Abstract
The tissue-engineered skin has emerged as a plausible alternative approach to the traditional wound dressing owing to its inherent advantages. Further, in tissue engineering applications, fibrous scaffold obtained through textile technologies is widely attractive. The present study focused on the fabrication of electrospun textile polyurethane wound dressing scaffold incorporated with grape extract. The fabricated composites showed smooth as well as reduced fiber (730 ± 127 nm) and pore (873 ± 51 nm) diameter than the control polyurethane (fiber diameter –890 ± 117 and pore diameter –1064 ± 74 nm) as revealed in the scanning electron microscopy. The formation of hydrogen bond in Fourier transform infrared spectroscopy revealed the interaction between the polyurethane and grape. The addition of grape enhanced the wettability behavior (86° ± 2) and the surface roughness (469 nm) of the polyurethane membrane. Thermal gravimetric analysis and mechanical testing revealed the enhancement of thermal and tensile strength with the incorporation of the grape into the polyurethane matrix. The in vitro blood compatibility and cytocompatibility studies revealed enhanced antithrombogenicity behavior and the non-toxic nature to human dermal fibroblast cells for the fabricated composites than the pristine polyurethane. Hence, the addition of grape into the polyurethane matrix had enhanced the physicochemical characteristics and biocompatibility parameters which could promote this candidate as a valid alternative for skin tissue engineering regeneration.
Keywords
Introduction
Skin is the largest organ of the human body which plays an important role in many physiological functions [1,2]. It acts as the interface between the body and the environment for self-renewing and self-repairing [2,3]. It shields the human from the toxins and microorganism present in the environment. The other functions of the skin were sensory stimuli detection and surveillance of immune [1,2]. Generally, the skin is made of epidermis and dermis. The epidermis consists of layers of keratinocytes, while the dermis underlies the epidermis which is made of several extracellular matrix (ECM) proteins. The ECM proteins were collagen, elastin, glycosaminoglycans and fibroblast cellular constituents [1]. The wounds caused to the epidermis could heal on their own but unfortunately any damage to the dermis cannot heal quickly [3]. Dermal wounds caused by any external factors were commercially treated utilizing autografts, allografts and xenografts. However, the above mentioned treatments were limited in the clinical applications because of their certain limitations. The limitations were an insufficient supply of donor sites and the risk of disease transmission [4]. With the growth in the technology, various advanced and improved approaches were implemented to achieve the better regeneration of the damaged skin tissue [5]. Recently, the tissue-engineered skin has emerged as a plausible alternative approach to the traditional wound dressing owing to its inherent advantages. Tissue engineering utilizes culturing of cells in a nanofibrous and microfibrous structure which resembles the three-dimensional organization of the native tissue and thus facilities the new tissue growth [4]. Scaffolds made from natural or artificial polymers were widely attractive in the tissue engineering application [6].
In tissue engineering applications, nanofibrous scaffold obtained through textile technologies is widely attracted owing to its versatility. The nanofibers obtained through textile-based technologies could be easily controlled by the varying parameters such as fiber size, orientation, pore size, geometry and surface morphology. The various techniques utilized for developing the scaffolds were electrospinning, phase-separation, freeze drying, self-assembly, etc. [7]. In a recent study, production of nanofibers from pressurized gyration technique has attracted a wide audience. The nanofibers obtained from this technique have been used for drug delivery, tissue engineering, filtration and wound-healing applications. Further, it also provides better scalability and productivity [8]. Among these techniques, the electrospinning technique which is a textile-based technology was used to fabricate the scaffold in this study. Electrospinning is a simple, cost effective and versatile technique which utilizes high voltage supply to the polymer solution to convert them into smooth fibers with diameters ranging from micrometre to nanometre ranges [9]. The main advantage is that fibers fabricated from electrospinning possess high surface area to volume ratio and high porosity [10]. Further, the nanofibers could mimic the native human ECM structure and support the cell adhesion and proliferation of new tissue growth. Several natural and synthetic polymers were utilized in the fabrication of nanofibers via electrospinning technique which are widely sought in tissue engineering applications such as wound dressing, cartilage, bone, vascular graft, cardiac valves, etc. [11]. In this research, Tecoflex EG-80A which is a polyether-based segmented polyurethane (PU) of medical grade was utilized to fabricate the wound dressing scaffold. Generally, PU possess biodegradable, biocompatible, good mechanical properties, better oxidation and thermal stability which makes them attractive choice in the medical applications [12,13]. Although there are several alternatives like polyglycolic acid, polylactic acid, poly(lactic-co-glycolic acid), polyvinyl alcohol and polycaprolactone (PCL), PU evolved as a good textile fiber because of its flexibility and resilience [14–17].
