Abstract
Poly (vinyl chloride)/graphene composites with different graphene concentrations were prepared by wet spinning and spin-coating methods. The thermal properties of the poly (vinyl chloride)/graphene composites are investigated through the analysis of the activation energy by using Kissinger and Friedman-Reich-Levi methods. The activation energy of poly (vinyl chloride)/graphene composites obtained by the two methods is consistent and it can be estimated from the non-isothermal kinetic results. In addition, the activation energy value of poly (vinyl chloride)/graphene composites is higher than that of the pure poly (vinyl chloride) fiber, indicating that the addition of graphene can significantly improve the thermal stability of poly (vinyl chloride). Moreover, the activation energy values of 9 wt% poly (vinyl chloride)/graphene composite obtained by Kissinger and Friedman-Reich-Levi methods are highest at 114.90 kJ/mol and 145.00 kJ/mol, respectively, and the result reveals that poly (vinyl chloride)/graphene composite with 9 wt% concentration of graphene has the best thermal stabilities. The prepared graphene-based poly (vinyl chloride) composites with good thermal properties have a promising application in the thermal protective materials.
Introduction
Poly (vinyl chloride) (PVC) is considered as one kind of important commercial thermoplastic materials and is widely used in industrial fields due to its good properties, such as low flammability, high mechanical strength and low cost [1]. However, the thermal stability of PVC is poor during its processing, which limits the applications of PVC and its composites [2,3]. As a result, many additives, such as thermal stabilizers and functional nanofillers, are used to enhance the thermal stability of PVC. Incorporation of nanoparticles into PVC to form nanocomposite is an effective method for improving the thermal properties of PVC [4,5]. Xie et al. [6] synthesized PVC/calcium carbonate (CaCO3) composites by in situ polymerization and their results showed that CaCO3 can improve the thermal stability of PVC. Pagacz et al. [7] prepared PVC/montmorillonite (MMT) nanocomposite and found that the addition of MMT significantly increased the thermal stability of PVC. Hasan et al. [8] prepared PVC/carbon nanotube (CNT) nanocomposite fibers and discovered that the glass transition temperature of the composite fibers was higher than that of the pure PVC fibers.
Graphene (G), a monolayer material of sp2 atoms arranged into a two-dimensional (2D) honeycomb lattice, has attracted great interests in the field of composite materials in the past several years because of its preeminent thermal [9], electrical [10,11] and mechanical properties [12]. It has been demonstrated that the incorporation of well-dispersed graphene sheets into polymers can remarkably improve the thermal stability of the polymer host materials. Yang et al. [13] fabricated G/poly (vinyl alcohol) (PVA) nanocomposites and found that the glass transition temperature and the thermal stability of nanocomposites were improved. Patole et al. [14] prepared G/polystyrene (PS) nanocomposite by in situ microemulsion polymerization, and their results showed that thermal properties of the PS were improved with the incorporation of graphene in the composite. Vallés et al. [15] prepared graphene oxide (GO)/poly (methyl methacrylate) (PMMA) nanocomposites and found that the addition of GO considerably improved the thermal stability of the nanocomposites.
Yuan et al. [16] fabricated functionalized graphene oxide (FGO)/polypropylene (PP) nanocomposite by grafting maleic anhydride grafted polypropylene (MAPP) to GO surface, and the results showed that a significant improvement of thermal stability of the nanocomposite was obtained and the maximum thermal decomposition of the nanocomposite improved about 94℃ when the concentration of the FGO was 1 wt%. Yu et al. [17] prepared FGO/phosphoramide oligomer (FRs) flame retardant, and then incorporated into PP. The results indicated that the thermal stability was improved at elevated temperature with higher char yields by the addition of FRs-FGO into PP.
In this study, poly (vinyl chloride)/graphene (PVC/G) composites blended with different concentrations of graphene particles were prepared by wet spinning and spin-coating method. This work focused on the investigation of thermal stability of PVC/G composite fibers. Thermal decomposition behaviors of the PVC/G composites at different typical temperatures were studied quantitatively. Kinetic analysis was applied to explain the thermal dehydrochlorination of PVC/G composites. The activation energies (E) of PVC/G composites were also calculated according to the Kissinger and Friedman-Reich-Levi methods. It was found that the presence of graphene could greatly improve mechanical property and thermal stability of PVC/G composite fibers. The PVC/G composite fibers with excellent thermal stability find promising roles in thermal protective materials.
