Abstract
To study the release behavior and the existing position of Domiphen® in its its complexed poly-L-lactic acid (PLLA) micro-fibers, two series of fibrous films were prepared by electrospinning solution and W/O emulsion, which was added to Domiphen® directly and emulsified by Domiphen®, respectively, by complexing methods. The mass ratio of Domiphen® to PLLA was 0.25%, 0.5%, 0.75%, and 1% for both methods. According to the scanning electron microscopic observation, W/O emulsion and fibers of narrow-distributed diameters were obtained by W/O emulsion electrospinning. Fluorescence microscopic observation suggested that Domiphen® was incorporated within fibers by W/O emulsion electrospinning. X-ray diffraction and differential thermal analysis characterization suggested that Domiphen® complexed well with PLLA according to noncrystalline Domiphen® existing within and on PLLA fibers prepared by both methods. Domiphen® release curves were prepared and fitted after the amount of Domiphen® released from the PLLA fibers was determined by high-performance liquid chromatography. It was discovered that, with regard to the fibrous films prepared by homogeneous solution electrospinning, Domiphen® released from fibers in accordence with the Freundlich’s equation suggesting that Domiphen® almost existed within the outermost border of the fiber cross section and its release behavior could be considered as the counter adsorption process, while with regard to the fibers prepared by W/O emulsion electrospinning, the release curves were S shape under the effect of proteinase K degradation and could be fitted to the corrosion-mechanism Peppas’ equation of n value >0.5, which suggested that Domiphen® existed both on the surfaces in small proportion and the inner border of the core-sheath fibers in big proportion.
Introduction
Nano/micro-fibers prepared by electrospinning are very useful and promising materials in many fields, especially for medical use including tissue engineering matrixes, drug delivery systems, anti-bacterials covers, and so on [1–3]. And fibers of aliphatic polyesters (APE), for example, polylactic acid, polyglycolic acid, poly-ɛ-caprolactone, and their copolymers, are among the most desirable materials for these purposes due to their low toxicity, bio-degradability, and bio-compatibility [4–6].
With regard to solution-electrospinning APEs, the most desirable solvents are small-molecule-weight halohydrocarbons. There are three reasons for that. First, solutions composed of APEs-halohydrocarbon are of appropriate viscosity for fibers forming from Taylor cone and desirable evaporating speed to maintain stereoshape of fibers during spinning. Second, halohydrocarbons are merely the organic solvent, which are able to dissolve the APEs easily and unable to dissolve with the other water-insoluable small molecule compounds when W/O emulsion method is used for preparing core-sheath fibers for forming the external oil phase and sheath layer. Third, some halohydrocarbons, such as dichloromethane and trichloromethane, possess very low toxicity and can be almost removed by freeze-drying method hardly resulting in any residual for the final product. Nevertheless, in order to electrospin these halohydrocarbon- polymers smoothly, polar organic solvents, such as acetone and N, N-dimethyformamide, are indispensible additives to lowering the solutions’ surface tension because halohydrocarbons are all hydrophobic and their single-organic-solvents solutions are all of high surface tension, otherwise, fibers of unhomogeneous diameter or even beads would occur in the products, which cannot be used for the purpose due to their undesirable morphology [7]. But, when W/O emulsion electrospinning is chosen for forming core-sheath fibers, polar organic solvents cannot be adopted since they co-dissolve within aqueous solution, which would result in separation of polymers.
Evidently, there is a contradiction between the formation of core-sheath fibers of narrow-distributed diameter electrospinning without the addition of polar organic solvents and inevitable usage of polar organic solvents for the formation of fibers of narrow-distributed diameter. In fact, there are no reports about fibers prepared by W/O emuslion electrospinning using dichloromethane/trichloromethane as the single solvent.
Thus, one easy method was applied for solving this problem where researchers used surfactants to both form of W/O emulsions and ensure the desirable shapes of fibers of narrow-distributed diameter. In the previous reports, some surfactants such as, trimethly benzyl ammonium chloride, sulfate dodecyl sodium, hexadecyl phosphatidyl choline, polyethylene glycol-b-alkyl ether, polynonylphenoxy-polyethoxyethanol, PEG blocked poly-α-hydroxyl octanoic acid, ethylene glycol-b-polylactic acid, were used for hemogenizing and lowering the diameter of fibers [8–13], and sulfate dodecyl sodium, polyethylene polypropylene copolymer, ethylene glycol-b-poly ɛ-caprolactone were used for preparing the emulsion-induced core-sheath fibers [14–18]. From the physico-chemical aspect, anionic surfactants merely dissolve within APEs/halohydrocarbons solutions resulting in bad complexing with the material system, and from the medical aspect, anionic surfactants are irritant to mucosa and hemolytic to cells. While nonionic surfactant, due to their water-miscible PEG blocked structure, can dissolve either in APEs-halohydrocarbons solution or in aqueous solution resulting in undesirable premature physical disintegrating and chemical degradating susceptible to environment facilitated by fast water penetration into fibers. In practical application, these may cause fiber structure fragility, drug burst release, drug unsustained release, and so on.
