Abstract
Nowadays, the synthetic biodegradable polymeric scaffolds were widely utilized for skin tissue engineering applications. The usage of nanofibrous scaffold manufactured through textile technologies has gained a widespread cynosure in wound healing applications. In this study, an electrospun textile scaffold based on polyurethane polymer incorporated with ginger extracts is hypothesized to possess unique properties facilitating wound healing. The fabricated electrospun membranes were characterized through scanning electron microscopy, Fourier transform infrared, thermogravimetric analysis, atomic force microscopy, and contact angle measurements. Coagulation assays such as activated prothrombin time, partial thromboplastin time, and hemolysis assay were performed for the electrospun membranes to determine their antithrombogenicity nature. Further, the cytocompatibility studies were evaluated using human dermal fibroblast cells to determine the toxicity of the electrospun polyurethane and their blends. Scanning electron microscopy revealed the reduced fiber (615 ± 154.31 nm) and pore (706 ± 90.07 nm) than the control (fiber diameter 1159 ± 147.48 nm and pore diameter 1087 ± 62.51 nm). Upon ginger incorporation, the wettability (80.33° ± 4.51) and surface roughness (389 nm) of the polyurethane was improved revealed in the contact angle and atomic force microscopy analysis. Infrared spectroscopy and thermogravimetric analysis study revealed the interaction of ginger with polyurethane by showing hydrogen bond formation and leaving an excess residue. The fabricated nanocomposites exhibited faster blood clotting time indicated in the activated prothrombin time and partial thromboplastin time. Further, hemolytic index and cytocompatibility studies revealed that the fabricated nanocomposites (Hemolytic index—0.96% and proliferation rate—159 ± 5.57%) showed nontoxic nature to the red blood cells and enhanced proliferation rate of human dermal fibroblast cells compared to the pristine polyurethane (hemolytic index—2.56% and proliferation rate—132 ± 3.61%). Our findings showed that polyurethane-based fibrous scaffolds comprising ginger exhibited desirable characteristics suitable for skin tissue engineering applications.
Introduction
Wound healing is a complex process and it is a mainstay for scientists around the globe for finding efficient ways to address this issue [1]. Generally, the process of acute wound healing follows four stages like inflammatory, proliferation, maturation, and remodeling. The examples of the acute wounds are traumatic injuries and wounds created through surgery [2]. Current modalities for treatment of acute wounds involve the applications of dressing materials in the form of gauzes and bandages, which can be tailored to assist the acute wound to heal [3]. These dressing materials provide necessary mechanical support and also act as a barrier for the invading bacteria into the wounds. Moreover, they facilitate the gas exchange and fluid transport facilitating the ambient environment for the wound to cure [4]. However, these aids are not effective enough in achieving the closure of wounds which mandates new options for the acute wound healing. The current wound care market accounts for USD 18.35 Billion and it is estimated to reach USD 22.01 Billion by 2022. China will be accounted for the fastest growth in wound management products demand compared to all countries. New materials with advanced properties are in huge demand for this field [5].
With the advent of nanotechnology, wound healing using nanofibrous scaffold evolved as a promising strategy with improved competence and enhanced closure rates [6]. Recently, the nanofibrous scaffold obtained using textile technologies is widely utilized in different tissue engineering applications [7]. The nanofibers obtained through textile technology provides massive versatility in which different parameters such as size and orientation of fibers, pore size, geometry and interconnectivity and surface morphology were easily controlled. Further, these parameters play a significant role in improving the physicochemical properties and cellular performance of the developed membrane [8]. Though several textile techniques exist, the role of electrospinning in tissue engineering is undisputed. The electrospinning is simple, cost-effective, and efficient process of manufacturing nanofibers. Electrospinning setup comprises a syringe loaded with the polymer solution. The solution is pumped through the syringe needle which is connected to the high voltage supply. When the applied voltage exceeds a certain threshold, a polymeric jet is ejected from the droplet overcoming the surface tension of the polymeric droplet. This jet is further whipped leading to the formation of nanofibers which is eventually collected at the grounded collector [9]. Electrospinning has been successfully employed in fabricating nanofibers from several natural and synthetic polymers. These fabricated nanofibers are utilized in tissue engineering because these fibers mimic the natural extracellular matrix (ECM) morphology and also allows better cell proliferation, migration, and differentiation [10].
