Abstract
Polyvinyl alcohol/agar/maltodextrin composite membranes at different ratios are investigated for the removal of the antibiotic ciprofloxacin from water. The factors affecting the adsorption capacity of the as-synthesized membranes include time, pH, temperature, concentration, and dosage of film. The PAM244 and PAM433 membranes are subjected to optimization using response surface methodology model employing the Box–Behnken experimental design model. The optimal model has shown compatibility between the experimental value and the predicted value of the model. The correlation coefficients of PAM244 and PAM433 are recorded as R2 = 0.999 and R2 = 0.991, respectively. The model also proposes the optimal parameters. For PAM244, the adsorption capacity predicted from the model is 11.52 mg g–1 (42% of efficiency) at pH 5.89, a concentration of 41.28 mg L–1, over a time period of 19.87 min, and dosage of film 2.08 g L–1. For PAM433, the adsorption capacity predicted from the model is 10.46 mg g–1 (40% of efficiency) at pH 6.07, a concentration of 41.24 mg L–1, over a time period of 19.59 min, and dosage of film 2.16 g L–1. To enhance the potential of the material, the membrane reusability is also examined with the number of reuses being three times with the PAM244 membrane and twice with the PAM433 membrane.

Introduction
The rapid development of aquaculture in Vietnam is associated with immediate and long-term environmental problems, such as biological imbalance, environmental pollution, and the development of diseases.1,2 The use of antibiotics in aquaculture that exceeds the regulations on type and doses over a prolonged period of time can affect the quality of aquatic products as well as causing environmental pollution.3–5 The removal of antibiotic residues has been a topic of interest for many scientists. Therefore, developing effective bio-adsorbent materials that are easy to recover after use is considered a necessary and urgent task to meet the needs of green and sustainable development. Due to the special adsorption properties of charge-carrying polymers on the surfaces of solid materials, many polymers have been used to modify the surfaces of materials to improve the efficiency of organic pollutant removal. 6 Charge-carrying polymers are generally divided into two categories: negatively charged polymers and positively charged polymers. Charged polymers have many applications in the coatings industry, in anticorrosion, and as coagulants.7–9 In addition, charge-carrying polymers are now widely used in environmental remediation. The antibiotics typically studied and for environmental purposes are: ciprofloxacin (CFX), tetracycline (TCC), amoxicillin, oxytetracycline (OTC), and doxycycline.6,10–15 Polyvinyl alcohol (PVA) composites are well studied materials with diverse combined components. Therefore, researchers have used optimization models to evaluate the ability to combine components and to study the applicability of PVA composites for the treatment of pollutants.
A PVA composite is a composite material containing a PVA component to replace the polymer matrix inside the composite material structure.16–19 Basically, the PVA production process includes polymerization of vinyl acetate monomer to polyvinyl acetate (PVAc) and hydrolysis of the acetate groups to produce PVA.20,21 Based on the catalyst used, three hydrolysis methods have been applied for the preparation of PVA. These include acidification, aminolysis, and alkaline hydrolysis. On an industrial scale, the conversion of PVAc into PVA is usually achieved with alkaline alcohol. In this hydrolysis method, the ester is exchanged with methanol, in the presence of sodium hydroxide, to hydrolyze the acetate groups. PVA is a hydrophilic synthetic polymer with a flat, semi-crystalline zigzag structure, and good mechanical properties. 20 PVA is chemically and thermally stable, and is resistant to degradation in most physiological environments. PVA exhibits the advantages of good film-forming performance, outstanding mechanical strength and excellent biocompatibility, and has been widely used as a protective material for goods and tools.22–24 Moreover, it is soluble in water due to its high polarity, is non-toxic, exhibits high biocompatibility, and can be handled easily. PVA also has the potential to produce promising biodegradable films.25–28
In this study, the composition ratio of the PAM membrane was evaluated and shows excellent great potential for the selective adsorption of antibiotics. In addition, the selective adsorption performance of synthetic membranes for antibiotics was evaluated. The effects of the contact time, the solution pH, the PVA dosage, and the initial antibiotic concentration on the adsorption capacity of composite films have also been studied. In addition, the adsorption mechanism of the PVA/agar/maltodextrin (PAM) films was evaluated by adsorption kinetics and isothermal modeling. However, in the previous studies, the adsorption capacities were not optimized and the reusability was not evaluated.
Results and discussion
Evaluation of selective adsorption
The proportional membranes were evaluated for their selective adsorption capacities. The results show that the adsorption capacity for CFX was the highest, ranging from 2.6 to 4 mg g–1, followed by TCC, which was only absorbed by PAM622 and PAM811 with limited capacities of 0.57 and 0.37 mg g–1, respectively, while chloramphenicol (CPC) and OTC were almost non-adsorbable (Figure 1). These results show that the membrane has the ability to adsorb antibiotics. Therefore, CFX was used to conduct subsequent experiments.