Experimental
Materials
Graphene powder (grain size < 20 µm) was supplied by Xinhe New Materials Co., Ltd, Fujian. Polyvinyl chloride (PVC) powder with average polymerization degree of 700 was purchased from Sinopec Group Co., Ltd, China. Formyldimethylamine (DMF) with molecular weight of 94.5–95 g/mol was provided by Sinopharm Chemical Reagent Co., Ltd, Shanghai. All chemicals were of analytical grade and were used without further purification.
Preparation of PVC/G Composites
Preparation of PVC/G composite solutions.
Parameters of PVC composite solutions with different graphene concentrations.

PVC: poly (vinyl chloride); DMF: Formyldimethylamine.
Preparation of PVC/G composites
Pure PVC fiber and PVC/G composite fibers were prepared by a wet spinning apparatus (Figure 1) and then dried in oven for 24 hours to complete the thermosetting process and remove the residual solvent. PVC/G fibers with various graphene concentrations of 0 wt%, 1 wt%, 3 wt%, 5 wt%, 7 wt% and 9 wt% were obtained for the systematical investigation. Moreover, the pure PVC membrane and PVC/G composite membranes were also prepared by using SJT-B digital spin-coating instrument for the study on the distribution of graphene in PVC. The preparation procedures of fiber and membrane can be referred to in our previous studies [18,19].
Schematic diagram of wet spinning process.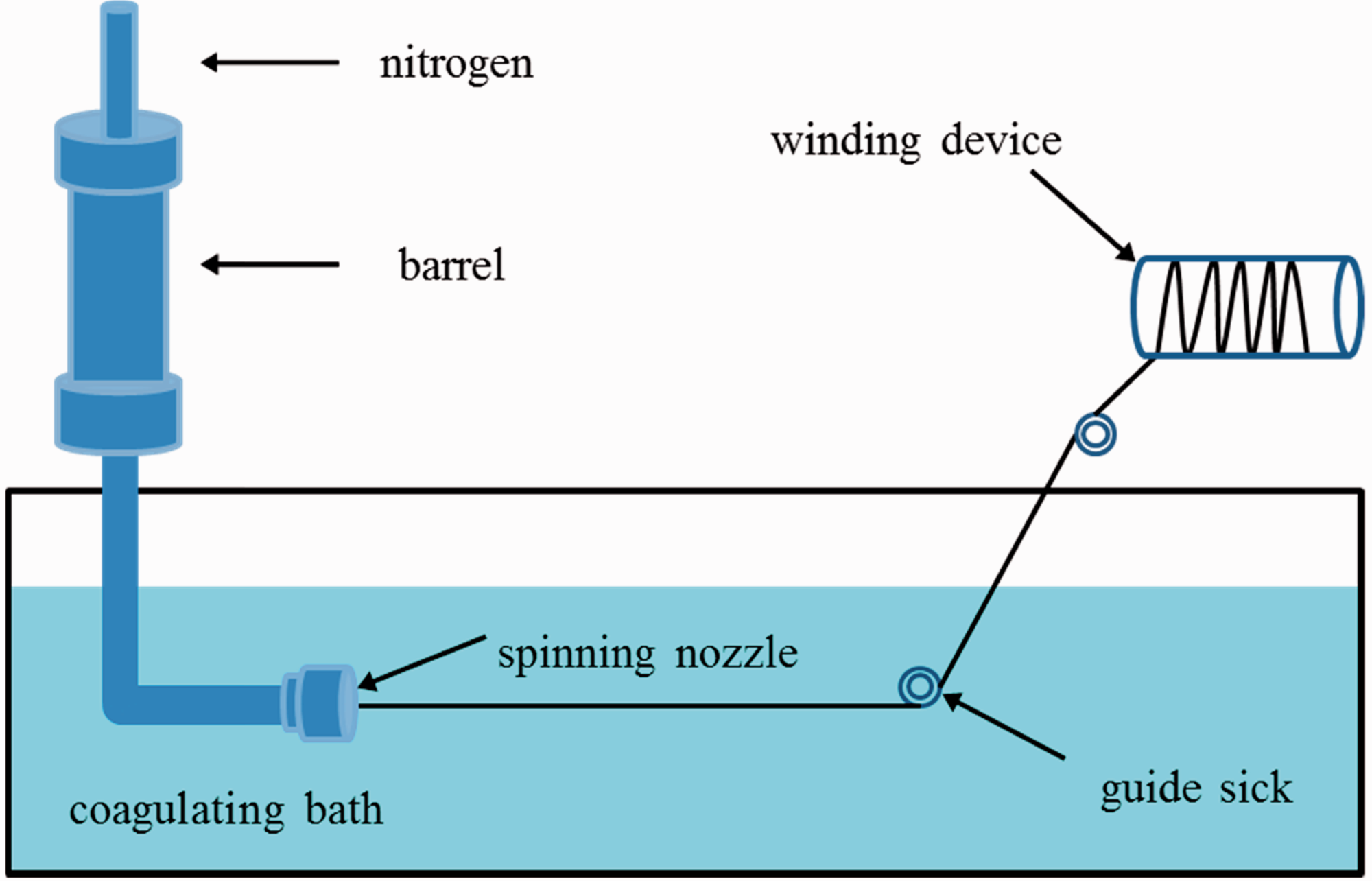
Equipment and measurements
The optional microscope with digital color camera (CX41RF, Olympus, Japan) was used to analyze the dispersion properties of graphene in the PVC matrix. Scanning electron microscope (SEM, SU8010, Hitachi, Japan) was employed to examine the surface morphology of the PVC/G composite fibers. Fourier Transform Infrared (FTIR, PerkinElmer Spectrum Two, USA) spectroscope was used to characterize the molecular structure