Abstract
In this study, the potential effects of N, N’-dodecanedioic bis(cyclopentanecarboxylic acid) dihydrazide (BCADD) as a new additive in poly(L-lactic acid) (PLLA) was estimated. The comparative study on the melt-crystallization showed that the BCADD as heterogeneous nuclei facilitated crystallization of PLLA in cooling, which indicated by the obvious crystallization exotherms and sharp melt-crystallization peak. Unfortunately, with increasing of BCADD from 0.5 wt% to 3 wt%, it is unexpected that the melt-crystallization peak of the BCADD-nucleated PLLA shifted toward the lower temperature and became flatter, evidencing the importance of BCADD loading for PLLA’s crystallization. Additionally, the cooling rate and the final melting temperature were also proved to be important influence factors during PLLA’s melt-crystallization process, but in contrast with the effect of the final melting temperature on the melt-crystallization, a higher cooling rate could more seriously weaken crystallization ability of the BCADD-nucleated PLLA. The chemical nucleation mechanism was proposed to explain the promoting effect of BCADD on the crystallization of PLLA via the analysis of frontier orbital energy. The melting behaviors after crystallization further confirmed the crystallization accelerating role of BCADD, and the melting behaviors were affected by the heating rate, crystallization temperature and BCADD loading. Although the onset thermal decomposition of the BCADD-nucleated PLLA occurred at lower temperature comparing with the pure PLLA, the intermolecular interaction of PLLA with BCADD attempted to prevent the decrease of thermal stability. Overall, the addition of BCADD resulted in the complicated effect on the tensile modulus and tensile strength of PLLA, but the elongation at break continuously decreased when increasing BCADD loading.
Keywords
Introduction
For petroleum-based plastics, it is very difficult to complete degradation in nature in a short time, resulting in the increasingly serious environmental problems; moreover, a large-scale usage of petroleum resources further accelerates the shortage of fossil resources. Thus, developing degradable polymers derived from the renewable resources is a good way of replacing traditional petroleum-based polymers to maintain the sustainable development of human society. Poly(L-lactic acid) (PLLA) was produced through the polymerization of lactic acid monomers which obtained from renewable resources such as corn, potato, cassava, etc1,2; and PLLA can be eventually degraded into carbon dioxide and water, showing that the total process is environmental-friendly. In addition, PLLA also exhibits excellent biocompatibility, easy processability, and transparency.3–5 Thus, PLLA is considered to be the most promising biodegradable materials. 6 Nowadays, the application of PLLA as packaging,7,8 agricultural films,9,10 automobile, 11 electronics12–14 has been extensively reported and possesses remarkable comparative advantages and industrialization value. Yang et al. 15 reported that the poly(D-lactide) acid-g-polyethylene glycol-g-poly(D-lactide) acid triblock polymer and polyethylene glycol were introduced to improve the melt strength, film blowing stability, toughness and gas barrier properties of PLA. The relevant results showed that the modified PLLA exhibited excellent stable during the process of blown film, and the elongation at break of the modified PLLA could reach over 250%, and the O2 permeability coefficient decreased by 61%.
However, PLLA suffers from the defect of a very poor crystallization ability resulting from its low levo content, and a lot of amorphous formed during manufacturing must lead to the low crystallinity and poor heat resistance, limiting the PLLA’s large-scale application.16,17 Hence, improving crystallization rate is very critical for extend and apply of PLLA, as well as commercial competitive with the traditional thermoplastics. Adding nucleating agent is thought to be one of the most effective and easiest way to accelerate crystallization in actual manufacture,18,19 and a large number of inorganic or organic additives as heterogeneous nucleating agents were employed for enhancing crystallization ability of PLLA, these popular additives include talc,20–22 montmorillonite,23,24 metal phenylphosphonate,25,26 salts amino acids, 27 TMC,28,29 cellulose,30,31 etc. And these additives were proved to be the effective nucleating agents for promoting crystallization of PLLA via the evaluation of the crystallization process, even some additives could cause the half time of overall crystallization to decrease into below 1 min.27,32
Even then, the crystallization accelerating ability of nucleating agent still need to be further improved to meet the greater industrial requirements. Accordingly, it is very important to find out the definite nucleation mechanism to develop the efficient nucleating agent, although there exist currently two nucleation mechanism of epitaxial and chemical nucleation. 33 Whereas determining nucleation mechanism needs to be based on the study on the influences of the more additives on PLLA’s crystallization process, and the more in-depth analysis on influence factors, including the lattice size, molecular structure, key groups, etc. may be conducted. Thus, developing more additives as nucleating agents is urgent to solve the aforementioned problem. Compared to the inorganic additives, the organic additives as nucleating agents can avoid the defect of poor compatibility of PLLA with inorganic additives, and the molecular designability and excellent nucleation effect are also two important advantages of organic additives. Thus, more and more works focus on synthesizing new organic nucleating agents for PLLA.34,35 However, the category and quantity of organic nucleating agents are still insufficient, which is seriously not conducive to reveal nucleation mechanism of organic additives for PLLA.
The goal of this work was to synthesize N, N’-dodecanedioic bis(cyclopentanecarboxylic acid) dihydrazide (designated here as BCADD) to serve as a new organic nucleating agent for PLLA. Apart from the melt-crystallization processes from the different melt temperatures and cooling rates, the melting behavior, thermal decomposition process and mechanical properties of PLLA containing various amounts of BCADD were also investigated by differential scanning calorimeter (DSC), thermogravimetric analysis (TGA) and electronic tensile tester. Additionally, the probable nucleation mechanism was proposed through the analysis of frontier orbital energy.
Experimental section
Reagents and materials
All analytical pure reagents were obtained from Chongqing Huanwei Chemical Company of China, they were cyclopentanecarboxylic acid, thionyl chloride, dodecanedioic dihydrazide, triethylamine and N, N-dimethylformamide. The PLLA in this study was produced by Nature Works LLC of USA, and the grades of PLLA product was 4032D.