Polyurethanes (PUs) are the class of polymers, which are widely utilized in medical applications. They possess excellent biocompatibility, high strength, and processing versatility which makes them a putative choice in biomedical applications [11,12]. Tecoflex 80 A is ether based PU and it is explored in fabricating scaffolds for a wide range of tissue engineering applications. Recent research fabricated nanofibers from Tecoflex 80 a mixed with carbon nanotubes with the intention of improving the mechanical strength. The developed composite exhibited a significant increase in Young’s modulus and breaking stress and also compatible with fibroblast cells portraying its suitability for tissue engineering applications [13]. In this research, a similar attempt is made to improve the physicochemical and mechanical properties of Tecoflex 80 A and test its suitability for acute wound healing.
Generally, the biological properties of the PU were reported to be less and so it was added with ginger to improve their biological properties. Ginger belongs to the family Zingiberaceae which originated in the tropical rainforests mainly in Indian subcontinent to Southern Asia [14]. Raw ginger contains over 400 different compounds and major constituents were 50–70% of carbohydrate, 3–8% of lipids, and remaining are terpenes and phenolic compounds. Terpene components present in the ginger are zingiberene, β-bisabolene, α-farnesene, β-sesquiphellandrene, and α-curcumene, while gingerol, paradols, and shogaol are the phenolic compounds of the ginger. Besides these components, the ginger also reported having amino acids, raw fiber, ash, protein, phytosterols, vitamins, and minerals [15]. Further, active compounds found in the ginger are reported to have antibacterial, antiviral, and antifungal activities [14,16]. Bhagavathula et al. reported the combination of curcumin and ginger extract has wound-healing potential and they showed improved healing of abrasion wound [17]. Mohamed et al. reported that the enthanolic extract of Zingiber Officinale showed significant reduction of the bacterial number and also exhibited faster wound healing rate [18]. Hence, these studies motivated us to use ginger in our study. The aim of the study is to fabricate a novel scaffold based on PU incorporated with ginger was fabricated using electrospinning technique. After fabrication, their physiochemical properties, blood compatibility assessments and cytocompatibility studies were tested to examine its effectiveness for skin tissue engineering.
Methodology
Materials and methodology
Materials
The medical grade Tecoflex EG-80A PU was obtained from Lubrizol and dissolved in dimethylformamide (DMF) solvent (Sigma Aldrich, UK). The raw ginger was used to make the extract and it was procured from AEON, Malaysia. The chemical phosphate buffered saline (PBS) and sodium chloride physiological saline (0.9% w/v) were purchased from Sigma-Aldrich, Kuala Lumpur, Malaysia. The reagents such as rabbit brain activated cephaloplastin, calcium chloride (0.025 M), and thromboplastin (Factor III) used in the blood compatibility studies were purchased from Diagnostic Enterprises, Solan, India.
Preparation of ginger extract
Initially, the outer surfaces of the ginger were peeled off and cleaned with distilled water. After, it was grinded using kitchen blender to obtain extracts from it. Finally, the obtained extract was filtered using filter paper and stored in the refrigerator for further use.
Preparation of solution
PU solution (9 wt%) was prepared by mixing 0.450 g in 5 mL of DMF for overnight at room temperature. Further, the ginger solution was prepared at a concentration of 4 wt% by dissolving 120 µL of ginger extract in 3 mL DMF and stirred for 1 h maximum. After, the prepared homogeneous solution was added with the ginger homogeneous solution at a ratio of 8:1 (v/v) and stirred for 2 h maximum.
Electrospinning process
Fabrication of pure PU and composite PU was done using electrospinning setup. For this, 10 mL syringe was filled with the solutions of PU and composite. The parameters of electrospinning were setup as follows: voltage of 10.5 kV with flow rate 0.2 mL/h and collector to syringe tip distance as 15 cm for both cases. The fibers were harvested at the collector which was wrapped with aluminum foil. Finally, the fibers were vacuum dried for 24 h to remove the remaining DMF in the fibers.
