Abstract
The effects of polycaprolactone (PCL) viscosity and carbon nanotubes (CNT) content on the microstructure, morphology development, tensile strength, and electrical percolation behavior of CNT/polylactic acid (PLA)/PCL blends were investigated. PLA/PCL blends with a volume ratio of 80/20 were prepared using a low viscosity PCL (PCL1) and a high viscosity PCL (PCL2). The microstructure analysis showed finer dispersion of PCL2 than PCL1 within the PLA matrix. The addition of CNT changed the PLA/PCL morphology from dispersed to co-continuous due to the localization of the nanotubes in the PCL domain (i.e. the minor domain). In addition, the introduction of CNT reduced the PCL domain size and improved the adhesion at the PLA/PCL interface. For the electrical percolation behavior, PLA/PCL2 blend exhibited a percolation threshold concentration (EPTC) of 0.5 wt.% CNT compared to 1.0 wt.% CNT for the PLA/PCL1 blend. This remarkably lower EPTC for the PCL2-based blend is in line with the microstructure analysis that indicated finer dispersion of PCL2 than PCL1 within the PLA matrix. The tensile strength analysis showed that adding up to 5 wt.% CNT does not influence the PLA/PCL blend’s tensile strength. However, remarkable adhesion and tensile strength enhancements were obtained at a CNT content of 10 wt.% and above.
Keywords
Introduction
Polymers are attractive materials for many applications due to their lightweight, ease of processing, versatility, low cost, and adaptatively to applications needs. 1 The unprecedented demand for polymeric materials has increased the environmental concerns regarding handling the post-consumed polymeric material. Almost 79% of plastics produced from 1950 to 2015 have been accumulated in the natural environment or landfills. 2 The problem of post-consumed polymers was partially handled by recycling or incineration. While this solution sounds great, a small fraction of the post-consumed plastics ended up in landfills. 3 Biodegradable polymers (BDPs), such as polylactic acid (PLA), are considered promising alternatives for conventional polymers. 4 In 2018, 0.36 million metric tons (MMT) of BDPs were consumed globally compared to almost 100 MMT of polyethylene (PE). 5 The market share of BDPs is tiny compared to that of conventional polymers. The relatively higher price and the incompetent mechanical and chemical properties of most BDPs compared to the conventional polymers are the most important reasons behind the limited use of the BDPs.