Synthesis of BCADD
As seen in Figure 1, the BACDD was synthesized through two step liquid-phase reactions of the acylation and amination. First, the cyclopentanecarboxylic acid was added into the mixed solution including the tiny amounts of N, N-dimethylformamide as catalyst and excess thionyl chloride as solvent and reactant, with slowly increasing of temperature to 80 ºC, the cyclopentanecarboxylic acid was dissolved with stirring, and the mixed solution was kept on stirring at 80 ºC to reflux for 36 h. After cooling, the aforementioned mixed solution was distilled in vacuum, and the residue was cyclopentanecarbonyl chloride.
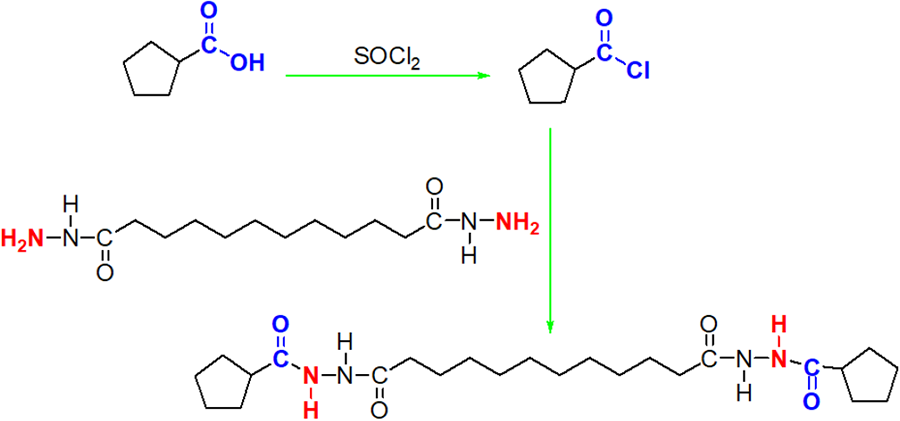
Synthetic scheme of BCADD.
Second, the dodecanedioic dihydrazide and triethylamine was dissolved into the N, N-dimethylformamide as solvent, and then the cyclopentanecarbonyl chloride was slowly added into the aforementioned mixed solution with stirring, and the solution was stirred at ice bath for 1.5 h, followed by heating up to 70 ºC to hold for 5 h. After that, the mixed solution was poured onto the deionized water of 300 mL, followed by filtrating and further washing using the deionized water. Finally, the product was dried over night at 35 ºC under vacuum. Fourier Transform Infrared Spectrometer (FT-IR) υ: 3443.1, 3216.3, 3043.5, 2921.5, 2849.1, 1662.7, 1601.4, 1485.5, 1470.3, 1448.2, 1412.0, 1376.6, 1348.0, 1271.3, 1212.8, 1187.3, 1160.1, 1061.9, 955.8, 722.8, 669.3 cm−1; 1H Nuclear Magnetic Resonance (1H NMR) δ: ppm; 9.73 (s, 1H, NH), 9.60 (s, 1H, NH), 2.73∼2.80 (t, 2H, CH2), 2.06∼2.10 (t, 2H, CH2), 1.62∼1.98 (m, 4H, CH), 1.49∼1.58 (t, 8H, CH2, Cyclopentane), 1.24 (s, 2H, CH2).
Preparation of PLLA/BCADD
PLLA and various concentrations BCADD (0.5 wt%, 1 wt%, 2 wt% and 3 wt%) were blended at the temperature of 190 ºC. And the blending process was following: the melting blend was firstly performed on a counter-rotating mixer with the rotation speed of 32 rpm for 5 min; and then the rotation speed was increased to 64 rpm for 5 min; finally, the mixture was hot pressed and cool pressed to obtain the relevant samples in the pressure of 20 MPa.
Test and characterization
The BCADD synthesized in our lab was characterized by FT-IR and 1H NMR. The KBr pellet was empolyed to prepare the FT-IR testing sample of BCADD, and the testing wavenumber was from 4000 cm−1 to 400 cm−1; the deuterated dimethyl sulfoxide was used to dissolve BCADD to perform 1H NMR test. The crystallization and melting processes of pure PLLA and PLLA/BCADD samples after eliminating heat history were recorded by Q2000 DSC. The temperature and heat flow were calibrated using an indium standard before DSC testing, and the testing sample was about 5∼7 mg, the flow rate of nitrogen was set to 50 mL/min. The Q500 TGA performed the thermal decomposition test in air atmosphere, and the testing temperature was from room temperature to 650 ºC at a heating rate of 5 ºC/min. A comparative test on the mechanical properties was performed on a D&G DX-10000 electronic tensile tester with 1 mm/min stretching speed.
