Abstract
The aim of this study is to investigate the electrospinning of Nafion® nanofibers with poly(vinyl alcohol) (PVA) as a carrier polymer and to assess the thermal and chemical stability of resultant PVA/Nafion® nanofibers for the use in direct methanol fuel cells, in simulated conditions. Bead-free PVA/Nafion® nanofibers were produced using higher molecular weight PVA. Resultant PVA and PVA/Nafion® nanofibers were stabilized using two different methods which are BTCA crosslinking and thermal stabilization, followed by sulfonation of the PVA part. FT-IR analysis demonstrated that the membranes were stabilized and sulfonated successfully. Thermal, water, methanol and oxidative stability of the membranes were tested in addition to ion-exchange capacity. Morphological changes in the structure were analyzed using SEM analysis. Thermally stabilized PVA/Nafion® nanofibrous membrane was found to be stable against water, methanol and oxidative effects. The nanofibrous structure was well preserved after treatments, while the other membranes became a film-like material. Thermal stability of the PVA/Nafion® nanofibrous membrane was similar to that of commercial Nafion® 115 membrane up to 200℃.
Introduction
Energy interactive technical textiles have gained an increasing interest for their potential use in many areas [1,2] and one of them is energy conversion applications. Recently due to the increase in greenhouse gas and air pollutant emissions, researchers have been striving to find highly efficient energy conversion technologies based on renewable or alternative energy resources. Among the different alternatives, due to the advantages of using methanol as the fuel (e.g. easy to store and obtain and high energy density), the direct methanol fuel cell (DMFC) has found favor within this aspect, particularly in portable applications [3]. One of the most important components of DMFC is the membrane that conducts protons from anode to cathode. The standard membrane used today is perfluorinated polyelectrolyte-based commercial Nafion® films. Nafion® is made up of a hydrophobic polytetrafluoroethylene backbone and hydrophilic sulfonic acid groups [4]. Despite the Nafion® membrane having desirable properties for use in a DMFC, it also possesses some major shortcomings such as its high cost, dehydration at high temperatures resulting in decreased proton conductivity, and high methanol permeability, which in turn causes increase in cathodic activation polarization and decrease in fuel efficiency [5,6]. Therefore, new methods of producing polymeric membranes have received much focus and within these polymeric membranes, nanofibrous membranes are thought to be a potential candidate for the production of economic and high performance proton exchange membranes. Several recent results have indicated that scaling down Nafion® to the nanometer size scale may impact its properties and subsequently its performance in devices [7]. Electrospinning is one technique that can produce polymer fibers in the nanometer range by applying a high-voltage electric field to a polymer solution thereby ejecting out a nanofiber jet [8]. Nanofibers have been reported to increase the proton conductivity of a polymer exchange membrane by artificial formation of well-connected ionic clusters, i.e. proton conducting channels [6,9]. The higher total surface area of nanofibrous membranes due to nanometer sized fiber diameters might be advantageous for the use as proton exchange membranes. Dong et al. [7] demonstrated the increase in proton conductivity of an electrospun Nafion® nanofiber (99.9 wt. %) from the conventional 0.1 S/cm for a bulk Nafion® film to 1.5 S/cm for a Nafion® nanofiber.
Aggregate formation exists due to the electrostatic forces in Nafion®, therefore it has insufficient chain entanglements required for electrospinning [10]. Thus, a carrier polymer is required for the production of Nafion® nanofibers by electrospinning.
The production of Nafion®-based nanofibers using poly(vinyl pyrrolidone) (PVP) [11], polyphenylsulfone (PPSU) [12], poly(ethylene oxide) (PEO) [7,11,13,14], poly(acrylic acid) (PAA) [4], poly(vinyl alcohol) (PVA) [11,13,15–18] as carrier polymers has been reported. PVA is one of the most researched carrier polymers. In the literature, PVA/Nafion® nanofibrous membranes have been produced by either direct electrospinning of PVA/Nafion® solutions or impregnation of sulfonated PVA nanofibers with Nafion® solution. PVA has several advantages such as being able to be functionalized by sulfonation and electrospinnability with water as a solvent. On the other hand, for a membrane to be regarded as fully functional, it has to possess adequate chemical, electrochemical and thermal stability under operating conditions [3,11] as well as high proton conductivity. These properties of PVA containing nanofibrous membranes have not been fully studied. Moreover, since PVA is a water-soluble polymer, a stabilization process should be applied, which directly influences the characteristics of the resultant membranes. Although nanofibrous membranes have been reported to be advantageous for the use in fuel cells, it is important to investigate to what extent the nanofibrous structure is preserved under fuel cell working conditions. The novelty of this work stems from the investigation of (i) the effects of molecular weight of the carrier polymer on the maximum available Nafion® content in PVA/Nafion® nanofibers, (ii) preservation of the nanofibrous structure under simulated fuel cell conditions, and (iii) the effects of PVA stabilization method on the overall stability of resultant PVA/Nafion® nanofibers. For this purpose, in the first part of the study, the electrospinnability and morphology of PVA/Nafion® nanofibers were discussed. In the second part, stabilized and sulfonated PVA and PVA/Nafion® nanofibers were investigated in terms of chemical and thermal stability. Additionally, the ion-exchange capacity of the resultant membranes was also discussed.