Polylactic acid is on the top of the most promising BDPs. 6 PLA is a thermoplastic that can be processed using conventional equipment in the polymer industry. In addition, it has a high melting temperature, good processability, competitive tensile strength, and biocompatibility.7,8 However, the low toughness of PLA compared to conventional polymers is one of its drawbacks.6,9,10 This shortcoming of PLA has been addressed by blending it with an adequate polymer matrix. 11 Many researchers utilized polymer blending and compatibilization techniques to create PLA-based polymer blends with improved tensile properties.7,8,11–13 PLA/polycaprolactone (PCL) blends have been one of the most studied biodegradable blends because of the biodegradability, biocompatibility, and high ductility of PCL.11,14,14–16 PCL is a semicrystalline thermoplastic having −60oC glass transition temperature and 60oC melting temperature. 11 PLA/PCL blends have higher toughness and lower Young’s modulus than PLA.6,11
To broaden the applications of PLA-based materials, PAL and PLA-blends filled with electrically and thermally conductive nanofillers have been investigated and showed promising properties.13,17–19 Conductive biopolymer nanocomposites have potential applications in many sectors such as energy, sensors, and electronics.17,20 In addition to improving the electrical and thermal conductivities of PLA/PCL blends, conductive fillers are expected to have a compatibilization impact on the blend’s microstructure and, consequently, the tensile properties.13,21 Herein, the focus is on PLA/PCL reinforced with multi-walled carbon nanotubes (MWCNT). Multi-walled carbon nanotubes-filled PLA/PCL blends have been investigated by many researchers.22–26 Wu et al. 22 found that the location of carboxylic-MWCNT (c-MWCNT) in a PLA/PCL blend depends on the PLA/PCL viscosity ratio. At a PLA/PCL viscosity ratio of ∼1, the c-MWCNT particles were found in the PLA phase and at the PLA/PCL interface. However, the nanotubes were seen in the PCL phase for a PLA/PCL blend with a viscosity ratio of 16. For a 30/70 weight ratio PCL/PAL blend, Laredo et al. 24 found that the c-MWCNT preferred the PCL phase, and the composite electrical percolation threshold was 1.0 wt.%. Xu et al. 25 and Tan et al. 26 studied the effect of the PLA/PCL ratio on the location of the MWCNT particles and the electrical conductivity of MWCNT-filled PLA/PCL blends. The MWCNT particles were solely seen in the PCL phase regardless of the PLA/PCL ratio. The blend electrical conductivity was found to be a function of the PLA/PCL ratio revealing its dependence on the blend morphology and the effective concentration of nanotubes in the PCL domain. For an 80/20 PLA/PCL blend, Urquijo et al. 27 reported selective localization of CNT in the PCL domain. The blend’s electrical and rheological percolation thresholds were ∼1.2 wt.% MWCNT. 27