Results and discussion
Melt-crystallization behavior
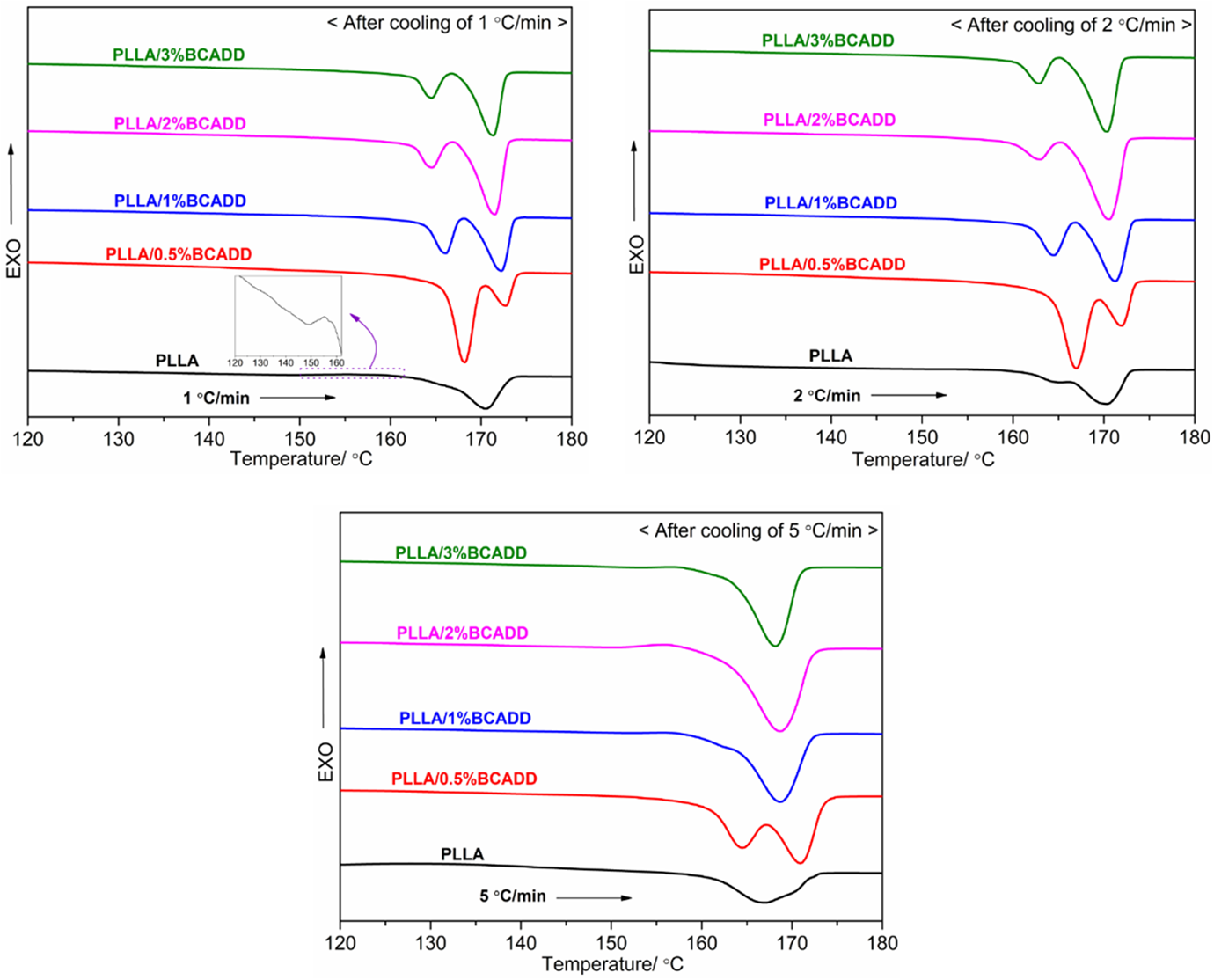
Figure 2 shows the melt-crystallization DSC curves of pure PLLA and PLLA/BCADD samples after melted at 190 ºC for 3 min at a cooling rate of 1 ºC/min. Usually, the slow cooling rate can cause nuclei to be activated in the high-temperature region to promote the appearance of crystal. 22 However, it is a great pity that, upon cooling of 1 ºC/min, pure PLLA does almost not have the melt-crystallization peak in DSC curve, which is thought to be because of the tremendously poor crystallization ability of pure PLLA. Whereas all PLLA/BCADD samples have very obvious crystallization exotherms in DSC curves, meaning that the presence of BCADD in PLLA leads to an enhancement in crystallization ability, directly resulting from the heterogeneous nucleation effect of BCADD. Additionally, as seen in Figure 2, the BACDD loading is an important factor that affects PLLA’s crystallization behavior. Both the onset crystallization temperature (Toc) and melt-crystallization peak temperature (Tc) shift to the lower temperature region with increasing of BCADD loading from 0.5 wt% to 3 wt%, as well as the melt-crystallization enthalpy (ΔHc) decreases from 50.2 J/g to 45.7 J/g, which indicates that the PLLA/0.5%BCADD sample has the more powerful crystallization ability than other PLLA/BCADD samples, this result may depend on the effect of BCADD loading on the mobility of PLLA molecular chain, because the crystallization process often includes two stages of nucleation and crystal growth, the existence of BCADD in PLLA matrix can bring about very fast nuclear rate, under this circumstance, the mobility of PLLA molecular chain directly determines the crystal growth rate and final crystallization rate. When the BCADD loading exceeds the saturated nucleation density in PLLA matrix, the excessive BCADD can exhibit an inhibition for the mobility of PLLA molecular chain. In a word, when the melt temperature is 190 ºC, the PLLA/0.5%BCADD sample possesses the highest Toc and Tc, as well as the largest ΔHc.
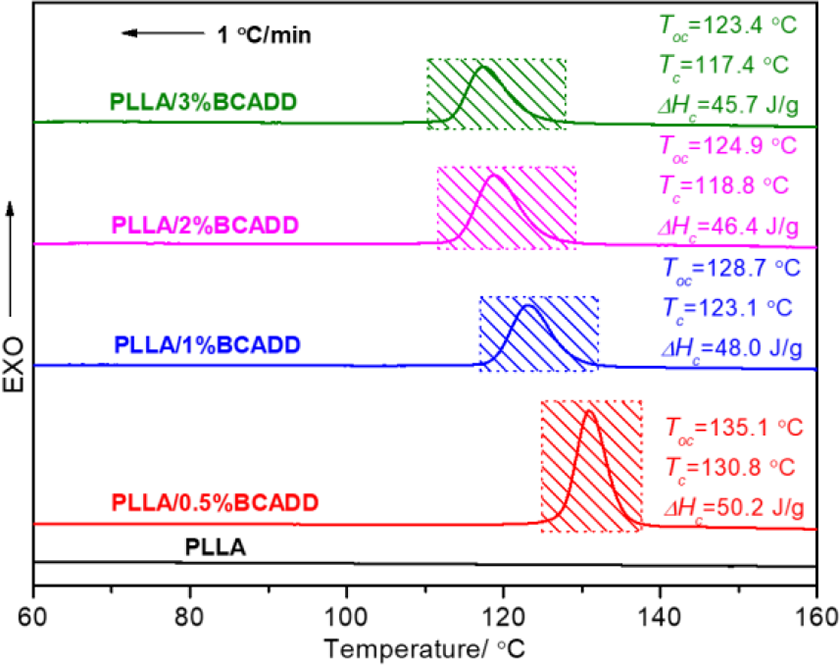
Melt-crystallization exotherms of pure PLLA and PLLA/BCADD samples from 190 ºC at a cooling rate of 1 ºC/min.