Physicochemical characterization
A thorough investigation of physicochemical and mechanical properties was performed. Initially, the morphology of the fibers was observed using scanning electron microscopy (SEM) unit and the plot of fiber diameter distribution, pore size (distance between two fibers) were obtained using Image J by choosing 50 locations randomly. Wettability of the samples was obtained manually by utilizing the video contact angle (VCA) instrument. Functional group scanning over the range of 600–4000 cm−1 was recorded using Fourier-transform infrared (FTIR) unit. The thermal properties of the fibers were studied using thermogravimetric analysis (TGA) unit (scanning was performed over the temperature range of 30℃–1000℃ with the rate of 10℃/min under nitrogen atmosphere). The surface roughness of the samples was estimated using atomic force microscopy (AFM) unit over the size of 20 µm × 20 µm.
Coagulation assays
Activated prothrombin time and partial thromboplastin time assay
Anticoagulant nature of the fibers was estimated using activated prothrombin time and partial thromboplastin time assay. Before performing this assay the fibers were cut and washed with PBS and then incubated at 37℃ for 30 min. The test was performed by following the procedure as described already [19]. Finally, the mean APTT and PT were plotted for both pure PU and composite fibers.
Hemolysis assay
The compatibility of the blood cells with the fabricated fibers was estimated using hemolysis assay. Before starting the procedure the membrane was incubated in physiological saline for 30 min at 37℃. The hemolytic index of the pure PU and the fabricated nanocomposites was estimated using the procedure as described already [19].
Cell culture and cell viability studies
The biocompatibility of the scaffolds was investigated using human dermal fibroblast (HDF) cells (Human Skin Fibroblast Cells 1184, ECACC, UK). The cells were maintained at 5% carbon dioxide (CO2) using Dulbecco's Modified Eagle's medium (DMEM) medium supplemented with 10% fetal bovine serum and 5% antibiotic penicillin. Before cell seeding, the prepared electrospun scaffolds were cut and placed in 24-well plates by sterilizing with 75% alcohol solution and washed with PBS solution. When the cells were confluent, they were scraped using trypsin–ethylenediaminetetraacetic acid (EDTA) solution and 10 × 103 cells/cm2 were seeded on each well containing the prepared samples. Then, HDF cells were placed in CO2 incubator for 72 h before analyzing cell viability. After three days incubation, the medium was extracted and added with 20% of MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H tetrazolium, inner salt) and further incubated for 4 h. After 4 h, the absorbance was measured at 490 nm using spectrophotometric plate reader to evaluate the cell proliferation rates.
Statistical analysis
All experiments were performed three times independently and unpaired
Result and discussion
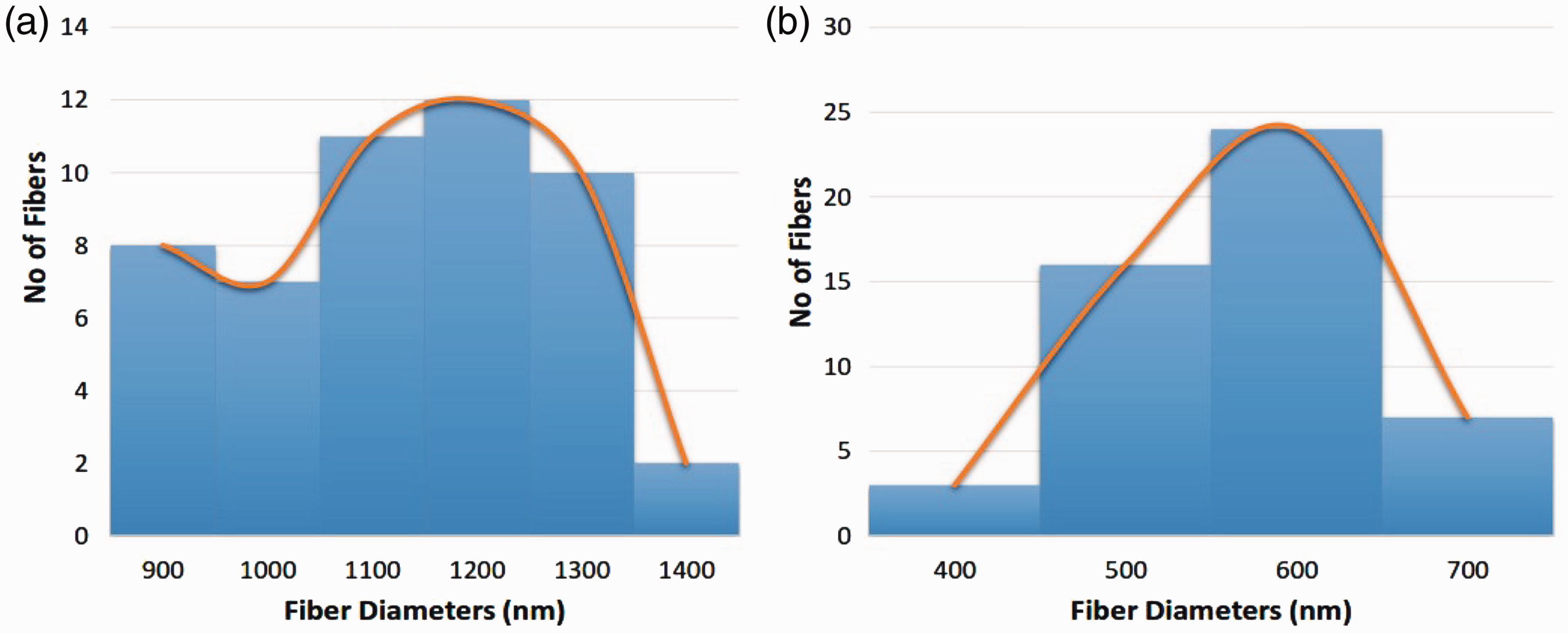