While, CNT-filled PLA/PCL blends have been investigated by many reseachers, there are no studies related to the influence of PCL viscosity on the electrical percaoltion behaviour of CNT/PLA/PCL blends. In addition, most of the studies focused on blends filled with moderate level of CNT content, i.e. up to 5 wt.% CNT. Herein, the focus is on understanding the influence of MWCNT content and PCL viscsoity on the morphology development, microstructure, electrical percolation threshold, and tensile strength of 80/20 PLA/PCL blends. Blends filled with up to 15 wt.% CNT were fabricated and tested. The PLA/PCL 80/20 volume ratio was selected since it is around the optimal composition to achieve a balance properties. 28
Experimental Details
Materials and Fabrication Methods
Information about polymers used in this study.
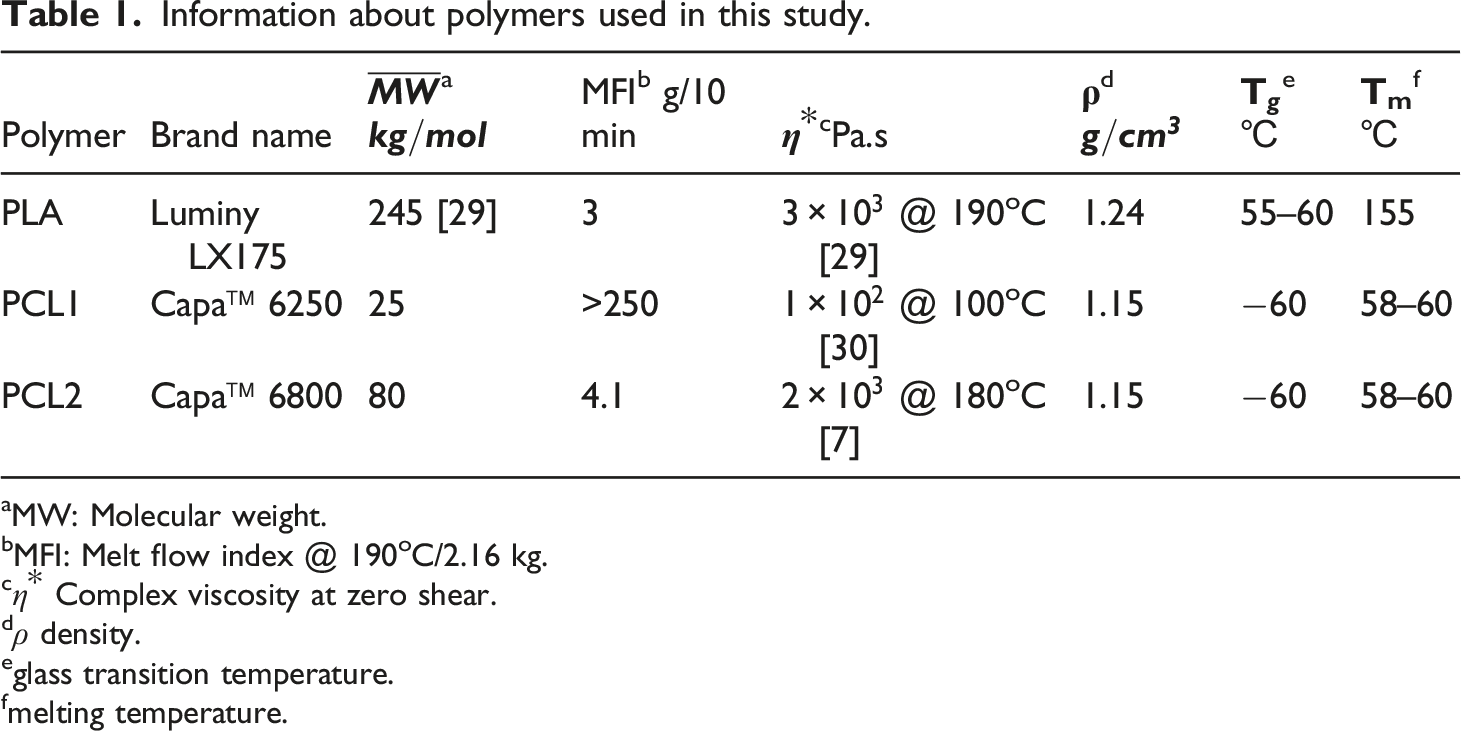
aMW: Molecular weight.
bMFI: Melt flow index @ 190oC/2.16 kg.
c
d
eglass transition temperature.
fmelting temperature.
The MWCNT-filled PLA/PCL blends were fabricated by melt mixing (Torque Rheometer Plastograph EC, Brabender, Germany) at 175oC and 100 r/min. The blend components were dried overnight in a vacuum oven before mixing. The drying temperatures for MWCNT, PLA, and PCL were 120oC, 70oC, and 50oC, respectively. For all blends, the PLA/PCL volume ratio was fixed at 80/20. This ratio was achieved by mixing 26.8 g of PLA pellets with 6.2 g of PCL pellets. In a typical experiment, the PLA/PCL pellets were introduced to the pre-heated mixing chamber and mixed for 3.0 min. Then X g of MWCNT powder was added to molten polymer blend and mixed additional 10 min. Parts for volume electrical resistivity and tensile strength testing were prepared using a hot press machine (Model CH (4386), Carver Inc., USA) at 180oC for 10 min under 27.6 MPa. After the 10 min annealing step, the heating source was turned off, and the mold platens were cooled by a water stream flowing inside the platens. The specimens for electrical resistivity characterization were rectangles 1.0 mm in thickness, 40 mm in length, and 20 mm in width. For the tensile testing, the samples were dumbbells according to ASTM D638-03 type IV dimensions.
Characterization Details
The blends microstructure analysis was conducted using a scanning electron microscope (SEM) (Quanta 450 FEG, FEI, USA). Before imaging, the selected specimens were freeze-cracked in liquid nitrogen. The obtained surfaces were then sputtered with a nanolayer of gold. The composites volume electrical resistivity (
Results and Discussion
CNT location within the PLA/PCL blend
Figure 1 depicts representatives SEM micrographs of 1.0 wt.% MWCNT-filled PLA/PCL1 and PLA/PCL2 blends. Regardless of the PCL viscosity, MWCNT is seen in the PCL phase (the minor phase in the image). This observation aligns with the theoretical predictions and the experimental observations reported by Urquijo et al.
27
and Zhu et al.