Several researchers are exploring natural substances incorporated membranes (fruits, honey, propolis, plant oils) for wound-healing applications. Balaji et al. developed PU wound dressing scaffold added with honey and papaya. The electrospun composites exhibited better hemocompatible properties insinuating its potential for treating burn injuries [18]. Kim et al. fabricated electrospun PU/propolis scaffold and observed that the incorporation of propolis into PU showed good cell compatibility suggested its suitability in wound-dressing applications [19]. In another study, Mani et al. developed a fibrous wound dressing membrane based on PU added with neem oil. The fabricated composites showed better physicochemical properties and blood compatibility behavior suggesting it as a potential candidate for wound-dressing applications [20]. Finally, Amna et al. fabricated electrospun PU scaffold added with olive oil/copper oxide. It was reported that the fabricated composites showed non-toxic to fibroblast cells, thereby concluding it as a potential candidate for wound dressing [21]. In this scenario, it would be interesting to explore the role of grape extract in wound-healing applications. In this research, the PU was added with grape extract to improve its biocompatibility properties. Fresh grape extract consists of 70 to 80% water and many organic and inorganic compounds such as sugars (glucose and fructose), organic acids (tartaric, malic and small amount of citric), phenolic compounds (anthocyanins and tannins), nitrogenous compounds (amino acids, peptides and proteins), aroma compounds (2-methoxy-3-isobutyl pyrazine, 4-vinylguaiacol and 4-vinylphenol and terpenes), minerals (sodium, potassium, phosphates, iron, chloride and sulfate) and pectic substances [22]. The chemical composition of grapes is indicated in Figure 1. There has been enormous literature signifying the diverse applications of grapes in the biomedical field. Few studies have been reported the antioxidant, anticancer, antimicrobial and anti-inflammation activity of the grapes which reported phenolic constituents of grapes as a putative candidate [23–25]. However, there is no single study comprising the grapes (crude extract) into the polymer matrix. In this work, electrospinning is used which is the cost effective and versatile technique for producing nanofibers. Further, the constituent added is a natural grape extract which is commonly available in all seasons globally. Hence, this fabricated electrospun composite seems to be an economically viable and effective alternative for wound dressing. The physicochemical properties, blood compatibility assessments and cytocompatibility studies of the fabricated PU and PU/grapes composites were determined to examine its effectiveness for skin tissue engineering.
Chemical composition of grapes.
Materials and methodology
Materials
The medical grade Tecoflex EG-80A PU was purchased from Lubrizol and dissolved in dimethylformamide (DMF) solvent (Sigma-Aldrich, UK). The Australian grapes were obtained locally from AEON, Malaysia. The chemicals, phosphate buffered saline (PBS) and sodium chloride physiological saline (0.9% w/v) utilized in the coagulation assays, were purchased from Sigma-Aldrich, Kuala Lumpur, Malaysia. The reagents for activated partial thromboplastin time (APTT) and prothrombin time (PT) assay, such as rabbit brain activated cephaloplastin, calcium chloride (0.025 M) and thromboplastin (Factor III), were obtained from Diagnostic Enterprises, Solan, India.
Preparation of grape extract
Initially, the fresh grapes were washed with clean water and then air dried. After drying, it was grinded with the aid of kitchen blender until the juice was obtained. Finally, the juice was filtered to obtain the grape extract which is stored in a refrigerator for further experiments.
Preparation of solution
PU solution (9 wt%) was prepared by mixing 0.450 g in 5 ml of DMF overnight at room temperature. Similarly, the grape solution (4 wt%) was obtained by mixing 120 µl in 3 ml of chloroform and stirred for 2 h at room temperature. The PU/grapes homogeneous solution was prepared at a ratio of 8:1 v/v and stirred for 1 h maximum at room temperature to ensure even dissolution.
Electrospinning process
Electrospinning process converted the homogeneous solution into scaffolds and carried out in room temperature with humidity of 55%. Initially, 10 ml of prepared homogeneous solution were taken separately inside the plastic syringe of 18-G stainless steel needle (outer diameter of 1.28 mm and the inner diameter of 0.92 mm, Unicontrols Singapore Pte. Ltd) and placed on the syringe pump of the equipment. The solutions were electrospun at a flow rate of 0.5 ml/h with the applied voltage of 10.5 kV. The collector drum used was static which was placed and collector distance was set at 20 cm away from the needle. The obtained fibers deposited on the aluminum foil were detached carefully and was dried under vacuum at room temperature to remove any residual DMF in the fibers. The fabricated PU and PU/grapes composites are shown in Figure 2.
Image of electrospun (a) PU and (b) PU/grapes composite.
Physicochemical characterization
The fabricated PU and composite membranes were subjected to several characterization techniques. Scanning electron microscope images of the electrospun membranes were obtained using the Hitachi Tabletop scanning electron microscope to investigate the morphology of the fibers. Image analysis was used to measure fiber and pore diameter from the obtained images. The pore size was measured from the inter-fiber spaces [26,27]. Further, the porosity of the electrospun membranes was determined using density bottle method. Next, the samples were inspected between the wavelength of 600 and 4000 cm−1 in Fourier transform infrared spectroscopy (FTIR) unit to analyze the characteristics bands present in the electrospun membranes. The contact angle measurements of the electrospun membranes were determined using video contact angle (VCA) optima contact angle in order to predict their wettability nature. Initially, a small cut sample was placed on the measuring surface and a water droplet with a size of 0.5 µl was deposited on the cut membranes. After 2 s, the static image of the droplet was captured using high resolution camera, and the manual contact angle was determined via computer integrated software. Thermal behavior of the electrospun membranes was performed to determine their thermal degradation behavior. To begin, 3 mg of the cut membrane was placed on the aluminum pan and the heating was done under a nitrogen atmosphere at a rate of 10℃/min with temperature range between 30℃ and 1000℃, respectively. AFM analysis of the electrospun membranes was performed under the normal room temperature using AFM equipment. Samples were scanned in 20 µm × 20 µm size, and the images 256 × 256 pixels were captured using JPJSPM software. Using the uniaxial testing machine, the tensile strength was measured with a load cell of 500 N at a strain rate of 5 mm/min. The specimen was tested according to ASTM D882-10 with sample dimension of 40 mm × 15 mm.