Selective antibiotic adsorption by the PAM244, PAM433, PAM622, and PAM811 composite membranes.
Effect of time
Several factors affecting the adsorption process, including the adsorption time, the solution pH, the ambient temperature, the material content, and the antibiotic concentration, were evaluated to optimize the conditions and adsorption capacity of the materials. As shown in Figure 2, the adsorption process of all membranes occurs rapidly between 0 and 20 min, during which the PAM244 membrane has the highest adsorption capacity, followed by the PAM433 and PAM622 membranes. The adsorption process achieved equilibrium in 20–30 min, then desorption occurs between 30 and 60 min. After 60 min of adsorption, the adsorption capacity of the PAM811 membrane was 0.89 mg g–1, which is higher than those of PAM244, PAM433, and PAM622, which range from 1.5 to 2 mg g–1. This result indicates that adsorption by the PAM811 film was slower than those of the other membranes. Therefore, 20 min was selected as the optimum time to further evaluate the effect of the membranes.
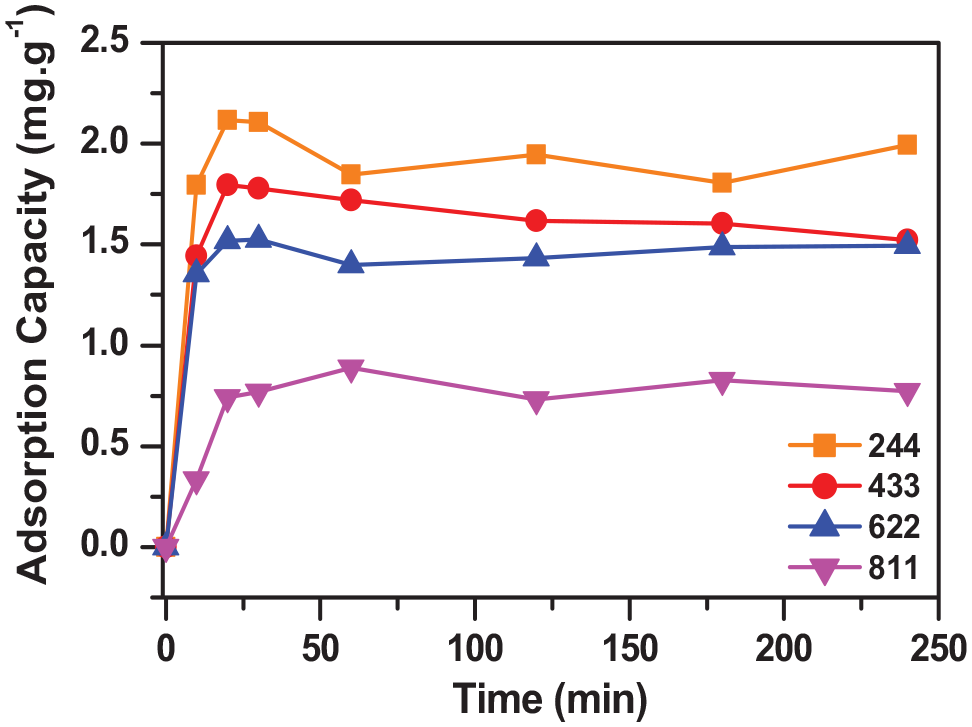
Effect of the adsorption time on the adsorption capacity of the PAM membranes.
Effect of the pH in solution
One of the important parameters in adsorption studies is the solution pH, which refers to the surface charge of the adsorbent and the antibiotic molecules. Before evaluating the effect of the solution pH on the membrane adsorption capacity, the measured pH point zero charged (pHpzc, pHz) value implies that pHz < solution pH indicates a negatively charged membrane surface, whereas pHz > solution pH indicates that the membrane surface is a negatively charged surface with a positive charge. Theoretically, based on electrostatic repulsion, the pHpzc value can be used to explain the influence of pH on the adsorption process. 29 The pHz values of the PAM244, PAM433, PAM622, and PAM811 membranes were recorded as pH 5.875, pH 5.625, pH 6.315, and pH 6.125, respectively. In Figure 3, the influence of the pH medium is evaluated from pH 2 to pH 10 over a period of 20 min. The results show that except for PAM811, the adsorption capacity of the membranes reaches its highest value at pH 6. It can be seen that the material works well in a neutral environment. While pHz < pH of the solution shows that the surface of the material was negatively charged, it easily adsorbs positive ions from CFX. Therefore, the electrostatic interaction between the positively charged CFX molecules and the negatively charged functional groups on the PAM244 and PAM433 films can promote the adsorption capacity. When pHz > solution pH, the material surface tends to be positively charged for the PAM811 and PAM622 films. Meanwhile, the free radicals of the membrane are mainly from hydroxy groups on the surface. Therefore, the PAM811 and PAM622 membranes can undergo physical adsorption through the pores inside the membrane. Therefore, pH 6 was selected as the most suitable pH to perform further experiments.

Effect of the solution pH on the adsorption capacity of PAM membranes.
Effect of the dosage of composite membranes
The content factors were then evaluated at 0.1, 0.5, 1, 2, and 3 g L–1. The results show that a high dosage lowers the adsorption capacity (Figure 4). This decrease was attributed to the formula for calculating the adsorption capacity. Membranes PAM244 and PAM433 gave their highest adsorption capacities at 2 g L–1. PAM811 and PAM622 gave their highest adsorption capacities at 3 g L–1. In the study by Hoang, it was shown that the film ratio had an effect on the adsorption capacity for the pigment methylene blue. 30 It can be seen that the material content that affects the adsorption process is partly controlled by the solubility of the film. 31 Due to the high water solubility, PAM622 and PAM811 had lower adsorption capacities than PAM244 and PAM433. The adsorption capacity at the above two dosage ratios did not change too much. Therefore, a dosage of 2 g L–1 was selected to evaluate the temperature factor.