As shown in Figure 3, for pure PLLA, no crystals are formed when increasing cooling rate from 5 ºC/min to 10 ºC/min; for any PLLA/BCADD sample, the melt-crystallization behavior still occurs as the cooling rate increased, however, the crystallization peak of PLLA/BCADD becomes wider, and the Toc and Tc shift toward the lower temperature region, suggesting that a stronger supercooling can promote the polymer melt to crystallize, and the similar results can be found in other nucleated PLLA systems.17,36 On the other hand, upon cooling rate of 10 ºC/min, it is disappointing that the melt-crystallization peak has become relatively flat, which implies that the promoting effect of BCADD for PLLA’s crystallization is weaker than other nucleating agents such as NT-20, 21 BTCA, 18 N1, N1′-(ethane-1,2-diyl)bis(N2-phenyloxalamide), 37 cadmium phenylmalonate, 38 etc.
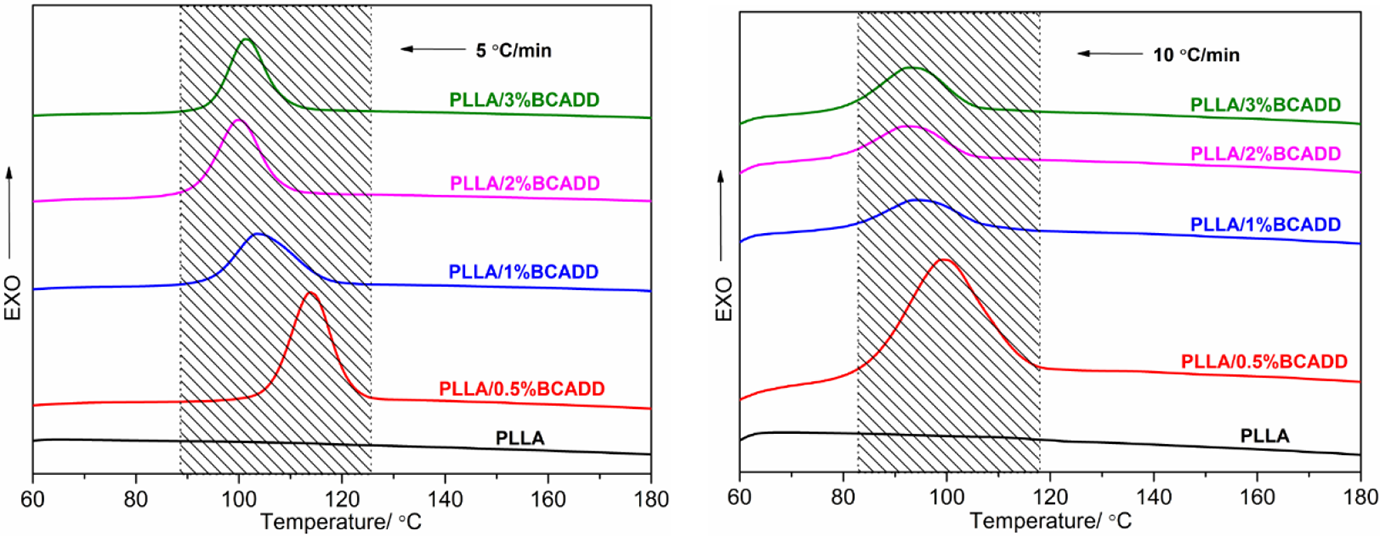
Melt-crystallization exotherms of pure PLLA and PLLA/BCADD samples from 190 ºC at different cooling rates.
As reported, 39 the solubility is a key factor that greatly influences nucleation efficiency for PLLA crystallization, and the final melting temperature (Tf) solely determines the solubility of the additive in PLLA resin. A higher Tf must lead to the greater solubility of the additive in PLLA, further exhibiting the better compatibility and stronger interaction. However, on the other hand, an increase of solubility results in the decrease of undissolved additive, which decreases the nucleation density. Figure 4 displays the melt-crystallization exotherms of pure PLLA and PLLA/BCADD samples in cooling at a rate of 1°C/min after the melt at different Tf for 3 min. As seen in Figure 4, when the Tf is 180 ºC, pure PLLA exhibits a very wide and weak melt-crystallization peak, the reason may be that a few crystals formed during processing cannot be thoroughly melted at 180 ºC, and these un-melted crystals as nucleus accelerate PLLA’s crystallization in cooling. Whereas all crystals are thoroughly melted at 200 ºC, and the nucleation rate of pure PLLA is very slow in cooling, as a result, the melt-crystallization peak can almost not be observed. For PLLA/BCADD samples, the influence trend of BCADD loading on crystallization process of PLLA is almost irrelevant with Tf, that is to say, the melt-crystallization peak appears in the lower temperature region with increasing of BCADD loading in general, when the Tf is identified. Through the comparative analysis of DSC curves and relevant data, it is found that, except for PLLA/3%BCADD sample, the To, Tc and ΔHc of other PLLA/BCADD samples generally increase as Tf is increased from 180 ºC to 200 ºC, meaning that a greater solubility is more conducive to crystallization comparing with the negative effect of a decrease of undissolved BCADD in this study. Additionally, it should be noted that, when the Tf is 200 ºC, the ΔHc of PLLA/1%BCADD is raised to the largest value of 53.0 J/g, indicating that PLLA/1%BCADD has the highest crystallinity after melt-crystallization.
