Abstract
The objective of the present study was to investigate the effect of coating of different concentrations and pH of aqueous casein suspension on thermo-oxidative properties, flame retardant behavior as well as physiological comfort of cotton fabrics. The flame retardant behavior was found to increase with increase in concentration of casein suspension in both alkaline and acidic pH conditions. The burning rate of 2.1 mm/s and 2.4 mm/s was measured for the samples of 150 g/L casein suspensions in acidic and alkaline pH, respectively. Moreover, better thermo-oxidative properties were obtained for the samples of casein suspension prepared in acidic pH than alkaline pH. The 150 g/L casein produced 19% and 28% residue, respectively for alkaline and acidic suspensions at 600℃. Furthermore, the phenomenon of intumescence was largely observed in case of samples coated with acidic casein suspension due to easier release of ammonia from protonated casein. These samples further exhibited relatively better durability against water washing and UV ageing as compared to those treated with alkaline casein suspension, however delivered poor physiological comfort properties due to blockage of fabric porosity by higher viscosity of casein in acidic pH.
Keywords
Introduction
Cellulosic fiber is one of the most important biopolymers in the world due to its availability in large quantities, good mechanical properties, biodegradability, and hydrophilicity [1,2]. However, for further applications in transportation, automotives, protective garments, military, furniture upholstery, bed linen, and nightwear, it is necessary to improve the thermal degradation, ignition, and burning behaviors of cellulosic materials [3,4]. The flame-retardant treatments of halogen, nitrogen, and organic-phosphorus compounds are generally applied on cellulosic materials for the prevention of fire and protection of human life [5,6]. These compounds act as flame retardants because they influence the pyrolysis reaction, prevent the formation of levoglucosan and flammable volatiles, and increase the formation of char [7,8]. Nowadays, the search for environment-friendly flame retardants that ensure fire performances comparable to the conventional flame retardant compounds have gained significant importance [9–11]. This is because of persistent, bioaccumulative, and environmentally toxic nature of some molecules employed in the formulation of conventional flame retardants [12].
Besides environment friendly behavior, flame retardant finishes are also required to maintain the acceptable physiological comfort and mechanical properties of textiles. This is important because the personnel involved in risk of fire hazards are expected to put on fire resistant clothing for longer time. In hot and humid conditions, trapped moisture heats up; while in cold conditions, trapped moisture results in temperature drop and causes hypothermia [13]. The temperature rise and higher body sweat can further cause discomfort, heat stress and skin damage for the wearer [14]. Therefore, the additional properties such as air permeability, water vapor permeability, thermal conductivity and surface texture are important for fire resistant textiles.
In recent years, intumescent systems are considered as the most performing solution available to withstand the fire threat [15,16]. The term intumescence refers to the substances which are able to grow and increase in volume against the heat. Usually, an intumescent material consists of three main components: an acid source, a carbon source and a blowing agent [17]. However, for maximum effect of intumescent formulation as flame retardant, the three components must react in proper time/temperature conditions. To overcome these limitations of intumescents, search for alternative environmentally-friendly intumescent materials containing all three components altogether (i.e. acid source, carbon source, and blowing agent) became important. In this context, the effectiveness of different biomacromolecules (whey proteins, caseins, hydrophobins, and deoxyribonucleic acid) has been assessed for textiles recently [18,19]. However, the effect of pH of casein suspension on intumescence phenomenon has never been discussed before. The present work deals with the effect of coating different concentration and pH of aqueous casein suspensions on thermo-oxidative properties, flame retardant behavior as well as physiological comfort of cotton fabrics. To the best of the authors’ knowledge, this study presented new findings on occurrence of more intumescence from protonation of casein under acidic pH conditions. Furthermore, their physiological comfort and durability properties are also compared for the first time. In this way, the present research could be very useful for valorization of dairy industry by utilization of casein wastes in applications of semi-durable but comfortable textile flame retardants.
Experimental methods
Materials
The plain woven 100% cotton bleached fabric having 145 g/m2 aerial density was selected for the study. Bovine milk casein (technical grade) was received from Sigma–Aldrich, Czech Republic with approximate composition (g/L) of: α–s1 12–15, α–s2 3–4, β 9–11 and κ 2–4. Sodium hydroxide and hydrochloric acid of analytical grade were obtained from Lach-Ner, Czech Republic.
Preparation of casein suspension
The aqueous suspensions of casein under alkaline and acidic pH conditions were prepared at three different concentrations (i.e. 50 g/L, 100 g/L, and 150 g/L). At first, casein powder was added into distilled water and pH was adjusted at 3 and 9 using 1M HCl and 1M NaOH, respectively. Then, the suspensions were heated at 80℃ in thermostatic bath and later cooled to 30℃ before applying on cotton fabric samples.