Effect of the content of PAM811 (a), PAM622 (b), PAM433 (c), and PAM244 (d) on the adsorption capacities.
Effect of the antibiotic concentration
After evaluation of the dosage factor, the antibiotic concentration factor was evaluated between 10 and 100 mg L–1. The results show that the adsorption capacities of PAM244 and PAM433 both decreased after the antibiotic concentration exceeded 40 mg L–1. The PAM811 and PAM622 membranes reached their highest values at concentrations of 80 and 100 mg L–1, respectively (Figure 5). The process reached equilibrium when the adsorption capacity remained almost unchanged when increasing the antibiotic concentration. It can be seen that at different film ratios, the adsorption capacity at concentration ranges was also different. This can be explained by evaluation of the adsorption isotherm model. A prediction of the adsorption mechanism of the film can also be evaluated through the adsorption kinetics and isothermal modeling. Therefore, optimization of the adsorption process of the component films by the response surface method (RSM) model is performed based on separate parameters.
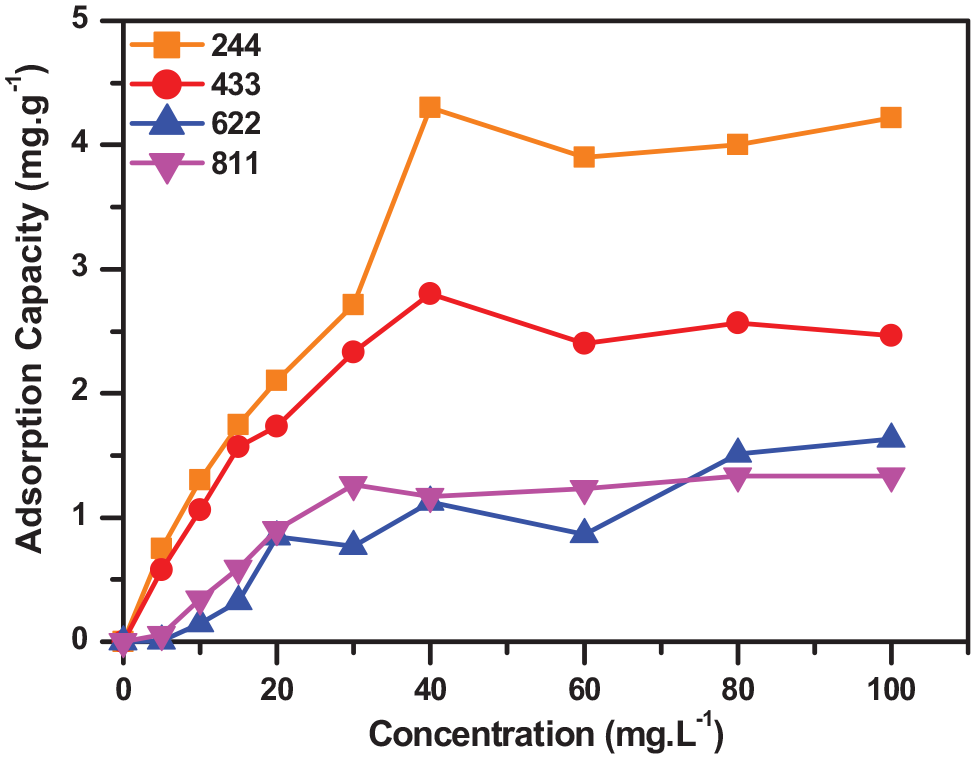
Effect of the antibiotic concentration on the adsorption capacity of the membranes.
Adsorption kinetics
The interactions between the membrane materials and pollutants are represented by kinetic models, such as pseudo first-order (PFO), pseudo second-order (PSO), the Elovich and the Bangham models. These models are described in Figure 6 with the parameters summarized in Table 1.

Adsorption kinetic models of PAM811 (a), PAM622 (b), PAM433 (c), and PAM244 (d).
Kinetic parameters for the adsorption of CFX on PAM244, PAM433, PAM622, and PAM811.