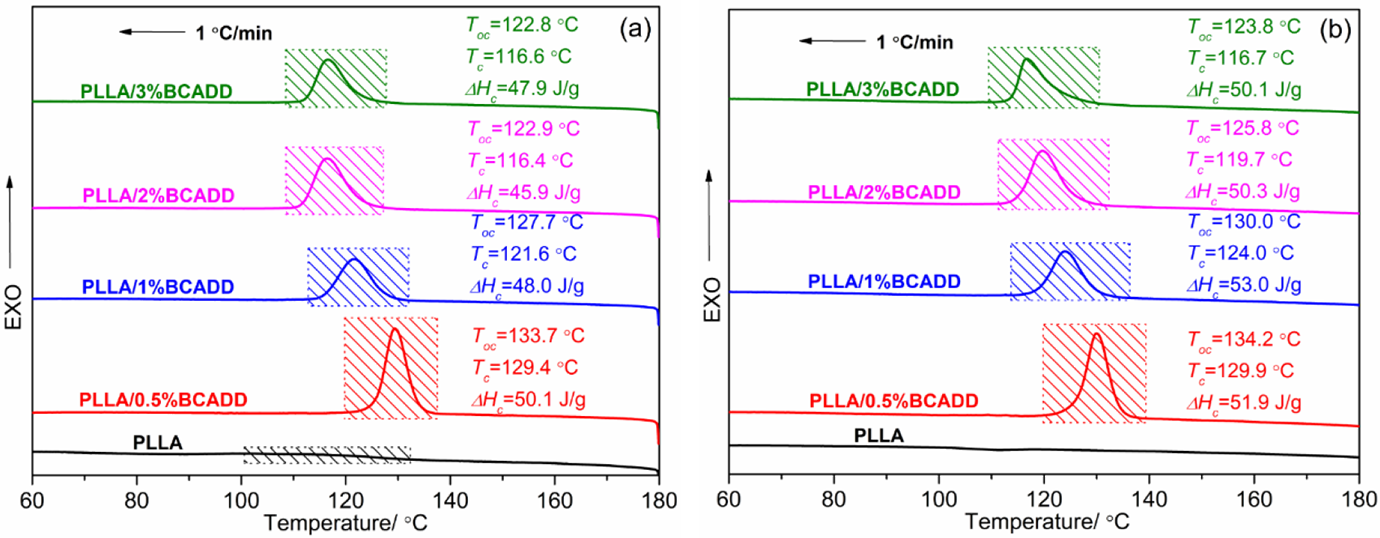
Melt-crystallization exotherms of pure PLLA and PLLA/BCADD samples after melt at 180 ºC (a) and 200 ºC (b).
For the melting blend process, both BCADD and PLLA molecules are activated via the thermal transmission, resulting in the very high molecular activity. If there existed the lower energy gap between PLLA and BCADD, it is very easy to form the intermolecular interaction during blending, which may be used to explain the nucleation mechanism of BCADD for PLLA. In addition, the literature has reported the fact that the polar-NH group can improve intermolecular interaction with PLLA. 39 Thus, the calculations on the frontier orbital of PLLA with 10 units and BCADD were performed by DMol3 density functional theory method, and the calculation result was shown in Figure 5. And the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of PLLA are −11.082 eV and 0.251 eV, and the HOMO and LUMO are −0.199 eV and −0.046 eV for BCADD. It is very clear that the energy gap of LUMO of BCADD with HOMO of PLLA is lower than that of PLLA itself, indicating that the electron of HOMO in PLLA more easily flows to the LUMO of BCADD. Moreover, it is observed form Figure 5 that the LUMO of BCADD focus on the amide group, whereas the HOMO of PLLA focus on the end group of repeating monomer, which is also instrumental in reducing space resistance. Through analysis of molecular structure, the intermolecular interaction may be presented in the N-H of BCADD and C=O of PLLA via hydrogen bond, and the chemical nucleation is proposed as nucleation mechanism of BCADD for PLLA.
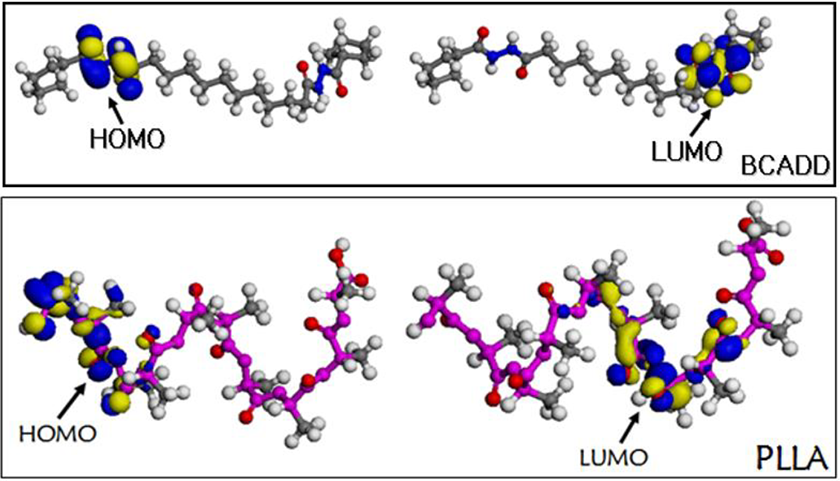
HOMO and LUMO of BCADD and PLLA.