Experimental
Materials
Materials used in the study.

Electrospinning
Stock solutions of 10% (w/w) for higher molecular weight PVA and 12.5% (w/w) for lower molecular weight PVA were prepared by stirring in distilled water at 80℃ for at least 3 h. To reduce the surface tension of the PVA solutions, 0.05 ml/gpolymer Tween 80® was added. The concentrations of PVA stock solutions and electrospinning parameters were chosen depending on the previous studies [20]. PVA solutions were subsequently mixed with 5% Nafion® solution in the following PVA/Nafion® solution ratios (v/v): 100:0, 90:10, 80:20, 70:30, 60:40 and 50:50 for higher molecular weight PVA and 100:0, 90:10, 80:20 and 70:30 for lower molecular weight PVA. It was not possible to electrospin lower molecular weight PVA/Nafion® solutions for the Nafion® solution ratios higher than 30% (v/v), due to very low viscosity. Nanofibers were produced by the electrospinning set-up which consisted of a 10 ml syringe with a stainless steel needle (1.5 cm long, 22 gauge and flat tip) and a high-voltage supply (Simco, MP Series CM5 30 P). A grounded stationary rectangular metal collector was used for the nanofiber deposition. Electrospinning was carried out at a voltage of 10–12 kV, a tip-to-collector distance of 15 cm, a feeding rate of 0.2–0.4 ml/h and a collection period of 24 h.
Stabilization
Two stabilization methods were applied for the stabilizing of the PVA within the nanofibers: chemical crosslinking with BTCA and physical stabilization by heat treatment. For chemical crosslinking, 5% (w/wpolymer) BTCA was added into the PVA stock solution with sodium hypophosphite monohydrate as a catalyst in a ratio of 2:1 (w/w) followed by stirring for 15 min. After electrospinning, PVA/Nafion® nanofibers were heat treated at 180℃ for 15 min. For physical stabilization, PVA/Nafion® nanofibers were heat treated at 180℃ for 1 h. BTCA crosslinked and thermally stabilized nanofibrous membranes were encoded throughout the paper as PVA-BTCA, PVA/Nafion®-BTCA and PVA-HT, PVA/Nafion®-HT, respectively.
Sulfonation
Sulfonation of PVA functional group within the PVA/Nafion® nanofibers was carried out according to the method given in Mollá et al. [18], to enhance the proton conductivity. The sulfonating solution was prepared by dissolving 0.04 M 4-formyl-1,3-benzenedisulfonic acid disodium salt and 0.1 M HCl in isopropanol/water (70:30, v/v). PVA/Nafion® nanofibers were then immersed into sulfonating solution for 2 h at 60℃ followed by immersion into a bath of isopropanol/water (70/30 v/v) containing 0.3 M HCl. Nanofibrous membranes were washed with isopropanol/water (70/30 v/v) and pure isopropanol, respectively.
Characterization
Viscosity, conductivity and pH of the electrospinning solutions were measured using a Brookfield DV III rheometer with a SC4-21 spindle at 30 r/min, a J.P. Selecta (CD-2004) conductivity meter and a J.P. Selecta pH meter, respectively.
Morphology of the resultant nanofibers was examined by scanning electron microscopy (SEM, FEI Quanta 250 FEG). Nanofiber diameters were measured using Image J software. Fiber diameters were investigated by statistical analysis by one-way ANOVA followed by a multiple comparison by the Tukey HSD post hoc test. Differences of
Fourier transform infrared spectroscopy (FT-IR) analysis was carried out using a PerkinElmer, Spectrum 100 FT-IR spectrometer.
Thermogravimetric analysis
The thermal stability of resultant nanofibers was investigated by thermogravimetric analysis (TGA) (TA Instruments, SDT Q600) by heating samples from room temperature to 600℃ under continuous nitrogen purge with a heating rate of 10℃/min.
Swelling in water
Swelling ratio of the nanofibers was tested by immersion into distilled water at 25℃, 65℃ and 100℃ for 24 h. After water treatment, the excess surface water was removed by filter paper and the wet weight of the samples was measured. They were subsequently dried at room temperature. The swelling ratio was calculated using equation (1)

Weight loss in water and methanol
Weight losses of the nanofibers in water and methanol were tested by immersion method. First, the dry weight of the samples was recorded. For weight loss in water, the samples were immersed into water at 25℃, 65℃ and 100℃ for 24 h. To test the weight loss in methanol, the samples were immersed into 5 M methanol solution at 25℃ and 65℃ for up to 72 h. After drying at room temperature, weight loss of the samples was calculated as given in equation (2)

Oxidative stability
Fenton's reagent was used to test the oxidative stability of the samples. 3 ppm Fe+2 was added into 3% H2O2 solution and samples were immersed into the solution at 65℃ for 24 h. Weight loss of the samples was calculated using equation (2).
Ion-exchange capacity
Ion-exchange capacity (IEC) was tested by a standard acid exchange and base titration method [12,16]. The nanofibrous membranes were immersed into 2 M NaCl solution for 48 h for the exchange of protons with Na+. The solution was titrated against 0.01 M NaOH solution using phenolphthalein as an indicator and IEC was calculated by equation (3)

Results and discussion
Electrospinning of PVA/Nafion® nanofibers
Parameters of electrospinning solutions and diameters of resultant nanofibers.