31
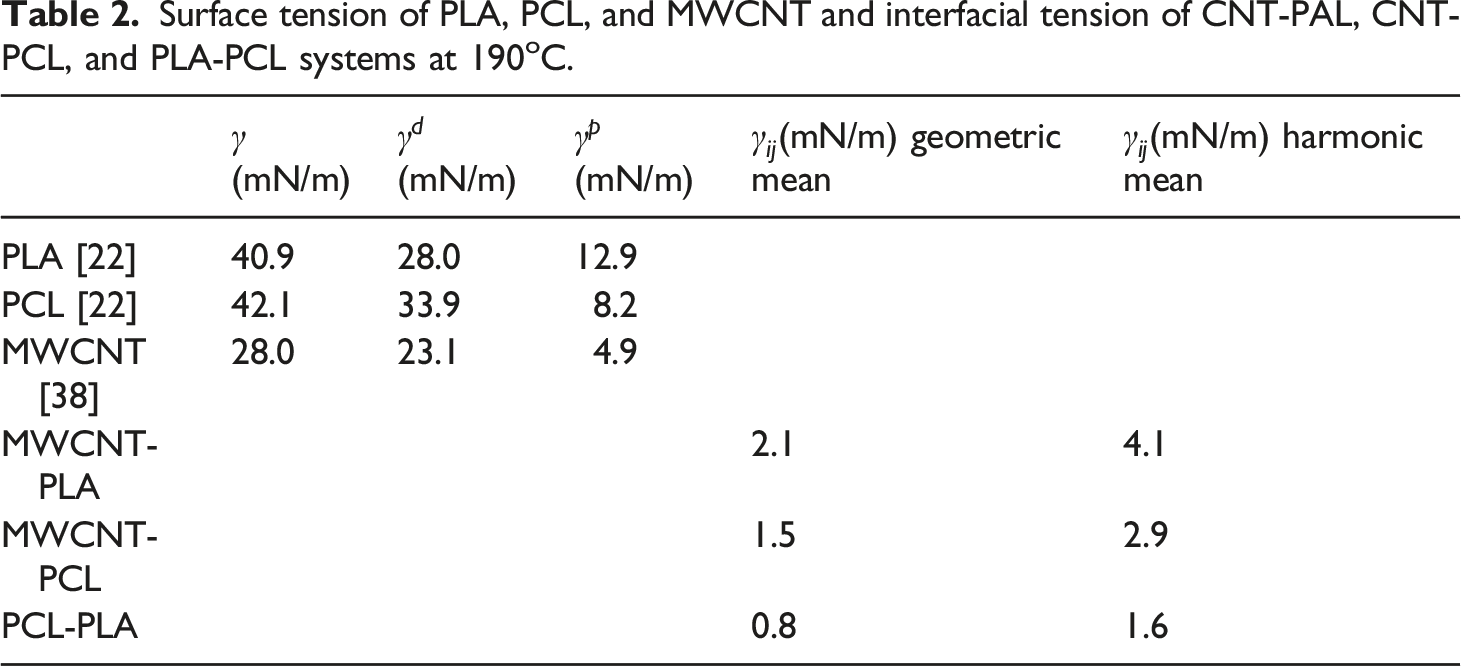
for MWCNT-filled PLA/PCL blends. The thermodynamic affinity of MWCNT towards the PCL domain in the PLA/PCL blend can be predicted based on the MWCNT-PLA and MWCNT-PCL interfacial tensions. In an immiscible polymer blend, nanofillers have an affinity toward the phase with the lost nanofiller-polymer interfacial tension.
32
In limited and rare cases, nanofiller particles can reside at the blend’s interface.13,33,34 The presence of filler particles at the interface is possible if the value of Young’s Model, equation (1), wetting coefficient ( Scanning electron microscope micrographs of 1 wt.% multi-walled carbon nanotubes filled (a) polylactic acid/polycaprolactone1 blend and (b) polylactic acid/polycaprolactone2 blend.

