Abstract
In the present study, a polyvinyl chloride-based barium tungstate ion-exchange membrane was synthesized by sol–gel method. The structure of membrane was studied in terms of Fourier transform infrared spectroscopy, X-ray diffraction and scanning electron microscopy. X-ray diffraction analysis confirms crystalline form of the composite membrane without any other impurity. Scanning electron microscopy and Fourier transform infrared spectroscopy analysis show the uniform arrangement of particles in the membrane with crack-free surface structure and presence of different functional groups of the organic-inorganic materials. The electrochemical properties like surface charge density (D), transport number and mobility ratio of the ion-exchange composite membrane were theoretically evaluated and compared with observed values using “Teorell, Meyers and Sievers” method. Transport number follows the order as KCl <NaCl < LiCl < NH4Cl, while the surface charge density showed reversed order. The results showed that the low concentration of electrolytes favors the high mobility of univalent cation in the present study. The above result proves the analytical utility of polyvinyl chloride-based barium tungstate ion-exchange membrane in environmental management.
Introduction
The word membrane is explained as a specifically penetrable obstruction that isolates one part from the blend. For this reason, numerous organic membranes are available inside our body which isolates the inside of the cell from outside condition and accordingly help in transport of particles in and out. In this similar pattern like biological membranes, synthetic membranes have been manually synthesized within the laboratories. These synthetic membranes serve as a model for different separation process [1]. They help in transporting diverse particles specifically to the coveted area and isolating the unwanted items. This is the primarily preferred standpoint of membrane innovation that is related to transport selectivity [2]. There are different sorts of layers that are manufactured in nature out of which ion-exchange layers are the essential kind of partition membrane [3]. These are used to separate ionic species from aqueous solution and charged components. So as to watch that these readied manufactured layers are relevant to which specific process, electrochemical and transport properties have been contemplated. It is observed that the ideal ion exchange membranes should have high permeability, low membrane resistance, good thermal and chemical stabilities with tensile strength. Keeping all the above properties in mind, ion exchange cation membrane was prepared in the utilization of waste water treatment [4,5].
The presence of inorganic Pb, Cd, Hg and Zn metal ions in industrial waste water is the major concern to the chemists, chemical engineers, and scientists worldwide due to their toxic effects on environments, plants, and animals. The metal ions present in the waste water stream, discharged from various industries such as food, chemical, metal plating, smelting, refining, etc. continued even after the serious considerations of various environmental protection agencies about their discharge that reflects ill effects on the ground water [6,7].
Environmental protection agencies have proposed different permissible limits of metal ions intake beyond which they become very toxic causing various health hazards for human kinds such as cancer, kidney and brain damage, etc. Thus, it is very important to treat the waste water before discharging to public sewage [8–10].
For this purpose, a variety of ion exchange membranes such as organic, inorganic and composite membranes have been used [11–16]. However, inorganic material plasticizes in an organic polymer such as polyvinyl chloride (PVC) in the form of the composite are very stable at high temperature as well as in acidic and basic media [17–20]. PVC is stable in all acid /base and all inorganic chemicals [21]. Also in another article [22], barium adds for the stabilization of PVC.
In the present study, a new inorganic ion-exchange composite membrane of PVC, mixed with barium tungstate (BT), was prepared. Various electrochemical parameters such as membrane potential, transport number, mobility ratio and surface charge density have been optimized by using TMS method even these methods are classical but still proving its strong results [23,24] and Nernst equation to check the validity of the membrane for the separation of various industrial processes. Among these parameters, the surface charge density was most important and dominant parameter because it was used for controlling the membrane phenomena [25–32].
Experimental
Materials and reagents
Sodium chloride, potassium chloride, lithium chloride, ammonium chloride, HCl, NaCl, HNO3, H2SO4, NaOH, sodium tungstate and barium chloride, KMnO4 and PVC were obtained from E. Merck, Mumbai, India. All the reagents used were of AR grade without further sanitization and their solutions were prepared in deionized water.
Preparation of BT inorganic ion-exchanger
BT was prepared by mixing equimolar solutions (0.3 M) of barium chloride and sodium tungstate in a conical flask with constant stirring for around 2 h until a white precipitate was obtained. The resultant precipitate was kept for 24 h and then filters were washed repeatedly wiith double distilled water to remove the free electrolytes [33,34]. After complete washings, it was kept in an oven at 40℃ for overnight drying. The BT is now ready for further studies.
Preparation of PVC-based BT ion exchange composite membrane
