Abstract
Poor solubility and low dissolution rate of ibuprofen (IBU) in the aqueous gastro-intestinal fluids restrict its application, absorption, distribution, target organ delivery, and bioavailability. For improvement of aqueous solubility of IBU, supramolecular nanocontainers of IBU/cyclodextrin were prepared via formation of inclusion complex between ibuprofen and cyclodextrins (α-cyclodextrin and β-cyclodextrin) at various conditions (at room temperature at 25℃ and under sonic energy). The formation of inclusion complex between IBU and cyclodextrins can be confirmed by hydrogen nuclear magnetic resonance, differential scanning calorimetry, fourier transform Infrared spectroscopy (FTIR), X-ray diffraction, and scanning electron microscopy study. FTIR of pure IBU and cyclodextrins is similar to the obtained complex, which indicated intactness of drug in the complex. The encapsulation of IBU in cyclodextrins cavity improved its solubility, phase solubility, and in vitro dissolution and also controlled its release which ensures the long-term delivery. Electro-spun nanofibers of poly-ɛ-caprolactone containing IBU/cyclodextrins is a promising method for controlled drug delivery electro-spun which is bead-free without any aggregation on the surface.
Keywords
Introduction
IBU is a nonsteroidal anti-inflammatory drug which is used for treating pain, fever, inflammation, painful menstrual periods, migraines, and rheumatoid arthritis. IBU shows limitations such as low solubility and dissolution rate, incorporation in formulations, skin permeation, and in vitro skin permeation [1,2]. By improving water solubility and bioavailability of IBU such as by using a series of co-solvents and surfactants, the limitations of IBU are eliminated [3].Bioactivity and antioxidant properties of guest molecules such as IBU dependent to stability of guest and chemical, physical, and biological conditions. Encapsulation technology on natural compounds has gained great interest and necessary to apply delivery or carrier systems in order to maximize the potential medical benefits of antioxidants. Encapsulation protects guest molecules from devastating environment effects such as undesirable effects of light, chemical reactions, moisture, oxidation, and dehydration which reduce the shelf life of compounds and improves processing step, delivery of bioactive and stability of the guest molecules [4]. There are many techniques for encapsulation of nanoparticles such as spray-drying, centrifugal extrusion, hot melt coating, coacervation, solvent evaporation–extraction, in situ polymerization, and formation of inclusion complex with cyclodextrins (CDs) [5–8]. CDs and their derivatives can be used for encapsulation of various hydrophobic and hydrophilic guest molecules which improve their solubility, stabilize light sensitive substances, increase shelf life and catalytic activity of CD with guest molecules. CDs are a family of compounds made up of cyclic oligosaccharides molecules bound together in a ring. Typical CDs contain a number of glucose monomers ranging from six to eight units in a ring, creating a cone shape which leads to a hydrophobic cavity and hydrophilic outer surface [4]. The most important property of CDs is the formation of inclusion complexes with a wide range of bioactive molecules, organic, and inorganic compounds. Hydrophobic bioactive molecules are interesting for food, cosmetic, and pharmaceutical applications but are not available for release and delivery to target area [5–8]. CD acts as a host molecule and polyphenols act as the guest molecule and by incorporation of the obtained nanoparticles from inclusion complex formation in electrospun nanofibers during electrospinning, various functionalities can be achieved such as antibacterial and anti-relief nanofibers for wound dressing. Some researches were done on the incorporation of nanoparticles into the nanofibers such as incorporation of neomycin into polyvinyl alcohol (PVA) [9], ciprofloxacin into electrospun scaffolds of dextran and polyurethane (PU) by electrospinning [10], triclosan/CD inclusion complexes into the poly(lactic acid) (PLA) nanofibrous [11], cinnamon essential oil/β-CD inclusion complex into the electrospun polylactic acid [12], and triclosan into CDs nanofibers [13].
Our work focused on the synthesis and characterization of the IBU nanocontainers via inclusion complex formation with CDs (α-CD and β-CD) at room temperature (25℃) and under sonic energy (Scheme 1).Obtained containers proven by hydrogen nuclear magnetic resonance (1H-NMR), FTIR, scanning electron microscopy (SEM), differential scanning calorimetry (DSC), and X-ray diffraction (XRD).The encapsulation of IBU into CDs cavity is an effective delivery system with slow and controlled release system from the molecular cavity of CDs ensures the long-term delivery. Solubility, phase solubility, and in vitro dissolution of obtained IBU/VD nanocontainers were determined. Then these nanocontainers were loaded in poly(ɛ-caprolactone) (PCL) nanofibers via electrospinning and their structure was characterized by SEM and EXD. After characterization of nanofibers structure, the drug released rate was determined.
