Abstract
The crystallization activation energy (ΔE) of a polymer comprises the nucleation activation energy ΔF and the transport activation energy ΔE*. In this paper, the ΔE of poly (L-lactic acid) (PLLA) nucleated with nucleating agent p-tert-butylcalix[8]arene (tBC8) was calculated. The results showed that the ΔE of nucleated PLLA was 165.97 kJ/mol, which is higher than that of pure PLLA. The reason why ΔE of PLLA increased when incorporating nucleating agent was studied. The increment of glass transition temperature (Tg) for nucleated PLLA revealed that the polymer chain mobility was restricted by tBC8, which was considered as the reason for the increase of ΔE*. Further, polyethylene glycol (PEG) was added to improve the chain mobility, thus eliminated the variation of the transport activation energy ΔE* caused by tBC8. Then the effect of the increment of crystallization temperature range on the increase of ΔF was also taken into consideration. It was concluded that both decreasing the mobility of chain segments and increasing the crystallization temperature range caused an increase of ΔE for PLLA/tBC8.
Introduction
The past 50 years has witnessed extensive researches in crystallization of polymers1–4, which consists two steps: nucleation of crystals and their subsequent growth5,6. In the crystallization process of polymers, nucleation often is hampered by the covalently bonded, long-chain structure of the crystallization species. Then it has become commonplace to employ suitable additives to promote this process7,8. As a result, additives such as nucleating agents are usually added to polymers in order to enhance the crystallization rate 9 .
The activation energy of crystallization (ΔE) which could be determined by the equation proposed by Kissinger 10 is always used to investigate the crystallization behavior of polymers. An interesting phenomenon is usually observed when ΔE is calculated:11–14 The ΔE is increased after adding nucleating agents, although the crystallization rate has been accelerated. According to previous conjecture11,13, the effect of nucleating agent on polymer crystallization is twofold: on one hand, nucleating agents can serve as heterogeneous nucleating sites and favor crystallization growth of molecular interface, corresponding to the decrease of ΔF; on the other hand, nucleating agents would baffle the transfer of macromolecular segments from PLLA melt to crystal growth surface due to the interaction between nucleating agents and segments of PLLA, which is related to the increase of ΔE*. However, nucleation is the rate-controlling step during crystallization, as a result, increasing the nucleation rate results in an increase of the overall crystallization rate.
Although a lot of research has focused on energy barrier for crystallization of polymers15,16, there are still no articles which explained the reason for the increase of ΔE after adding nucleating agent clearly. According to our previous study, tBC8 is an effective nucleating agent for PLLA, which exceeds talc the commercial NA for PLLA. In this article the ΔE of PLLA nucleated with tBC8 was investigated. To further disclose the mechanism of the increase of ΔE, polyethylene glycol was added to PLLA, which can improve the chain mobility.
Experimental
Materials
The PLLA (MW =100000 g mol−1) used in this paper was manufactured by Jiu Ding Company, China. All the other chemicals were of reagent grade, purchased from commercial sources and used without further purification.
Preparation of Nucleating Agent tBC8
The p-tert-butylcalix[8]arene was synthesized according to the method of Munch 17 . The mixture of 50 g (0.33 mol) of p-tert-butylphenol, 17.5 g (0.55 mol) of paraformaldehyde, 300 mL of xylene and 0.415 g (0.01038 mol) sodium hydroxide was put in a 1 L flask with a water separator. Then it was heated to reflux for 4 h under the circumstance of nitrogen. After the mixture was cooled to room temperature, the precipitate was obtained after filtration. The crude product was washed, in succession, with 200 mL portions of toluene, ether, acetone, and water and then dried under reduced pressure. It was added into ethylbenzene. The mixture was kept refluxing for two hours. The boiling solution was concentrated to a quarter of its volume. The solution was cooled to room temperature, and the precipitate was collected by filtration. Then colorless powder nucleating agent tBC8 was obtained after drying under vacuum at 60°C for 12 h.
Preparation of PLLA Samples
PLLA was mixed with 1 wt% nucleating agent in a self-designed flask, which had an inlet and outlet for nitrogen. After removing air with nitrogen for 10 min, the temperature was raised to 200°C and held for 5 min to reach equilibrium. The mixture was stirred at 200°C for 5 min. The neat PLLA was prepared in the same procedure.
