Abstract
The separation of air (O2/N2) via a polymeric membrane has recently piqued the interest of academic researcher as well as the industrial sector. Because of its remarkable characteristics, the polymeric membrane has emerged as one of the innovative and fast growing technology. However, two major problems faced by membrane technology, which hinder its growth in the commercial sector are, 1): The trade-off between permeability and selectivity. 2): Maintaining physical and chemical structural stability in a long-term commercial scale process. Recent advancements in membrane material, structural, and process design have enabled the development of dual-layer composite (DLC) membranes. This concept combines the benefits of both thinner mixed matrix membranes (MMMs) based active layer and porous support substrate. Due to these properties, the membrane exhibits higher perm-selectivity with enhanced mechanical strength as compared to single layer polymeric membrane. This review article mainly focused on the developmental progress of DLC membrane throughout the years. In which membrane structural details, selection criteria, fabrication methodologies, application [e.g., air (O2/N2) separation] were critically reviewed. In addition, challenges arising in the DLC membrane production and future prospects for the development of these membranes were also thoroughly discussed in this literature. This creates a paradigm for future research in the commercial development of these membranes for the air (O2/N2) separation process, which can be utilized in both medical and industrial sectors.

Keywords
Introduction
Membrane-based gas separation (MBGS) and purification technology has attracted the interest of both academic researchers and industrial sector in recent years. In-order to meet the end requirements of products, this technology can achieve sustainable industrial growth. Since 1970, membrane technology has become a viable substitute for the conventional technologies. Because of its remarkable characteristics, such as efficient and energy saving process, membrane technology is considered as the best alternative method for gas separation (GS) applications. Up till now, polymeric based membrane technology has been widely used for various commercial GS processes, which include natural gas purification (CO2/CH4) for methane enrichment, carbon capture from flue gas (CO2/N2), air separation (O2/N2) for oxygen enrichment, and syn-gas separation (CO2/H2) for hydrogen enrichment. 1
Over the past few decades, air (O2/N2) separation, for the purpose of oxygen (O2) enrichment, has gained quite a substantial value in both the industrial and medical sector. In the industrial sector, O2 gas has been utilized in various chemical oxidation processes, such as gasification, natural gas purification and oxy-fuel combustion process. 2 In the medical sector, enrichment of O2 gas has also gained extensive importance. O2 gas has been used in various medical treatments and therapy, such as oxygen therapy for the treatment of toxicity in blood (e.g., carbon monoxide toxicity reduction). 3 Also used in the treatment of novel corona virus (COVID-19), 4 open heart surgeries and lung transplantation. 5 Aside from these uses, it has also been used in waste water treatment, 6 whereas byproduct (retentate) nitrogen (N2) gas can also be used in a variety of applications, such as in food industries, pharmaceutical and chemical industries, which include packaging medicine and fertilizer production processes. 7
Therefore, a wide range of conventional methods have been used for the air (O2/N2) separation, such as pressure swing adsorption (PSA) process and cryogenic distillation process unit.1,2 However, these conventional technologies have a number of drawbacks, such as these processes are highly energy intensive processes, require large areas (large footprint), and are difficult to scale-up due to their high cost of unit, whereas membrane technology has numerous advantages over the conventional technologies for gas separation in the industrial sector, such as, 1): It required no complex instrumentation and knowledge to install the process. 2): Gas separation is carried out continuously. 3): Energy consumption is lower as compared to conventional technologies. 4): Membrane technology can easily be combined with other separation technologies to form a hybrid technology for gas separation. 5): It is easily scalable. 6): Required no mass transfer agent or additive for the separation process. 7): Membrane technology has greater flexibility in adjusting variables and designing systems. 8): It is a clean technology with operational ease, also known as one of the green technology systems.7–9
Advances in membrane materials, configuration, and fabrication methods, for example, can broaden the market for MBGS applications. These areas of studies are still in the research phase, which need to be fully understood in-order to enhance MBGS performance. A wide range of dense polymeric membranes have already been investigated. Many improvements have been achieved, such as higher selectivity and higher gas permeate flux in-order to separate the desired gas component from a mixture. However, the gas permeability of a polymeric dense membrane also depends on its thickness. Generally, these membranes are made around a thickness of 50 μm–150 μm. Due to its thicker structure, gas diffused slowly. This leads to one of the major problems of lower permeation rate of gas components through membrane. However, its thickness can be further reducible to a highly thin dense layer. But due to its fragile polymeric structure, it cannot be directly used for the high pressure GS process. 10 For this purpose, the concept of dual-layer composite (DLC) membrane was introduced. In these membranes, a porous support layer is attached beneath the thin, dense membrane, which provides mechanical support to the thin selective membrane structure and has negligible resistance toward gas transport.