Schematic mechanism showing the formation of inclusion complex between CDs and IB.
Experimental
Material
IBU (Aldrich, >98%), α-CD (α-CD) (Applichem, >98%), β-CD (β-CD) (Aldrich, >98%), poly(ɛ-caprolactone) (PCL, Merck, ≥98%), dimethyl sulfoxide (DMSO, Merck, ≥98%), dichloromethane (DCM, Merck, ≥98%), and ethanol (EtOH) (Merck, >98%) were used as received without further purification.
Characterization
Solid-state 1H-NMR spectra were recorded with a Bruker DRX 500 spectrometer (500 MHz) in deuterated chloroform. Infrared spectra were recorded with a Bruker IFS 48 FTIR spectrometer. XRD was used to study the diffraction patterns of CD/IBU crystals at ambient conditions; a Xpert Philips diffractometer (USA) with nickel filtered Cu Kα radiation was used in this work. Data were collected at a rate of 2°θ min over the 2θ = 5–60° range. SEM, Philips XL30 (Poland), was used to study the Au-coated microstructures of complexes. UV–Vis spectrophotometer (Shimadzu UV-1650 PC) at the wanted adsorptions was used [5–8].
Inclusion complex formation of CDs/IBU at room temperature (25℃)
The inclusion complex of CDs (α-CD and β-CD) with IBU was prepared by adding IBU solution in ethanol (5.0 mL of 14.5 wt%) to α-CD solution in ethanol (5.0 mL of 5.0 wt%). The suspension was stirred at room temperature for seven days, then maintained at room temperature (25℃) for further 24 h. The obtained solid was collected and dried at 50℃ in an oven for 24 h. The dried complex was characterized by XRD, SEM, FTIR, and DSC [5–8,14,15].
Inclusion complex formation of CDs/IBU under sonic energy
The inclusion complex of CDs (α-CD and β-CD) with IBU was prepared by adding IBU solution in ethanol (5.0 mL of 14.5 wt%) to α-CD solution in ethanol (5.0 mL of 5.0 wt%). The suspension was sonicated for 30 min while the temperature of the solution was kept at 25℃ using ice-water bath, then maintained at room temperature (25℃) for further 24 h. In this research, Q500 Ultrasonic Probe Sonicator was used with 500-watt power for nanoparticle dispersion and homogenization. The obtained solid was collected and dried at 50℃ in an oven for 24 h. The dried complex was characterized by XRD, SEM, FTIR, and DSC [5–8,14,15].
Phase solubility analysis
The phase solubility study was carried out by the method reported by Higuchi and Connors [16,17]. For this purpose, different concentrations of aqueous CD solutions were prepared, filled in test tubes; then IBU solution was added to these solutions to bottles until saturation was attained. Bottles were capped and placed in an ultrasound bath for 48 h at 27℃ ± 2℃, and then obtained solutions were filtered using 0.45 -µm nylon disk filter. After that the solutions were diluted and measure for the total dissolved IBU content was measured by UV analysis at 264 nm. Each sample was determined in triplicate and the samples were protected from light. The phase solubility diagram was constructed by plotting concentrations of dissolved IBU against CDs concentration [18].
Dissolution profile of IBU and its inclusion complex at various conditions
The inclusion complexes containing 20 mg of IBU (calculated based on the content of IBU found in the inclusion complexes) were IBU accurately weighed and placed in a vessel containing 250 mL of simulated dissolution medium and the temperature was controlled at 37℃ ± 01℃ and the paddle was set at 100 r/min throughout the study. At fixed time intervals (0, 5, 10, 15, 30, 45, and 60 min), 5 mL aliquots were withdrawn and equal volumes of fresh medium were replaced. The samples were filtered, suitably diluted, and assayed for IBU content by using UV-visible spectroscopy at 264 nm. The release profiles of IBU and the inclusion complexes at various conditions were also evaluated for comparison [18].