According to the aforementioned facts, seeking certain cationic surfactants to prepare electrospun fibers is necessary and promising. Many cationic surfactants have good solubility within APEs-halohydrocarbons solution, high surfactivity for lowering their surface tension, and high emulsifying ability to form stable W/O emulsions under mechanical works. Admittedly, most cationic surfactants are of high toxicity, yet, there are still considerable amount that could be selected as electrospun fiber additives because of their well-tolerated toxic dose. Among them, dodecyl-dimethy(2-phenoxyethyl)-ammonium bromide, commercially named Domiphen® is a good candidate. Practically and clinically, Domiphen® is a disinfectant for buccal cavity, vagina, and surgical instrument.
In this paper, Domiphen® was complexed with one of the commonly used and simplest-structure APEs, poly-L-lactic acid (PLLA, Figure 1(b)) by forming transparent homogeneous solution and W/O emulsion before electrospinning. The obtained fibers were characterized by field-emission scanning electron microscopy (FESEM) and laser scanning confocal fluorescence microscopy (LSCFM). The combination of Domiphen® with PLLA was investigated by X-ray diffraction (XRD) and differential thermal analysis (DTA). The released Domiphen® was determined by high-performance liquid chromathgraphy-ultra violet (HPLC-UV). The time-dependent Domiphen® release curves were drawn and fitted from equations for analyzing the existing position of Domiphen® molecules within fibers. Especially, after it was found that considerable proportion of complexed Domiphen® had not been released from the W/O emuslion electrospun fibers, proteinase K was adopted to degrade the fibers in order to release Domiphen® and to speculate the existing position of Domiphen® within the core-sheath fibers by analyzing the release curves.
Chemical structure of Domiphen® (a) and poly-L-lactic acid (b).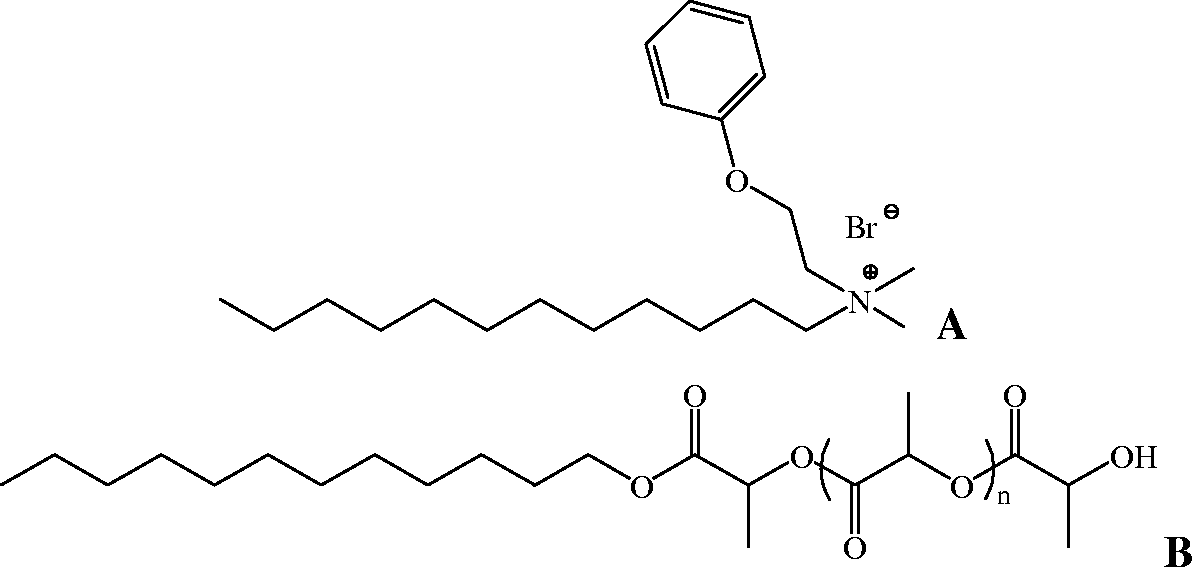
After understanding of certain facts from the experiments, these novel Domiphen® fibers could be used for loading and controlled releasing theraputic drugs, entrapping environment contaminants, immobilizing functional enzymes, which is our intent for their practical application [2–4].