and chemical composition of the composite fibers. The mechanical properties of the composite fibers were measured by electronic single-fiber strength tester (YG006, Dahe, China). The thermal stability of composite fibers was characterized by a thermal gravimetric analyzer (TGA, PerkinElmer TGA4000, USA). The TGA experiments were carried out in nitrogen atmosphere and the gas flow was 80–100 mL/min. Samples were heated from room temperature to 700℃ and the heating rate was set to be 5, 10, 15, 20, and 25℃/min.
Results and discussion
Morphological structure of PVC/G composites
Optical microscopy images of PVC and PVC/G composite membranes are depicted in Figure 2. It can be observed that the distribution of graphene with low concentrations of 1 wt%, 3 wt% and 5 wt% is relatively uniform in PVC, although a small amount of aggregation can be discovered. The aggregation of graphene becomes serious as the concentration further increases from 5 wt% to 9 wt%.
Optical microscopy images of PVC and PVC/G composite films with different graphene concentrations. (a) 0 wt% PVC/G; (b) 1 wt% PVC/G; 3 wt% PVC/G; (d) 5 wt% PVC/G; (e) 7 wt% PVC/G; (f) 9 wt% PVC/G. PVC/G: poly (vinyl chloride)/graphene.
SEM images of PVC fiber and PVC/G composite fibers are illustrated in Figure 3. As can be observed, all of the fibers exhibit a non-circular cross-section and a porous structure. The porous interior morphology of fibers is attributed to liquid-liquid phase separation process. The initially homogeneous polymer solution is separated into two coexisting liquid phases in thermodynamic equilibrium, i.e. solvent-rich phase and polymer-rich phase [20]. During spinning process, the porous structure of fiber begin gradually to form when the polymer solution is in direct contact with the coagulant (i.e. non-solvent or solvent/non-solvent mixture).