Electrospinning
Electrospinning was carried out by preparation of PCL and IC solutions. PCL solutions were prepared in DMSO/DCM (35/65 v/v%) mixture. After formation a homogenous solution, 0.075 g CDs/IBU inclusion complex were dissolved in PCL solution. The solution was drawn into a 5-mL glass syringe with a 30 G needle and electrospinning was carried out at 15 kV and 2000 r/min on a cylindrical collector with fixed flow rate at 1 mL/h. Distance between the needle tip and the collector (aluminum foil covered collector) was fixed as 18 cm. Electrospinning experiments were carried out in an enclosed box at 23℃–26℃ and 18% relative humidity [16].
Results and discussion
Figures 1 and 2 show the FTIR spectrum of pure IBU, α-CD, β-CD, and their inclusion complex with IBU at room temperature and under sonic energy. The FTIR spectrum of the inclusion complex is similar to the pure CD. Characteristic peaks of CDs are seen at 3340 cm−1 (O–H stretching, vibration); 2920 cm−1 (O–H stretching); 1648 cm−1 (O–H bending); 1425 cm−1 (O–H deformation); 1160 cm−1 (C–O–C stretching and O–H bonding); 1035 cm−1 (C–O–C stretching). Characteristic peaks of IBU are seen at 1740 cm−1 which is related to carbonyl stretching. Formation of the inclusion complex via hydrogen bonding caused to narrow the broad peak corresponding to hydroxyl band at 3370.72 cm−1 which is an indication of the formation of the inclusion complex [17,19–21]. Due to the formation of inclusion complex between hydroxyl groups of the CDs cavity and carbonyl groups of IBU, stretching vibrations of the –OH groups in CD considerably disappeared. C=O peak of IBU at 1740 cm−1 disappeared which is due to the formation of hydrogen bonds. Due to the formation of hydrogen bonds, carbonyl radicals weakened and shift the stretching band to lower frequencies. Also, the and OH peak at 1275 cm−1 shifted to higher frequencies which indicated intermolecular hydrogen bonding and interaction through hydrogen bonding with CD [5–8, 17,19–21].
FTIR of (a) α-cyclodextrin (α-CD), (b) ibuprofen (IBU), (c) α-CD/IBU at room temperature, and (d) α-CD/IBU under sonic energy. FTIR of (a) β-cyclodextrin (β-CD), (b) ibuprofen (IBU), (c) β-CD/IBU at room temperature, and (d) β-CD/IBU under sonic energy.

XRD results
XRD is a more useful method for the detection of formation of inclusion complex via sharpening of the existing peaks, the appearance of a few new peaks and shifting of certain peaks. CDs inclusion complex are divided into channel-type and cage type. By incorporation of IBU guest molecules in CDs cavity in the structure of channel-type, characteristic peaks are seen at 2θ = 7.5, 11, 13, 14, 18, 20, 22, 25. In the cage type, each cavity is blocked off on both sides by adjacent CDs and characteristic peaks are seen at 2θ = 5, 8, 12, 23, 30 [5–8,17,19–21]. The XRD patterns of pure IBU, α-CD, β-CD, and the inclusion complexes are illustrated in Figures 3 and 4 and the characteristic peaks are shown in Table. 1. The XRD results confirmed that the inclusion complexes formed are crystalline. Results showed that under sonic energy inclusion complexes of CDs are head-to-head channel-type and CDs molecules are stacked along an IBU axis to form a cylinder. Sonic energy showed a major effect on the formation of discrete size crystals with channel-type structure. At room temperature, the obtained crystals are a mixture of cage type and channel-type crystals [5–8].
X-ray diffraction of (a) α-cyclodextrin (α-CD), (b) ibuprofen (IBU), (c) α-CD/IBU at room temperature, and (d) α-CD/IBU under sonic energy. X-ray diffraction of (a) β-cyclodextrin (β-CD), (b) ibuprofen (IBU), (c) β-CD/IBU at room temperature, and (d) β-CD/IBU under sonic energy. X-ray diffraction peaks of α-CD, β-CD, IBU, and their inclusion complexes. α-CD: α-cyclodextrin; β-CD: β-cyclodextrin; IBU: ibuprofen.


SEM images
Formation of the inclusion complex between CDs and IBU at various conditions is also indicated by SEM images. Figures 5 and 6 show the SEM images of pure IBU, α-CD, β-CD, and their inclusion complexes at room temperature and under sonic energy. Comparing the pure compound morphology with inclusion complexes indicated the formation of inclusion complexes. Thus, the data obtained from SEM supported previous results, suggesting the inclusion complex formation from the processing of the IBU with CDs at various conditions [5–7].