Materials and methods
Materials
Domiphen® was purchased from Meilun® (99.2%, Dalian China). Fluorandiol was purchased from Adamas® (95%, Shanghai China) and was dissolved in 0.01M NaOH pure water solution of 2% mass/volumn ratio before use. PLLA was synthesized with lauryl alcohol as the initiator and its Mw was about 150,000 g/mol and PDI was about 1.25. Analytical pure dichloromethane, chromatographic pure methanol, and double deionized water were used.
Electrospinning
First, homogeneous solution and W/O emulsion were prepared for electrospinning. With respect to the homogeneous solution, 1 g PLLA and 0.25%, 0.5%, 0.75%, and 1% Domiphen® (mass ratio to PLLA) were added to dichloromethane by magnetic stirring forming a final volume of 25 mL solution, which was transparent and homogeneous. The fibers spun from these solutions were named Fs1, Fs2, Fs3, Fs4 accordingly. With regard to W/O emulsion, series of the same 25 mL transparent and homogeneous solutions as external phase were prepared with 2 mL pure water as the internal phase, which was dropped into and magnetically stirred at 200 r/min for 30 min to obtain coarse emulsions. And consequently, the final highly dispersed emulsion was prepared by probe-ultrasonicating the coarse emulsions with a power of 60 W for 30 times where ultrasonicating lasted for 3 s with 1 s interval. The fibers spun from these highly dispersed emulsions were named Fe1, Fe2, Fe3, and Fe4, accordingly. In order to investigate and prove the core-sheath structure of W/O emulsion electrospun fiber, 2 mL fluorandiol–0.01M NaOH solution of 2% mass/volume ratio was used as the internal phase and obtained fibers were named Fe1-f, Fe2-f, Fe3-f, and Fe4-f, accordingly.
The electrospinning set-up was similar to the previous reports elsewhere, which was based on electrostatic generator (Dongwen Apparatus Co., Tianjin China) [9]. The prepared solutions and emulsions mentioned above were transferred into glass syringes attached with a stainless needle of 0.6 mm inner diameter. A pressure was applied to maintain a steady flow from the needle outlet in the range of 1.5 ± 0.2 mL/h. The distance between the needle tip to the grounded static 100 × 100 cm2 tin foil for collecting was 50 cm and the electric field strength applied was 15,000 V. All electrospinning experiments were carried out at 25 ± 2℃ in air, and the relative humidity was 60 ± 5%. In order to remove the residual dichloromethane, the fiber mats collected were dried at 30℃ under vacuum for 24 h [14–16].
Morphology and Domiphen® entrapment
The morphology of the electrospun fibers were observed under FESEM (Hitachi® S-4800, Japan) at an accelerating voltage of 5 kV, and all samples were mounted on steel plate by using a double-sided adhesive electroconductive tape and treated by vacuum evaporating-Aurum depositing process prior to examination.
W/O emulsion electrospun fibers with fluorandiol entrapped were observed under a laser scanning confocal fluorescence microscopy (LSCFM, Olympus® CKX53, Japan).
Domiphen® loading on and within fibers were respectivley investigated by wide-angle XRD (Rigaku UltimaIV, Japan) in which all samples were scanned from 5° to 50° at a scanning rate of 2°/min at 25℃, and by differential thermal analysis (DTA, Perkin Elmer, Pyris Diamond, USA) where all samples were scanned from 25° to 500° at a scanning rate of 5°/min under N2 atmosphere [10,17,18].
HPLC and Domiphen® release curve
Domiphen® were quantitively determined by a set of HITACHI ELITE Lachrom HPLC system equipped with a column (Diamonsil C18, φ4.5 × 200 mm, 5 µm), a L-2455 ultraviolet-detector, and a L-2130 pump. The eluent was composed of 80/20 mixture (v/v) of acetonitrile and 0.012 mol/L SDS aqueous solution (with SDS 3.46 g, NaH2PO45.0 g dissolved in 1000 mL pure water) of pH 3.0 adjusted by 30% H3PO4 solution. The flow rate was 1.0 mL/min, the detection wavelength was 269 nm, and the column temperature was 30℃ [19].