As shown in Figure 6, all the models were relatively similar, except for PAM811. The parameters of the model also show that the PAM811 film followed the PFO model with the correlation coefficient (R2 = 0.931). It can be seen that the PAM811 film adsorbs by way of a physical adsorption mechanism, which is described by the interactions between antibiotic molecules and the adsorbent membrane over time. 32 The PAM622 and PAM433 membranes showed high correlation with the kinetic models, especially the PSO kinetic model as evidenced by the high correlation coefficients (R2 = 0.987 and 0.953) compared to the models. The adsorption process can occur via many different mechanisms, the main of which is chemisorption. This can be attributed to the formation of pores and free functional groups on the surface of the material. These results are in agreement with previously published scanning electron microscope and Fourier transform infrared analysis results.30,31,33 In the case of the PAM244 membrane, it shows compliance with the models with the same correlation coefficients. In addition, the Elovich and Bangham models showed the presence of diffusion of antibiotic molecules into the membrane.
Adsorption isotherms
The adsorption process has been described through adsorption isotherm models, such as the Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich (D–R) models. These models are described and shown in Figure 7, with the parameters summarized in Table 2.
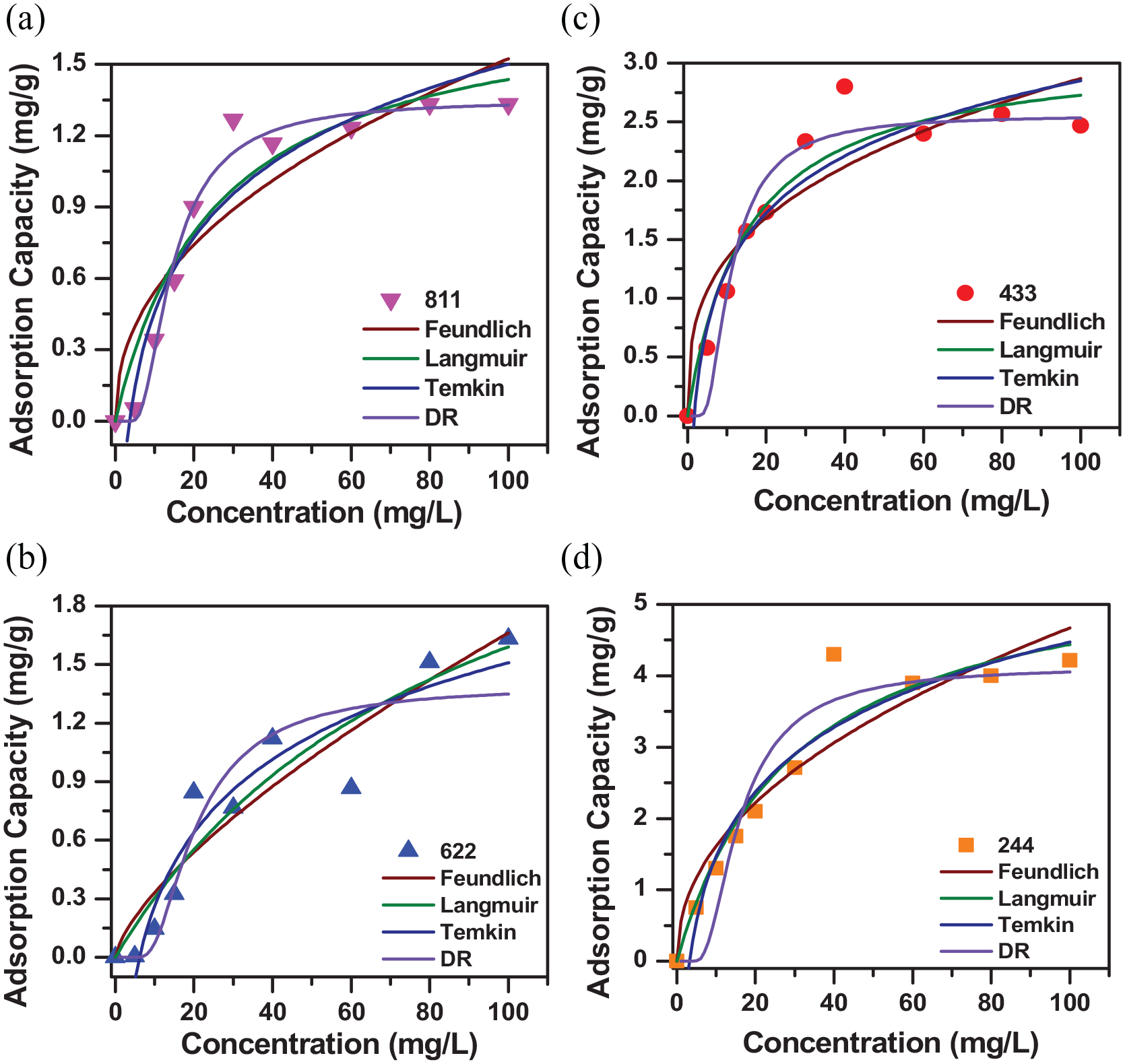
Adsorption isotherm models of PAM811 (a), PAM622 (b), PAM433 (c), and PAM244 (d).
Isothermal parameters for the adsorption of CFX on PAM244, PAM433, PAM622, and PAM811 films.

Looking at Figure 7, it can be seen that the experimental values are randomly distributed. For the PAM811 film, it can be seen that the DR model well describes the adsorption process with a high correlation coefficient (R2 = 0.984). The process describes a heterogeneous surface adsorption mechanism with a Gaussian energy distribution. The adsorbent size is thought to be equivalent to the micropore size and this is consistent with previously published Brunauer–Emmett–Teller measurements. For the PAM622 film, the Temkin model well describes the adsorption process with the correlation coefficient R2 = 0.901. The process describes uniform distribution and binding energies between the membrane and the antibiotic. Increasing the interaction of the bonds on the surface will reduce the thermal linearity of all the molecules inside the adsorbent layer. This is in agreement with the results of the temperature evaluation. For PAM244 and PAM433, the Langmuir model shows high compatibility with R2 values of 0.930 and 0.937, respectively. The adsorption process follows the Langmuir model, showing that it occurs on the surface of the adsorbent with uniform adsorption points and represents the model for monolayer adsorption. The separation coefficient of the model has been calculated and the results show that the adsorption process is favorable (0 < RL < 1). The adsorption capacities of PAM811, PAM622, PAM433, and PAM244 films were recorded as 1.801, 3.006, 3.136, and 5.758 mg g–1, respectively.
Optimization of the adsorption conditions
In order for the adsorption process to take place in the best way, the RSM model was used to optimize the parameters. To obtain a suitable evaluation, the PAM244 and PAM433 membranes, with the same good adsorption conditions were selected to evaluate the RSM model. The best conditions recorded for each membrane were PAM244 (20 min, pH 6, 2 g L–1, 40 mg L–1), and PAM433 (20 min, pH 6, 2 g L–1, 40 mg L–1). The parameters of the factors are designed experimentally using the Box–Behnken design (BBD) model and are divided into three levels: central variable (0), low level (–1), and high level (+1) (Table 3) 34 . To design the experiment, the total number of experiments of the BBD model is calculated according to the formula:
Matrix of independent variables and levels.


From the best adsorption conditions, the RSM model was built based on the central and boundary values. Based on the BBD model, the experimental results were used to evaluate CFX adsorption efficiency of the material samples (Table 4). From the RSM matrix, a total of 30 experiments were set up.
Table of BBD values of 30 independent experiments.