Figure 1(a) and 1(b) shows the SEM micrographs of electrospun PU and PU/ginger nanofibers mat. The morphology of the electrospun membranes revealed the presence of smooth fibers without any bead defects. It was observed that the PU/ginger nanofibers mat shows the decrease in diameter compared to the pristine PU. The fiber diameter of the PU/ginger is reduced compared to the PU and this reduction in the fiber diameter may be due to the decrease in the polymer concentration utilized. The fiber diameter of pristine PU was found to be 1159 ± 147.48 nm, while the PU/ginger nanofibers mat have fiber diameter of 615 ± 154.31 nm, respectively and the corresponding fiber distribution curve are shown in Figure 2(a) and 2(b). Unnithan et al. developed wound dressing scaffold based on PU blended with dextran nanofibers mat. It was observed that the addition of dextran into PU matrix reduced the fiber diameter and exhibits enhanced fibroblast adhesion and proliferation compared to the pure PU [20]. The developed nanocomposites in the present work also have reduced fiber diameter which could be anticipated with enhanced fibroblast proliferation for the new tissue growth.
SEM images of (a) PU membrane and (b) PU/ginger nanocomposites. Fiber diameter distribution of (a) PU membrane and (b) PU/ginger nanocomposites.
Using Image J software, the pore size measurements for the electrospun pure PU and PU/ginger nanofibrous scaffolds were measured. The PU/ginger nanocomposite showed pore size in the range of 706 ± 90.07 nm, while the pure PU exhibited pore size of 1087 ± 62.51 nm, respectively and the corresponding graphical representations are illustrated in Figure 3(a) and 3(b). It was reported that the membranes with low pore size reduce the growth of bacteria and our fabricated nanocomposites possessed smaller pore size which might reduce the bacterial growth [21].
Pore diameter distribution of (a) PU membrane and (b) PU/ginger nanocomposites.
The functional groups present in the fabricated PU and PU/ginger nanofibers mat are indicated in the Figure 4. In the spectra of PU, a broad peak was observed at 3328 cm−1 assigned to the NH stretching and the peaks observed at 1596 cm−1 and 1530 cm−1 attributed to the vibrations of NH group. The sharp peaks are seen at 2938 cm−1 and 2853 cm−1 indicates the CH stretching and the peak at 1413 cm−1 represents their vibrations. The C=O stretching attributed to the carboxylic groups were observed at 1730 cm−1 and 1702 cm−1 and C–O corresponding to alcohol were seen at 1220 cm−1 and 1105 cm−1 [22,23]. In the absorption band of PU/ginger nanofibers mat, it was observed that no new additional bands were formed, but the intensity of the peak was increased due to hydrogen bond formation [24]. The formation of hydrogen bond was identified by the increase in the intensity of the peak at 3328 cm−1. It was reported that the combining of two different macromolecules will produce a strong inter-hydrogen bonds than those formed between the molecules of the same polymer [25].
