics industries. Most of the colours contain monoazo, disazo, and trisazo dyes, where each colour was dominated by unmetallized structures. Azo dyes comprise diazotized amine combined with an amine or a phenol with one or more azo connections. Among all azo dyes used in the synthetic process contribute about 60-70% with variety of colours [53].
MO dye reduction
Dyes degradation and detoxification by using excessive sodium borohydride solution as a reducing agent in the presence of an effective catalyst is commonly used to evaluate the catalytic activity of different catalysts. Reduction of MO by reducing agent NaBH4 alone is thermodynamically favorable but kinetically unfavorable. The deterioration of azo dye MO is of considerable interest due to a number of health and environmental related concerns.
In detail, 3 mL of dye solution were inserted to a quartz cuvette. At pH 7 aqueous MO is orange red in color and give strong absorption at 465 nm in the visible region and small peak at 285 nm in the UV region. The absorption of MO at 465 nm is due to the azo linkage. The position and intensity of MO peaks remain unchanged with the addition of NaBH4. The reduction of MO by NaBH4 in the absence of catalyst is thermodynamically favorable but extremely slow in term of kinetics. Earlier reports also recommend that MO peak at 465 nm in the absence of suitable catalysts remains unchanged for a long time [17,54]. Both peaks of the MO at 285 and 465 nm begin to decline after the addition of Ag/ZnO-CH catalyst. The peak of maximum absorption at 465 nm slowly decrease, however, the peak λmax at 285 nm moved slightly to lower wave length, observing blue shift and reaching 251 nm with an increase in the intensity as revealed in Figure 11(b). The decrease in peak λmax at 465 nm with the addition catalyst is attributable to the initiation of dye reduction. Ag/ZnO-CH in the presence of NaBH4 reduce the MO molecule at (–N = N–) azo linkage by generating low molecular amino compounds. Consequently, the peak at 251 nm was from the free amino group molecules produced by MO reduction [5].
The percentage of MO reduction was determined using equation (5) from their UV-visible spectra. Ag/ZnO-CH cotton fibers supported catalyst show excellent catalytic reduction of MO and reduced 96.5% of MO in 12 minutes. The calculated pseudo first-order kapp for MO at room temperature was 1.663 × 10−3 s−1. Ag/ZnO-CH nanocomposites in the reduction of MO show improved activity, then the earlier study of Cu/CH-FP strip which took 13 minutes [17]. This highlights Ag/ZnO-CH nanocomposites exceptional catalytic activity over MO dye.
CR reduction
The azo dye CR is another commonly used hazardous dye that causes water pollution. Therefore, the existence of CR dye in aquatic environments is undesirable even in small concentration. It is therefore necessary to remove such toxic chemicals from wastewater. An aqueous solution of CR gave two strong peaks at 491 and 337 nm in the UV-visible spectrum. Both peaks remain constant in the presence of NaBH4 for half an hour. However, with the insertion of Ag/ZnO-CH nanocomposites, gradual decline at both peaks 491 and 337 nm started. The gradual disappearance of peaks at 491 and 337 nm with the addition of Ag/ZnO-CH catalyst and appearance of a new strong peak at 251 nm with increasing intensity and a small peak at 293 nm was due to the initiation of CR reduction as shown in Figure 12(a). Ag/ZnO-CH in the presence of sodium borohydride reduce CR molecule at azo linkage by generating aromatic amines of low molecular weight. Thus, the appearance of two peaks at 251 and 293 nm was from the compound of the free –NH2 group.

UV–visible reduction spectra of (a) CR by Ag/ZnO-CH, (b) MR by Ag/ZnO-CH (c) percent reduction and (d), plot of the ln(At/Ao) vs time.
Here, Ag/ZnO-CH shows an outstanding catalytic activity and completed 89.77% of CR reduction in 11 min (Figure 12(c)). The pseudo first-order rate (kapp) for CR was 1.43 × 10−3 at room temperature. Our prepared Ag/ZnO-CH nanofibers catalyst in the reduction of CR shows superior activity, then our earlier study of Ag/TP catalyst which takes 26 min for a 94% reduction of the CR dye [55]. This highlights Ag/ZnO-CH nanofibers exceptional catalytic activity over CR dye.
MR reduction
Methyl red is a classic organic azo compound which has been used broadly in a number of laboratory techniques. Aqueous solution of MR gave a strong peak at 412 nm as demonstrated in Figure 12(b). The peak at 412 nm remains constant with the addition of borohydride. However, with the addition of Ag/ZnO-CH catalyst, the decline in the intensity of peak 412 nm started. The decline of peak intensity at 412 nm and formation of two novel peaks at 305 and 241 nm, which was due to the initiation of MR reduction as shown in Figure 12(b). NaBH4 in the presence of a suitable catalyst (Ag/ZnO-CH) reduces MR at azo linkage and thus aromatic amines are generated by low molecular weight. Thus, the new peak formation at 305 and 241 nm was due the free –NH2 group compound produced during the MR reduction. In this study, Ag/ZnO-CH shows excellent catalytic efficiency and reduced 95.29% of MR in 16 minutes. The rate constant kapp calculated for MR was 1.577 × 10−3. Our catalyst Ag/ZnO-CH displays excellent activity, then recently reported Psidium guajava leaf extract capped and hydrazine mediated Cu nanoparticles catalyst which takes 14 min for a 75.022% reduction of MR dye [56].