In this review paper, the progress of DLC membrane for the application of O2/N2 gas separation has been thoroughly discussed, which provides information about structural details of DLC membranes, selection criteria, different fabrication methods, application such as air (O2/N2) separation, current challenges with its solution, and future research possibilities for the improvement of DLC membrane.
Overview of dual-layer composite membrane and its classification
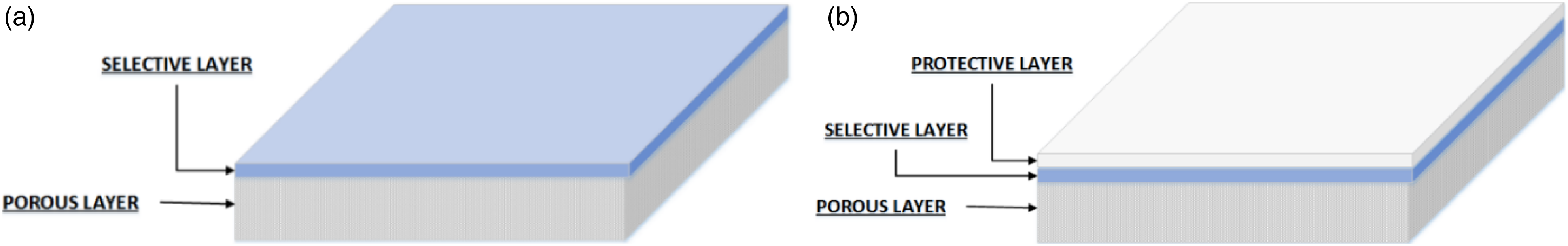
The DLC membrane has a similar structure to an asymmetric membrane, which consists of a thin dense layer on top of a porous support. However, unlike asymmetric membrane, both layers of these membranes are composed of different materials. The top thin dense layer acts as a selective layer, which helps in separating components of the gaseous mixture, whereas the bottom porous layer provides mechanical support to the top thin selective layer, having negligible resistance toward gas transport as illustrated in Figure 1(a). To enhance the shelf life of the DLC membrane, an additional protective layer is also applied, which prevents damage to the selective layer, as depicted in Figure 1(b). Due to this structural design, DLC membranes showed high selectivity and high permeability in the separation process. These membranes are preferred and can be used in high pressure range GS applications. These membranes also have numerous advantages over asymmetric membranes, such as a): independent selection of materials for each layer (e.g., selective layer and porous support layer) are possible. b): Both selective and porous layers can be independently prepared and then combined together. c): As only a thin selective layer is required on top of porous support in DLC membranes, for this reason, expensive materials for the selective layer can be used in lower quantity.
11
Composite membrane types a): DLC membrane and b): DLC membrane with protective layer.
12
Dual-layer composite membrane criteria for gas separation applications
The selection of DLC membrane mainly relies on the material’s physical and chemical properties. Since these membranes are used for GS applications, they should have a number of properties, which are mentioned below.12,13 • The selective layer should have high selectivity for the required component of the gas mixture. • The dual-layer of the membrane should have good adhesion and compatibility. • It should have high permeability/flux toward the selected components of the gas stream mixture. • It should be physically and chemically stable. • Having a long shelf life. • Fouling resistance. • Higher mechanical strength, in-order to withstand high pressure GS process. • Plasticization resistance. • It should be cost effective (economically good). • It should be inexpensive to manufacture and can be produced into a variety of membrane modules.
Fabrication methodologies of dual-layer composite membrane
DLC membranes are generally fabricated by deposition of a thin selective layer on a porous support layer, which is composed of different materials. Throughout the years, various methods have been developed for the fabrication of DLC membranes, which include solution casting, dip-coating, spin-coating, interfacial polymerization, and chemical vapor deposition method. Their detailed descriptions are mentioned below.