Experiments of Domiphen® release from PLLA fibers were carried out by incubating pieces of fibrous mats of about 300 ± 5 mg into 10 mL pure water as the release medium in a thermostatic oscillator of 37℃ with oscillation of 100 r/min. At predetermined time intervals, total release medium was withdrawn and analyzed by HPLC. After this, 10 mL pure water was re-added and sampling procedures were re-done afterwards [10,14,16].
The release percent of Domiphen® was calculated by the following
Results and discussion
Fiber morphology and Domiphen®–PLLA complexing
All fibers appeared as pure white fibrous films to the naked eye observation and from Figure 1, they were all almost less than 2 µm fibers under FESEM and there was no crystal-like substance emerging on the fibers surface. With regard to the fibers prepared by solution electrospinning (Fs1, Fs2, Fs3, Fs4 in Figure 2), there were few tiny holes exsiting on the fibers surface, suggesting some occurrence of phase separation that is common in APEs-halohydrocarbons electrospinning. With respect to the fibers prepared by W/O emulsion electrospinning (Fe1, Fe2, Fe3, Fe4 in Figure 2), there were a lot of tiny holes on the surface of fibers, which might be caused by different water and dicloromethane evaporation speeds from W/O emulsion droplets on the fiber surface when PLLA concreted from polymeric solution forming cheese-like tiny holes and water completely drained out. Under LSCFM, linear green fluorescence emission was discovered, which suggested that W/O emulsion electrospun fibers had entrapped fluorandiol in the hollow continuous core. Since fluorandiol itself could not be dissolved in the PLLA–dichloromethane solution, it could only exist either in aqueous phase or on the surface of the fibers during emulsification of W/O. As there were no fluorandiol diffraction peaks existing on the fibers, fluorandiol could only exist in the internal hollow core of the core-sheath fibers formed along with dichloromethane evaporation during electrospinning.
Field-emission scanning electron microscope photographs of Domiphen®–poly-L-lactic acid electrospun fibers (scale bars in Fs1, Fs2, Fs3, Fs4 and Fe1, Fe2, Fe3, Fe4 represent 20 µm; scale bars in Fs1 and Fe1 represent 1 µm); fluorescence microscope of Fe1 and Fe4 (Fe1-f and Fe4-f, respectively).
After the measurement of diameter, it was known that the diameters for Fs1, Fs2, Fs3, Fs4 were 752 ± 152 nm, 629 ± 82 nm, 563 ± 76 nm, 551 ± 89 nm, respectively, and Fe1, Fe2, Fe3, Fe4 were 641 ± 137 nm, 627 ± 115 nm, 793 ± 106 nm, 781 ± 117 nm, respectively. Obviously and interestingly, as the loading amount of Domiphen® increased, the fiber diameter decreased for solution electrospun fibers, which is due to the surface-tension-decreasing effect usually reported previously in APEs-halohydrocarbons solutions electrospinning with complexed surfactant, and the fiber diameter increased instead for W/O emulsion electrospun fibers. The reason for this is not clear and is needed to be further investigated. Although, we surmise that more Domiphen® would facilitate faster movement of H2O molecules from the fibers’ interior to the exterior for final evaporation, and since evaporating is endothermic, the dichloromethane of the external phase would evaporate more slowly resulting in longer lasting of the shape and volume of the dichloromethane–PLLA pseudo-solution to be formed in the fibers sheath finally. But, with regard to the W/O emulsion electrospun fibers with less Domiphen® loaded, H2O molecules evaporated slower enough to maintain thorough heat exchange between fibers and the surroundings where considerable heat helped the faster movement of dichloromethane resulting in volume shrinkage of sheath layer and smaller diameters during electrospinning.