IR spectrum of PU membrane and PU/ginger nanocomposites.
In our developed PU/ginger nanocomposites, the development of stronger inter-hydrogen bonds was due to the interaction between the functional groups of OH and NH functional groups in the ginger and PU respectively.
The contact angle measurements of the fabricated nanofibrous mats were measured through VCA optimal unit. From the obtained contact angle measurements, the PU matrix contributes to the hydrophobic nature, which shows the contact value of 100° ± 0.58, while the electrospun PU/ginger nanofibrous mat showed the decrease in contact angle to around 80.33° ± 4.51. Unnithan et al. developed a electrospun scaffold for wound dressing applications by utilizing PU added with cellulose acetate and zein nanofibers. It was reported that the addition of cellulose acetate and zein into the PU matrix enhanced the wettability of the electrospun scaffold and exhibits enhanced fibroblast adhesion and proliferation [26]. Hence, in this present study, the addition of ginger into PU membrane showed hydrophilic nature which might favor the enhanced fibroblast adhesion and proliferation for new tissue growth applications.
The thermal analysis of the electrospun PU and PU/ginger nanofibers mat is depicted in the Figure 5(a) and 5(b). It was observed that the PU membrane starts to degrade at 276℃, whereas in the case of electrospun PU/ginger nanofibers mat, it showed the initial onset degradation temperature at 256℃ indicating the decreased thermal stability. The decrease in the thermal stability might be due to the presence of water molecules present in the ginger. Further, at 1000℃, the pure PU membrane showed remaining weight percentage of 0.47%, while the electrospun PU/ginger nanofibers mat, it was observed to be increased to about 2.93% indicating the presence of ginger in the PU matrix. As discussed previously, major components of garlic are carbohydrate, lipids, terpenes, and phenolic compounds. Apart from these constituents, they also contain certain amount of amino acids, raw fiber, ash, protein, phytosterols, vitamins, and minerals [15]. Hence, the presence of raw fiber and ash content in the ginger might have favored the enhanced weight residue for the fabricated PU/ginger nanocomposites.
TGA analysis of (a) PU membrane and (b) PU/ginger nanocomposites.
Moreover, Figure 6(a) and 6(b) depicts the results of derivative weight loss curve for the electrospun membranes, which suggested that the addition of ginger reduce the weight loss of PU membrane. The DTG result of PU membrane showed a two large weight loss peak in which the first loss happens at 223℃ and ends at 348℃ and second weight loss occurs at 223℃ to 348℃, respectively. Further, a small weight loss occurs at 557℃ to 684℃ indicating the third weight loss. In the case of electrospun PU/ginger nanofibers mat, it was observed two weight loss in which the first loss begins at 223℃ to 369℃, the second loss from 369℃ to 563℃, respectively. It was observed that the intensity of the weight loss peaks of the fabricated nanocomposites was found to be reduced than the PU membrane indicating the presence of ginger in the PU matrix. Jaganathan et al electrospun wound dressing scaffold comprising PU added with murivennai oil nanofibers. It was reported that the addition of murivennai oil into the PU matrix showed improvement in the thermal behavior which correlates with our findings [27].
Weight residue percentage of (a) PU membrane and (b) PU/ginger nanocomposites.
Atomic force microscope (AFM) analysis was carried out to analyze the surface roughness of electrospun nanofibers as indicated in Figure 7(a) and 7(b). From the surface roughness measurements, it was revealed that PU/ginger nanofibers mat exhibited an enhancement in the surface roughness than the pristine PU. The calculated surface roughness for the electrospun PU membrane was found to 216 nm and the electrospun PU/ginger nanofibers mat, the surface roughness was observed to be 389 nm, respectively, which clearly indicates that the addition of ginger enhanced the surface roughness of the PU membrane. Ghorbani et al. fabricated polycaprolactone (PCL)/chitosan added zinc-doped hydroxyapatite nanoparticles (nZnHA). It was shown that the fabricated PCL/chitosan membrane added with zinc-doped hydroxyapatite nanoparticles (nZnHA) exhibited enhanced surface roughness [28] and obtained results were in line with our findings.
AFM images of (a) PU membrane and (b) PU/ginger nanocomposites.