Solution casting
Solution casting is one of the most commonly used method for the fabrication of DLC membrane on lab scale as illustrated in Figure 2. An automatic or manual membrane caster, which consists of doctor blade, is typically used in this technique to apply coating to the surface of a porous support membrane. The thickness of the coated layer can be controlled by adjusting the distance between the doctor blade and the porous substrate. The selective layer thickness can be controlled below 10 μm, but practically fabricating thickness below 1 μm is quite a challenging task.9,12
Dip-coating
The dip-coating method is the simplest technique for the fabrication of DLC membrane as illustrated in Figure 3, in which a selective layer is deposited by dipping the porous support layer in the polymeric solution for some time and then lifted at a controlled speed. The main parameters to control the thickness of the coating layer are as mentioned below: (a) Polymeric solution concentration. (b) Dipping time. (c) The speed for the withdrawal of porous substrate. (d) Evaporation rate and environmental impact.

On the basis of these parameters, several predictive mathematical models have been developed to predict the coated layer thickness, such as the Landau and Levich model as mentioned in equation (1) below
14

In which h is the coating thickness, c is a constant (0.944 for Newtonian liquids),
Interfacial polymerization
Interfacial polymerization, is another method for the fabrication of DLC membrane as illustrated in Figure 4, proposed by John Cadotte, which was further developed by North Star Research. In this method, a porous support layer is dipped in a reactive coating solution in-order to deposit it in the pores of the porous layer. This coated support layer was then immersed in a reactant bath for some time, forming a highly cross linked selective layer on the surface of the porous substrate. At the end, heat treatment is often applied in order to complete the fabrication process. Through this method, the dense layer thickness can be achievable up-to a range of 0.1 μm or less. The major problem in the DLC membrane prepared through this method is less cross linked hydrogel formation in the pores of the support membrane. When dried, it becomes rigid and adds resistance toward gas transport. Another problem is the fragility of thin layer developed through interfacial polymerization. However, applying a coating layer reduces this issue, whereas less cross linked hydrogel formation can be prevented by adding a gutter layer on the porous support before immersion in the reactive solution.9,15 Schematic diagram of interfacial polymerization fabrication method.
9

Spin-coating