Domiphen® and PLLA complexing was characterized by XRD and DTA, which suggested the existing state of Domiphen® on and within the fibers, respectively. From the XRD patterns in Figure 3, it was discovered that pure Domiphen® is of crystalline form by demonstrating five obvious sharp characterisitic peaks at special 2θ angles and PLLA raw powder is semi-crystalline where more than 10 small peaks sequentially emerged from about 2θ = 10° to 2θ = 25°. After dispersion within solution or emulsion and sequent electrospinning, both peaks of Domiphen® and PLLA disappeared, which suggested that there was no crystalline Domiphen® existing on the surface of PLLA fibers; however, it could not be ruled out that Domiphen® could exist as a certain assembling state on the surface of PLLA fiber. Moreover, it was also discovered that as the Domiphen® loading amount increased in PLLA when the fibers were prepared by solution-electrospinning, XRD intensity increased from 2θ = 10° to 2θ = 25°, and the maximum value of diffraction shifted to bigger 2θ. Specifically, for solution-electrospun fibers, diffraction intensity increased when the loading amount of Domiphen® was 0.5%, 0.75%, and 1%, while for W/O emulsion-electrospun fibers, diffraction intensity increased when the loading amount of Domiphen® was 0.75% and 1%. We perceive that this kind of diffraction intensity varying phenomenon is related to Domiphen®’s assembling state within the fibers where although Domiphen® dispersed within PLLA fibers is not in the crystalline state on the fibers surface, there was still considerable amount of Domiphen® existing as a certain assembled state of themselves on or within the very border of fiber surface. In fact, this was further proved by Domiphen® release experiment. Besides this, on comparison of solution-electrospun fibers with W/O emulsion-electrospun fibers of different Domiphen® loading amount, it was seen that neither of the two showed increase in the XRD intensity when the Domiphen® loading amount was 0.25%, and when the Domiphen® loading amount was 0.5% the solution-electrospun fibers showed increase in the diffraction intensity while that of the W/O emulsion electrospun fibers did not. This might suggest that when the Domiphen® loading amount was considerably low, it would not emerge onto the fiber surface to show the effect of XRD for both solution-electrospun fibers and W/O emulsion-electrospun, while when the Domiphen® loading amount increased to a certain amount of 0.5% in this research which is not high enough, the Domiphen® molecules would accumulate at the very border of the fiber surface for the solution-electrospun fibers causing an increase in the XRD intensity. However, with respect to the W/O emulsion-electrospun fibers of the same amount, under the precondition of enough proportion of Domiphen® molecules accumulated at the border of the fiber surface performing surface-tension-lowering effect for formation of the homogeneous diameter fibers, considerable proportion of Domiphen® molecules were drawn into the deeper position of the fiber cross section by performing the emulsifying function and lastly around the hollow core after the core-sheath structure was formed. And with regard to the proportion of total Domiphen® molecules accumulated on the fiber surface and the amount drawn into deeper position of the fibers, further investigation was needed, for which the drug release behavior study was employed for the purpose in our research. As the loading amount increased to 0.75% and 1%, there seemed to be enough proportion of Domiphen® for emulsifying and considerable proportion of Domiphen® for assembling onto the fiber surface for causing an increase in the XRD intensity. Generally, W/O emulsion-electrospinning drew Domiphen® into deeper position of fibers than the solution-electrospinning did. This conclusion need to be proved and, in fact, was proved by DTA and Domiphen® release curve presented in the after content of this research [10,14,16,18].
From top to bottom: XRD pattern of Domiphen® raw powder, fluorandiol, Fs1, Fs2, Fs3, Fs4, and Fe1, Fe2, Fe3, Fe4.
DTA curves shown in Figure 4(a) suggested that Domiphen® had three significant endothermal peaks, at 61.8℃ of de-assembling, 116.7℃ of de-arrangement, and 200–260℃ of degradation. And with regard to the PLLA raw material and fibers, they had also three significant endothermal peaks, at 54.8℃ of glass transformation, 173.9℃ of melting and about 200–260℃ of degradation. After the two components were electrospun into fibers, the characteristic peaks of Domiphen® had already disappeared owing to its well-complexed and considerable small loading amount within PLLA. PLLA glass transforming temperature had nearly disappeared, while melting temperature and degradation temperature still appeared with little shifting. This suggested that Domiphen® seemed to be complex with PLLA to a certain extent where Domiphen® affected the noncystalline glass state of PLLA, which was at low energy level. Moreover, it was also noticed that in comparison to the solution-spun fibers, PLLA degradation of emulsion-spun fibers at the same Domiphen® loading amount level needed much less heat, which was indicated by the decrease in the area of the endothermal peaks. This can be explained as follows: in emulsion-spun fibers, Domiphen® complexed with PLLA more thoroughly than in solution-spun fibers due to ultra-sonification and emulsification effect, which means Domiphen® destructed the PLLA crystalline structure by lowering the degree of crystallinity when Domiphen® accumulated onto the border of the fiber cross section leaving the inner crystalline PLLA unchanged or less changed. This conclusion accorded with the conclusion from XRD and was proven by the Domiphen® release curve mentioned in the after content of this research. But, it is must be understood that although Domiphen® interfered with the PLLA structure, it seemed to be combined with PLLA by other forces like hydrogen bond, which is much stronger than van der Waal forces commonly seen in polymer–small molecule interaction. Thus, only by this way, it could be accorded with the conlusion obtained from the release experiment [10,16,18,20].