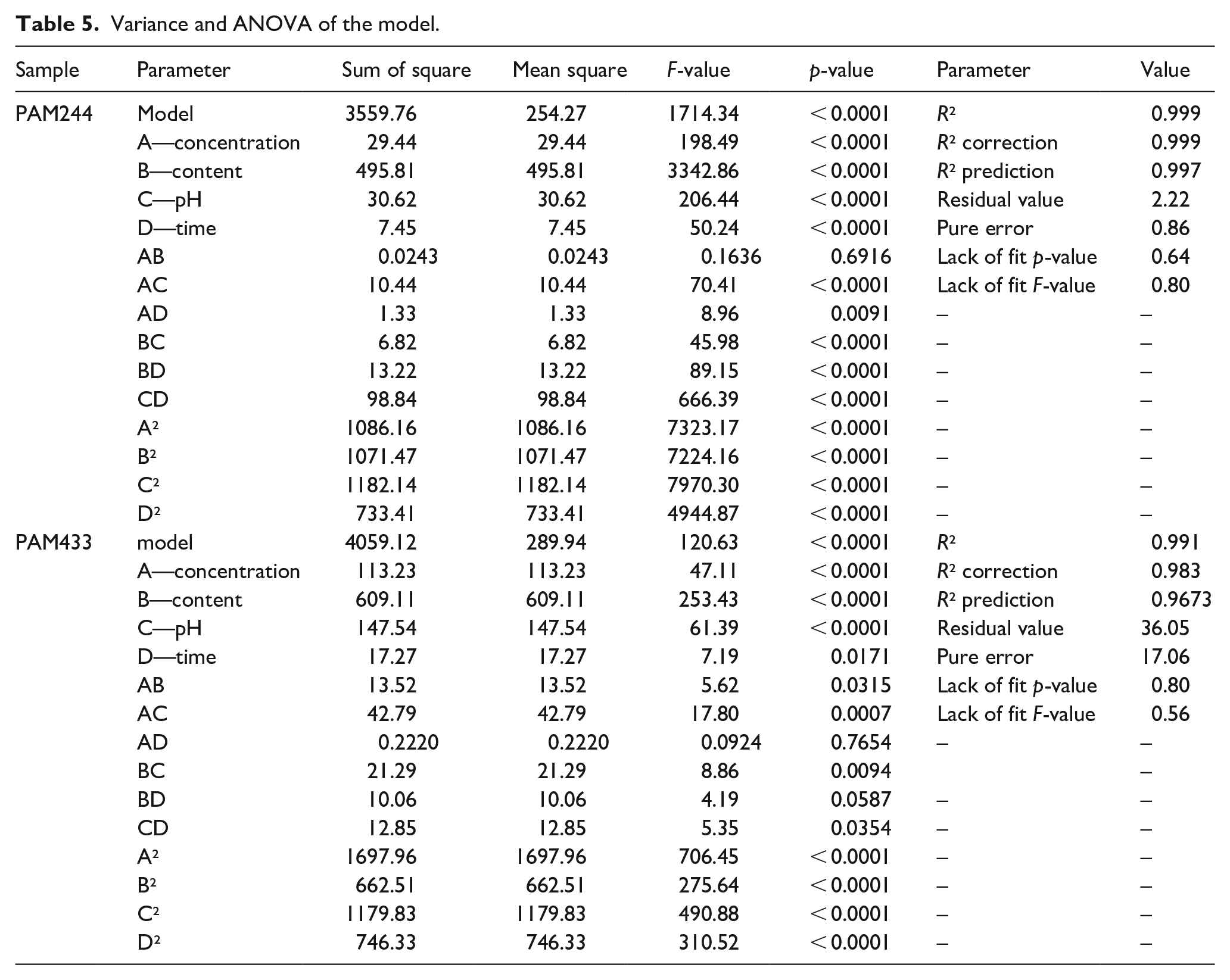
Analysis of variance (ANOVA) was calculated using Design-Expert software (version 11, State Ease, Minneapolis, USA). ANOVA of the quadratic linear regression model is used to analyze the influence of the input and output variables as well as the correlation of response functions and independent variables. From the RSM model, the recorded results include the model’s quadratic equation of the model, the residual value plots, the line and 3D inter-factor plots, and the variance analysis table. From the practical experiments, the residual value charts are shown in Figure 8. It can be seen that the experiments are independently distributed and the quadratic equation is used to calculate the variance values.
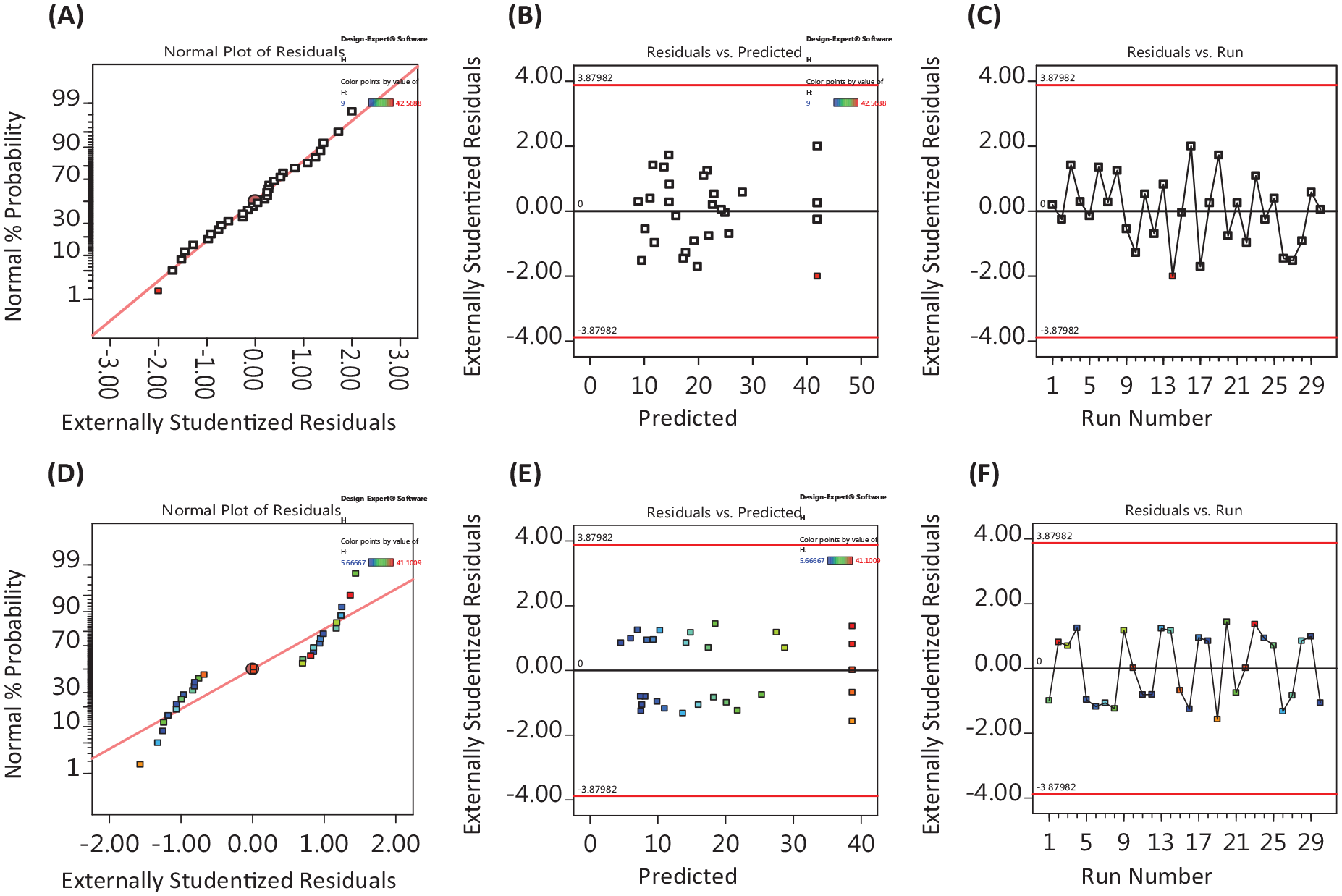
Graphs of the residual values of PAM244 and PAM433.
The quadratic equation describing the correlation between the response factor (y) and the independent variables is determined as follows:


The ANOVA data for the regression equations are presented in Table 5. The significance of the regression models is determined by the correlation coefficient (R2), the p- and F-values. In general, a smaller p-value and a larger F-value indicated the statistical significance of the model. The model is considered effective when p < 0.05, and not effective when p > 0.05. The model F-values of 1714.34 (PAM244) and 120.63 (PAM433) imply that the model is significant. The lack of fit of the F-value of 0.80 (PAM244) and 0.56 (PAM433) implies that the degree of mismatch has no effect on the standard error of the model. The lack of fit value from the model is good to help the model predict a better outcome. The value of p < 0.05 shows the statistical significance of the effect factors (95% confidence level). In this case, A, B, C, D, AC, AD, BC, BD, CD, A², B², C², and D² are the important conditions of the model. A p-value > 0.1 indicates that the model conditions are not significant. If many model conditions are not significant, reducing the number of experiments can improve the model. The results of ANOVA analysis show that the model has statistical significance (p < 0.05), the regression coefficients of PAM244 and PAM433 are R2 = 0.999 and 0.991, which are both greater than 0.95, thus proving that the model is compatible with the experiment.
Variance and ANOVA of the model.
The RSM model was used to optimize the experimental conditions, such as the antibiotic concentration (mg L–1), the dosage of PAM244 or PAM433 adsorbent (g L–1), the time, and the pH value. The response surface plots also show the optimal region of the model (Figures 9 and 10). After evaluating the RSM model, it can be seen that the PAM244 material has a convergence point for the pH value in the range of pH 5.5–pH 6.5, the concentration from 35 to 45 mg L–1, the time from 15 to 25 min, and the content ranges from 1.75 to 2.75 g L–1. For the PAM433 material, there is a convergence point with a pH value in the range of pH 5.5–pH 6.5, the concentration is about 35–45 mg L–1, and the concentration ranges from 2 to 3 g L–1. The surface graphs all show the optimal area of the evaluation model with the interaction between factors.

The plots show the correlation between the factors (a) dosage and concentration, (b) pH and concentration, (c) time and concentration, (d) dosage and pH, (e) dosage and time, and (f) pH and time of the PAM244 membrane.