Scanning electron microscopy image of (a) ibuprofen (IBU), (b) α-cyclodextrin (α-CD), (c) α-CD/IBU under sonic energy, and (d) α-CD/IBU at room temperature. Scanning electron microscopy image of (a) ibuprofen (IBU), (b) β-cyclodextrin (β-CD), (c) β-CD/IBU under sonic energy, and (d) β-CD/IBU at room temperature.

Formation of inclusion complex between IBU and CDs changed morphology particle shape and size of pure IBU and CDs indicating that there was an apparent interaction between the IBU and CDs during the formation of the inclusion complex. The shape and size of the inclusion complexes are completely different from those of the free α-CD (Figure 5(a)), β-CD (Figure 6(a)) and IBU (Figures 5(b) and 6(b)) at the same magnification. SEM images also indicated that the size of crystals strongly depended on ultra-sonication and formed discrete crystals. At room temperature and without sonication, the inclusion compounds tend to be randomized without discrete homogenous crystals being created [5–8].
1H-NMR
1H-NMR images of the IBU, α-CD, β-CD, and the inclusion complex at various conditions are shown in Figure 7. All signals of the 1H-NMR spectra were assigned to their corresponding monomers and it can be declared the synthesis of IBU/α-CD and IBU/β-CD inclusion complexes. IBU is a hydrophobic guest and entrapped in CDs cavity so interior protons (H-3, H-5, and H-6) of CDs significantly shifted compared to exterior protons (H-2 and H-4).
Hydrogen nuclear magnetic resonance of (a) ibuprofen (IBU), (b) α-cyclodextrin (α-CD), (c) α-CD/IBU at room temperature, (d) β-CD, and (e) β-CD/IBU under sonic energy.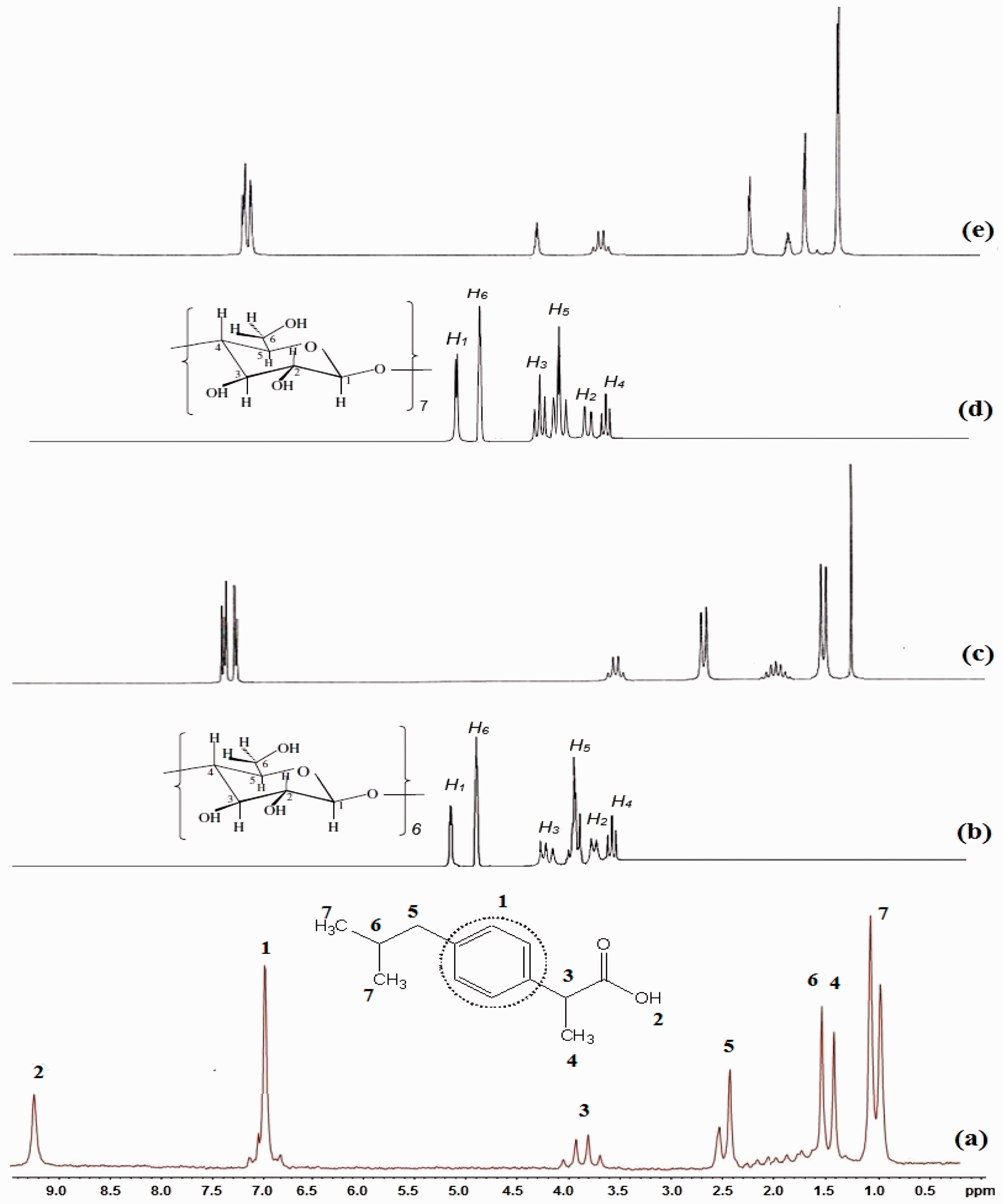