(a) DTA curve of Domiphen® raw powder, PLLA raw powder, Fs1, Fs2, Fs3, Fs4, and Fe1, Fe2, Fe3, Fe4. (b) TG of Domiphen® raw powder (dash line), PLLA raw powder (thin black line), Fs4 (thick gray line), and Fe4 (thick black line).
Domiphen® release behavior
In order to explain the Domiphen® release behavior in pure water, both absolute release amount and relative release percentage with respect to the total loaded Domiphen® in fibers were presented respectively in Figure 5(a) and (b), under the condition that the fibers were incubated without degradation by proteinase K. The former gives a direct know-how of the amount of Domiphen® released in pure water at a certain sampling time, while the latter was re-calculated to obtain further understanding of how Domiphen® loading amount affected the existing state of itself on which Domiphen® release behavior was based upon. It is obvious that Domiphen® was released from solution electrospun fibers (Fs series) and emulsion electrospun spun fibers (Fe series) in a quite different way. Specificly, the burst release of Domiphen® from solution-spun fibers were 34.1%, 27.5%, 19.8%, 12.9%, accordingly. And after the burst release, sustained release lasted for nearly 12 h, and then there was seldom release of Domiphen® from the fibers within 48 h, which is usually called the plateau release phase. After 48 h, 94.3%, 90.1%, 86.7%, 83.3% were achieved of the accumulated maximum release to the loaded Domiphen® of every fiber sample, respectively. While, the burst release of Domiphen® from W/O emulsion electrospun fibers were seldom which were respectively 8.2%, 6.9%, 5.3%, 4.5%, considerably less than solution electrospun fibers. After the burst release, sustained release also lasted for nearly 12 h, and then it was the plateau release phase where Domiphen® was seldomly released within 48 h. After 48 h, there were still 63.9%, 67.4%, 71.8%, 73.8% remaining in the fibers, respectively. Obviously, which is same as the situation of the burst release, emulsion electrospun fibers released considerably less than the solution electrospun fibers. The sustained release period of these two fibers were fitted by a number of equations, which would suggest and reveal the mechanism of the molecule release. It was found that both Fs and Fe series fibers release Domiphen® following the Freundlich equation of ln(100 − R%) = alnt − b where the regression coefficient is considerably high. The release behavior provided the information about where and how Domiphen®existed in the PLLA fiber and the effect of Domiphen® facilitating the formation of the diameter-homogenous and core-sheath structure fibers (Table 1).
Domiphen® accumulated release curve of absolute amount (a) and percentage (b) from Domiphen®–PLLA electrospun fibers, and the fitting of release curves by Frendlich’s equation (c). Every value was presented as the avarage value, with all the relative standard deviations <5%, which is not shown in the graph. Frendlich’s equation fitting of Domiphen® release curves.

Specifically, because Domiphen® is both water soluble and organic solvents soluble, when Domiphen® was dissolved within PLLA-dichoromethane forming homogeneous solution, which was electrospun, Domiphen® accumulated mostly near the very border of the fiber’s cross section to exert its surfactivity. Thus, there would be considerable amount of burst release right after the fibrous film was immersed in the water release medium and, with time, the remaining Domiphen® moved a very short distance from the not-so-deep position to the fiber surface, and finally into water resulting in the sustained release period following the Freundlich equation. This could be even considered as the border-loaded Domiphen® molecules being desorbed from the fibers, which is not like the other small molecule compound that is much more lipophilic than Domiphen® where they are released from the fiber to water with little burst release and more sustained manner afterwards [11,16,21] (Figure 6).