Melting behavior
In the second heating scan, both the previous crystallization and heating rate dramatically affect melting behavior of semi-crystalline polymer.40,41 Thus, investigating on the melting process of the modified PLLA can further reveal the role of BCADD in PLLA matrix. Figure 6 is the melting processes of pure PLLA and PLLA/BCADD samples at different heating rates corresponding to the rate of melt-crystallization at different cooling rates. As shown in Figure 6, the DSC curves have obvious double melting peaks, but the double melting peaks gradually merge into the single melting peak with a wide melting range with increasing of rate, the probable reason is that a higher heating rate cannot make the melted crystals to have no enough to form new crystals in heating according to the melting-recrystallization theory, 42 leading to the appearance of the single melting peak. For PLLA without BCADD, when the rate is 1 ºC/min, the low-temperature melting peak is very weak comparing with the high-temperature melting peak, the reason may be that it is difficult for pure PLLA to form nucleus via homogeneous nucleation in cooling, leading to the formation of few crystals, which further confirms the aforementioned DSC results. Whereas a low heating rate is often beneficial for crystallization in heating, resulting in the relative larger high-temperature melting peak area; but an increase of heating rate causes crystallization ability to weaken, exhibiting the decrease of peak area of high-temperature melting peak comparing with the low-temperature melting peak. For PLLA with BCADD, the presence of the heterogeneous nucleating agent BCADD can induce crystallization to occur at the higher temperature in cooling, which results in that, when the cooling rate is slow, a large number of crystals are formed because of the synergistic effect of BCADD (provide fast nucleation rate) and excellent mobility of PLLA molecular chain (assure the crystal growth), which is reason for that the low-temperature melting peak has the larger melting enthalpy with decreasing of rate. However, upon a given rate, PLLA/0.5%BCADD has the largest area of low-temperature melting peak, meaning that PLLA/0.5%BCADD has more crystals in cooling, which is consistent with the melt-crystallization DSC results. Whereas the PLLA/3%BCADD sample exhibits the largest area of high-temperature melting peak.
Melting behavior of pure PLLA and PLLA/BCADD samples at different heating rates corresponding to the rate of melt-crystallization at different cooling rates.
The melting behavior of PLLA/BCADD samples after isothermal crystallization was further studied by DSC. Figure 7 displays DSC melting curves of pure PLLA and PLLA/BCADD samples at a heating rate of 10 ºC/min after isothermal crystallization at different crystallization temperatures for 180 min. It is still observed that there exists the phenomenon of the double melting peaks after crystallization at 115 ºC, although the crystallization time has reached 180 min, which thoroughly confirms the crystal growth rate is the rate-determining step for PLLA/BCADD sample, because the BCADD can provide fast nucleation rate, under this circumstance, a low crystallization temperature leads to poor mobility of PLLA molecular chain, which affects crystal growth rate, as a result, the crystallization cannot be fully carried out and the formed crystals possess different perfectability. When these crystals are heated, less perfect crystals are firstly melted, and these melted crystals can form new crystals again through being attached to the surface of an un-melted crystal, leading to the appearance of double melting peaks. However, when the crystallization temperature is 120 ºC, pure PLLA and PLLA/0.5%BCADD exhibit the single melting peak comparing with the double melting peaks of other PLLA/BCADD samples, furthermore, the single melting peak of pure PLLA is sharper than that of PLLA/0.5%BCADD sample, meaning that pure PLLA has more perfect crystals. When the crystallization temperature can ensure that the PLLA molecular chain has excellent mobility, the crystallization can be fully carried out at the corresponding temperature, as well as the relative perfect crystals are formed, resulting in that the melt occurs at the higher temperature, and it is difficult to have un-melted crystals as nucleus to recrystallize in heating, which is used to explain the single melting peak when the crystallization temperatures are 125 ºC and 130 ºC.
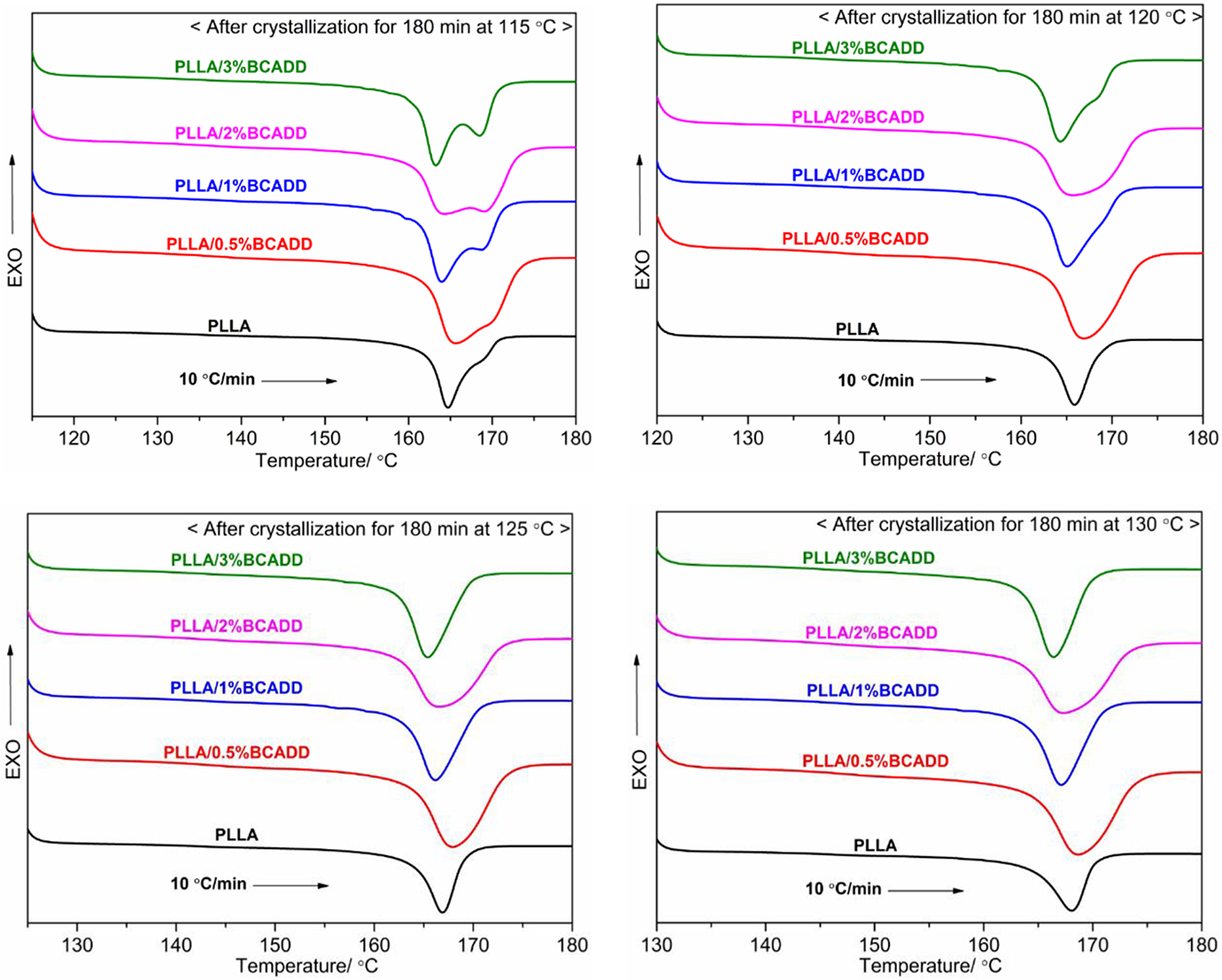
Melting behavior of pure PLLA and PLLA/BCADD samples after isothermal crystallization at different temperatures.