Coagulation assays
APTT and PT assays
APTT and PT assays were used to investigate the anticoagulant nature of the electrospun membranes. APTT and PT assays were used to predict the intrinsic and extrinsic pathways of the coagulation cascade. Prior to performing these assays, the samples were cut and incubated at 37℃ for 30 min in physiological saline. Both assays were carried out using a similar protocol as explained in the previously published article [28].
Hemolysis assay
Hemolysis assay was carried out to assess the compatibility of the red blood cells with the electrospun membranes. A small piece of sample was cut and was soaked in physiological saline at 37℃ for 30 min. The protocol followed in this assay was explained in the previous research article [28]. Further, the hemolytic percentage or index was measured.
Cell culture and cell viability studies
The cell viability of the electrospun membranes was determined using human dermal fibroblast (HDF) cells which were cultured in the medium of DMEM supplemented with 10% fetal bovine serum and 5% antibiotic penicillin. The culture medium was incubated at 37℃ under 5% carbon dioxide (CO2) and the medium was refreshed for every three days. Prior to cell seeding, the fabricated electrospun scaffolds were cut into small pieces and placed in 96-well plates. Further, they were sterilized with 75% alcohol and cleaned with PBS. Then, HDF cells with a density of 10 × 103 cells/cm2 seeded on each well in the samples were placed and kept in CO2 incubator for 72 h. After 72 h culture, the medium was added with 20% of MTS and placed in the 5% CO2 for 4 h. After 4 h, absorbance was measured at 490 nm through spectrophotometric plate reader to determine their viability rates.
Statistical analysis
All experiments were carried out three times independently and the statistical significance was determined through an unpaired t-test. The obtained results from all experiments are conveyed as mean ± SD. A representative of three images is shown for qualitative experiments.
Result and discussion
Morphological analysis
Figure 3(a) and (b) indicates the surface morphology of the electrospun pure PU and PU/grapes fibrous membrane. The electrospun membranes showed smooth, ultrafine, non-woven and bead free fibers indicated in the scanning electron microscopy (SEM) images. Further, it was observed that the fiber diameter of the electrospun PU/grapes fibrous membrane is reduced compared to the PU fibers. The average fiber diameter of the electrospun PU/grapes fibrous membrane was found to be 730 ± 127 nm and for the pristine PU, the average fiber diameter was observed to be 890 ± 117 nm. The graphical fiber distribution curve is indicated in Figure 4(a) and (b).
SEM images of (a) PU membrane and (b) PU/grapes composites. Fiber diameter distribution of (a) PU membrane and (b) PU/grapes composites.

Further, it was evident from the SEM image in which PU flat morphology was changed to round shape in PU/grapes composite. The presence of grape on the surface can be attributed to this morphology change. Shi et al. reported that reducing the polymer concentration could result in the small diameter of fibers [29]. In the fabricated PU/grapes composites, the reduction of fiber diameter might be due to the decreasing polymer concentration by the addition of grapes into the PU matrix. Jaganathan et al. fabricated electrospun fibrous scaffold based on PU scaffold added with corn and neem oil fibers. It was noted that the incorporation of corn oil and neem oil into the matrix of PU caused a reduction in the fiber diameter and their observations correlate with our findings. Further, the developed PU/corn oil and PU/corn/neem oil scaffold exhibited enhanced fibroblast cell adhesion and proliferation than the pristine PU [28]. In our study, the incorporation of grapes into the PU resulted in the reduction of fiber diameter which might help in improved fibroblast cell adhesion and proliferation of new tissue growth.
Pore size investigation
The density bottle method was used to determine the porosity of the electrospun membranes. The obtained results showed that the electrospun PU/grapes membranes showed an average porosity of 73%, while the pristine PU showed 76%. Further, the pore size measurement of the electrospun PU and PU/grapes composites was calculated through Image J. It was observed that the incorporation of grapes into the PU matrix resulted in the pore size reduction. The average pore size of pristine PU was observed to be 1064 ± 74 nm and for PU/grapes composites, it showed pore size in the range of 873 ± 51 nm. Prabhakaran et al. developed a fibrous scaffold based on poly (DL-lactide-co-glycolide) blended with gelatin fibers. It was revealed that the incorporation of gelatin into the poly (DL-lactide-co-glycolide) resulted in the reduction of pore size which correlates with our findings. Further, they suggested that the reason for the reduction in the pore size was due to the smaller fiber diameter exhibited by the fabricated membranes [30] which is similar to our observations. Further, Kim et al. reported that the fibrous membranes with a pore size in the range of 500–1000 nm showed better protection against the bacteria [31]. Our pore size of the fabricated composite was observed to be within this range suggesting it as a potential membrane for protection against the bacterial invasion.
FTIR analysis
The interaction between the PU and the grapes was determined through the FTIR spectra as indicated in Figure 5.
(a) IR spectrum of PU membrane and PU/grapes composites and (b) peak intensity increase in the developed composites.