Reduction of mixture of dyes and nitrophenols
Mostly, the catalyst’s reduction and remediation were carried out on a single substance, i.e., either on the nitrophenol or dye. The performance of the Ag/ZnO-CH catalyst toward a mixture of organic pollutants was also analyzed. Our synthesized catalyst shows exceptional catalytic degradation activity toward a mixture of dyes and p-NP.
Reduction of CR and MO mixture
Ag/ZnO-CH catalyst was assessed against mix CR and MO solution. For the experiment, 1.5 mL solution of each CR and MO were added to a UV cuvette and collected their UV-visible spectrum. Mixture of CR and MO gave a single strong absorption peak at λmax of 471 nm as shown in Figure 13(a). The strong peak at 471 nm was attributed to the absorption of CR and MO dyes. The intensity and position of λmax at 471 nm was stable after the addition of 0.5 mL of NaBH4. However, the decline in intensity of peak at 471 nm started with the addition of 15 mg of catalyst Ag/ZnO-CH. The decline of peak intensity at 471 nm was due to the reductive degradation of azo dyes CR and MO. Here, Ag/ZnO-CH completed 100% of the CR and MO reduction in 9 minutes.
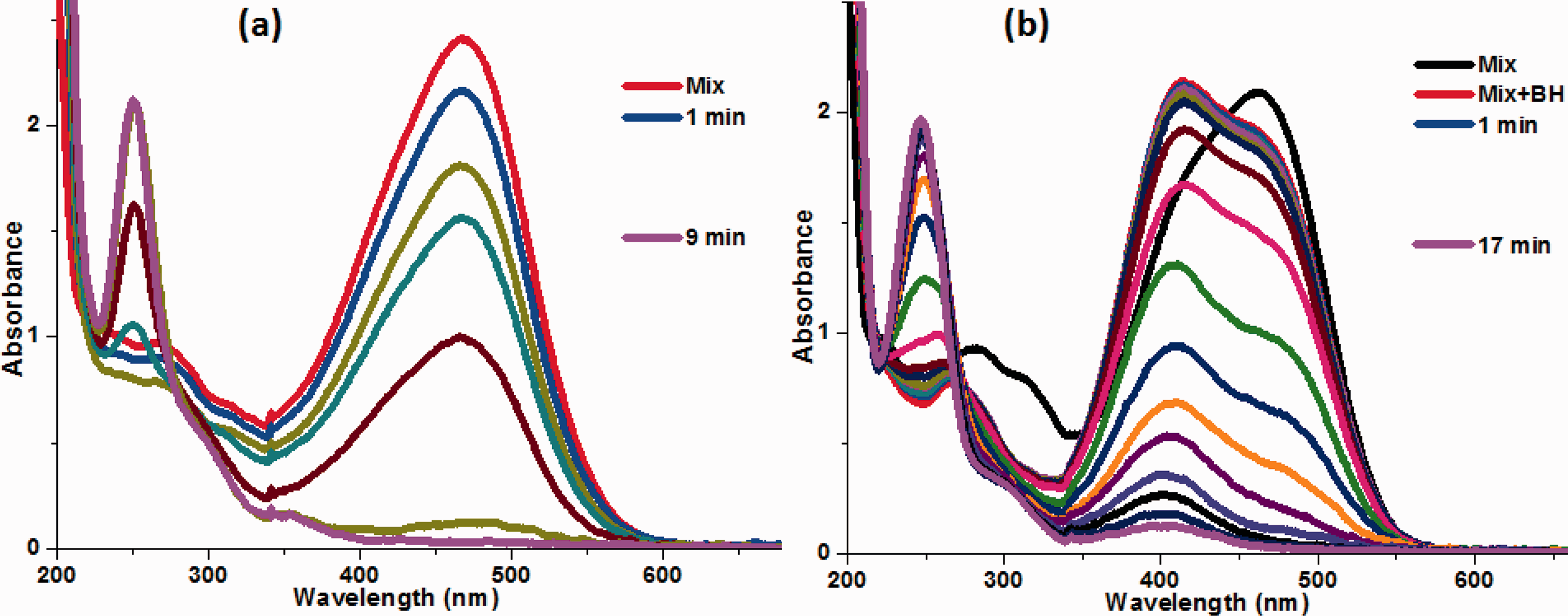
UV–visible reduction spectra of mixture of (a) MO and CR by Ag/ZnO-CH and NaBH4, and (b) MO and o-NP by Ag/ZnO-CH and NaBH4.