Interior protons of CDs took part in the interaction due to the deep penetration of the IBU molecule into the β-CD cavity and the site of interaction could be the secondary alcoholic group at H-3 [5–8,17,19–21]. Therefore, this shift provides the indication for the formation of the inclusion complex between CDs and IBU, which indicated FTIR results of inclusion complexes. Also, hydroxyl groups of IBU which is seen at 9.1 ppm disappeared in inclusion complex which indicated the formation of hydrogen bonding between IBU and CDs [5–8].
DSC study of complex
The DSC thermogram of pure α-CD, β-CD, IBU, and their inclusion complex at various conditions are shown in Figure 8. The thermogram of α-CD and β-CD showed a characteristic endothermic peak at 90℃ and 100℃, respectively which is related to water loss from CD. Second peak of CDs is seen at 310℃ and 320℃ for α-CD and β-CD, respectively which is due to thermal decomposition of CDs. Pure IBU showed an endothermic peak at 78℃. By formation of inclusion complex between CDs and IBU, this peak relatively dispersed which indicated partially insertion of IBU in CDs hydrophobic cavity. Under sonic energy, higher amount of IBU encapsulated in CDs cavity [5–8].
Differential scanning calorimetry of (a) α-cyclodextrin (α-CD), (b) β-cyclodextrin (β-CD), (c) ibuprofen (IBU), (d) α-CD/IBU at room temperature, (e) α-CD/IBU under sonic energy, (f) β-CD/IBU at room temperature, and (he) β-CD/IBU under sonic energy.
Phase solubility
The content of IBU was determined using UV-visible spectroscopy at a wavelength of 264 nm. The standard IBU solution was prepared in ethanol and diluted to the desire concentrations by 50% v/v ethanol. CDs did not show any absorption at the wavelength of 264 nm which is the λmax of IBU absorption. The calibration curve of IBU was linear [17,19]. Figure 9 shows that the concentration of IBU increased linearly with increasing CD concentration with the slope between 0 and 1 which indicated an AL-type of phase solubility according to Higuchi and Connors concepts [16,17].