In the spectra of PU, the NH stretch mode was observed at peak of 3328 cm−1 and the peaks at 1596 cm−1 and 1530 cm−1 represent the vibration of the NH mode. The sharp bands seen at 1702 cm−1 and 1730 cm−1 indicate CO stretch group and the peaks at 1220 cm−1 and 1105 cm−1 denote the carbonyl group corresponding to the alcohol group. The other absorption bands observed at 2938 cm−1 and 2853 cm−1 were attributed to the CH stretch group and the peak at 1413 cm−1 indicated its vibration [28,32]. In the absorption band of PU/grapes fibrous membranes, there were no new peaks but the peak intensity was observed to be increased due to hydrogen bond formation as shown in Figure 5(b) [33]. As discussed in the Introduction section, the grapes are reported to contain 70 to 80% water and many organic and inorganic compounds which could have a wide range of OH and CH molecules. Hence, the strong increase in the intensity of the peak at 3328 cm−1 and 2938 cm−1 may be due to the reaction of grape extract's OH and CH groups with the molecules of pristine PU. Further, it has been reported that the combining of two different macromolecules will yield a formation of strong inter-hydrogen bonds than bonding between the molecules of the same polymer [33]. In PU/grapes composites, the strong inter hydrogen bond formation was because of the combination of OH and CH molecules in the grapes with the molecules of the pristine PU.
Wettability analysis
The wettability is a vital criterion in deciding the application of the novel materials [34–36]. The contact angle measurements of the pure PU and PU/grapes fibrous membranes were presented and their images are shown in Figure 6(a) and (b). The mean contact angle of neat PU fibrous mat was found to be 100° ± 1 indicating hydrophobic nature, while the PU/grapes fibrous membranes showed 86.33° ± 2 indicating a hydrophilic behavior. The presence of various constituents in the grapes might be responsible for the hydrophilic nature exerted by the scaffold. The observed findings correlate with the FTIR analysis in which the band at 3328 cm−1 is widened in the fabricated composite compared to pristine PU which corresponds to the OH group imparting wettability to the fabricated scaffold. Unnithan et al. prepared a PU skin dressing scaffold blended with cellulose acetate and zein. It was found that the PU matrix added with cellulose acetate and zein displayed enhanced hydrophilic nature compared to the pristine PU which resembles our observations. They also observed that the hydrophilic surfaces exhibited enhanced adhesion and proliferation of fibroblast cells [37].
Contact angle images of (a) PU membrane and (b) PU/grapes composites.
Hence, in our study, the incorporation of grapes enhanced the wettability of the PU membrane which will be conducive to the fibroblast adhesion and proliferation for new tissue growth.
Thermal behavior
In tissue engineering applications, the thermal analysis of the developed composites performed to analyze their degradation behavior. Further, it was utilized to determine the thermal stability, the existence of added constituents and integrity of the developed materials [38]. The thermal behavior of the electrospun PU and PU/grapes fibrous membranes were indicated in Figure 7.
TGA analysis of PU membrane and PU/grapes composites.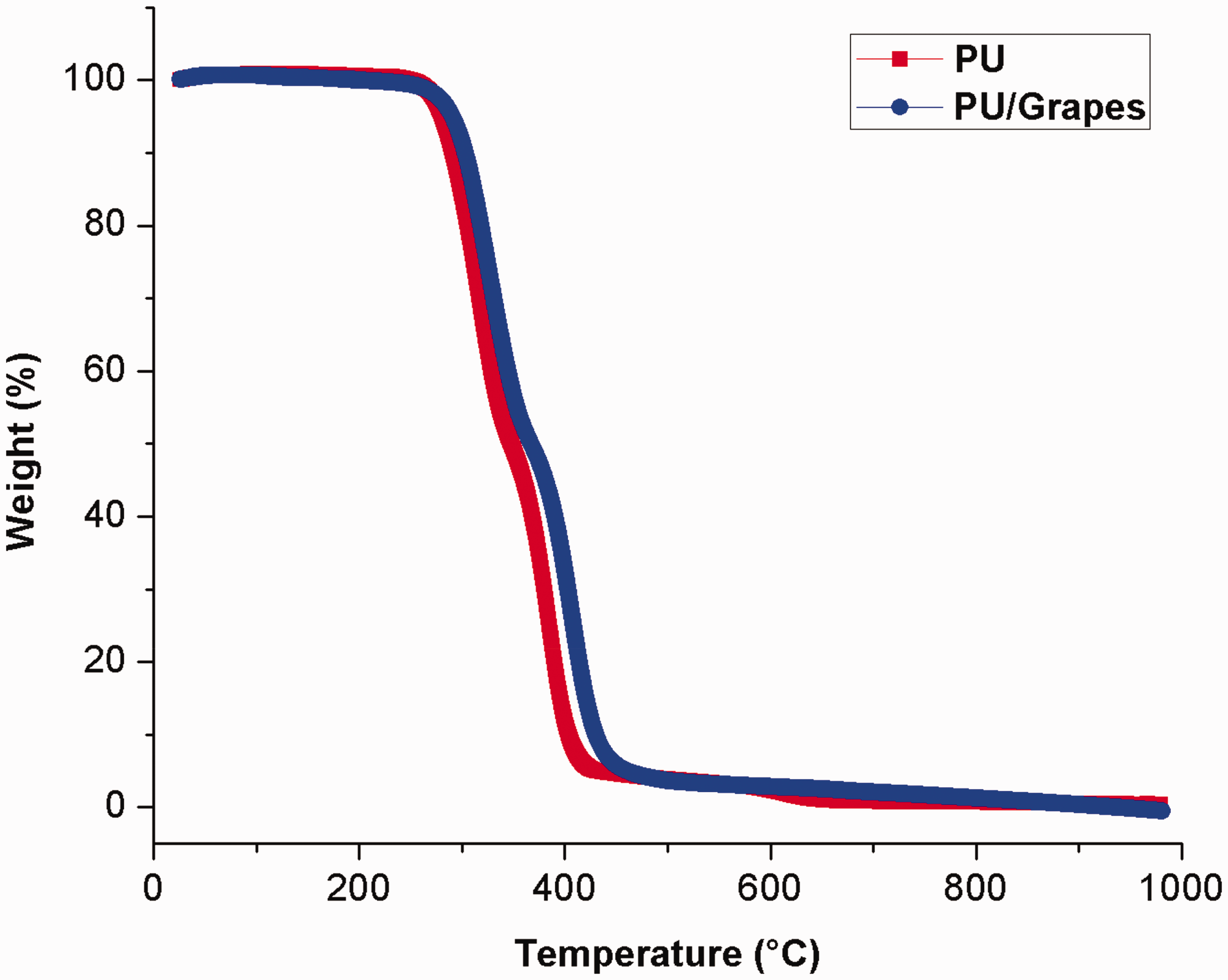
The pristine PU membrane showed initial decomposition begins at 276℃ whereas in case of PU/grapes fibrous membranes, the initial degradation temperature was increased to 289℃ indicating its enhanced thermal stability on adding the grapes into PU. As discussed previously, Jaganathan et al. fabricated PU scaffold with the addition of corn oil and neem oil fibers and they observed PU scaffold added with corn and oil neem oil showed enhanced thermal behavior compare to the pristine PU which resembles our findings [28]. Gibson et al. fabricated PAN fiber and studied the thermal resistance for various fiber diameters. It was observed that the PAN fiber with smaller fiber diameter tends to increase the thermal resistance [39]. Further, it has been reported that the smaller fiber diameter favors the decrease in the pore size [30]. Hence, reduced fiber and pore size of the developed PU/grapes composite might be attributed for the improvement in