Reduction of p-NP and MO mixture
Similarly, another reaction was carried out of mix dye and nitrophenol solution. For the reduction, 1.5 mL of each p-NP and MO solution were placed in a cuvette. Mixture of p-NP and MO gave a strong peak at 461 nm and small peak at 316 nm as shown in the Figure 13(b). The strong peak at 461 nm is attributed to the absorption of MO, while the small absorption peak at 316 nm is attributed to the p-NP. Both peaks of 316 and 461 nm shifted to 416 nm after the addition of sodium borohydride as indicated by red arrow in Figure 13(b). The formation of new peak at 416 nm after sodium borohydride addition was due to the generation of nitrophenolate ions. The intensity of the peak remains unchanged at 416 nm without catalyst. However, with the addition of a catalyst, the decrease in peak intensity at 416 nm started. The gradual decline of peak λmax at 416 nm and formation of new peak at 249 nm demonstrates the reduction of MO and p-NP. Herein, Ag/ZnO-CH reduces 98% of MO and p-NP mixture in 17 minutes. From the above results, it was concluded that prepared catalyst Ag/ZnO-CH is not only efficient in the remediation of only single contaminant but also against a mixture of various pollutants.
Reusability of catalyst
Catalyst recyclability is a very important and critical issue during catalysis. Majority of catalysts becomes deactivated after one or two cycles. Reusability of catalyst is very significant because it decreases the cost of the process. That is why it is very important to separate the active catalyst easily and simply from the reaction for reusability.
CR, MO and p-NP were used for the reusability experiment in the current study. Figure 14 showed the selective reusability reduction study of CR, MO and p-NP. Reusability study of the prepared catalyst Ag/ZnO-CH was assessed five times for the complete reduction of CR and MO and p-NP. The catalyst Ag/ZnO-CH reusability was performed by washing the catalyst twice with DDW after the reaction and reused for the next reaction on the same day. Figure 14 exhibits the complete reduction of p-NP, CR, and MO using Ag/ZnO-CH in different cycles.

Reusability of Ag/ZnO-CH for the complete reduction of CR, MO and o-NP.
In the first cycle of CR reduction, Ag/ZnO-CH took 12 minutes, for 2nd cycle it took 13 minutes, while for 3rd use it took 15 minutes. However, for the 4th and 5th cycles, Ag/ZnO-CH took 17 and 19 minutes, respectively. Similarly for the MO, the complete reduction of 1st cycle took 12 minutes, for the 2nd, and 3rd cycles, Ag/ZnO-CH took 13 minutes and 14 minutes, respectively. While, for the 4th and 5th cycles, it took 15 and 17 minutes, respectively. Hydrogenation of p-NP, in the 1st cycle, it completed in 12 minutes. For the 2nd and 3rd cycles, Ag/ZnO-CH took 14 and 15 minutes, respectively. While, for the 4th and 5th cycles, Ag/ZnO-CH took 17 and 20 minutes, respectively, for the complete reduction of p-NP dye as shown in Figure 14.
The recyclability results suggest that Ag/ZnO-CH showed superior catalytic ability and could be used as an active catalyst in a reduction reaction for several times. The Ag/ZnO-CH outstanding catalytic activity could be due to the high adsorption capacity of ZnO-CH cotton support for the silver ions with high inorganic nanoparticles contents as shown by EDX analysis. The developed Ag/ZnO-CH catalyst therefore has real potential being used as an economic catalyst. The decrease in the catalytic efficiency of the Ag/ZnO-CH catalyst may be due to aggregation after catalysis or due to the formation of silver oxide as discussed in our reported silver supported catalyst [23].
Conclusions
We investigated a simple approach to zero-valent AgNPs on supported composites of chitosan-cotton (biodegradable and readily available natural polymer) and ZnO (high absorption, less toxic, and inexpensive). XRD, EDX and UV-visible analyses confirmed the successful synthesis of ZnO and Ag/ZnO-CH nanocomposites materials. We demonstrate that catalytic performance of Ag/ZnO-CH nanocomposites were effective in reduction of CR, MO and MR organic dyes and nitroarenes TNP, o-NP, m-NP, and p-NP. The reduction of CR, and MO dyes were completed by Ag/ZnO-CH within 11 min, MR reduced in 16 min and moreover p-NP was also reduced in 11 min. Besides, it also reduced the mixture of dyes MO and MR in only 9 min showing its outstanding activity for the reduction of all types of organic pollutants individual as well as collectively. The reusability of Ag/ZnO-CH was investigated for five repeated cycles in the catalytic reduction of MO, CR, and p-NP. The catalyst separation was performed easily by just pulling the catalyst from the reaction mixture. The development of a new strategy by preparing nanocomposites using textile cotton fibers as a support material has great promise for the synthesis of other kinds of pure and bimetallic catalysts for quick separation and elimination of organic pollutants from wastewater.
Footnotes
Acknowledgments
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. G: 1345-130-1440. The authors, therefore, acknowledge with thanks DSR for technical and financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. G: 1345-130-1440.