Spin-coating is a film coating method that is commonly used in the fabrication of DLC membranes, as shown in Figure 5. The device used for this process is called a spin-coater. In this method, a thin uniform layer is coated by spreading polymeric solution on a rotating porous substrate through centrifugal forces, whereas the solvent evaporation during this process is highly dependent on the properties of the solvent and speed of spinning. As the evaporation process is accelerated by air flow induced mechanism through high rotation speed. For this reason, the film solidified within a minute during the spin-coating process. In industries, the spin-coating method is usually used for the manufacturing of advanced electronic devices. The coating layer thickness can be controlled by changing the rotation speed and concentration of polymeric solution.
16
Schematic diagram of spin-coating fabrication method.
10

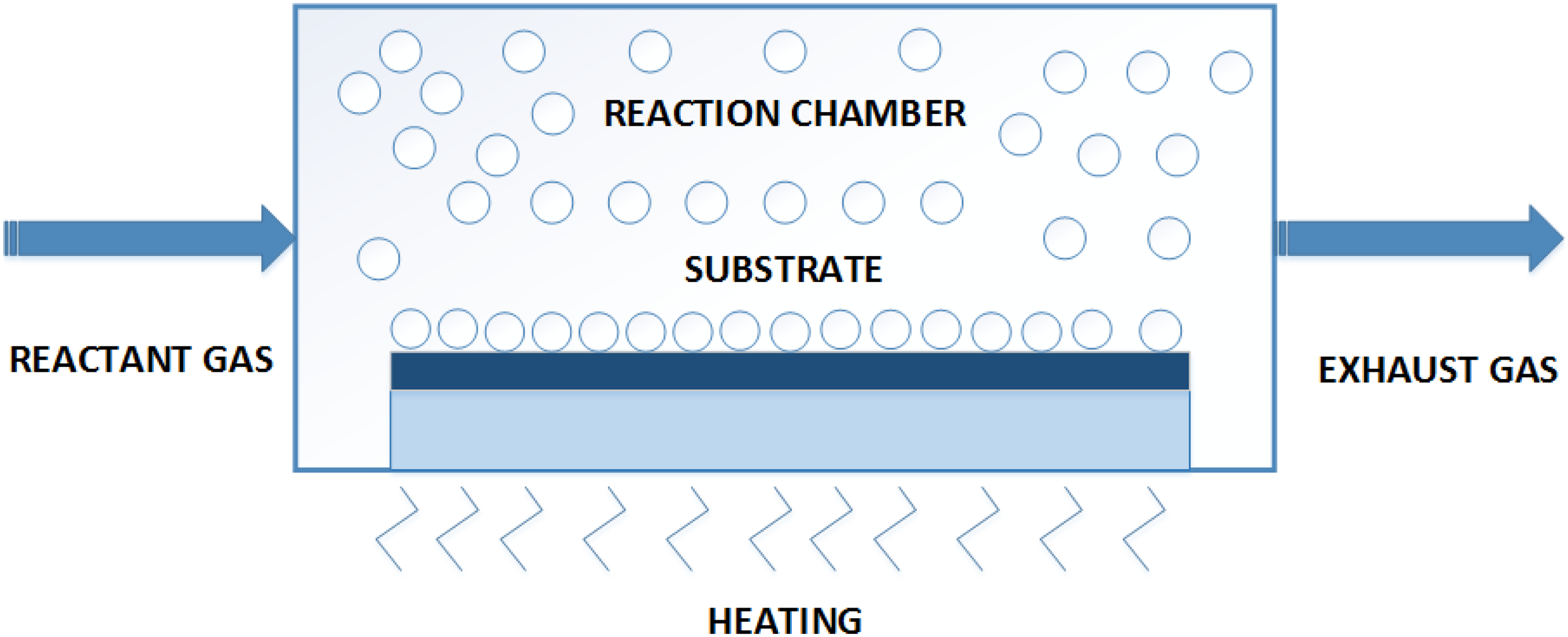
Chemical vapor deposition
Chemical vapor deposition (CVD) is a film deposition technique as illustrated in Figure 6, in which through chemical treatment, vapors of materials are deposited on the surface of a porous substrate. The film deposition can be controlled through changing the condition applied in a chemical reaction, which include materials of substrates, temperature, total pressure applied, flow rate, and composition of gas mixture used for reaction etc.
17
CVD can also be used for material deposition processes that are insoluble in nature. Another advantage of this method is that it can be used for surface modification of substrate, such as narrowing the pore size by deposition of material on the surface of a porous substrate.
18
Gas transport mechanism of dual-layer composite membrane
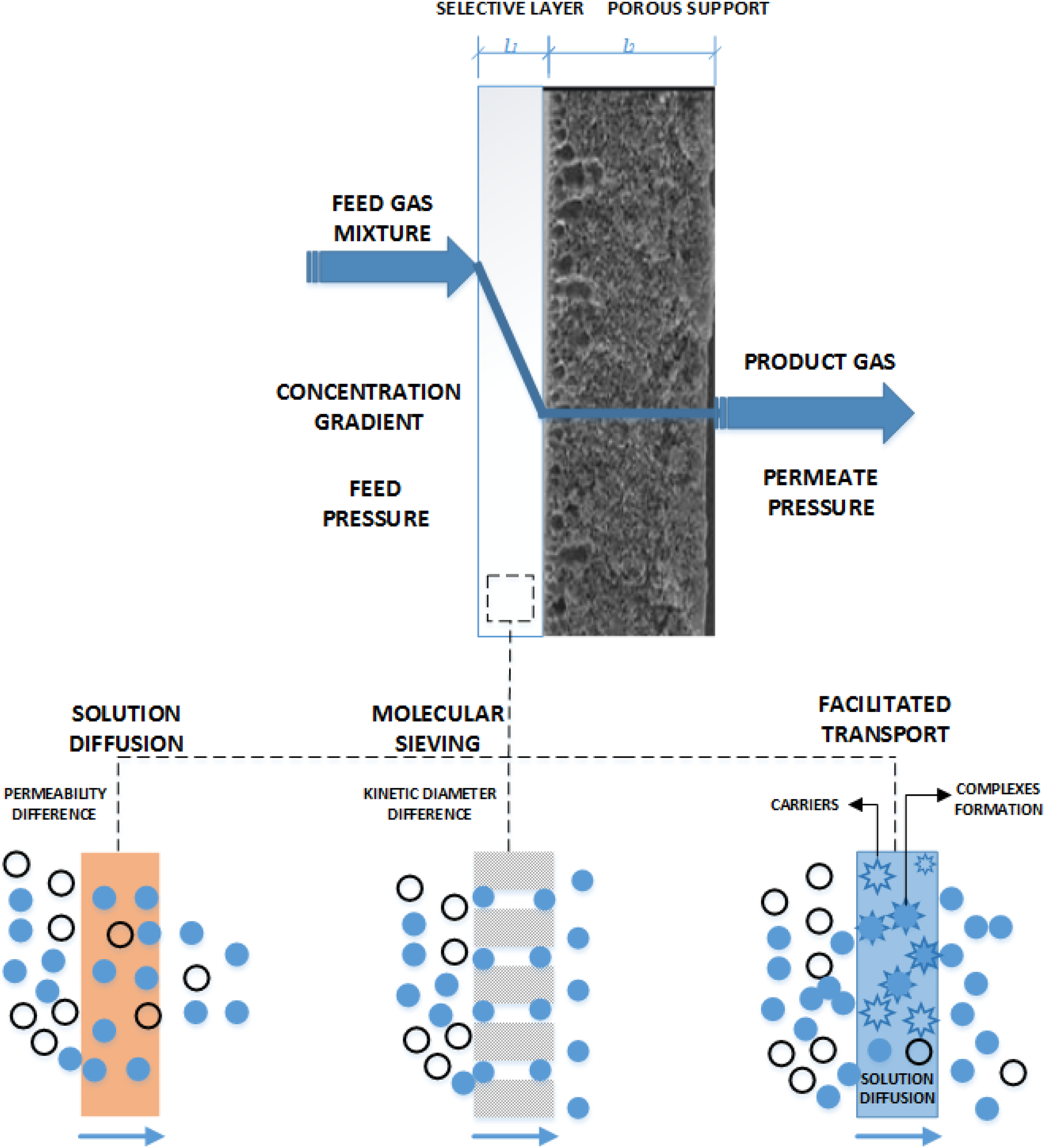
A DLC membrane separates a gas mixture because different components of the gas mixture exhibit different permeability through a selective layer of membrane. A greater difference between the components of the gas mixture should be preferred to achieve higher MBGS performance, whereas this difference comes from their physical and chemical properties as well as their interaction with membrane material. By surface modification or incorporating nano-particles ( MOFs, COFs, ZIFs, MWCNTs, etc.) in polymeric membrane matrix, DLC membranes can exhibit various gas transport mechanisms, which include solution diffusion, molecular sieving, and facilitated transport as illustrated in Figure 7. Their detailed descriptions are mentioned below. Gas transport mechanism of dual-layer composite (DLC) membrane.
1
Solution diffusion
A dense membrane has no pores or channels for gas transport, so it follows a solution diffusion mechanism. There are three main steps occurred in this transport mechanism, as illustrated in Figure 7. a): It absorbs a specific gas component of the mixture. b): In the second step, due to the concentration gradient, absorbed gas diffuses through the membrane. c): The gas desorbs at the permeate side under lower pressure in the final step. In this mechanism, the gas components of the mixture separate on the basis of their difference in permeability through the membrane.9,19