Thermal stability
Figure 8 is the thermal decomposition process of pure PLLA and PLLA/BCADD at a heating rate of 5 ºC/min in air atmosphere of 60 mL/min. In heating, any PLLA/BCADD as pure PLLA only exhibits one thermal weight loss stage, and the weightlessness appears at 270 ºC∼370 ºC, which originates from the chain scissions and loss of ester groups.43,44 Meantime, the thermal decomposition profile of any PLLA/BCADD is also similar with pure PLLA. In a word, the influence of BCADD and its loading on thermal decomposition profile of PLLA is negligible. However, the presence of BCADD affects the onset thermal decomposition temperature (Tod) of PLLA as shown in Figure 8. The Tod located at 341.3 ºC, 321.2 ºC, 329.6 ºC, 328.7 ºC, 329.2 ºC, 328.7 ºC and 329.2 ºC belongs to pure PLLA, PLLA/0.5%BCADD, PLLA/1%BCADD, PLLA/2%BCADD and PLLA/3%BCADD, respectively. It is found that the Tod of pure PLLA is higher than that of any PLLA/BCADD through the data comparison, implying that the thermal stability of PLLA becomes poor because of the existence of BCADD. Among PLLA/BCADD samples, PLLA/0.5%BCADD exhibits the lowest Tod, the reason may be that PLLA/0.5%BCADD with the best crystallization ability can form more crystals upon fast cooling, but there exist great differences between these imperfect crystals, which results in the formation of more defects in modified PLLA. As a result, PLLA/0.5%BCADD firstly decomposes at the lower temperature. However, compared to the significant reduction of Tod of PLLA/BCADD with pure PLLA, there is a small difference in Tod among PLLA/BCADD samples, and the maximum difference is 8.4 ºC. Additionally, it is also observed that the Tod does not continuously decrease as BCADD concentration is increased from 0.5 wt% to 3 wt%, an important reason may be the interaction of PLLA and BCADD, because this intermolecular interaction has an inhibition for the decomposition in heating.
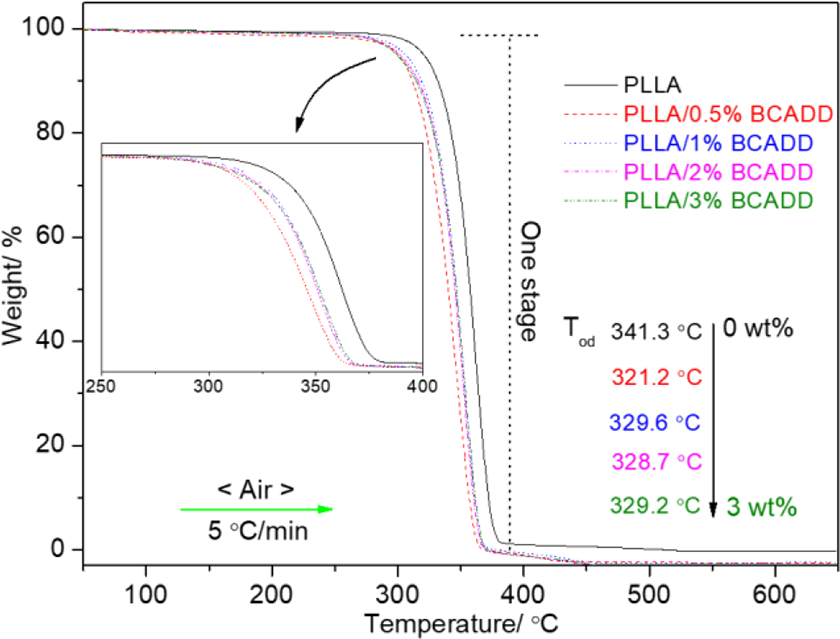
TGA of pure PLLA and PLLA/BCADD in air atmosphere.
Mechanical properties
The influences of BCADD and its loading on the tensile modulus, tensile strength and elongation at break of PLLA were displayed in Figure 9. Except for the elongation at break, the tensile modulus and tensile strength exhibit an irregular change with increasing of BCADD loading. Specifically, the tensile modulus firstly decreases and shows a minimum value at 1 wt% BCADD loading, and then increases as BCADD loading further increased to 2 wt%, finally decreases again. The addition of BCADD can plasticize PLLA to some extent, which is thought to be due to smaller size crystals; but the addition of BCADD probably results in an increase of rigidity because of rigidity of BCADD itself. In this case, the tensile modulus of PLLA/BCADD depends on the aforementioned competitive relationship. However, compared to all PLLA/BCADD samples, pure PLLA has the largest tensile modulus value, showing that pure PLLA has the strongest rigidity. As for the tensile strength, 0.5 wt%∼2 wt% BCADD loading can increase the tensile strength of PLLA whereas a higher BCADD loading (like 3 wt% in Figure 9) will cause the modified PLLA material to become brittle. Additionally, Figure 9 clearly shows that the introduction of BCADD continuously decreases the elongation at break of PLLA, one reason is thought to be crystals formed during compression molding; another reason may be the phase-separated BCADD in PLLA matrix, leading to the serious defects which firstly break during tensile process.