In Young’s Equation,
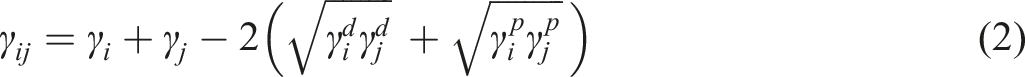
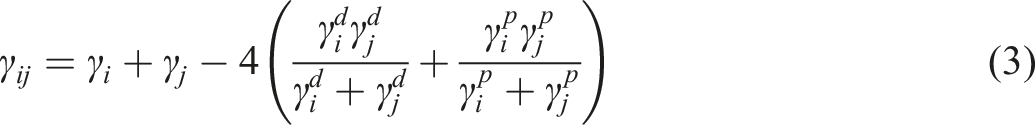
Surface tension of PLA, PCL, and MWCNT and interfacial tension of CNT-PAL, CNT-PCL, and PLA-PCL systems at 190oC.
Morphology Development
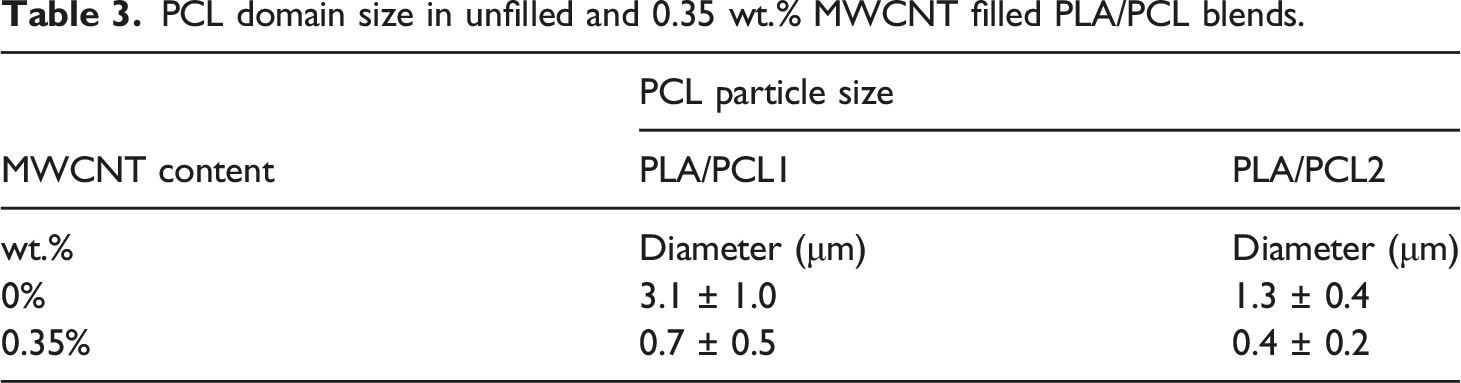
Figures 2 and 3 show representative SEM micrographs for the impact of MWCNT weight fraction on the microstructure and morphology development of PLA/PCL1 and PLA/PCL2 blends, respectively. Many observations can be drawn from the micrographs. The first observation is related to the size of the dispersed PCL domain in the unfilled PLA/PCL blends. The size of the dispersed PCL domain in the PLA/PCL2 blend (Figure 3(a)) is smaller than its size in the PLA/PCL1 blend (Figure 2(a)). Quantitative analysis for the PCL domain size in PLA/PCL1 and PLA/PCL2 blends indicated that the average size of the PCL domain was 3.1 μm and 1.3 μm, respectively. This means that with the increase in the dispersed phase viscosity, for a system with a dispersed phase/continuous phase viscosity ratio less than unity, the domain size of the dispersed phases decreases. Scanning electron microscope micrographs of (a) 80/20 polylactic acid/polycaprolactone1 blend and 80/20 polylactic acid/polycaprolactone1 blends filled with (b) 0.35 wt.% multi-walled carbon nanotubes, (c) 1.0 wt.% multi-walled carbon nanotubes, (d) 5.0 wt.% multi-walled carbon nanotubes, and (e) 15.0 wt.% multi-walled carbon nanotubes. Scanning electron microscope micrographs of (a) 80/20 polylactic acid/polycaprolactone2 blend and 80/20 polylactic acid/polycaprolactone2 blends filled with (b) 0.35 wt.% multi-walled carbon nanotubes, (c) 1.0 wt.% multi-walled carbon nanotubes, (d) 5.0 wt.% multi-walled carbon nanotubes, and (e) 15.0 wt.% multi-walled carbon nanotubes.