the thermal stability. Moreover, Figure 8 illustrates the derivative weight loss curve for the electrospun PU and PU/grapes fibrous membranes. The pure PU exhibited three weight loss peaks in which the first weight loss occurs at 223℃ and ends at 348℃, the second loss begins at 348℃ and ends at 446℃ and the third loss begins at 557℃ and ends at 684℃ respectively. These weight loss peaks are due to the decomposition of the hard and smooth segments of the PU. In the case of electrospun PU/grapes fibrous membranes, it exhibited only two losses indicating the lower weight loss compared to the pristine PU. The first loss begins at 228℃ and ends at 368℃ and the second loss starts at 368℃ and ends at 523℃ respectively. Further, it was observed that the peak intensity of the electrospun PU/grapes fibrous membranes was decreased than the PU membrane which confirms the interaction of the PU matrix with grapes. Further, in both PU and PU/grapes composites, the obtained weight loss peak was found to be different which was identified by the peak shift in temperature scale. In first loss peak, the end temperature seen at 348℃ in pristine PU was shifted to 368℃ in PU/grapes composites and in the second loss peak, the end temperature observed at 446℃ in pure PU was moved to 514℃ in PU/grapes composites. Hence, the peak shift clearly indicates the presence of grapes constituents (as discussed in the introduction section) in the PU matrix.
Weight residue percentage of PU membrane and PU/grapes composites.
Surface roughness measurements
The measured Ra for the electrospun PU and PU/grapes fibrous membranes are illustrated in Figure 9(a) and (b). It was observed that the incorporation of grapes improved the surface roughness of the pristine PU. The average roughness of the pristine PU was found to be 313 nm and the average roughness of the electrospun PU/grapes fibrous membranes was observed to be 469 nm (Ra) respectively. The improvement in the surface roughness on adding grapes was evident from the morphology of the fibers. The fibers of the composite were rounded and also rougher compared to the pristine PU. Jaganathan et al. fabricated PU scaffolds added with mustard oil. They observed the improved surface roughness and reported this might due to the presence of mustard constituents in the PU matrix [40]. In our developed composites, the improved surface roughness was might due to the grapes constitutes present in the PU matrix. Prasad et al. prepared PCL/chitosan fibrous scaffold for skin tissue engineering. It was observed that the prepared PCL/chitosan scaffold exhibited an increase in surface roughness and it showed the enhanced density of the fibroblast cells compare to the pristine PCL [41]. In our study, the addition of grapes into the PU matrix facilitated the increase in surface roughness which may be conducive for skin tissue engineering.
AFM images of (a) PU membrane and (b) PU/grapes composites.
Tensile strength analysis
The mechanical testing for developed PU and PU/grapes composites were performed through uniaxial a tensile testing machine and the results are depicted in Figure 10. The results of mechanical testing showed that the tensile strength of PU matrix was enhanced with the addition of grapes. The tensile strength of the electrospun PU/grapes composites was found to be 19.55 MPa, while for pristine PU, it exhibited a tensile strength of 7.12 MPa respectively. Unnithan et al. fabricated PU scaffold added with emu oil fibers for wound-dressing applications. It was observed that the addition of emu oil resulted in the enhancement of the tensile strength of the pristine PU. They concluded the reason for the enhancement was due to the formation of hydrogen bond between the molecules of PU and emu oil [33]. As reported in FTIR study, the formation of strong hydrogen bond in the fabricated composites might have favored the enhancement of the mechanical strength. Certain studies reported that the smaller fiber diameter favors the enhancement of the tensile strength [42,43]. Further, Prabhakaran et al. observed that their fabricated fibrous scaffold exhibited reduced pore size due to the smaller fiber diameter [30]. The developed PU/grapes composites showed reduced fiber and pore diameter which might had favored the improved tensile strength.