Molecular sieving effects
In this mechanism, the separation of gas components takes place due to difference in their kinetic diameter as mentioned in Table S1. By incorporating different nano-particles in a dense polymeric membrane, the selective layer can also exhibit molecular sieving effects with the combination of solution diffusion mechanism. As different nano-particles, such as (MOFs, COFs, MWCNTs etc.) have different sizes of pore aperture, which acts as a molecular sieve toward the gas components of the mixture as illustrated in Figure 7.20,21
Facilitated transport
This mechanism of gas transport also works with the combination of solution diffusion in DLC membrane as illustrated in Figure 7. In this mechanism, the gas components of the mixture are separated, by forming a complex through temporary bonding with the carriers. These complexes then diffuse through the membrane due to the concentration gradient. Afterward, complexes undergo a reversible reaction, which releases gas component at the permeate side under lower pressure.1,22
Factors affecting membrane-based gas separation
Membranes act as a selective barrier between the components of a gas mixture, as illustrated in Figure 8, which represents the separation of binary gas mixture through a DLC membrane. The product stream is enriched with the desired gas component, whereas the retentate stream is enriched with the remaining (undesired) component of the gas mixture. The GS performance of a membrane is represented by two main factors, i): Permeability of desired gas component (Pi) and ii): Selectivity Schematic diagram of MBGS system applied on gas mixture.
13




Progress of dual-layer composite membrane for air (O2/N2) separation
Air is the main source of both oxygen (O2) and nitrogen (N2) as mentioned in Table S1. Air consists of 20.95% of O2, 78.09% N2, and the remaining 0.96% comprises of other gaseous mixture.23,24 Both oxygen (O2) and nitrogen (N2) gas have many chemical and medical applications. For its enrichment, various conventional technologies have been developed, such as cryogenic distillation and pressure swing adsorption (PSA) process unit.1–7 But in recent years, through the latest technology and research, a large number of commercial grade membranes have been produced, which showed promising performance results in pilot scale tests. The progress details of reported membranes are mentioned in Table S2, which shows information about the membrane materials, fabrication methods, module/configuration, operating conditions, permeability, and selectivity of membrane.