The plots show the correlation between the factors (a) dosage and concentration, (b) pH and concentration, (c) time and concentration, (d) dosage and pH, (e) dosage and time, and (f) pH and time of the PAM433 membrane.
The optimal values are calculated and selected based on the optimal area from the influencing factors. The optimal values of PAM244 and PAM433 are clearly shown in Figure 11. For the PAM244 material, the optimal value was recorded at pH 5.89, a concentration of 41.28 mg L–1, a time of 19.87 min, and with a dosage of 2.08 g L–1. Under optimal conditions, the adsorption capacity predicted from the model was 11.52 mg g–1 with an efficiency of 42%. For the PAM433 material, the optimal value was recorded at pH 6.07, a concentration of 41.24 mg L–1, a time of 19.59 min, and with a dosage of 2.16 g L–1. Under optimal conditions, the adsorption capacity predicted from the model was 10.46 mg g–1 with an efficiency of 40%. From the optimal results, it can be seen that PAM244 has better adsorption capacity than the PAM433 membrane.
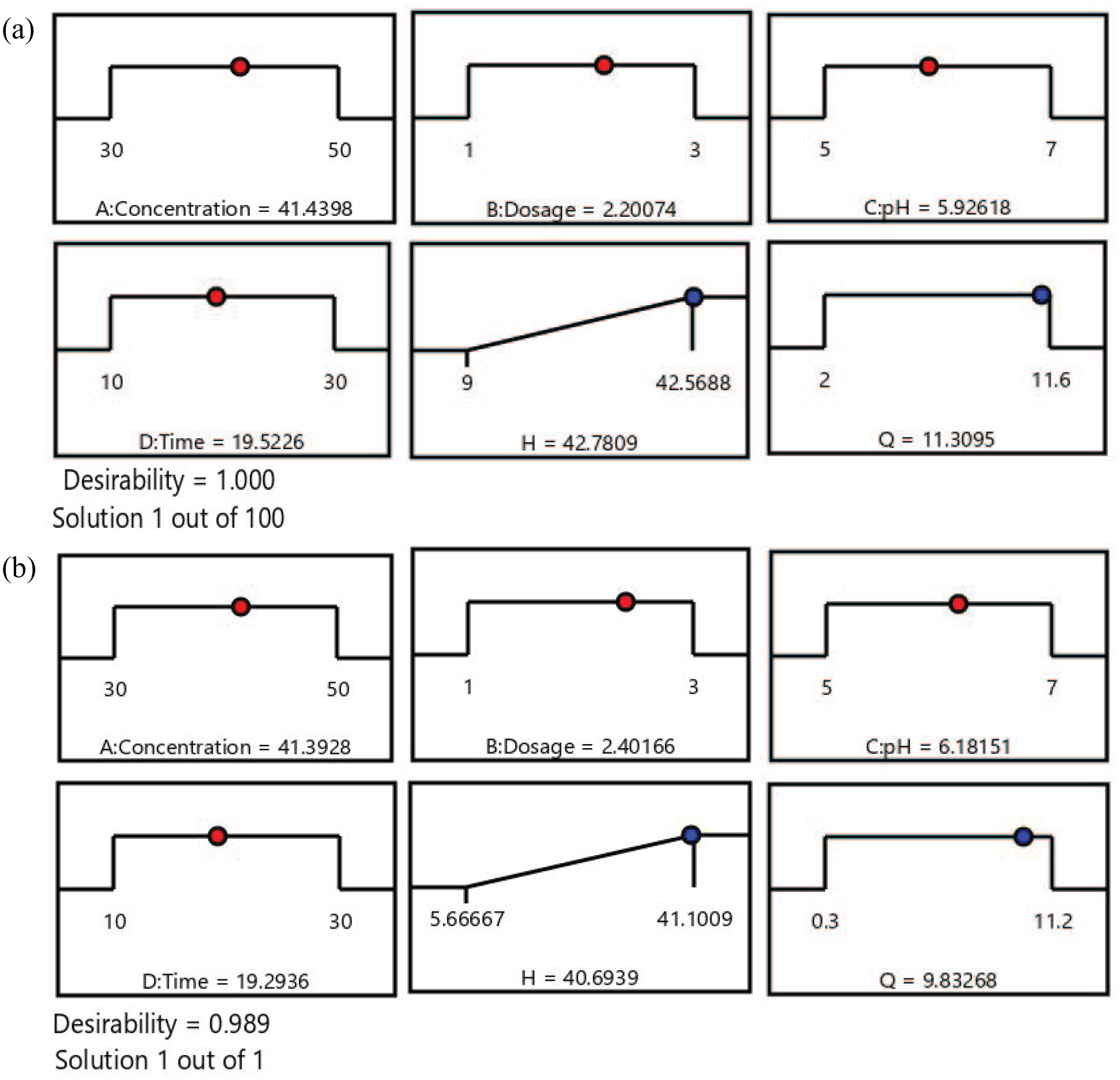
Optimal values from the RSM models for membranes PAM244 (a) and PAM433 (b).
Reusability
The PAM244 and PAM433 membranes were evaluated for their reusability with water as the solvent. The reuse process was evaluated by employing the optimal adsorption conditions of the PAM244 film (pH 5.89, concentration 41.28 mg L–1, time 19.87 min, and content 2.08 g L–1) and the PAM433 film (pH 6.07, concentration 41.24 mg L–1, time 19.59 min, and content 2.16 g L–1). The first adsorptions (reuse zero) were recorded at 12 mg g–1 (PAM244) and 10 mg g–1 (PAM433), respectively. It can be seen that the adsorption capacities under the optimal conditions are close to the previously predicted adsorption capacities. During the first reuse, the adsorption capacities of the PAM244 and PAM433 membranes were significantly reduced to 10 and 6 mg g–1, respectively. During the second reuse, the adsorption capacity of the PAM433 film was reduced by more than 50% and for the third reuse, the membrane was almost incapable of adsorption. For the PAM244 membrane, during the third reuse, the adsorption capacities were reduced to 5 mg g–1. These results show similarities with other types of component membranes. In studies by Pham’s group, their nanosilica membrane was reused three times to remove the antibiotic amoxicillin and four times for the antibiotic OTC.35,36 This shows that the reusability of the membrane to remove antibiotics is more limited than for the dye removal described in previous studies.30,31