(a) Calibration curve and (b) relationships between ibuprofen and cyclodextrin concentrations.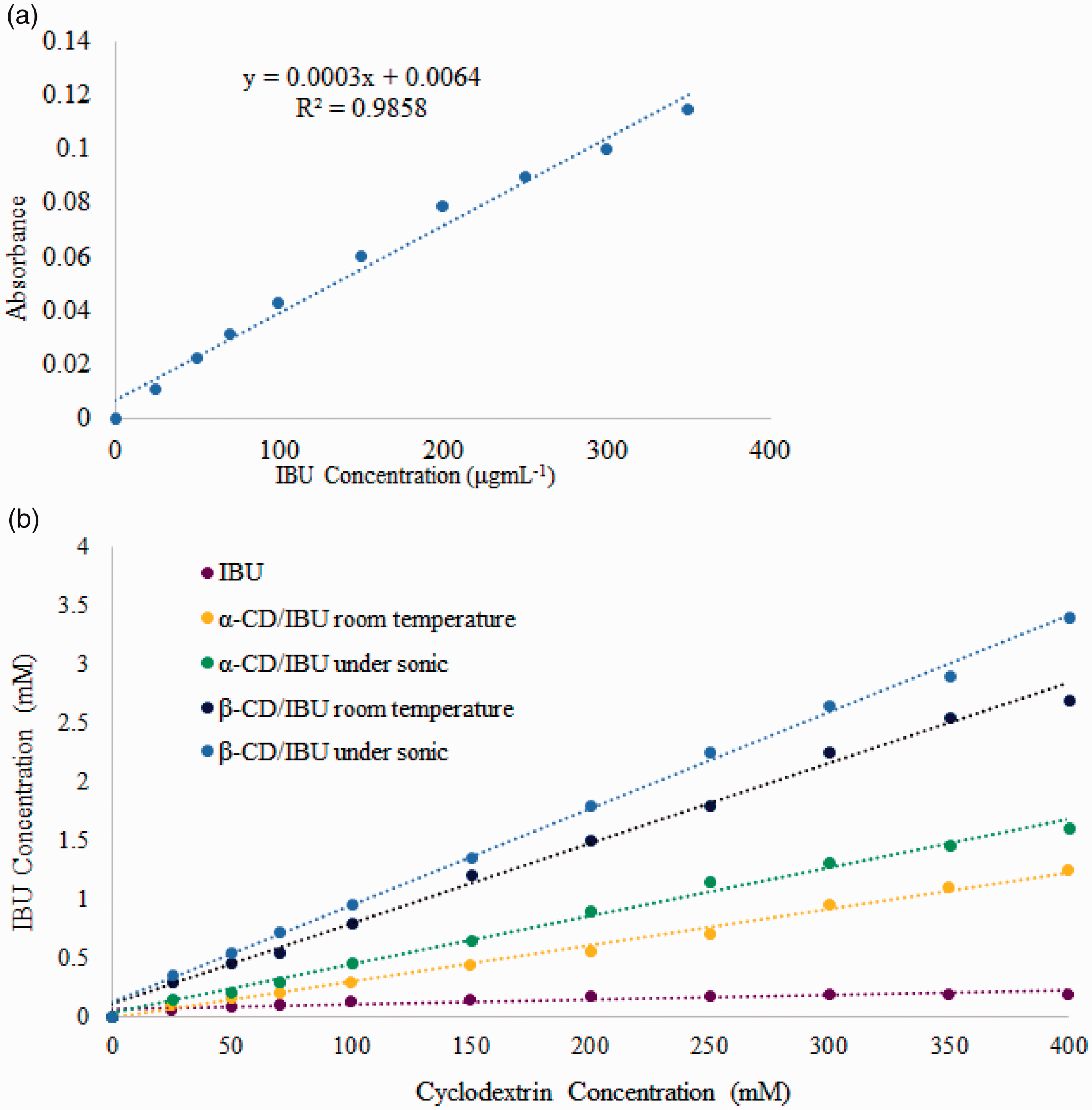
By formation of inclusion complex between CDs and guest molecules, guest fully or partially covered and let to a stable product and changed physical and chemical properties of the guest molecule and CDs may enhance the aqueous solubility of highly insoluble drugs or insoluble compounds because of their ability to form hydrogen bonds with insoluble drugs and form a partially water soluble inclusion complex, which is the most important applications of CD inclusion complexes along with controlled release of the drug and inhibitor corrosion compounds [7–9].
The content of IBU in the inclusion complex was determined using UV-visible spectroscopy at a wavelength of 280 nm. The standard ciprofloxacin solution was prepared in ethanol and diluted to the desired concentrations by 50% v/v ethanol. CDs did not show any absorption at the wavelength of 280 nm which is the λmax of ciprofloxacin absorption. The calibration curve of the method was linear [11,12]. The dissolution of IBU and its inclusion complex with CDs was investigated to evaluate the IBU release profiles (Figure 10). Dissolution medium was simulated at pH = 1.2 (0.1 M HCl). In acidic pH = 1.2 (enteric conditions), hydroxyl groups of CDs are protonated, load a positive charge and more intense release as compared to pure IBU (2.2%) at every time point [22,23].