Measurements
Differential Scanning Calorimetry (DSC)
DSC (Diamond, Perkin-Elmer) was carried out to study the crystallization of PLLA. Temperature was calibrated before the measurements by using indium as a standard. All DSC operations were carried out under a nitrogen atmosphere.
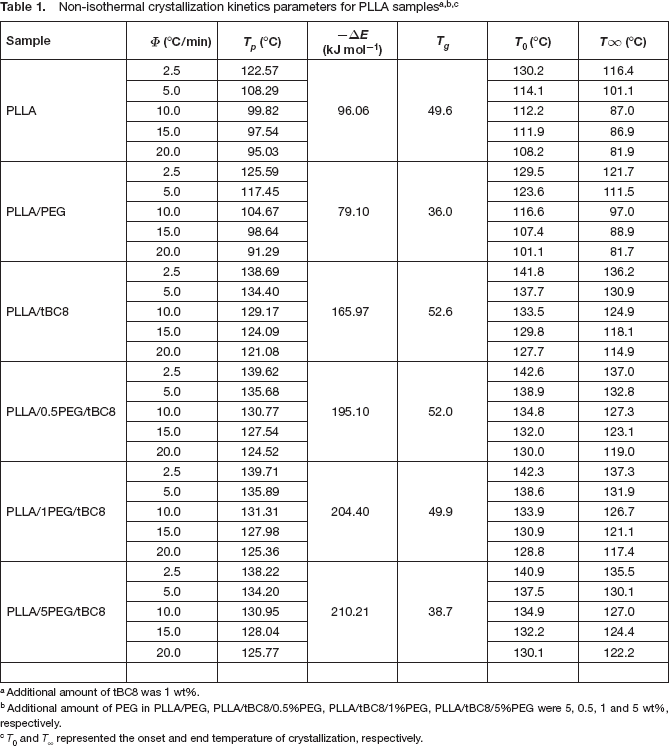
For the study of crystallization activation energy, a sample with a weight in the range of 3 to 4 mg was heated from 50°C to 200°C at the rate of 20°C min−1, held at 200°C for 5min, and then cooled from 200°C to 50°C at a cooling rate of 2.5, 5, 10, 15 and 20°C /min. Data were recorded in the process. The crystallization peak temperatures (T p ) can be obtained from this curve.
To study the cold crystallization and glass transition temperature (T g ) of PLLA samples were initially heated to 200°C, and held at 200°C for 5 min to erase previous thermal history. After subsequently rapidly cooling, samples were heated at 10°C/min, data were recorded in the process. T g was taken both at the midpoint of the specific heat increment and at the onset of a step, determined as the intercept between the baseline preceding the transition and the steepest tangent to the step.
Polarized Optical Microscopy (POM)
Optical micrographs were obtained with an Olympus BX51 (Japan) polarized optical microscopy attached with a DP70 digital camera and a THMS600 hot-stage. Samples were placed between two microscopy slides, melted and pressed at 200°C for 5 min to erase any trace of crystals, and then rapidly cooled to a predetermined crystallization temperature.
Results and Discussion
The Activation Energy of Crystallization for PLLA/tBC8
Crystallization peak temperatures (T
p
) of PLLA and PLLA/tBC8 under different cooling rate are listed in
The activation energy of crystallization for PLLA samples can be obtained by using the Kissinger equation
10
:

Kissinger plots for calculating non-isothermal crystallization activation energies for PLLA samples
Additional amount of tBC8 was 1 wt%.
Additional amount of PEG in PLLA/PEG, PLLA/tBC8/0.5%PEG, PLLA/tBC8/1%PEG, PLLA/tBC8/5%PEG were 5, 0.5, 1 and 5 wt%, respectively.
T0 and T∞ represented the onset and end temperature of crystallization, respectively.
As listed in
ΔE consists of nucleation activation energy ΔE and the transport activation energy ΔE*12. ΔE is the free energy of formation of the critical-size crystal nuclei, and ΔE* refers to the activation energy required to transport molecular segments across the phase boundary to the crystallization surface. According to previous presumption11,13, it can be attributed to the fact that the addition of nucleating agents would baffle the transfer of macromolecular segments from melt to crystal growth surface. It was caused by the interaction between nucleating agents and segments of PLLA, which was related to the increase of ΔE*.