The coagulation assays such as APTT and PT were utilized to determine the blood clotting time of the electrospun PU and PU/ginger nanofibers. The coagulation assays revealed the decreased blood clotting time of the electrospun nanocomposites than the PU membrane as indicated in Figures 8 and 9. The PU/ginger nanofibers mat exhibited blood clotting time of 156.7 ± 2.52 s, while the PU scaffold showed blood clotting time of 166.33 ± 3.06 s revealed in the APTT assay. Similarly, the PT revealed the blood clotting time for the electrospun PU/ginger nanofiber mat was found to 68.33 ± 1.53 s, while the PU scaffold showed blood clotting around 72 ± 1 s. The addition of ginger into the PU matrix reduced the blood clotting time and the decrease in the blood clotting time was due to the trade-off between the polar and apolar regions of the PU/ginger scaffold [29].
APTT assay of (a) PU membrane and (b) PU/ginger nanocomposites. PT assay of (a) PU membrane and (b) PU/ginger nanocomposites.

Further, the hemolytic assay was carried out to analyze the toxicity of the electrospun membranes with red blood cells. The results of hemolytic assay depict that the hemolytic index of the electrospun PU/ginger nanofibers mat was observed to be decreased than pristine PU. The hemolytic index of PU was observed to be 2.56% and for the electrospun PU/ginger nanofibers mat, it showed 0.96% as denoted in Figure 10. It was reported, if the hemolytic value was above 2%, the material was considered as hemolytic and if the value was below 2%, the material was classified as non-hemolytic [27]. The calculated value of the hemolytic index for the developed nanocomposites was found to be below 2% and hence it possessed improved blood compatibility than the PU membrane. The healing of acute wound healing occurs in different stages such as inflammatory phase, proliferation and maturation. The first stage of the wound healing initiates blood-clotting cascade in order to prevent the excess blood bleeding from the wound zone and also helps in wound closure to avoid the infection [30]. The fabricated nanocomposites exhibited faster blood clotting times which might be beneficial in preventing the blood bleeding and also helps in wound closure to prevent the infection.
Hemolytic index of (a) PU membrane and (b) PU/ginger nanocomposites.
The fibroblast cells present at the wound site plays an important role in supporting the wound healing process. Further, the HDF cells were actively take part in ECM and collagen production for regeneration of new tissue growth [31]. Hence, evaluating the influence of the fabricated scaffolds on fibroblast cell adhesion and proliferation will promote them for wound healing application. Figure 11 illustrates the cell proliferation rate of HDF cells in the electrospun membranes using MTS assay. After three days of cell culture, it was shown that the HDF cells were well grown and proliferated well on the electrospun membranes than the control plates. Further, the proliferation rate of the HDF cells on the electrospun PU/ginger membrane was observed to enhance than the control. The proliferation rate of PU membrane was found to be 132 ± 3.61 %, while the electrospun PU/ginger oil nanocomposites showed proliferation rate of 159 ± 5.57 % respectively. The reason for the enhanced proliferation of HDF cells in the fabricated nanocomposites was might be due to the hydrophilic behavior than the pristine PU [32]. Hence, the newly developed PU/ginger oil scaffolds showed enhanced proliferation of HDF cells and might be suitable for skin tissue engineering.
MTS assay of (a) PU membrane and (b) PU/ginger nanocomposites.
Conclusion
In this research, the PU scaffold incorporated with ginger was successfully electrospun for skin tissue engineering applications. Morphology investigation showed reduced fiber and pore size for the fabricated composites than the control. Upon ginger incorporation, the wettability and surface roughness of the PU was improved revealed in the contact angle and AFM analysis. FTIR and TGA study revealed the interaction of ginger with PU by showing hydrogen bond formation and leaving an excess residue. The fabricated nanocomposites exhibited faster blood clotting time indicated in the APTT and PT assay. Further, hemolytic index and cytocompatibility studies revealed that the fabricated nanocomposites showed nontoxic nature to the red blood cells and HDF cells. Our findings showed that PU-based fibrous scaffolds comprising ginger exhibited desirable characteristics which might be a promising candidate for skin tissue engineering applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education Malaysia with the Grant no. Q.J130000.2545.17H00 and Q.J130000.2545.20H00.