aBTCA included PVA/Nafion® solution.
SEM images of resultant nanofibers are illustrated in Figures 1 and 2 for lower and higher molecular weight PVA, respectively. Nanofiber diameters obtained from SEM images are given in Table 2. With lower molecular weight PVA, finer nanofibers were obtained; however, bead-free nanofibers could not be obtained. The use of higher molecular weight PVA led to the production of bead-free and smooth nanofibers. The maximum obtainable Nafion® concentration was found to be 50% (v/v) with higher molecular weight PVA and above this concentration electrospinning of the solutions could not be achieved, possibly due to the very low viscosity of the solution as a result of insufficient chain entanglements. The nanofiber diameters were found to be between 127 and 162 nm for higher molecular weight PVA. Although the solution viscosity decreased and the conductivity increased with the increase in Nafion® content, statistical analysis revealed that Nafion® content had insignificant effect on the resultant nanofiber diameter ( SEM images and nanofiber diameter distribution histograms of lower molecular weight PVA/Nafion® nanofibers obtained at different solution mixture ratios. SEM images and nanofiber diameter distribution histograms of higher molecular weight PVA/Nafion® nanofibers obtained at different solution mixture ratios.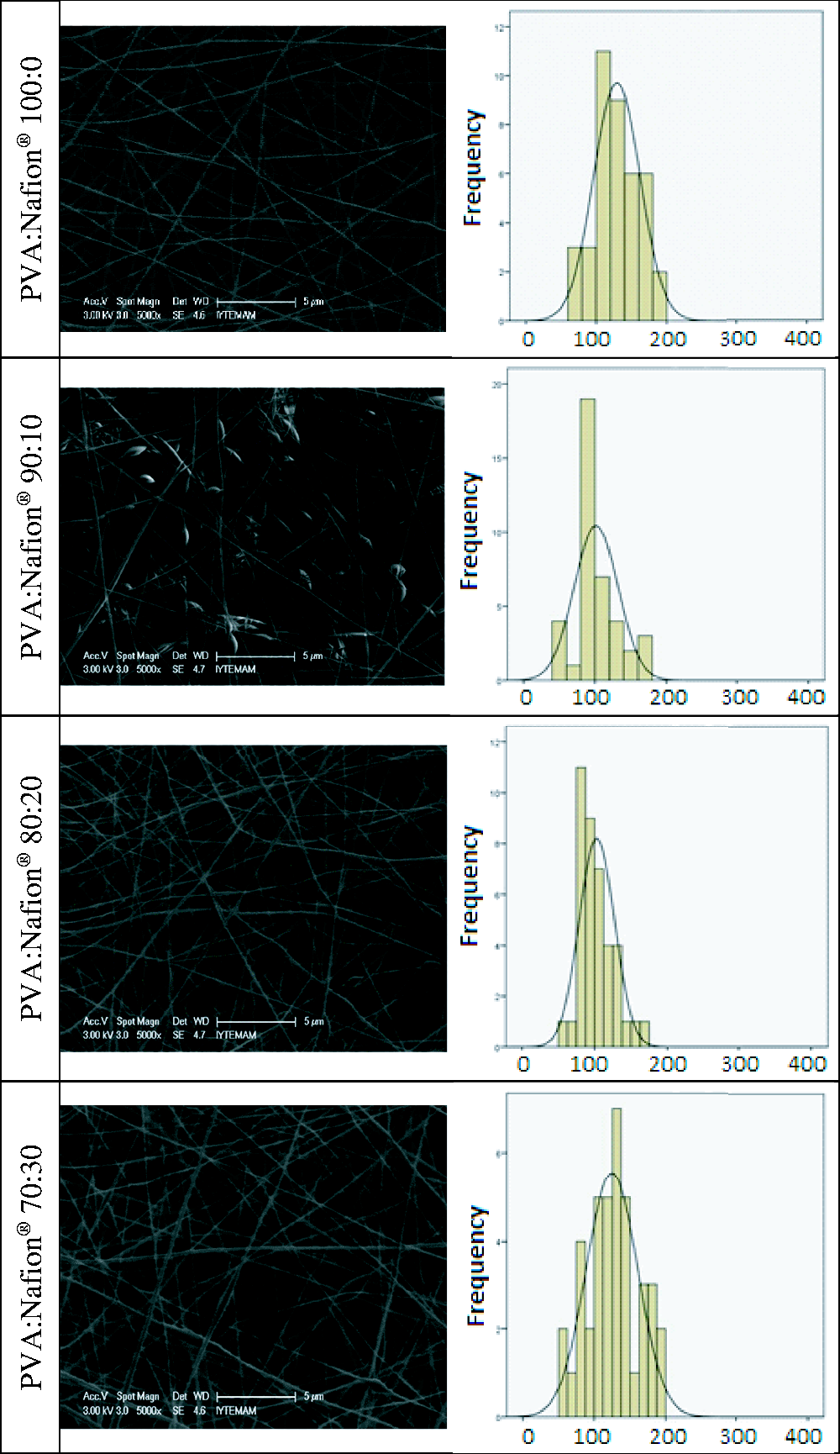
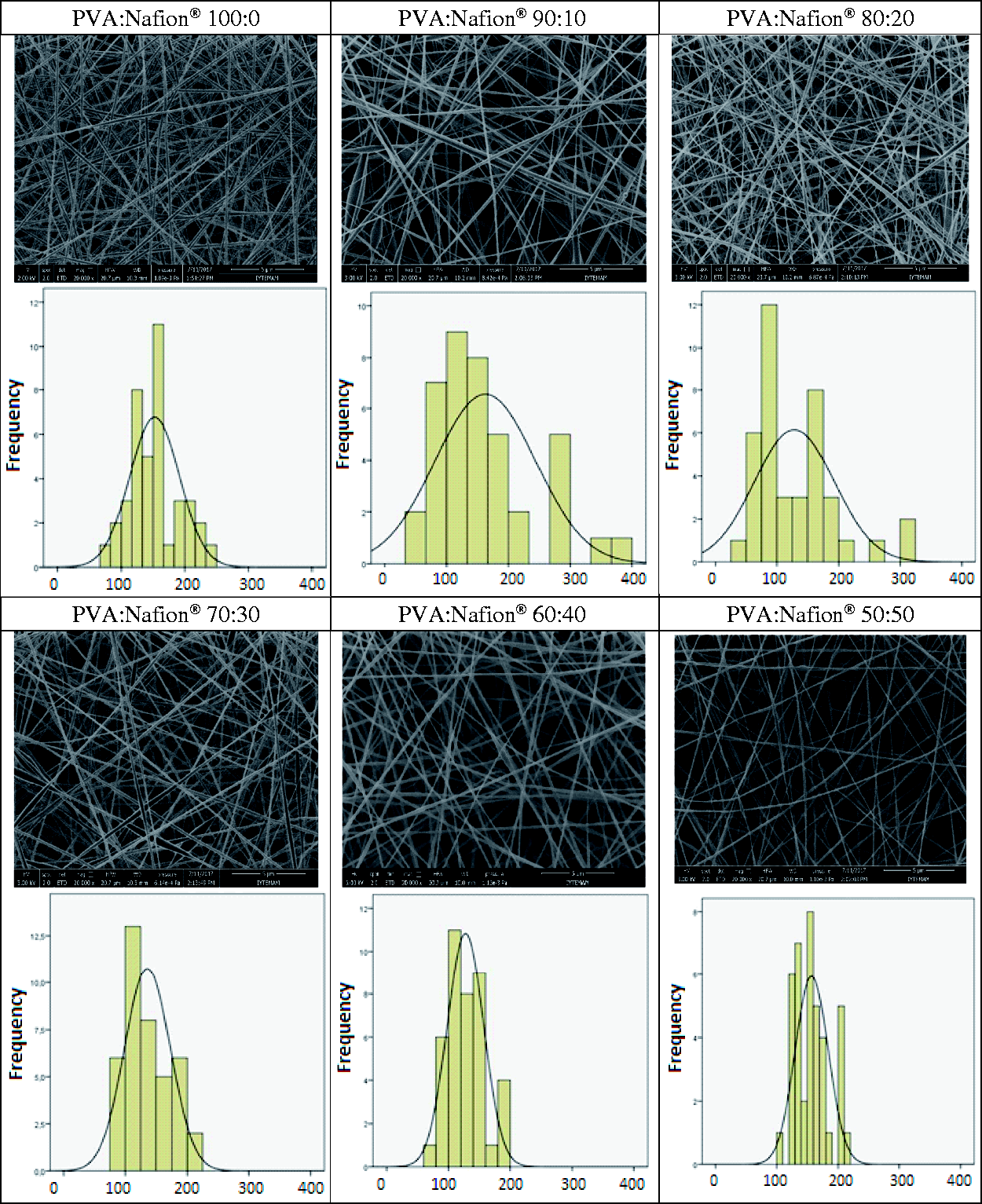