Throughout the years, DLC membranes have been manufactured using different techniques such as solution casting, dip-coating, interfacial polymerization, spin-coating, and CVD techniques, as mentioned in Table S2. In-order to improve their GS performance, various methods were developed, such as blending of different polymers, incorporating nano-particles in the polymeric membrane matrix, and functionalizing the active layer, which increases its affinity toward the selective gas component of the mixture.
Recently, Nikpour N. et al., 25 fabricated a flat-sheet DLC membrane, in which the active layer is based on mixed matrix membranes (MMMs), composed of Pebax-1657 incorporated with BaFe12O19 nano-particles. It was coated on a PES porous support layer by using a solution casting method. The GS performance of these membranes showed O2/N2 selectivity of 3.34–4.01 and O2 gas permeance in the range of 0.08,233–0.06,377 (GPU) at a magnetic field (H = 0.5 T) and temperature of 25°C. J. Han et al., 26 studied highly selective O2/N2 separation membrane based on facilitated transport mechanism by using a porphyrin based O2 carrier (T (p-OCH3) PPCoCl). In this study, dip-coating method was used to coat a 1 μm thick active layer on flat-sheet porous support membrane (100 μm) based on Pebax-2533. The resulted membrane showed O2/N2 gas selectivity in the range of 2.2–7.6 with O2 gas permeability of 6.3–12.3 (Barrer), at a pressure range of 0.35–8 bar and a constant temperature of T = 18°C.
Another research group, K.C Chong et al., 27 developed a DLC hollow fiber membrane for oxygen enrichment through a dip-coating method, which consists of an active layer of Poly (ether block amide) having a thickness of 50 ± 4 μm, whereas the support layer is composed of PSF polymeric material. The membrane showed O2/N2 gas selectivity of 3.71 and O2 gas permeance of 39.81 (GPU) at a pressure of 5 bar. However, further studies on PEBAX polymer as coating material needed to be considered for the improvement of air (O2/N2) separation performance. M. Salehi Maleh et al. 20 produced three different flat-sheet DLC membranes, using a solution casting method. These membranes consist of MMMs based on active layers and porous support, in which three nano-fillers, such as NaX, ZIF-8, and SiO2 particles were incorporated into each Pebax-1657 polymer matrix (active layer) on top of the PES support membrane. These DLC membranes showed O2/N2 gas selectivity of 6.06, 3.85, and 3.52, respectively, whereas O2 gas permeance of 2.87, 4.01, and 3.17 (GPU) were observed, respectively, at a temperature of 25°C and pressure of 4 bar.
Chong KC et al., 28 compared two different coatings of 1 wt.% PEBAX and 3 wt.% PDMS active layer on PSF hollow fiber membrane for the O2/N2 GS process. The dip-coating method was used for the preparation of these DLC membranes. The fabricated membranes were tested on a pilot scale. According to their results, a PEBAX coated hollow fiber composite membranes showed a selectivity of 3.94 and an O2 gas permeability of 12.23 (Barrer), whereas the PDMS coated composite membrane showed the maximum GS performance result, having O2/N2 selectivity of 4.56 with O2 gas permeability of 18.31 (Barrer) at a constant temperature T = 28°C and pressure of 5 bar. Another research by C.Z. Liang et al. 29 also fabricated a high-performance hollow fiber DLC membrane through dip-coating method for the air (O2/N2) separation. These membranes were composed of PDMS (active layer) on top of PAN porous hollow fibers, which showed a maximum O2/N2 gas selectivity of 2.1 and had O2 gas permeance of 1005 (GPU). Mohammad R.M et al., 30 fabricated a hollow fiber multi-layer composite (MLC) membrane, by applying PDMS coating having a thickness of (11.85–11.17 μm) on recycled/used RO membranes, that were composed of PSF/Polyester layer by layer assembly. A dip-coating method was used to fabricate the coated hollow fiber. The MLC membranes showed maximum O2/N2 gas selectivity of 5.92, having O2 gas permeance of 0.7104 (GPU).