Based on the equation (2), ΔE* is regarded to be inversely proportional to the glass transition temperature (T
g
)19–21. To verify that whether ΔE* of PLLA/tBC8 was higher than that of PLLA, the effect of tBC8 on mobility of chain segment was studied by means of measuring T
g
of the samples. The results of T
g
also listed in
To further disclose the mechanism of the increase of ΔE, PEG was added to this system as a nucleation promoter to eliminate the rise of ΔE* caused by nucleating agent tBC8. As reported22,23, PEG can improve the mobility of PLLA chain segments and increase the crystallization rate by reducing the inter-chain interaction, which results in a decline of ΔE*.
First, the crystallization behavior of PLLA/PEG/tBC8 samples was studied to reveal the effect of PEG, so as to help us determine whether inducing PEG was good for the crystallization of PLLA/tBC8 or not (

Cold crystallization behavior of PLLA samples

CPOM images of isothermal crystallization of PLLA (a) PLLA/5PEG/tBC8 (b) and PLLA/tBC8 (c) at 140°C
However, T
cp
of PLLA/0.5PEG/tBC8 was higher than that of PLLA/tBC8, from
Here, we eliminated the rise of ΔE* caused by nucleating agent tBC8 through adding PEG. In addition, we proved that once the additional amount of PEG was higher than 1%, the negative effect on nucleation would be covered by its contribution to the segmental mobility. The additional amount of PEG for PLLA/PEG was the same as that for PLLA/5PEG /tBC8. The T g for these two samples were both much lower than that of PLLA, furthermore, and the crystallization temperature range of these two sample both were higher than that of PLLA. Therefore, the ΔE* of these two samples should be lower than that of PLLA. Interestingly, the ΔE of PLLA/PEG was lower than that of PLLA, but on the contrary, the ΔE of PLLA/5PEG/tBC8 was 210.2 kJ/ mol, which was much higher than that of PLLA. That is to say, the increased ΔE can not only be accounted for by the rise of ΔE* caused by tBC8.
Based on the previous study, both nucleation and crystal growth are related to T c , the increment of crystallization temperature would be hindered and enhance the nucleation and crystal growth process, respectively26,27. In other words, along with the increment of T c , crystallization of PLLA occurs at higher temperature, then ΔF would be increased. In this case, tBC8 acting as nuclei accelerated the crystallization rate of PLLA, which made the crystallization occur at a high temperature during the cooling process. At the cooling rate of 2.5°C min−1, PLLA/5PEG/tBC8 started crystallizing at 140.9°C, much higher than that of PLLA and PLLA/PEG. Additionally, ΔF increases with the crystallization temperature 27 . Thus, the increment of ΔE could be attributed to the increasing crystallization temperature of PLLA. In other words, if PLLA could crystallize at the same temperature range as PLLA/ tBC8, the ΔF of PLLA must be higher than that of PLLA/tBC8.
Therefore, we could draw a conclusion that the increment of ΔE for PLLA/tBC8 compared to PLLA was the result of the increased temperature range of crystallization and the lowered mobility of the chain segments. Then crystallization temperature range must be taken into consideration when ΔE is calculated by the Kissinger equation.
Conclusions
The crystallization activation energy, ΔE, of PLLA nucleated with nucleating agent tBC8 was calculated. The results showed that the ΔE of nucleated PLLA was 165.97 kJ mol−1, higher that of pure PLLA. The mechanism of increase of ΔE for PLLA with the addition of tBC8 was investigated. On one hand, the presence of tBC8 restricted the chain mobility. PEG was introduced to the system to eliminate the rise of ΔE* caused by the nucleating agent tBC8. Then it was concluded that the addition of nucleating agent increased the crystallization temperature range of PLLA, and that as a result, the ΔF increased. Therefore, we drew a conclusion that the increment of ΔE was the result of both reduction of chain mobility and increase of crystallization temperature when using the nucleating agent. Crystallization temperature range is another aspect that must be taken into consideration when ΔE is analyzed.
Footnotes
Acknowledgments
The authors thank the National Natural Science Funds of China (21306047), the Fundamental Research Funds for the central universities of China for financial support.