SEM images of PVC and PVC/G composite fibers with different graphene concentrations: (a) 0 wt%; (b) 1 wt%; (c) 3 wt%; (d) 5 wt%; (e) 7 wt% and (f) 9 wt%. PVC/G: poly (vinyl chloride)/graphene.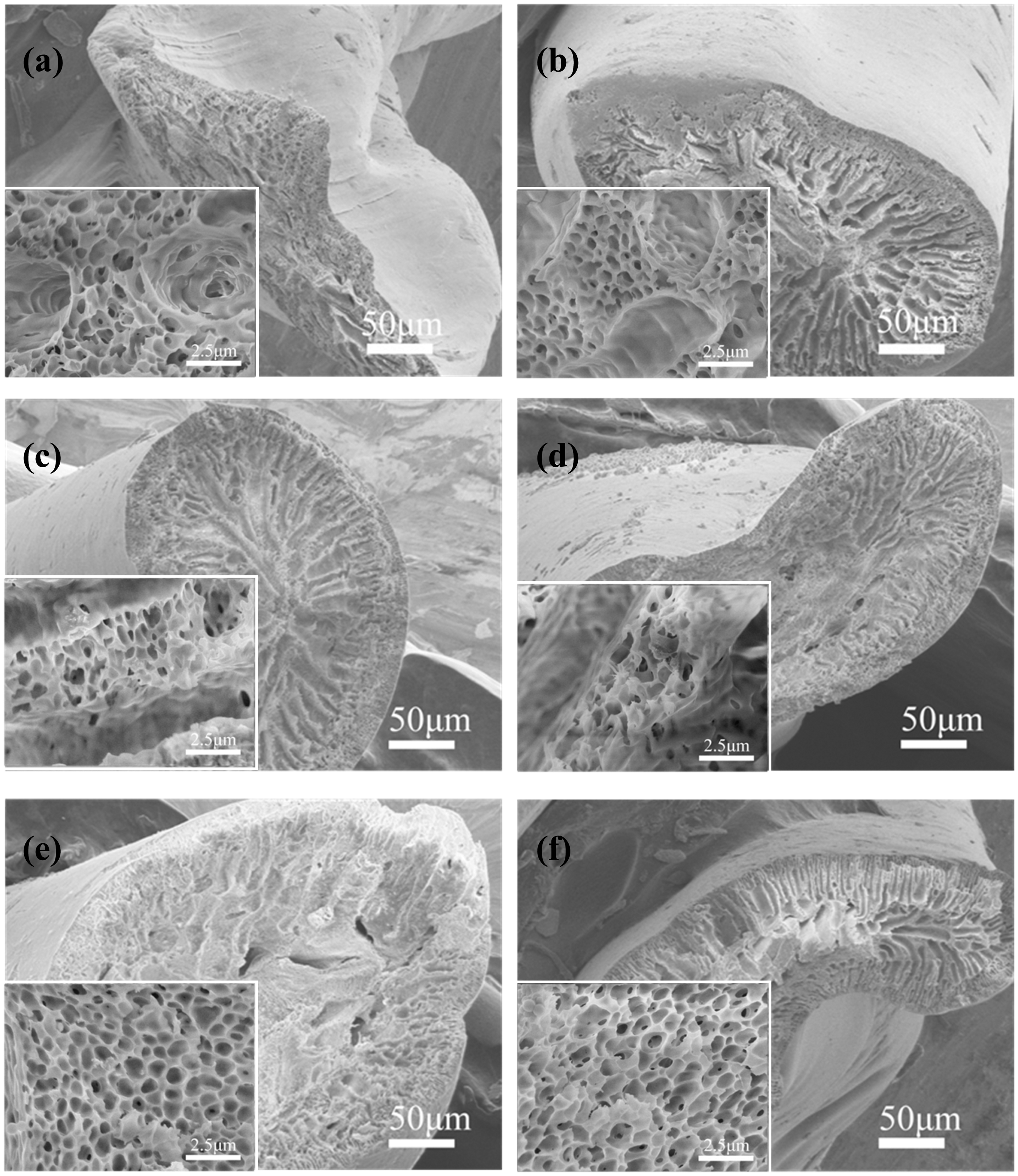
Fourier transform infrared analysis of PVC/G composite fibers
Figure 4 shows the FTIR spectra of the graphene, PVC and PVC/G composite fibers with different graphene concentrations. For the PVC fiber and PVC/G composite fiber, the peaks at 2917 cm−1 and 1430 cm−1 are due to the stretching vibration and the bending vibration of C-H, respectively [21]. The peak at 1249 cm−1 attributes to the bending vibration of C-H from CHCl groups of PVC polymer. The peaks at 1093 cm−1 and 690 cm−1 correspond to the stretching vibrations of C-C and C-Cl, respectively [22]. These results can confirm the existence of PVC. The FTIR spectrum of G shows peaks at 3431 cm−1 and 1588 cm−1, which correspond to O-H stretching vibrations and C = C vibrations, respectively. In addition, there is no obvious difference between the FTIR spectra of 9 wt% PVC/G and pure PVC. The result indicates that the addition of graphene has no significant influence on the chemical structure of PVC.