However, there is another method for the fabrication of flat-sheet DLC membrane, which was discussed by Z. Wang et al. 31 in their research work, which described that, ZIF-8 based intrinsically microporous TB-PI (TBDA2-6FDA-PI) polymeric membranes having different filler loading in the range of (7–30%), were coated with ultra-thin PD layer through interfacial polymerization forming a highly selective DLC membranes, which showed, O2/N2 gas selectivity of 7%, 20%, 30% filler loading were 4.50, 4.60, and 4.30, respectively, having O2 gas permeability of 90, 175, and 314 (Barrer) at a constant temperature of T = 35°C accordingly. S.A. Habibiannejad et al., 21 investigated MWCNTs nano-particles for GS separation by incorporating them into the Pebax-1657 polymeric matrix, which were then coated on the PES porous support membranes by using the solution casting method. Prior to embedding in the polymer matrix, the MWCNTs were modified by functionalized with carboxylic, amine groups and Triton X-100 as a strong surfactant. The thickness of the active layer and porous support membranes were 20 ± 5 μm and 50 ± 5 μm, respectively. The maximum O2/N2 gas selectivity in the range of 2.5–6.5 was achieved by CNTX-100 and CNT–NH3 DLC membrane, which showed O2 permeability in the range of 4.2–5 (Barrer) and 2.7–3.5 (Barrer) at a pressure range of 3–7 bar accordingly.
Prajapati PK et al., 32 investigated the preparation and characterization of hollow fiber DLC membranes. These membranes were composed of PDMS active layer, having a variable concentration of (5–20 wt.%), which were then coated on PSF hollow fiber by using a dip-coating method. The maximum GS performance was achieved at 15 wt.% at a constant temperature (T = 25°C) and a pressure range of 0.5–2 bar, PDMS coated composite membrane showed O2/N2 selectivity of 3.91, with O2 gas permeance of 2.75 (GPU). Another researcher, C. Lam long et al., 33 used porous support, composed of PVC-PEG polymer blend. Afterward, it was then coated with an active layer of cross-linkable carboxylated styrene butadiene rubber (XSBR) through a solution casting method, in-order to fabricate a DLC membrane for GS applications including O2/N2 separation, which showed O2/N2 gas selectivity of 13 and had O2 gas permeability of 1.3 (Barrer) at a pressure range of 3–7 bar. H. Nagar et al., 22 investigated air (O2/N2) separation through facilitated transport membrane. In which O2 gas permeated through Pebax polymeric membrane incorporated with cobalt (CoPc) complexes. In this investigation, thin film composite (TFC) membrane were fabricated by a thin coating layer of Pebax-1657 incorporated with a cobalt (II) pthalocyannine (CoPc) compound on an ultra-porous support membrane, composed of PES. Effects of feed pressure and concentration of CoPc compound were also evaluated, which showed enhanced selectivity from 2.9 to 8.5 by increasing the concentration of CoPc loading from 0–1 wt.% at a constant pressure of 2 bar, whereas at maximum selectivity, the O2 gas permeance of 1.12 ± 0.108 (GPU) was observed.
Challenges and possible solution for dual-layer composite membranes
Defects in dual-layer composite membrane assembly
Delamination, detachment, and defects sometimes appear in the layer by layer (LBL) assembly of DLC membrane, which affects the GS performance, such as decreased in selectivity and permeability. To solve this problem, different solutions were proposed by researchers, 1): Selection of suitable and compatible polymeric materials for each layer of DLC membrane. 2): By applying variable conditions during DLC membrane fabrication, such as change in temperature, pressure and using different solvent, in-order to ensure the compatibility, good adhesion, and defect free layer by layer assembly of DLC membrane. 34
Discontinuous nano-particle layer and inter-crystalline voids
These problems need to be tackled before using DLC membrane for GS applications such as air (O2/N2) separation. As uniform growth, dispersion, and deposition of nano-particle is necessary for high GS performance. Possible solutions for this problem include LBL deposition, 31 the use of nano-particles of various sizes, and the interfacial micro-fluidic membrane processing (IMMP) method.35,36
Non-selective voids in dual-layer composite membranes
Weak adhesion between DLC membranes produces non-selective void space. This causes a decrease in overall efficiency of the MBGS process, such as reduction of membrane selective nature toward the gas components. To resolve this problem, many researchers have proposed different solutions, which include, 1): by using seeded polymer substrates as porous support membrane. 31 2): Chemical modification of polymeric substrate in-order to provide the nucleation site for the deposition of active layer.35,37 3): Using electro-spun method for the fabrication of tunable polymeric support substrate, which provides higher mechanical strength and lower gas transport resistance.35,38
Detaching or shattering of active layer