In the case of Nafion®-based nanofibers, the desired state is the maximum amount of Nafion® that can be achieved by the present electrospinning conditions. For this study, the ratio of 50% (v/v) Nafion® solution was found to be the critical threshold. With this ratio, nanofibers with the highest Nafion® amount could be obtained, whereas above this value, it was not possible to produce nanofibers.
FT-IR analysis was applied to neat PVA and PVA/Nafion® nanofibers, and their stabilized forms as shown in Figure 3. For neat PVA and PVA/Nafion® nanofibers, the peak at 1094 cm−1 was assigned to C–O stretching of secondary alcohol groups of PVA [18]. Characteristic peaks of Nafion® at 1234 cm−1, 1054 cm−1 and 982 cm−1 indicating C–F2 stretching, S–O stretching and C–F stretching, respectively [22], were observed for PVA/Nafion® nanofibers, which categorically proved the existence of Nafion® in the resultant nanofibrous membranes.
FT-IR spectra of PVA and PVA/Nafion® nanofibers and their crosslinked-sulfonated forms (s denotes sulfonated).
Considering the electrospinning results, PVA/Nafion® nanofibers produced from 50/50% PVA/Nafion® solutions with higher molecular weight PVA, in which the maximum amount of Nafion® could be incorporated (approximately 33% in weight), were chosen for stability investigation. Additionally, since the type of carrier polymer will also have an effect on the membrane stability, the nanofibrous membranes produced from neat PVA was also tested for comparison to better investigate the behavior of the carrier polymer and its interaction with Nafion®.