Mechanical testing of PU membrane and PU/grapes composites.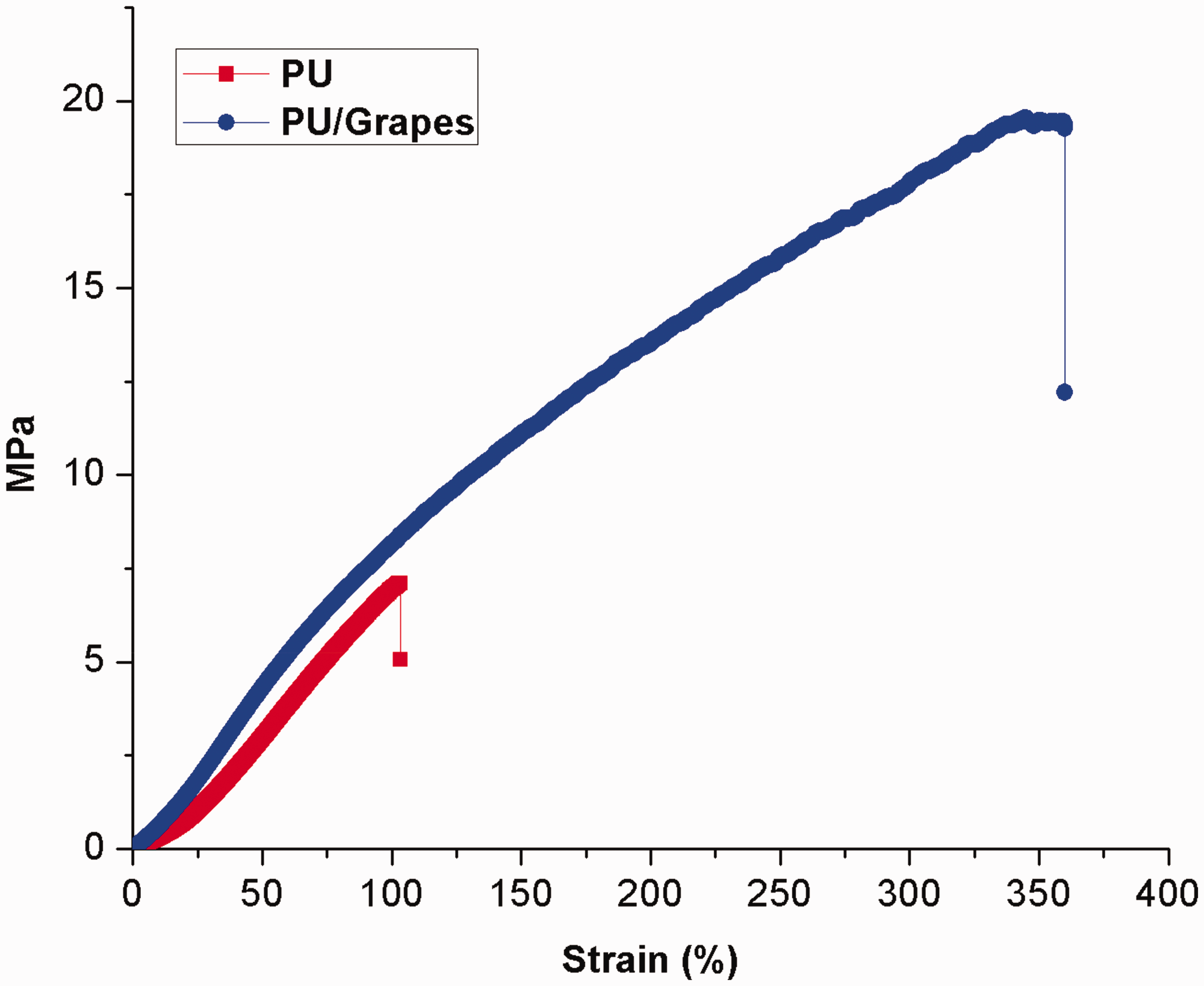
Blood compatibility investigation
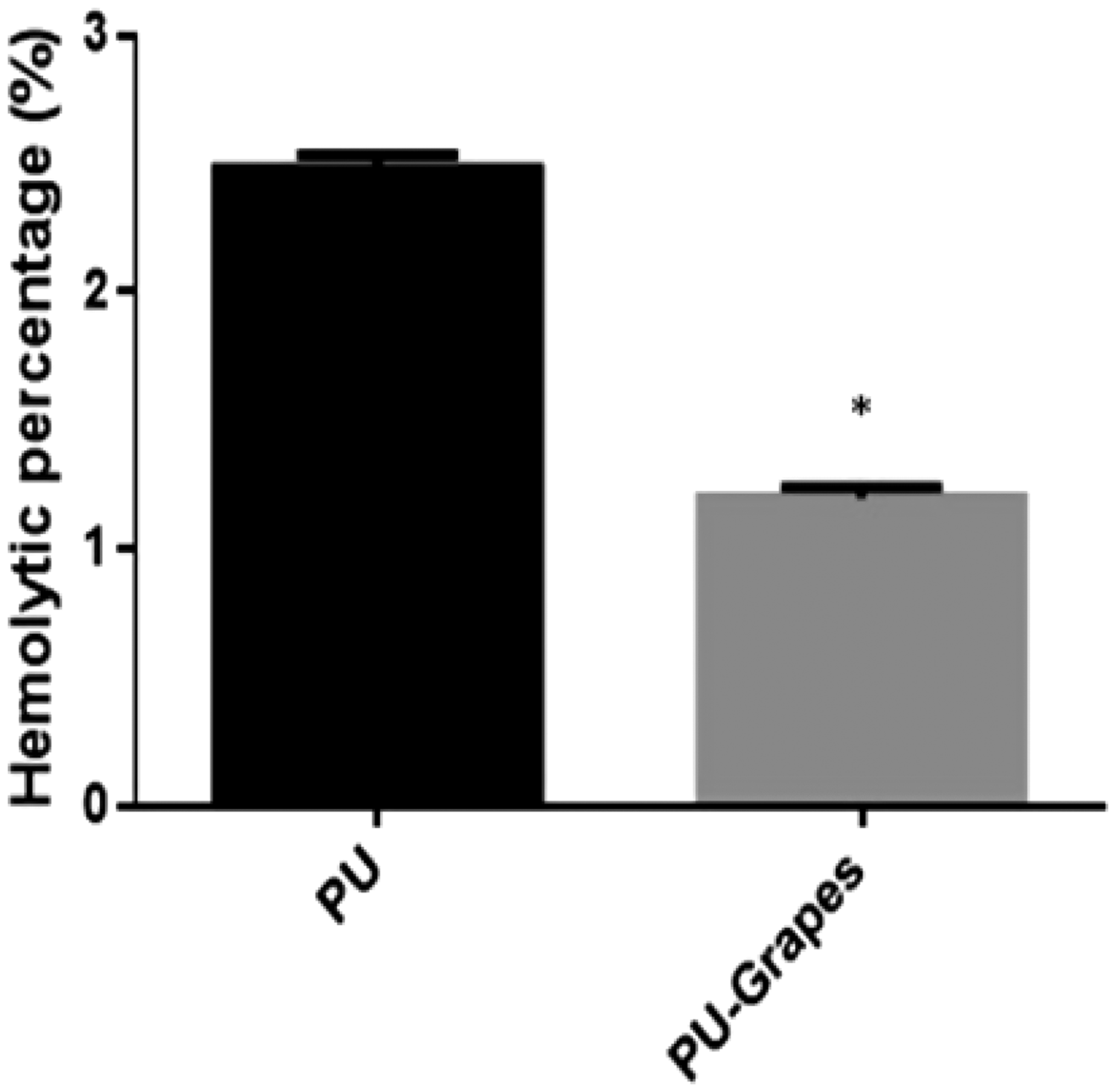
The anticoagulant nature of pure PU and PU/grapes fibrous membranes were determined through measuring the APTT and PT assay. The extrinsic coagulation pathway was determined through PT, while APTT assessed the intrinsic pathway of the coagulation time. The fabricated composites showed delayed blood clotting time in both APTT and PT assays as indicated in Figures 11 and 12. In APTT, the fabricated composites showed blood clotting time of 178 ± 3 s and for the PU membrane it was observed to be 175 ± 4 s. Similarly, in PT assay, the fabricated composites showed blood clotting time of 79 ± 2 s and the blood clotting time for the PU membrane was observed to be 76 ± 3 s. Hence, the addition of grapes in the PU matrix resulted in the delayed blood clotting time. Next, the hemolytic assay was performed to investigate the release of hemoglobin from the blood while interacting with the electrospun membranes. Hemolytic index of PU was observed to be higher than the developed PU/grapes fibrous membranes. The PU/grapes fibrous membranes showed hemolytic percentage of 1.20% which was less compared to the pristine PU (2.48 %) as indicated in Figure 13. If the hemolytic index is above 2%, the material is named as hemolytic and if the measured index is less than 2%, the material is considered as non-hemolytic materials [28]. The hemolytic index value for the developed PU/grapes composites was found to be 1.20% which was less than 2% and hence it was considered as a non-hemolytic material. The blood compatibility is a multifactorial process [44] which was influenced by various surface properties. The surface properties like enhanced surface roughness [40], smaller fiber diameter [45] and hydrophobic behavior [46] were reported to enhance the blood compatibility behavior. In our study, the developed PU/grapes fibrous membranes composites exhibited a similar trend as reported above which might have favored the higher blood clotting time.