PCL domain size in unfilled and 0.35 wt.% MWCNT filled PLA/PCL blends.
Electrical Percolation Behavior
Figure 4 shows the electrical percolation profiles of MWCNT-filled PLA/PCL1 and PLA/PCL2 blends. A typical electrical percolation behavior can be observed for both blends. At a critical MWCNT concentration, a sudden and remarkable drop in electrical resistivity occurs. The PLA/PCL2 blend’s electrical percolation threshold is 50% less than that of the PLA/PCL1 blend. In addition, beyond the percolation point, the electrical resistivity of the PCL2-based blends filled with up to 3.0 wt.% CNT is substantially lower than those of the PCL1-based blends. For example, at 2 wt.% CNT content, the electrical resistivity of the PCL2-based and PCL1-based blends were 27 and 115 Ohm.com, respectively. This finding is consistent with the morphological observations that suggested finer dispersion of the PCL2 domain in the PLA matrix compared to the dispersion of the PCL1 domain. A finer morphology of the PCL2 phase means less CNT to create the first MWCNT network within the blend. However, at MWCNT content of 5.0 wt.% and above, the electrical resistivities for the PCL1-based and PCL2-based blends were the same. This can be ascribed to the fact that at high MWCNT loading, both blends exhibited a co-continuous structure, and consequently, the electrical resistivity is function of MWCNT content only. Electrical percolation curves of multi-walled carbon nanotubes-filled polylactic acid/polycaprolactone1 and polylactic acid/polycaprolactone2 blends.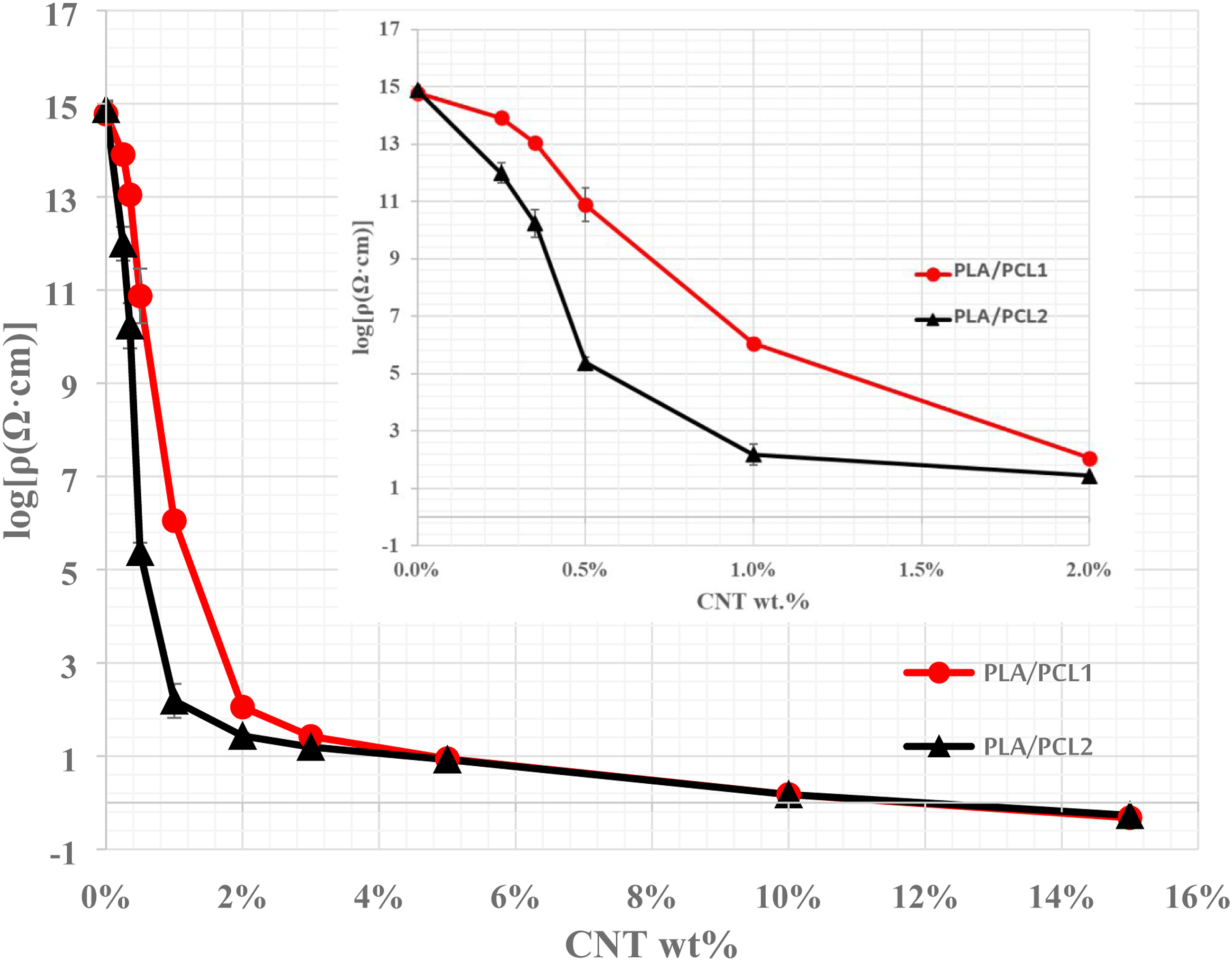