FTIR spectra of graphene, PVC and PVC/G composite fibers with different graphene concentrations. PVC/G: poly (vinyl chloride)/graphene.
Thermal stabilities of PVC/G composite fibers
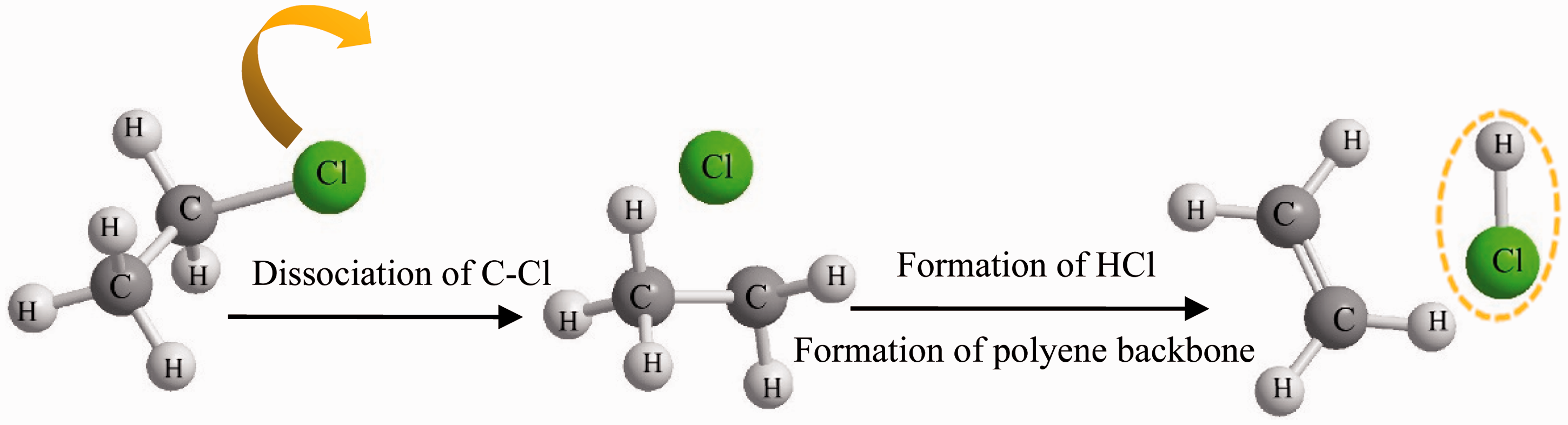
TG and DTG curves of PVC and PVC/G composite fibers are shown in Figure 5. As shown in Figure 5, the thermal decomposition of composites can be analyzed as a decomposition process of weight loss and it can be divided into two sections. The first process is observed from 240℃ to 350℃, which corresponds to the thermal dehydrochlorination of PVC and evolution of hydrogen chloride (HCl) as illustrated in Figure 6 [23]. The cleavage of C-Cl bond can easily and initially happen because of its weak binding energy [24]. The Cl radicals can be formed and bonded with H radical from the neighboring C-H bond resulting in the evolution of HCl molecules and the formation of polyene backbone [25]. The second process from 400℃ to 500℃ is mainly due to the degradation of polyene backbone, resulting in the formation of volatile aromatic compounds and a stable carbonaceous residue.
TG curves (a) and DTG curves (b) of PVC and PVC/G composite fibers with different graphene concentrations (heating rate: 10℃/min). PVC/G: poly (vinyl chloride)/graphene. Thermal dehydrochlorination of PVC. PVC: poly (vinyl chloride).
Parameters of different PVC/G composite fibers during the thermal decomposition.

PVC/G: poly (vinyl chloride)/graphene.
T5%—The temperature at weight losses of 5%;
T50%—The temperature at weight losses of 50%;
Tmax—The temperature of the maximum thermal decomposition rate.