APTT assay of PU membrane and PU/grapes composites. PT assay of PU membrane and PU/grapes composites. Hemolytic index of PU membrane and PU/grapes composites. *Mean differences were significant compared with pure (p < 0.05).

Cytocompatibility investigation
MTS assay was utilized to determine the cell viability of HDF cells in the fabricated pure PU and PU/grapes fibrous membranes. The MTS results were determined after three days culturing and it was observed that the electrospun membranes showed enhanced growth of HDF cells than the control. Further, the cell viability of PU/grapes fibrous membranes was found to be higher than the PU membranes. The pure PU showed cell viability of 133 ± 9%, while the electrospun PU/grapes fibrous membranes showed cell viability of 173 ± 1% as indicated in Figure 14. Hence, the addition of grapes in the PU matrix improved the HDF cell adhesion and proliferation. The reason for the improved fibroblast cell proliferation rate was due to their hydrophilic behavior which is reported to enhance the cell adhesion and proliferation [29]. Further, Mani et al. fabricated PU scaffolds added with ginger for wound-dressing applications. It was observed that the addition of garlic resulted in the reduction of fiber and pore size of the pristine PU. Further, the fabricated composites showed enhanced adhesion and proliferation of the fibroblast cells than the pristine PU [47]. In this research, the developed composites showed reduced fiber and pore size than the pristine PU which might also have a putative role in the enhanced fibroblast cell adhesion and proliferation. Hence, the improved fibroblast cell proliferation rate of the developed composites might be suitable for skin tissue engineering.