Stabilization and sulfonation of PVA and PNA/Nafion® nanofibers
The two different stabilization methods which were applied to PVA and PVA/Nafion® nanofibrous membranes were chemical crosslinking with BTCA and thermal stabilization through heat treatment. Each stabilization method has different mechanisms. BTCA forms ester bonds with hydroxyl groups of PVA [23]. This was proven from the FT-IR analysis results as shown in Figure 3, in which a new vibration band was observed for BTCA crosslinked samples at 1721 cm−1 that can be attributed to C=O stretching of esters. On the other hand, heat treatment stabilizes the structure by increasing the crystallinity through hydrogen bond formation between the hydroxyl groups of PVA [20]. An increase in absorption peak at 1144 cm−1 was observed for PVA-HT samples (Figure 3). It has been reported that the increase in the intensity of this peak shows the increase in crystallinity [24], which proves the effects of heat treatment. Unfortunately, these peaks are not visible in PVA/Nafion® nanofibers because they were obscured by the overlapping peaks of Nafion® in the same region. As shown in Table 1, the mean nanofiber diameter of BTCA crosslinked PVA/Nafion® nanofibers decreased (68 nm) due to the increase in the conductivity of the electrospinning solution as a result of the incorporation of BTCA into the solution. Post heat treatment for thermal stabilization did not have an effect on resultant nanofiber diameter.
After stabilization, sulfonation process was carried out to enhance the formation of sulfonic acid groups on the PVA functional groups of the membranes. FT-IR analysis in Figure 3 showed that new vibration peaks occurred for sulfonated PVA nanofibers (sPVA-BTCA and sPVA-HT) at 1032 cm−1 and 693 cm−1 ascribed to the S=O stretching and S–O stretching of sulfonic acid groups, respectively [25,26]. These results confirmed that the PVA was sulfonated successfully.
Thermogravimetric analysis
TGA curves of PVA and PVA/Nafion® nanofibers were shown in Figure 4. The TGA curve of commercial Nafion® 115 film membrane was also given for comparison. The weight loss below 100℃ corresponds to the dehydration of water within the nanofibers. The neat Nafion® membrane was thermally stable up to nearly 300℃ and showed two degradation steps; the degradation of sulfonic acid groups due to the decomposition of Nafion® side chains [16] started around 300℃ and the breakdown of the polymer backbone [16] started around 450℃. In comparison with Nafion® membrane, neat PVA nanofibers showed 30% weight loss around 200℃ and a sharp decrease was observed due to the elimination reactions followed by the degradation of polymer backbone [27]. On the other hand, PVA/Nafion® nanofibers were found to be thermally stable up to 200℃ and the effect of the stabilization method was not significant in terms of thermal stability. After around 200℃, PVA/Nafion® nanofibers showed a sharp weight loss due to the degradation of the PVA part within the nanofibers. Whereas, having thermal stability up to 200℃ indicated that PVA/Nafion® nanofibrous membranes are suitable for the use in direct methanol fuel cells, since these type of fuel cells have relatively low operating temperatures in the range of 50–120℃ [28].
TGA curves of PVA and PVA/Nafion® nanofibers.
Swelling and weight loss in water
Figure 5 illustrates the swelling behavior of PVA and PVA/Nafion® nanofibers at different temperatures. The swelling values of the Nafion® 115 membrane were also measured as control sample. In general, as the temperature increased, the degree of swelling decreased. It was found that the nanofibrous membranes obtained with 100% PVA have the highest swelling values. When Nafion® was incorporated in the membrane structure, the degree of swelling decreased. However, it was found that the degree of swelling of PVA/Nafion® nanofibers crosslinked with BTCA is significantly higher than that of those stabilized by heat treatment. This is due to the hydrogel character of the chemically crosslinked PVA. On the other hand, PVA/Nafion® nanofibers stabilized by heat treatment were found to have a very low (∼7%) swelling ratio in water compared to the other nanofibrous membranes. After heat treatment, the crystallinity of PVA nanofibers increases. Moreover, Deluca and Elabd [5] reported that heat treatment of PVA/Nafion® mixtures at high temperatures promotes the formation of hydrogen bonds between the hydroxyl groups in PVA and the sulfonic acid groups in Nafion®, rather than between the hydroxyl groups in PVA itself. Therefore, it was considered that the low swelling values obtained from the heat-treated PVA/Nafion® nanofibers resulted from both the increased crystallization of PVA and the interaction between PVA and Nafion®. Although PVA/Nafion® nanofibrous membranes had lower swelling values, it was still higher than the commercial Nafion® 115 membrane.