The statistical power-law Equation, equation (4), was used to estimate the composites' electrical percolation threshold volume fraction (
Summary of fitting results for CNT-filled PLA/PCL blends.

Tensile Strength
Figure 5 depicts the effects of PCL viscosity and MWCNT loading on the tensile strength of MWCNT-filled PLA/PCL blends. Blends filled with up to 5 wt.% MWCNT have a similar tensile strength to the unfilled blends. Nevertheless, at MWCNT loading of at least 10 wt.%, an apparent enhancement in the blend’s tensile strength was obtained. The enhancement in tensile strength is consistent with the morphology development analysis that showed noticeable gaps between the PLA and PCL phases for the unfilled blends and the blends at low MWCNT content. However, at high MWCNT content, the adhesion between the MWCNT-rich phase and the PLA phase was improved, as shown in Figure 2(e) for PLA/PCL1 blends. The improvement in adhesion facilitated the stress transfer from the PLA phase to the MWCNT/PCL phase, leading to the enhancement of the tensile strength. Similar observations were reported for MWCNT-filled 90/10 polypropylene/PE blends, where no influence of adding up to 5 wt.% MWCNT on the tensile strength was observed, and at high MWCNT content remarkable increase in tensile strength was reported with the increase in MWCNT loading.
35
Tensile strength of multi-walled carbon nanotubes-filled polylactic acid/polycaprolactone blends.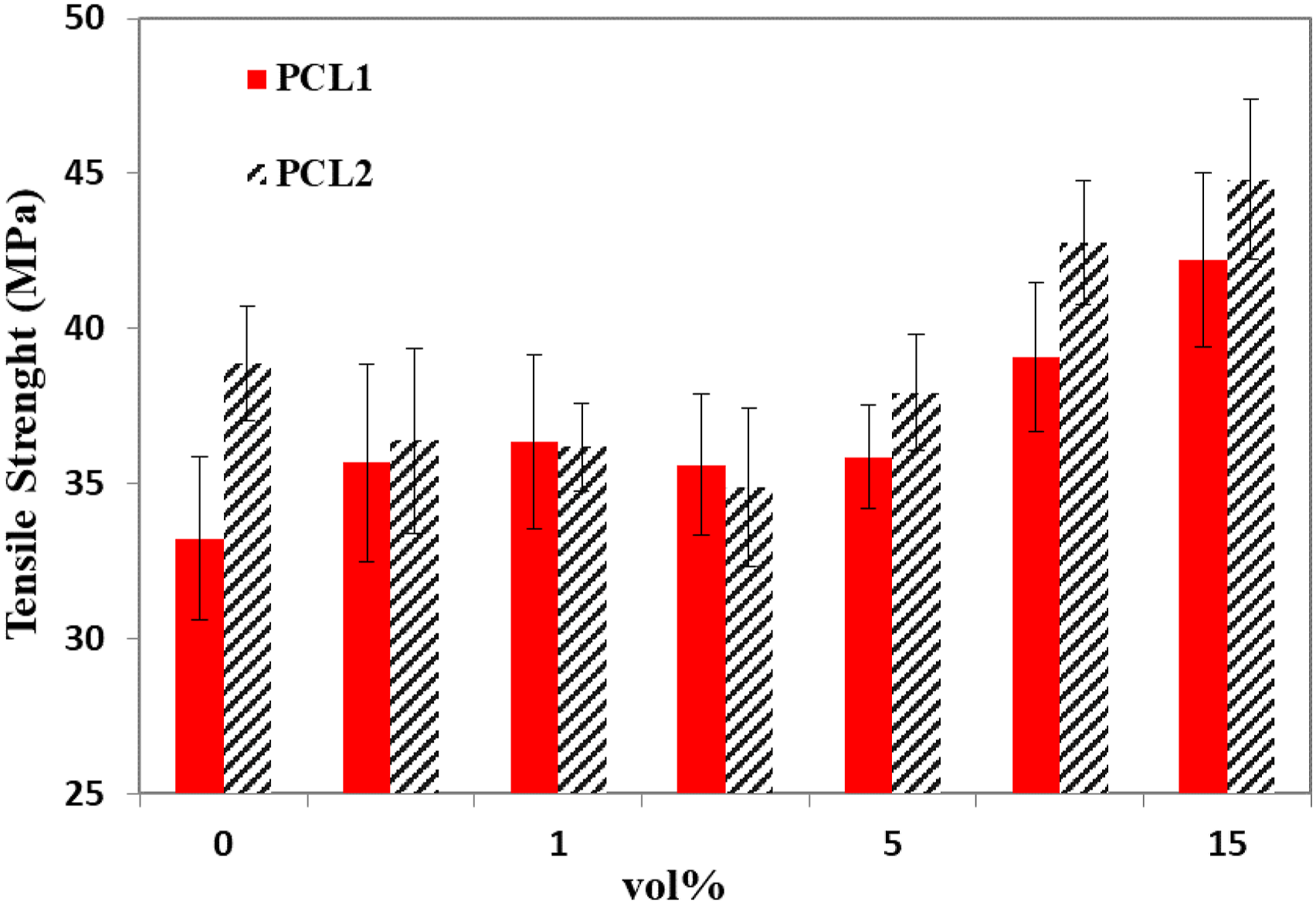
Conclusions
The influence of PCL minor phase viscosity and MWCNT weight fraction on interfacial adhesion, morphology development, electrical properties, and tensile strength of 80/20 PLA/PCL blends were investigated. The viscosity of the PCL minor phase was found to have a remarkable influence on the system’s electrical percolation threshold. MWCNT-filled PLA/PCL blend based non the high viscosity PCL (PCL2) exhibited a percolation threshold that was 50% lower than that for the blend based on the low viscosity PCL (PCL1). This remarkably lower electrical percolation threshold of the PCL2-based system is due to the finer dispersion (i.e. smaller domain size) of the PCL2 compared to the PCL1 within the PLA matrix. The addition of MWCNT was found to decrease the PCL domain size, change the morphology from dispersed into co-continuous, and improve the adhesion at the PLA/PCL interface. The improved adhesion was apparent at high MWCNT content, and due to this improvement, an enhancement in tensile strength was obtained. For example, the 15 wt.% MWCNT-filled PLA/PCL1 blend exhibited a tensile strength of 42.2 MPa compared to 33.2 MPa for the unfilled PLA/PCL1 blend.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Jordan University of Science and Technology, Grant Number 534/2019.