After being degraded by proteinase K, Domiphen® accumulated release curve of absolute mount (a) from Domiphen®–PLLA fibers, and the fitting of release curves by Peppas’ equation (b).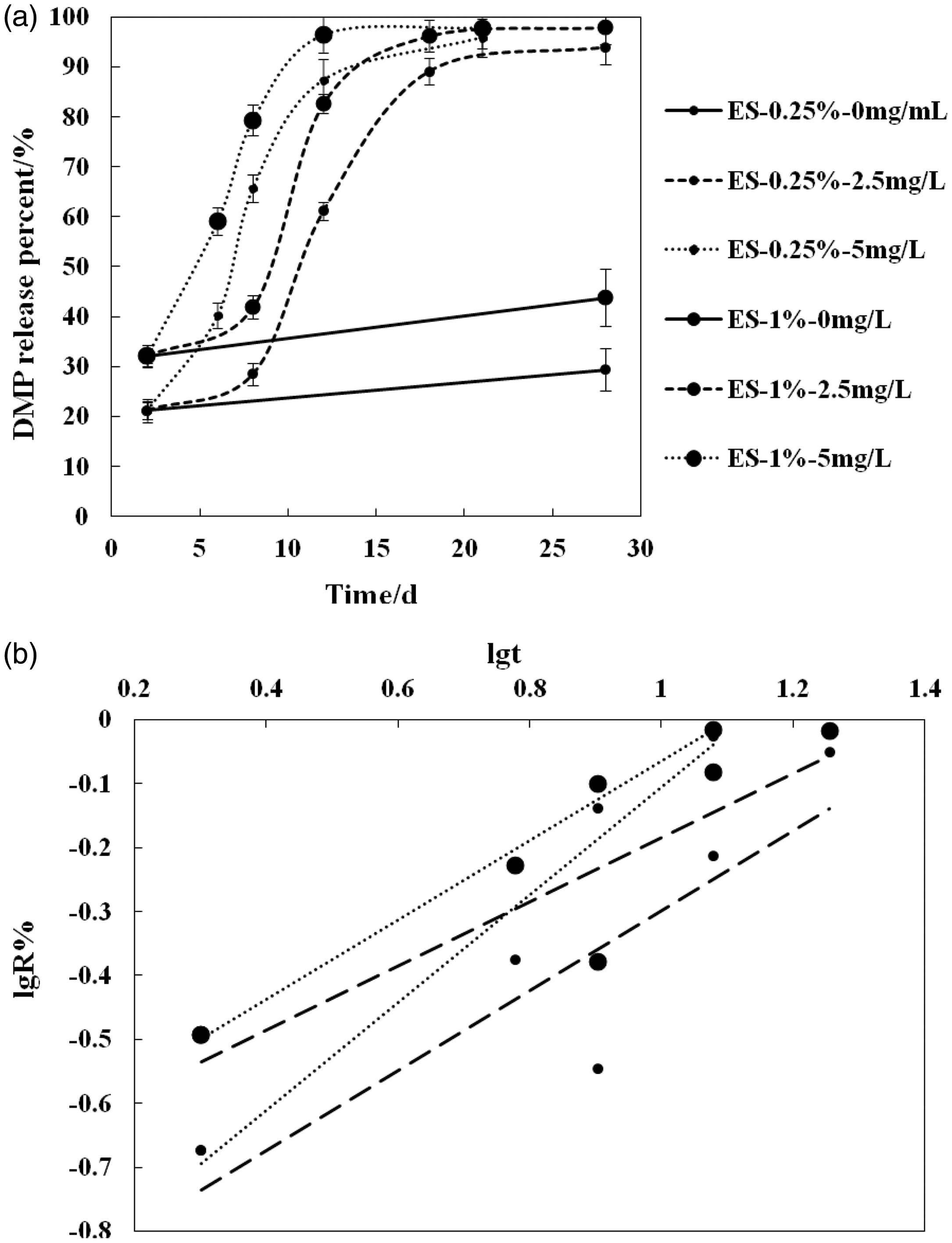
With respect to the plateau release phase, there was considerably small amount of Domiphen® remaining within Fs series fibers, which is respectively 19.7%, 14.3%, 10.9%, 5.7%. Meanwhile, there was still considerable amount of Domiphen®, which is respectively 78.8%, 74.8%, 71.4%, and 67.9% remaining in the fibers Fe1, Fe2, Fe3, and Fe4 when Domiphen® release experiment was carried out without the degradation of proteinase K.
The reason for this is that, with regard to the fiber electrospun from W/O emulsion with Domiphen® as the emulsifier, the Domiphen® molecules were located in a much deeper position of the PLLA fibers probably by the virtue of the internal water phase drawing Domiphen® to the border of organic solution phase and water phase, which is also the position on which Domiphen® had the emuslifing effect. The W/O emulsification also suggests that Domiphen® dispersed more uniformly within the fibers in comparison to the border-accumulating mode of the homogeneous solution-electrospun fibers. Moreover, further inspection of the existing state of Domiphen® in the fibers, proteinase K was used to degrade the fibers for nearly two weeks until Domiphen® had been totally released. Proteinase K of 2.5 mg/L and 5 mg/L were used within the water to degrade Fe1 and Fe4, and it was found that Domiphen®, which was conjectured being loaded and combined in deeper position of cross section of the fibers prepared from W/O emulsion electrospinning, almost released from fibers under the enzyme degradation finally after 21d and 28d.