Water swelling of PVA and PVA/Nafion® nanofibers at room temperature (a), 65 ℃ (b) and 100 ℃ (c).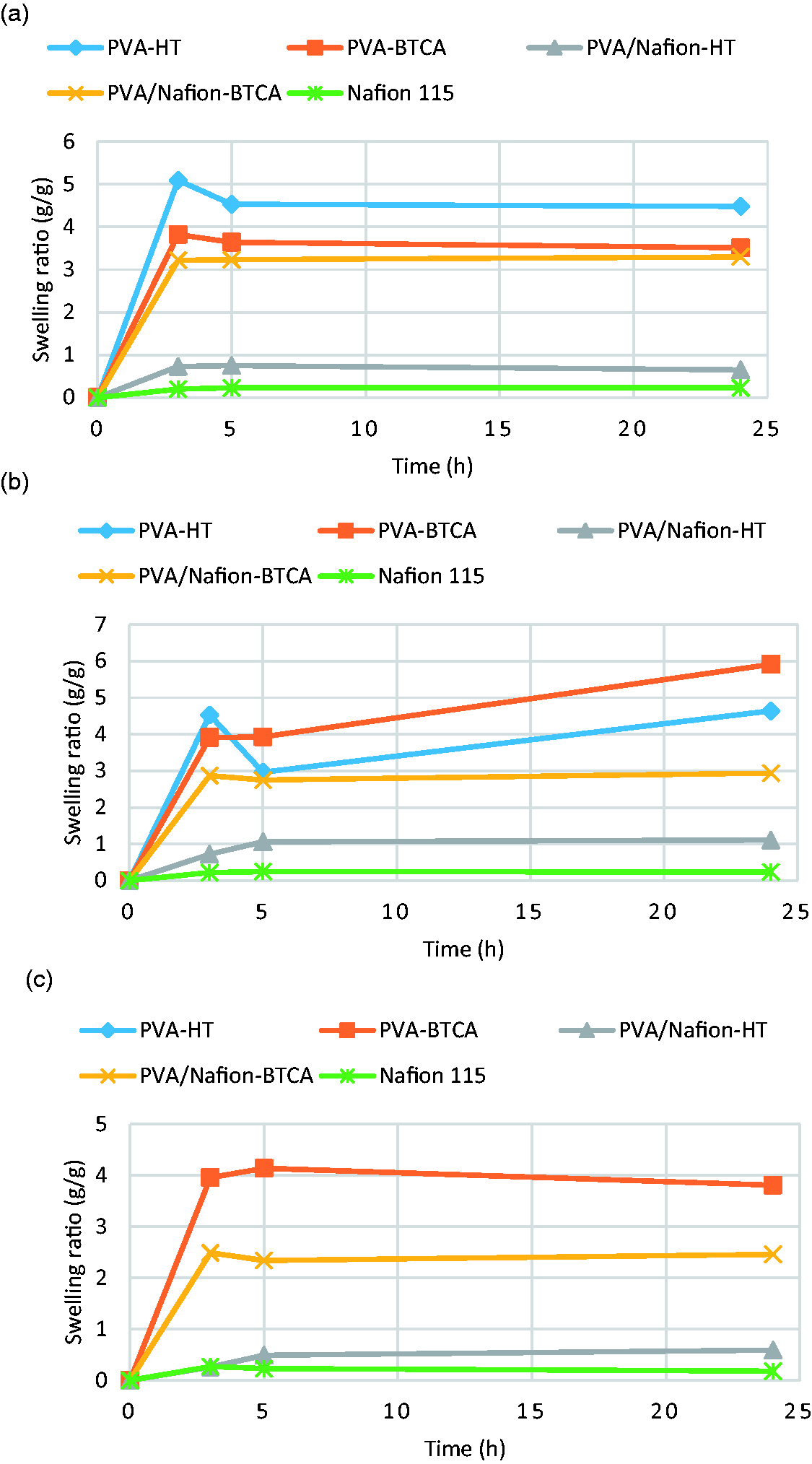
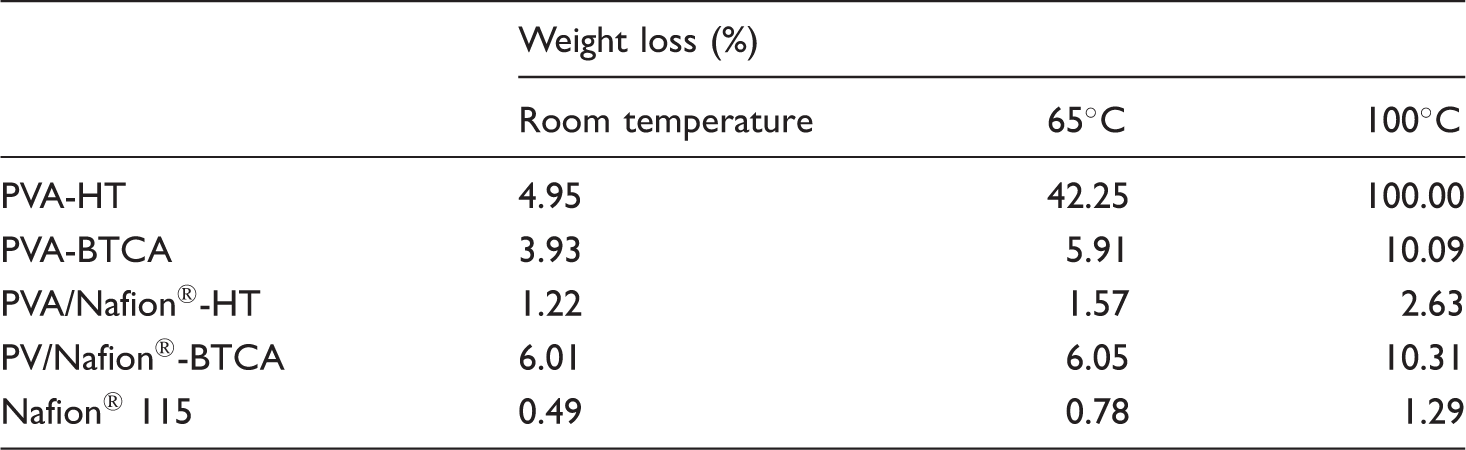
Table 3 shows the weight loss of the nanofibrous membranes after the treatment with water for 24 hours at different temperatures. Also, Figure 6 shows SEM photographs of PVA and PVA/Nafion® nanofibers after the treatment with water at 65℃ for 24 h. As water temperature increased, weight loss generally appeared to increase. When the 100% PVA nanofibers were examined, it was observed that the water resistance of the heat stabilized nanofibers (PVA-HT) was similar to that of BTCA crosslinked samples at room temperature, but with increasing temperature, PVA-HT samples showed very high levels of weight loss. SEM photographs show that the PVA-HT nanofibers are completely degraded after hot water treatment and became a film-like material. PVA-HT nanofibers completely dissolved at 100℃. On the contrary, for PVA/Nafion® nanofibers, it was seen that the water resistance of the samples stabilized by heat treatment was much better. The high resistance is believed to be due to the formation of hydrogen bonds between the hydroxyl groups of PVA and the sulfonic acid groups of Nafion® as described above. As seen in the SEM photographs, the nanofibrous structure of PVA/Nafion®-HT nanofibers was completely preserved after hot water treatment, and the resultant weight losses are close to those of commercial Nafion® 115 membrane. On the other hand, BTCA crosslinked PVA/Nafion® and PVA nanofibers showed similar weight loss, which is higher than PVA/Nafion®-HT nanofibers. After hot water treatment of BTCA-crosslinked nanofibers, a swollen, conjugated and beaded nanofibrous structure was observed to be formed.
Morphology of PVA and PVA/Nafion® nanofibers after water treatment at 65℃ for 24 h. Weight loss in water.
Weight loss in methanol