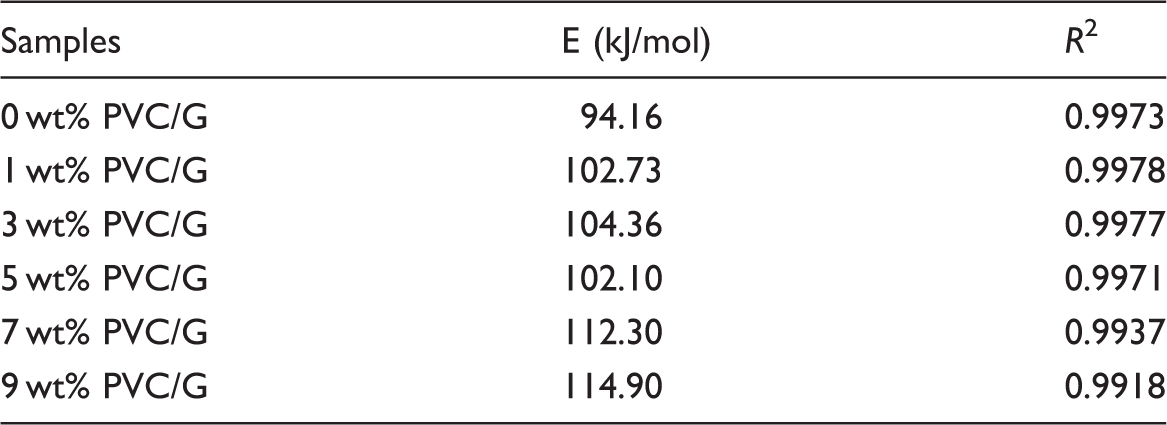
Kinetic analysis of thermal degradation
The thermal stabilities during thermal degradation and the thermal degradation kinetics of PVC and PVC/G composite fibers are further analyzed by using Kissinger and Friedman-Reich-Levi methods, which are the most popular approaches for determining the kinetic parameters of polymers, namely the activation energies of the thermal degradation [26,27]. Thermal degradation speed (dα/dt) can be calculated by equation (1) and it is commonly used in the kinetics process [28,29]



β is a constant and represents the heating rate (β = dT/dt) during the thermal degradation, then dα/dt can be expressed by equation (4)
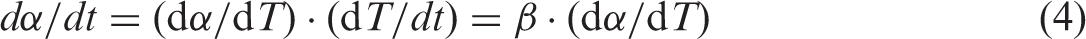
The prevalent non-isothermal reaction kinetic equation can be obtained by the combination equations (3) and (4) as below

Kissinger method
Kissinger method has been used to determine the activation energy from plots of the logarithm of the heating rate versus the inverse of the temperature at the maximum thermal degradation speed during the experiments with a constant heating rate [30,31]. Kissinger method is a differential method and has the advantage that does not need the knowledge of the exact thermo-degradation mechanism [32]. Equation (5) can be evolved into equation (6) when there is 
From the slope of the plot of Plots of Activation energy values of PVC and PVC/G composite fibers by Kissinger method. PVC/G: poly (vinyl chloride)/graphene.
Friedman-Reich-Levi method
Friedman-Reich-Levi method is a differential non-isothermal method [33]. It is simple and applicable to the random degradation of polymers and other complicated reactions. Friedman-Reich-Levi method involving multiple heating rates is expressed in equation (7) after the logarithm based on equation (5)