The BT was mixed with PVC granules by pestle and mortar and then the mixture was kept into a cast die having a diameter of 2.45 cm and placed in a digital furnace by maintaining temperature at 200˚C for about 1 h to equilibrate the reaction mixture and transfer to a pressure device (SL-89, UK), and the pressure was applied upto 120 MPa during the fabrication of the membrane. The mechanical stability of membrane depends on the membrane preparation ratio if 25% PVC was used for the preparation of membrane and then it was found to be mechanically most stable than other ratios according to the method provided by Khan et al. [34–36].
Thickness and porosity calculation
Firstly, the structural studies of composite membrane in terms of basic parameters such as porosity and thickness of the membrane were calculated according to the method provided by Khan et al.[34].
Instruments used for structural characterization
The membrane morphology was investigated by ZEISS EVO Series scanning electron microscope EVO 50 at an accelerating voltage of 20 kV. The sample was mounted on a copper stub and sputter coated with gold to minimize the charging. The FTIR spectrum of composite membrane was done by Interspec 2020 FT-IR spectrometer, spectral (UK). The sample compartment was 200 mm wide, 290 mm deep and 255 mm high. The entrance and exit beam to the sample compartment were sealed with a coated KBr window and there was a hinged cover to seal it from the environment. X-ray diffraction pattern of the membrane was recorded by Miniflex-II X-ray diffractometer (Rigaku Corporation) with Cu Kα radiation.
Electrochemical properties of PVC-based BT composite membrane
Potential measurement.
The membrane potential was measured with the help of a digital potentiometer (EI-118) as studied by Khan et al. [36]. This set up is usually used for the potential measurement for the univalent cation-exchange membrane.
Calculation of the transport number, mobility ratio, and surface charge density
The methods of Altug and Hair, Kobatake et al. Nagasawa and co-workers are available for the calculation of electrochemical parameters such as transport number, mobility ratio and surface charge density [28–32]. In the present study, the TMS method (equation (2)) and Nernst equation (equation (1)) were used for the determination of these parameters and show good results. The mechanism of ion transport through the composite membrane is easily understood by the electrochemical parameters [25–27].
Results and discussion
The thickness and porosity of BT composite membrane were found to be 0.070 cm and 0.022. The thickness and porosity play important role in controlling the diffusion across the membrane. A. Jovan Kamcev reported that as membrane thickness decreases, the time required for the membrane to equilibrate with the dipping solution becomes shorter. They have used the membrane they have also used the membrane thickness from 0.06 to .014 cm. May be it varies from different materials.
In this case, the low order of porosity which has been analyzed by SEM suggests that interstices are negligible and diffusion across the membrane would occur mainly through exchange sites [36].
The scanning electron microscopy (SEM) was used for the surface characterization of the membrane at a magnification of 5.00 KX. It is clear from SEM (Figure 1) image that the membrane was found crack-free, dense, homogeneous, uniform, compact which show good stability and flexibility. In addition, the particles of inorganic material (BT) were dispersed uniformly in the polymer matrix and the particle sizes of the inorganic material are interdependent.
SEM image of PVC-based BT ion-exchange composite membrane at different magnifications magnification of (a) 3.00 KX and (b) 5.00 KX). PVC: polyvinyl chloride; SEM: scanning electron microscope; BT: barium tungstate.
The FTIR spectrum of PVC-based BT composite membrane is shown in Figure 2. The intense peak in the region of 832–900 cm−1 indicates the presence of barium metal in the composite. The peaks at 475 and 595 cm−1 may be due to the presence of C–Cl bond. In the spectrum, the broad absorption peak at 3308 cm−1 indicates to the stretching vibration of a hydroxyl group and peaks at around 2925 and 2855 cm−1 may be due to C–H stretching. The peaks at around 1383 and 1675 cm−1 indicate the stretching vibration of –CH2. The rest of the peaks present at an appropriate position indicates only the interaction between inorganic and organic materials [34]. The peak at 1080 cm−1 may be due to the C–OH stretching in hydrated PVC composite.
FT-IR spectra of the PVC-based barium tungstate ion-exchange membrane showing the different functional groups and bonding the current membrane. PVC: polyvinyl chloride; FT-IR: Fourier transform infrared spectroscopy.
Figure 3 shows the XRD pattern of the PVC-based BT composite membrane. The membrane displayed the semi-crystalline nature owing to the presence of some sharp and broad peaks of BT in the diffractogram. There is no distinct peak which can be seen in the diffractogram correspond to the PVC component because of their amorphous nature. Figure 3 also demonstrates our recorded X-beam diffraction profiles at a couple of agent weights. Because of the disparity of the X-beam pillar, the recorded diffraction designs are sullied by a few diffraction tops. In any case, at surrounding conditions, 14 diffraction pinnacles of BaWO4 are unmistakably perceptible and the cell constants for the underlying stage are observed to be, a = 5:66 Å and c = 12.73 Å, which are in genuinely great concurrence with the distributed outcomes a = 5:61 Å and c = 12.71 Å; (ICDD card no 43-0646)
XRD of PVC-based BT ion-exchange composite membrane. PVC: polyvinyl chloride; XRD: X-ray diffraction.
Observed membrane potential (mV) of PVC-based BT composite membrane.