The methanol resistance of a membrane to be used in direct methanol fuel cells must be high. The concentration of methanol used in fuel cells can generally range from 0.5 to 2 M [29]. In this study, methanol resistance tests were carried out at a concentration of 5 M in order to expose the membranes to extreme conditions. Figure 7 shows the weight loss of PVA and PVA/Nafion® nanofibers after methanol treatment at different temperatures. Also, Figure 8 shows SEM photographs after methanol treatment at 65℃ for 72 h. Thermally stabilized PVA nanofibers (PVA-HT) showed 20% weight loss after 72 hours of treatment at room temperature. However, when the temperature rose, the membrane was almost completely dissolved. In the SEM photographs, it was seen that the nanofibrous structure completely deteriorated and formed a film-like structure. The weight loss of PVA nanofibers crosslinked with BTCA in methanol was measured as 10% at room temperature and 25% at 65℃ after 72 hours of treatment. PVA/Nafion®-BTCA nanofibers showed similar weight losses. Both nanofibers transformed into a highly swollen, conjugated and beaded structure after the treatment. In PVA/Nafion®-HT nanofibers, no weight loss was observed in methanol. Methanol resistance was found to be as good as commercial Nafion® 115 membrane. Moreover, it was observed that the nanofibrous structure was also protected.
Weight loss of PVA and PVA/Nafion® nanofibers in methanol at room temperature (a) and 65℃ (b). Morphology of PVA and PVA/Nafion® nanofibers after methanol treatment at 65℃ for 72 h.

Oxidative stability
Figure 9 shows the weight loss of PVA and PVA/Nafion® nanofibers after treatment with the Fenton's reagent. Figure 10 shows SEM photographs after the treatment. The weight loss of PVA nanofibers and PVA/Nafion®-BTCA nanofibers gradually increased with increasing time. PVA nanofibers were completely disintegrated after 24 hours of treatment. This suggests that the use of PVA nanofibers alone in fuel cells may be problematic. Also PVA/Nafion®-BTCA nanofibers showed a weight loss of more than 70% after 24 hours of treatment and the nanofibrous structure was appeared to be destroyed after the process. On the other hand, it was shown that PVA/Nafion®-HT nanofibers became flat and conjugated but the nanofibrous structure was preserved. The oxidative stability of the PVA/Nafion®-HT nanofibers was found to be close to that of commercial Nafion® 115 membrane.