Thus, by plotting Plots of The relationship of activation energy values on the conversion degrees (α) of pure PVC and PVC/G composite fibers according to Friedman-Reich-Levi method. PVC/G: poly (vinyl chloride)/graphene. Activation energy values by the Friedman-Reich-Levi method. PVC/G: poly (vinyl chloride)/graphene.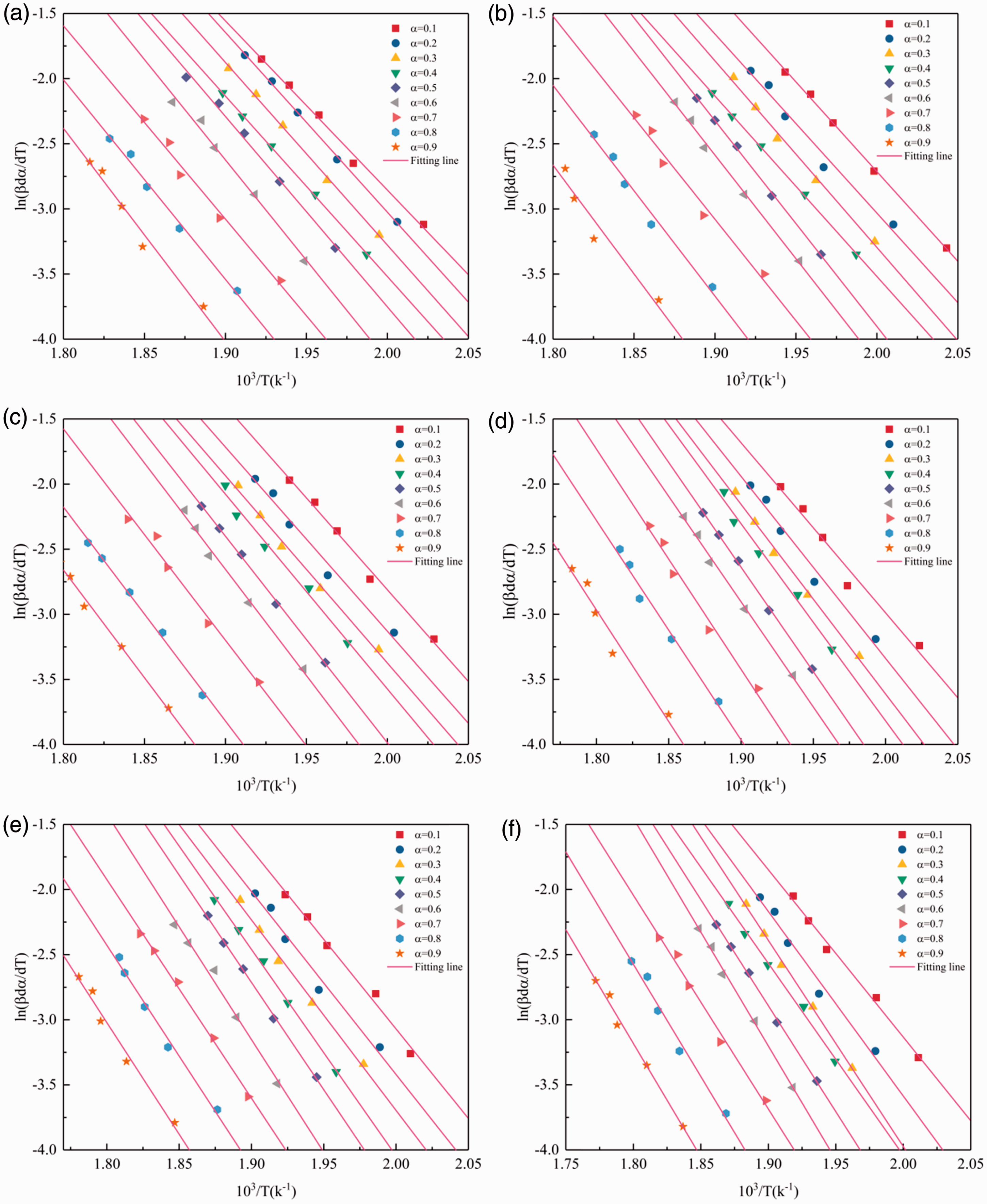


Conclusions
In this paper, PVC and PVC/G composite fibers were prepared by wet spinning and spin-coating methods. Thermogravimetric analysis has shown that the well-dispersed graphene can effectively delay the degradation process of PVC and then enhance its thermal stability by interfering with the formation of the conjugated polyene sequences. The dynamic thermal degradation of PVC and PVC/G composite fibers were further analyzed by the Kissinger and Friedman-Reich-Levi methods. The results show that the activation energy values of PVC/G composite fibers are higher than that of the pure PVC, and increase with the increasing of graphene concentrations. The kinetic analysis results demonstrate good agreement with the TGA results and reveal that the blending with graphene can effectively enhance the thermal properties of PVC, in addition, the thermal stabilities of the PVC/G composite fibers gradually improved with the raising concentration of graphene.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support by Research Initiation Funds of Shanghai University of Engineering Science (XQ2016-27) and Training Scheme of Young Teachers in Shanghai Universities (ZZGCD16028) and Graduate Research Projects for the Shanghai University of Engineering Science (17KY0906). This work was also supported by 2017 Talents Action Program of Shanghai University of Engineering Science (2017RC432017).