From the previous study, the release of small molecules from aliphatic polyesters matrix can be divided precisely into three main phases as burst release phase, sustained release phase, and plateau release phase [22–24]. In our research, after the release period without proteinase K during which the weak-combined Domiphen® released as much as possible, there were already three kinds of release phase. And from the 48th hour, Domiphen® released in a three-combined period after proteinase K was applied into the release medium, which were the slow release period, speeding release period, and plateau release period. Specifically, at the very beginning, there was no rapid release as commonly seen in release of many small molecules from APEs fibers; however, the release speed begins to be fast after the slow release phase with the ongoing degradation of proteinase K, where the slope of the release becomes bigger suggesting that there was hardly any Domiphen® on the surface of the fibers and not so much of it near the outermost border of the fibers cross section, otherwise, the release at the beginning would be more than the release later presenting the release curve bend to X-axis, and not like the release curves bending to Y-axis in this research. Along with the degradation of proteinase K, Domiphen® was released both by corrosion and diffusion machanism, and the molecules in the inner place of the cross section of the fibers release little by little, which is commonly seen in many reports where the small molecule loaded APEs fibers were prepared by homogeneous solution electrospinning. At the end with the depletion of Domiphen® in fibers was the plateau release period. So, S shape release curves were figuratively presented. Especially for the situation of 2.5 mg/L proteinase K’s effect where S shape release curves were evident because proteinase K of lower concentration had not much significant effect on the degradation and far less Domiphen® was released in comparison with the situation of 5 mg/L proteinase K within 10 days. So released Domiphen® is as to the sustained release period providing a key information that except for the Domiphen® molecules accumulated around the border of the fibers cross section perfroming burst release, rest of the Domiphen® molecules indeed were located in the rather inner place, otherwise, there would be a rapid release increasing within 10 h after proteinase K was added. The facts revealed by Domiphen® release curves were in accordance with the results obtained by XRD and DTA [25–27]. Of course, with regard to the more accurate and precise existing position of Domiphen® within inner fibers cross section, it needs more experiments and methods for further study.
Peppas’s equation fitting from Domiphen® release curves under proteinase K degradation.

Along with the depletion of most of the Domiphen® within fibers, the very slow decomposition of PLLA-tightly-combined Domiphen® from PLLA matrix is also the reason for short plateau phase, which is usually presented in the previous researches [9,11,14,21].
In this research, we also electrospun Domiphen®-PLLA complexed fibers with more than 1% ratio of Domiphen®, and it was discovered that spun fibers disorderly accumulated from needle tip to the collection foil forming fibrous snaggle, which comprised a lot of beads and fibers of unhomogeneous diameters under FESEM observation. This may be caused by incompletable drawing of Taylor’s cone of polymeric solution caused by considerably high electric conductivity of Domiphen® [1,9]. So, it is assumed that the Domiphen® amount complexed within PLLA fibers should be below a certain percentage for electrospinning.
Conclusion
This study has systematically revealed the morphology and complexing state of Domiphen®–PLLA electrospun fibers, especially based on the Domiphen®–PLLA interaction after homogeneous solution and emulsion electrospinning and the corresponding Domiphen® release behaviors, respectively. Due to desirable solubility in dichloromethane–PLLA solution and water, Domiphen® was easily complexed within PLLA fibers, though in a different way, where most of the loaded Domiphen® accumulated on the border of the cross section of fibers for homogeneous solution electrospinning either partially accumulated on the border or partially within the inner position of the cross section for W/O emulsion electrospinning. Domiphen® plays a pivotal role of both emulsifying the inner aqueous phase for emulsion, and yet it is suggested that considerable rate of Domiphen® should be accumulated around the cavum formed after water evaporation within the inner phase during electrospinning, and maybe even more than the ratio of Domiphen® existing within the sheath layer, which was proved by the release behavior. After understanding this, it is also clearly known that a suitable surfactant must be chosen for the homogeneous diameter core-sheath fibers prepared by W/O electrospinning where it must have emuslifying function forming the inner hollows and the surface-tension-lowering function between polymer solution and atmosphere to realize diameter homogeneity [28].
As a research group for the drug delivery system, we expect the Domiphen®-complexed PLLA fiber to be considerably desirable and used for loading and delivering either lipophilic drug by using solid fibers electrospun from homogeneous solution, or lipophobic-hydrophilic drug including proteins, peptides, DNAs, RNAs, by using core-sheath fibers electrospun from W/O emulsion [1–4,7].
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Liaoning Province (2014022041), People’s Republic of China, Foundation of China Postal Doctor (2014M561187), and National Natural Science Foundation of China (51102178).