Weight loss of PVA and PVA/Nafion® nanofibers in Fenton's reagent at 65℃. Morphology of PVA/Nafion® nanofibers after treatment with Fenton's reagent at 65℃ for 24 h (morphology of PVA nanofibers could not be given since they were dissolved completely after the treatment).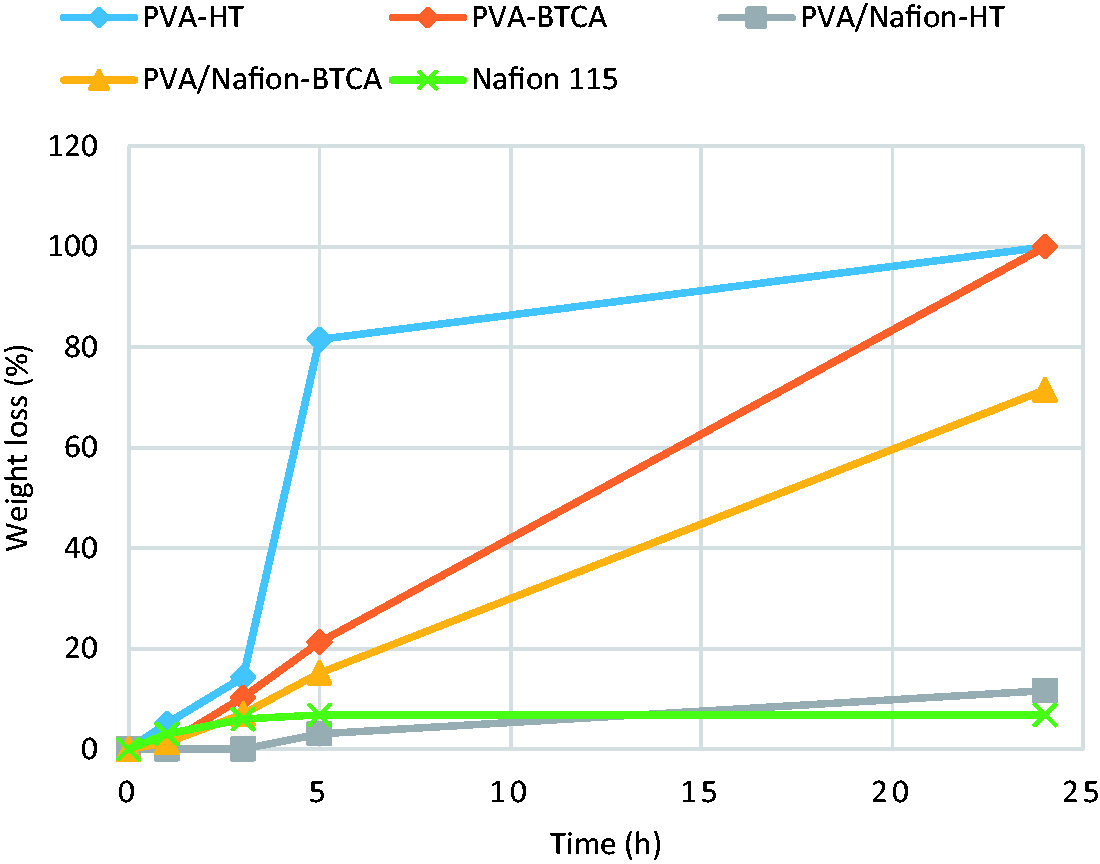

Ion-exchange capacity
Ion exchange capacity of PVA and PVA/Nafion® nanofibers.

Conclusions
Electrospinning of PVA/Nafion® nanofibers and their thermal and chemical stability were investigated to assess the potential use in direct methanol fuel cells.
Bead-free Nafion®-based nanofibers were successfully produced by electrospinning with higher molecular weight PVA. The highest possible Nafion® ratio was obtained by 50/50% PVA/Nafion® solution mixture. The effect of the Nafion® solution ratio on resultant nanofiber diameter was found to be insignificant. PVA/Nafion® nanofibers with the highest amount of Nafion® were selected for stability investigations and pure PVA nanofibers were also tested for comparison. The produced nanofibers were stabilized by two different stabilization methods; chemical crosslinking with BTCA and thermal stabilization. After stabilization, PVA functional groups in the membranes were sulfonated. The success of the stabilization and sulfonation process was proven by FT-IR analysis.
TGA results showed that crosslinked/sulfonated PVA/Nafion® nanofibers have similar thermal stability with commercial Nafion® 115 film membrane up to 200℃, which indicated the suitability for use in direct methanol fuel cells. On the other hand, the thermal stability of neat PVA nanofibers was observed to be lower.
Neat PVA nanofibers were found to be unsuitable for use in direct methanol fuel cells due to the lower thermal and chemical stability. The nanofibrous structure deteriorated after water and methanol treatment, and moreover they completely disintegrated after Fenton's reagent test.
The thermally stabilized PVA/Nafion® nanofibrous membrane was found to have the highest water, methanol and oxidative stability within all nanofibrous membranes. The nanofibrous structure of PVA/Nafion®-HT nanofibers was preserved while the other membranes became a film-like material. Moreover, the stability characteristics were found to be very similar to those of commercial Nafion® 115. The high resistance of PVA/Nafion®-HT nanofibers was thought to be due to the hydrogen bond interaction between the hydroxyl groups of PVA and the sulfonic acid groups of Nafion®. Although PVA/Nafion®-HT nanofibers had the highest stability, the IEC values were found to be lower.
In this study, the stability and preservation degree of the nanofibrous structure of PVA/Nafion® nanofibers under simulated fuel cell conditions were investigated and sufficient stability was proven. The water swelling value was found to be higher than that of the commercial Nafion® 115 film membrane. The proton conductivity and methanol permeability of thermally stabilized PVA/Nafion® nanofibers in relation to production parameters and membrane thickness will be investigated in further studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ege University Scientific Research Projects Coordination Unit (Project Number: 16-MÜH-042).