Detaching or shattering of the active layer is another problem of DLC membranes, which appears due to the stiffness and brittleness of the membrane. This causes non-selective channels in the membrane, which reduce the selectivity of the membrane. In-order to resolve this issue, different solutions were proposed, which include, 1) Addition of plasticizer in the active layer, which increases its flexibility. 2) Cross-linking the polymer matrix is necessary in-order to improve the stiffness and compression strength of the DLC membrane.35,39
Aging, long-term stability, and impacts of impurities
These are major issues, which cause hindrance in utilizing these membranes for commercial MBGS applications. GS membranes typically have a shelf life of more than 3–5 years. But after 2–3 years of shelf life, the membrane started to show a decline of 30% performance efficiency in the GS process. These statistical and recorded data were produced by many researchers through lab scale testing. However, there may be a chance that these membranes will have to withstand more harsh environmental conditions in industrial applications, which include high temperature, corrosive environment, high pressure, and impurities during the GS process. This may reduce the shelf life of membrane. 12 Several key factors that significantly impact on the physical aging mechanism and behavior of different polymers, which include membrane fabrication conditions, impurities, storage area, and applied operating conditions, such as temperature and pressure. The detailed descriptions of these factors are mentioned below:
1): Thinner membranes have higher physical aging as compared to thicker membranes. 2): Operating the membrane in a harsh environment, deteriorates the polymeric membrane matrix. 3): whereas some membrane materials were also affected by the humid environment, present in storage areas. In-order to resolve these issues, a protective layer is applied on the DLC membranes and is packed in moisture free environment in the storage area. Although there is another factor that impacts on the physical aging of the membrane, which is the impurities such as dust particles and other corrosive gases present in feed gas, which requires a pretreatment of feed gas before processing in a membrane separator. 12
Commercialization of the dual-layer composite membranes
High GS performance DLC membranes, usually consist of MMMs based on active layer. For its production, a method for nano-particles synthesis at a large scale needs to be developed, which is quite a challenging task. Because raw materials required for its production are costly and, in most cases, synthesis methods of nano-particle production are also complicated. However, through electrolysis and extruding method, mass production of some limited nano-particles is achievable. Similarly, continuous mass production of MMMs based on active layer is also challenging. In-order to resolve this issue, a group of researchers reported a scalable method, such as the solvent free hot pressing method. In this process, high temperature and high pressure were simultaneously applied, which promoted the rapid growth of nano-particles (e.g., MOFs) on various porous substrates. This novel, effortless and easy method can be used for the mass production of DLC membrane. Another researcher reported that, through roll and roll machine, the efficiency of the current fabrication method for the large-scale production of DLC membrane can be improvised.35,40
Conclusions
This review focused on a comprehensive overview of DLC membranes, for the application of air (O2/N2) separation, which include selection criteria, different fabrication methods used for the preparation of DLC membranes, gas transport properties, and a brief discussion of DLC membrane for air (O2/N2) separation application on the basis of years of development. This concludes that, as a result of the combined effect of both MMMs and the support substrate, these membranes exhibit high perm-selectivity with enhanced mechanical strength, which can be very useful in air (O2/N2) separation. Furthermore, the key challenges of DLC membranes were also outlined in this review article along with the potential solutions, which encourage further improvements and its implementation for commercial applications.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911211045852 – Supplemental Material for A comprehensive overview of dual-layer composite membrane for air (O2/N2) separation
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911211045852 for A comprehensive overview of dual-layer composite membrane for air (O2/N2) separation by Syed Shujaat Karim, Sarah Farrukh, Arshad Hussain, Tayyaba Noor and Mohammad Younas in Polymers and Polymer Composites
Supplemental Material
sj-pdf-2-ppc-10.1177_09673911211045852 – Supplemental Material for A comprehensive overview of dual-layer composite membrane for air (O2/N2) separation
Supplemental Material, sj-pdf-2-ppc-10.1177_09673911211045852 for A comprehensive overview of dual-layer composite membrane for air (O2/N2) separation by Syed Shujaat Karim, Sarah Farrukh, Arshad Hussain, Tayyaba Noor and Mohammad Younas in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the MEMAR lab at School of Chemical and Materials Engineering, NUST, Islamabad and HEC (Pakistan) grant No. 10032/Federal/NRPU/R&D/HEC/2017.
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