PAM244 and PAM433 membrane reusability.
Discussion
In this study, PAM composite membranes at different ratios were studied for the removal of the antibiotic of CFX from water. The ratios of the membranes were evaluated for their adsorption capacities and the factors affecting the adsorption process: time, pH, temperature, concentration, and content. The results show that a changing the ratios of the components leads to different adsorption capacities clear difference is shown for the PAM811 membrane as follows 60 min, pH 6, a dosage of 3 g L–1, a concentration of 80 mg L–1, and a temperature of 50oC. Meanwhile, films with other ratio have similar adsorption conditions: 20 min, pH 6, content of 2 g L–1, a concentration of 40 mg L–1, and a temperature of 30oC. This difference is also reflected in the kinetic and isothermal models of adsorption. The PAM811 membrane follows the DR isotherm and PFO kinetics. The PAM622 model follows the Temkin isotherm, and the PSO, Elovich, Bangham kinetics. The PAM244 and PAM433 films follow the Langmuir isotherm and the PFO, PSO, and Bangham kinetics. It can be seen that at different rates the adsorption process also takes place differently. The PAM244 and PAM433 membranes were selected for optimization of the adsorption process via the RSM model using the Box–Behnken experimental design model. The optimal model shows compatibility between the experimental and predicted values of the model. The correlation coefficient (R2) of the model has also been recorded, with PAM244 and PAM433 being > 0.95. The model proposed the optimal parameters. For the PAM244 material, the optimal values are as follows: pH 5.89, a concentration of 41.28 mg L–1, a time of 19.87 min, and a dosage of 2.08 g L–1. Under optimal conditions, the adsorption capacity predicted from the model is said to be 11.52 mg g–1 with an efficiency of 42%. For PAM43, the optimal values are a pH of 6.07, a concentration of 41.24 mg L–1, a time of 19.59 min, and a dosage of 2.16 g L–1. Under optimal conditions, the adsorption capacity predicted from the model is found to be 10.46 mg g–1 with a 40% efficiency. Furthermore, the PAM244 and PAM433 membranes showed potential reusability over two to three reuse. The findings from the present study provide an essential insight for further applications of PAM membranes in antibiotic removal on industrial scale.
Experimental
Chemicals
PVA (M = 160,000 g/mol, solubility 86.5–89%) and maltodextrin (C6nH(10n+2)O(5n+1)) were obtained from HIMEDIA Laboratories (Mumbai, India). Agarose was obtained from VWR BHD Prolabo Chemicals (Singapore). CFX, CPC, TCC, and OTC were obtained from Sigma-Aldrich (US). Ethanol (purity ⩾ 95%) was obtained from Xilong Science Co., Ltd. (China). All chemicals and reagents are of analytical grade.
Preparation of the composite membrane
The membrane films were synthesized by the casting method at different component ratios to give PAM244 (PAM at 20%/40%/40%, w/w), PAM433 (PAM at 40%/30%/30%, w/w), PAM622 (PAM at 60%/20%/20%, w/w), and PAM811 (PAM at 80%/10%/10%, w/w), based on our previous studies.30,31,33 Components were dissolved in 100 mL of distilled water at 70°C. 31 After mixing three solutions, 0.5% of glycerol (v/v) was added to the mixture and homogenized for 1 h using a magnetic stirrer. The mixture was poured into a mold and dried at 45ºC for 48 h to allow film formation. The films were dried at 40ºC before characterization. 30
Adsorption experiments
The as-synthesized membrane (0.1–3 g L–1) was added to 100 mL of the antibiotic solution (concentration 10–100 mg L–1) in an Erlenmeyer flask. The mixture of the PAM films and the antibiotic solution was agitated on a Thermal Incubation Shaker at 200 rpm for 180 min at 30oC. The absorbance of the TCC, CFX, OTC, and CPR solutions were measured using a UV−VIS spectrophotometer (Shimadzu-1601, Tokyo, Japan) at 273, 275, 275, and 280 nm, respectively. The dye adsorption capacity (Qe) was calculated according to equation (4):

where Ce is the post-adsorption concentration (mg L–1); Co is the pre-adsorption concentration (mg L–1); W (g) is the mass of the adsorbent, and V (L) is the solution volume.
Reusability
The reusability of hydrogel-based adsorbents is important to expand their practical applicability. Inspired by this aspect, PAM244 and PAM433 were used over several cycles. The post-adsorbed film was adsorbed under optimal conditions from the RSM model. Specifically, the regeneration procedure was initiated by washing PAM244 and PAM433 containing antibiotics with a mixture of ethanol and water three times. The regenerated film is the dried at 45oC and can be reused in subsequent cycles.
Adsorption kinetics
Based on by Tan and Hameed studies, the adsorption kinetic models were applied to predict the adsorption process. 37 Kinetic models were assumed for the adsorption process and the reaction rates for adsorption film. The experimental data are described by kinetic models, such as Elovich, Bangham, PFO, and PSO in the nonlinear forms shown in Table 6. 37
Nonlinear parameters of different kinetic models.

Adsorption equilibrium isotherm
Based on the study of Batool, isothermal models were applied to assess the behavior of the adsorption processes. 38 An isotherm is the basis for considering the interaction between adsorbents and adsorbents in gas/liquid/solid interfaces. The adsorption processes are provided by the adsorption isotherms. The advantages and behavior of the adsorption processes are precisely described by isothermal models, such as those developed by Langmuir, Freundlich, Temkin, and D–R. 38 these models are calculated in nonlinear forms in Table 7. 38
Nonlinear parameters of different isothermal models.

Response surface methodology
In this study, RSM was used to optimize antibiotic adsorption on activated carbon samples from peel and pulp. Several influencing factors were investigated, including solution pH, function amount of adsorbent and initial antibiotic concentration. The evaluation factors of the model are divided into three or five levels: central variable (0), low level (–1), high level (+1) and level ±α (±α). ANOVA was calculated using Design-Expert software (version 11, State Ease, Minneapolis, USA). ANOVA of quadratic linear regression model is used to analyze the influence of input and output variables as well as the correlation of response functions and independent variables [59]. The value of α and the total number of experiments of the central composite design (CCD) and BBD matrix are calculated according to the formula:


Footnotes
Acknowledgements
Author Contributions: Writing—original draft preparation, H.N.B. and H.D.T.; data curation, H.D.T; methodology, L.V.T.; writing—review and editing, L.G.B. and H.N.B. All authors have assented to the manuscript’s published version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Nguyen Tat Thanh University, Ho Chi Minh city, Vietnam with code (grant no. 2023.01.09/HĐ-KHCN).