MTS assay of PU membrane and PU/grapes composites. *Mean differences were significant compared with pure (p < 0.05).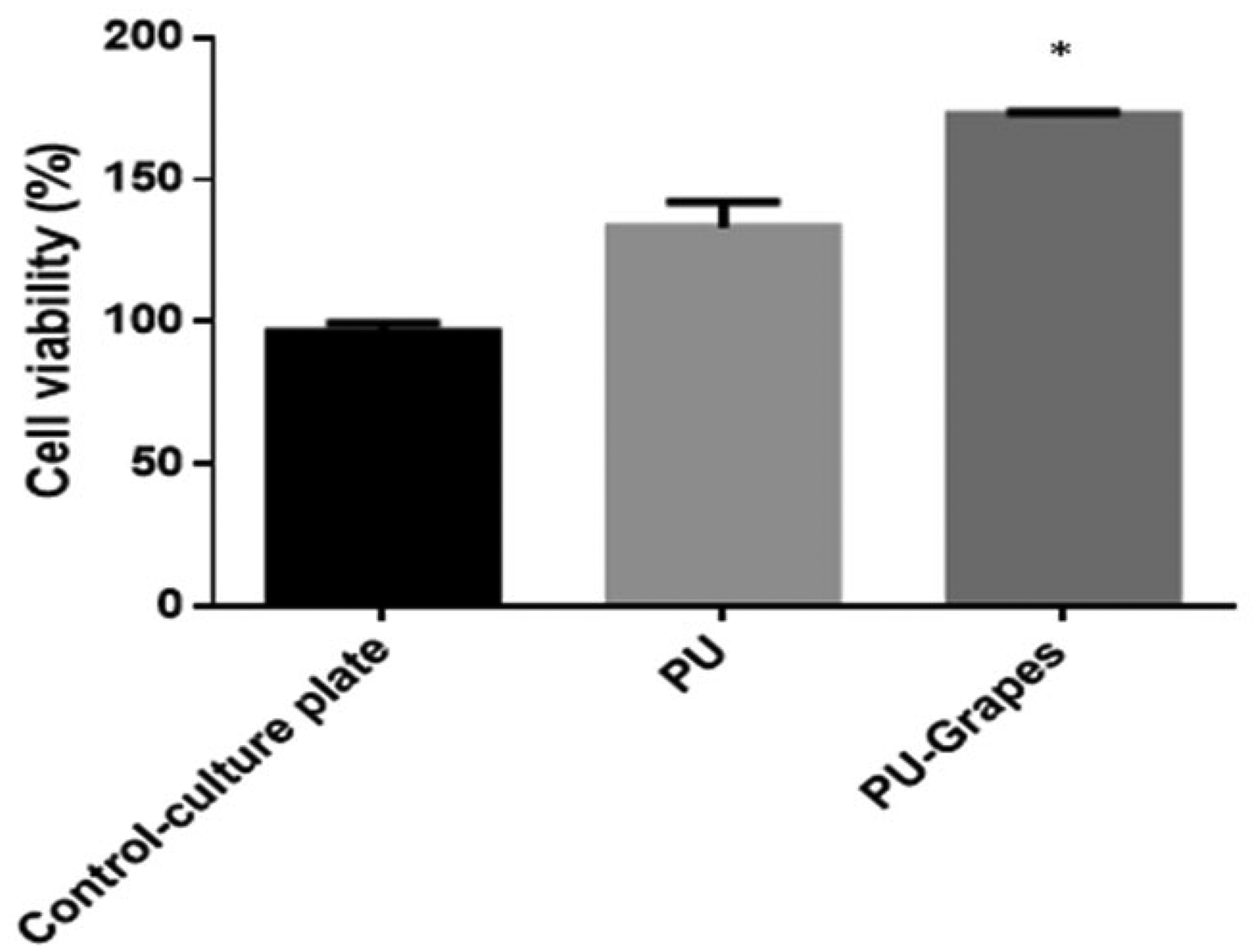
Conclusion
The present study summarized the fabrication and testing of electrospun textile PU wound-dressing scaffold incorporated with grape extract. The fabricated composites showed smooth fibers with reduced fiber and pore size than PU as revealed in the SEM. The formation of the hydrogen bond in the FTIR indicates an interaction between the PU and grapes. The addition of grapes resulted in the enhancement of the wettability behavior and surface roughness of the PU membrane. Thermal gravimetric analysis showed the improvement of thermal stability with the incorporation of grapes into the PU matrix. Mechanical testing indicated an improvement of the tensile strength of the PU/grapes. The in vitro blood compatibility and cytocompatibility studies revealed the slightly higher antithrombogenicity and the non-toxic nature of HDF cells for the fabricated composites than the pristine PU. Hence, the addition of grapes into the PU matrix had enhanced the physicochemical characteristics and biocompatibility parameters which could be potentially exploited for skin tissue regeneration. Further, there is a good scope for the fabricated membranes to be explored for other tissue engineering applications like bone, cartilage, cardiac and also for delivering drugs in biomedical applications. This study also opens the door for utilization of grape extract as an active constituent for blending with other polymers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education Malaysia with the grant nos. Q.J130000.2545.17H00 and Q.J130000.2545.20H00.