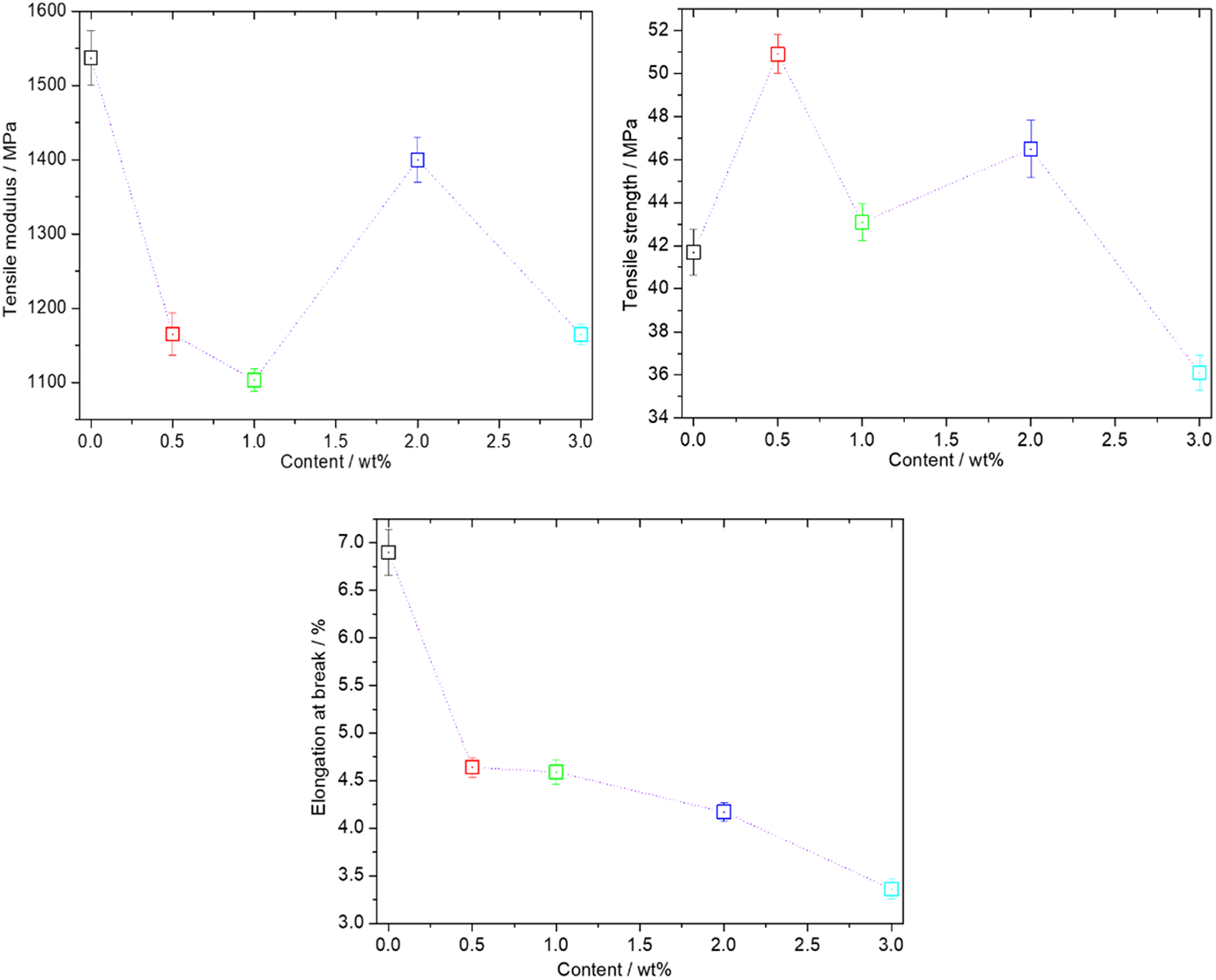
Mechanical properties of pure PLLA and PLLA/BCADD samples.
Conclusions
BCADD as a new organic heterogeneous nucleating agent for PLLA was synthesized to estimate its influences on melt-crystallization, melting process, thermal decomposition process, and the mechanical properties of PLLA. The melt-crystallization after the melt of 190 ºC showed that the BCADD could be used as an effective site of heterogeneous nucleation, leading to the enhancement of crystallization ability of PLLA/BCADD; and compared to other PLLA/BCADD, PLLA/0.5%BCADD exhibited the highest Toc of 135.1 ºC and Tc of 130.8 ºC, as well as the largest ΔHc of 50.2 J/g in cooling. Additionally, a higher Tf was generally conducive to crystallization. And the enhancement on crystallization ability of PLLA/BCADD was proposed to be the chemical nucleation via the calculation of frontier orbital energy of PLLA and BCADD, because there existed the smaller energy gap of PLLA with BCADD. Unfortunately, an increase of cooling rate weakened crystallization ability of BCADD-nucleated PLLA, proved by the more flat melt-crystallization peak located at the lower temperature. The melting processes of PLLA/BCADD depended on the previous crystallization, the heating rate, the crystallization temperature and BCADD loading; and these differences in melting behaviors of PLLA/BCADD also effectively reflected the different promoting effect of BCADD loading for PLLA’s crystallization, and the double melting peaks phenomenon in DSC curves was attributed to the melting-recrystallization. The addition of BCADD did almost not affect the thermal decomposition profile of PLLA, but the thermal stability was decreased because of the drop in Tod, however, the intermolecular interaction of PLLA with BCADD attempted to prevent the drop in Tod with increasing of BCADD loading. The resultant of mechanical properties showed that the presence of BCADD decreased the elongation at break of PLLA, but the tensile modulus and tensile strength could not be attributed simply to the introduction of BCADD.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (project number 51403027), Foundation of Chongqing Municipal Science and Technology Commission (cstc2019jcyj-msxmX0876), Scientific and Technological Research Program of Chongqing Municipal Education Commission (project number KJQN201801319).