Release profiles of ibuprofen and its inclusion complex with cyclodextrins at various conditions at pH = 7.2.
Morphology of PCL-based nanofibers containing inclusion complex of CDs/IBU
The SEM and EDX images of PCL-based nanofibers containing inclusion complex of CDs an IBU (Figures 11 and 12) showed bead-free nanofiber with average diameter around 360–486 nm and a wide range of IBU dispersion. Sonic energy provides energy for water withdrawal from CDs cavity and guest molecules can be trapped in the cavity so higher numbers of hydrogen bonding are formed. Inclusion complexes which are formed under sonic energy are smaller than complexes which are formed at room temperature by magnetic stirrer, so PCL nanofibers containing IBU/CDs inclusion show smaller diameter and higher distribution of IBU at the surface of PCL nanofibers [24,25].
Scanning electron microscopy image of poly(ɛ-caprolactone) electrospun containing (a) α-cyclodextrin (α-CD)/ibuprofen (IBU) at room temperature and (b) α-CD/IBU under sonic energy. Scanning electron microscopy image of poly(ɛ-caprolactone) electrospun containing (a) β-cyclodextrin (β-CD)/ibuprofen (IBU) at room temperature and (b) β-CD/IBU under sonic energy.

EDX analysis PCL nanofibers confirmed homogenous distribution of IBU at the surface of obtained nanofibers. In neat PCL, distribution of carbon and oxygen elements is less than PCL nanofiber containing IBU/CD nanocontainers. Also Figure 12 shows that IBU distribution in the presence of β-CD is higher than α-CD which is related to cavity size of CDs. Cavity size of β-CD is bigger than α-CD so can entrapped higher numbers of guest molecules in its cavity [24,25].
In vitro IBU release from the obtained nanofibers
By encapsulation of IBU in CDs cavity, IBU is protected from its surrounding environment for a specific period of time and IBU chemical and physical properties are modified, so its solubility, bioavailability, drug loading capacity, prolong the existence of the drug in systemic circulation may increase and toxicity reduces. increased. By incorporation of IBU/CDs inclusion complexes in PCL nanofibers, these systems can be used as smart bandage with slow and targeted drug release [7–9]. After incorporation of theses inclusion complexes into PCL nanofibers, release of IBU was showed at pH = 7.2 (Figure 13).
Release profiles of ibuprofen (IBU) from PCL-nanofibers containing IBU/CD at various conditions at pH = 7.2.
Due to increased solubility and bioavailability of IBU by formation of inclusion complexes, release of IBU from PCL nanofibers significantly increased with but its diffusion is controlled because of reversible hydrogen bonding between IBU and CDs, so obtained nanofibers showed controlled manner in IBU release. Incorporation of CDs inclusion complexes with various compounds into nanofibers matrix could control release of guest molecules which is attractive way for designing fibrous materials with advanced applications such as drug delivery and tissue engineering [13,14].
Conclusion
In the present study, the formation of inclusion complex between IBU and CDs (α-CD and β-CD) at two different conditions (room temperature and under sonic energy) was reported. The results obtained by 1H-NMR, SEM, XRD, DSC, and FTIR clearly indicate inclusion complex formation. FTIR indicated the presence of IBU and CDs in the inclusion complex, which was also confirmed by the 1H-NMR and XRD results. Entrapment of IBU in the hydrophobic cavity of CDs was confirmed by 1H-NMR via comparing chemical shifts of pure IBU and CDs with their inclusion complexes. XRD images indicated the formation of head-to-head channel-type structures and a mixture of cage-type and channel type crystal structures for CDs/IBU and CDs/IBU inclusion complexes under sonic energy and at room temperature, respectively. SEM image analysis also confirmed the formation of an inclusion complex between IBU and CDs via changes in the initial shape of pure IBU and CDs with their inclusion complexes. UV-spectra showed the controlled release of IBU from nanocontainers with time which is based on reversible hydrogen bonds formation in inclusion complex of CDs and CUR. Dissolution studies of the obtained nanocontainers and nanofibers suggested that the inclusion complexes of IBU with β-CD and α-CD provide higher dissolution rate compared with the IBU alone both in nanoparticles and nanofibers. Obtained nanofibers were bead-free and homogenous.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