PVC: polyvinyl chloride; BT: barium tungstate.
Transport number of PVC-based BT composite membrane.

PVC: polyvinyl chloride; BT: barium tungstate.
Mobility ratio of PVC-based BT composite membrane.

PVC: polyvinyl chloride; BT: barium tungstate.
The surface charge density of PVC based BT ion exchange composite membrane was determined by Teorell, Meyer, and Sievers (TMS) method. Theoretical concentration potentials existing across the membrane are calculated with the help of equation (1) as a function of concentration.
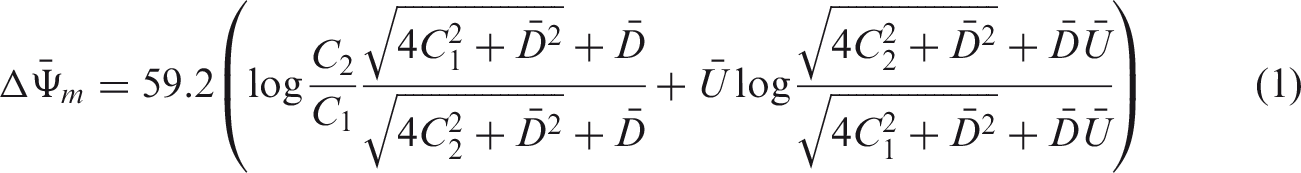

This method gave good results for the fixed charge density as compared to other methods [30–33]. The surface charge density is the central parameter and facilitates the understanding of the mechanism of the ionic and molecular process which occurs in membrane phase. The surface charge densities of PVC based BT composite membranes were determined with the help of observed and theoretical membrane potential data involving different univalent electrolytes. The theoretical membrane potential and observed membrane potential were plotted as a function of electrolytes concentration in Figure 4. The membrane potential (mV), measured across PVC-based BT composite membrane in contact with different monovalent electrolyte (KCl, NaCl, LiCl, NH4Cl) solutions at different concentrations at 25 ± 1℃ (Figure 4). The observed potential depends on the concentration of electrolytes and its value increased with dilution and follows the order KCl <NaCl <LiCl <NH4Cl. Finally, the coinciding curve for univalent electrolytes gave the value of the charge density D within the membrane phase as shown in Table 4.
Plots of membrane potential (theoretical and observed) (mV) versus (mol/l) at different concentrations of NH4Cl electrolyte solution for PVC-based BT ion-exchange composite membrane. PVC: polyvinyl chloride. The surface charge densities of PVC-based BT composite membrane. PVC: polyvinyl chloride; BT: barium tungstate.

Conclusion
New ion-exchange membrane of BT-PVC was prepared by mixing different valumes ration (1:1) and characterized by various spectrophotometric methods. This work presents the determination of transport number and electrochemical parameters from the structural information and observation of potential for ion-exchange BT membrane which can be used for the industrial separation process. The electrochemical properties of PVC-based BT ion-exchange membranes such as transport number, mobility ratio, and charge density were determined and help to understand the electrical phenomena. The transport number follows the order as KCl <NaCl < LiCl < NH4Cl, while the surface charge density showed reversed order. Then results showed that the low concentration of electrolytes favors the high mobility of univalent cation in the present study.
Footnotes
Acknowledgements
The authors are thankful to the Principal of University Polytechnic, Faculty of Engineering and Technology, and the Chairman, Department of Chemistry, Aligarh Muslim University, Aligarh (India) for providing necessary research facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