Application of casein onto cotton fabric
The prepared casein suspensions were applied on cotton fabric in a climatic chamber (30℃ and 30% R.H.). The casein suspensions were spread on the surface of cotton fabrics with a spatula, while the excess of the suspensions were removed gently by pressing with rotary drum. Then, the samples were dried at 100℃ in lab scale hot air dryer (Mathis AG, Switzerland). The uptake (add on percentage) of casein was calculated using the equation (1)
Characterization of viscosity of casein suspension
The viscosity of the casein suspensions was measured using rotational type Modular Compact Rheometer (Anton Paar, Austria) equipped with parallel plate system at room temperature. Viscosity curves were obtained by varying the shear rates from 0 to 250 s−1 at gap size of 1000 µm and the strain of 2%.
Characterization of surface structure of treated fabrics
Fourier-transform infrared (FTIR) analysis
The chemical structure of untreated and casein treated cotton fabric samples were assessed by attenuated total reflectance spectroscopy. It was performed on a FTIR spectrometer Perkin Elmer in the spectra range 4000–400 cm−1 using Nicolet iZ10 reflection ATR technique on an adapter with a crystal of ZnSe.
Scanning electron microscope (SEM) analysis
The surface morphology of untreated and casein treated cotton fabric samples were examined by an SEM (TS5130-Tescan SEM) at an accelerating voltage of 25 kV. The surface of samples was coated with conductive layer by sputtering before SEM analysis.
Characterization of thermo-oxidative stability
The thermo-oxidative stability of untreated and casein treated fabric samples were assessed by thermo-gravimetric analysis (TGA). The samples were heated in air from 30 to 600℃ at a heating rate of 10℃/min using TGA/SDTA 851 METLER TOLEDO analyzer.
Characterization of flame retardancy
In order to describe the realistic fire scenario, untreated and casein treated cotton fabrics were tested for the ignitability and the flame propagation behavior in horizontal configuration using Atlas HMV set up. The fabrics samples were conditioned in 65% relative humidity and 25℃ atmospheres for 24h before the testing. The 25 mm methane flame was applied for 3 s on the edges of specimen having width 50 mm and length 100 mm. These tests were repeated three times on each sample. The total burning time (s), burning rate (mm/s) and final residue (%) were measured. Furthermore, the surface morphology of residue obtained from burnt samples was examined on SEM (TS5130-Tescan SEM) at an accelerating voltage of 20 kV.
Later, an additional method of flame resistance evaluation using image analysis was employed to detect the small differences in flame propagation. The burning was captured under high speed video camera. The video file was composed from frames of size 1080 × 1920 × 3 pixels in RGB 8-bith depth in 25 frame rate. The MATLAB VideoReader function was used to extract the images from video file. The images were further modified using basic image processing operations such as conversion to gray level image; subtraction of non-burned sample for suppression of sample texture; global thresholding (threshold = 0.3) for conversion to binary image; morphological opening for removing small connected objects and closing for filling the holes in burned area. Finally, the length of burned region and burned area was estimated for every 1 s interval of burning time.
Characterization of durability properties
Washing durability
The washing durability was examined by vigorously stirring the casein treated fabrics in tap water at 30℃ with a stirring speed of 600 r/min. The washing was conducted for 1–5 cycles of 20 min each. After washing, all samples were dried and conditioned in a standard atmosphere (65% humidity; 25℃) for 24 h before testing. Further, the flame retardant properties were verified on Atlas HMV set up by recording the burn time and char residue for every washing cycles.
Ageing durability
The ageing durability was examined according to ASTM D 4587-91 standards with few modifications on Xenotest-type weatherometer under the xenon arc lamp with 500 W/m K intensity. The test was performed for 1–5 ageing cycles based on 10 min water spray, drying and 10 h exposure to UV radiation. Further, the flame retardant properties were verified on Atlas HMV set up by recording the burn time and char residue for every ageing cycles.
Characterization of physiological comfort and mechanical properties
Prior to testing of physiological comfort properties, the samples were conditioned in 65% relative humidity and 25℃ atmospheres for 24 h.
Air permeability
The Textest FX 3300 instrument was used to measure the air permeability of casein treated cotton fabrics according to standard ASTM D2986. The air flow passing through the fabric at 200 Pa pressure was measured.
Water vapor permeability
The Permetest device (Sensora Instruments) was used to measure the water vapor permeability (%) according to standard ISO 11092 [20]. This instrument is based on skin model, which simulates dry and wet human skin surface in terms of its moisture, water vapor, and evaporative heat permeation. The heat required for the water to evaporate from porous membrane, with and without a fabric covering, is measured.
Thermal conductivity
Alambeta measuring device (Sensora Instruments) was used to measure the thermal conductivity of fabric samples having diameter of 12 cm [21]. It operates on the principle of heat convection emitted by hot upper plate in one direction through the examined sample to the cold bottom plate adjoined to the sample. The thermal conductivity was calculated using the equation (2)
Stiffness
The TH-7 instrument was used to measure the bending force of samples and then stiffness was calculated [22]. The TH-7 instrument is similar to standard Kawabata KES-FB 2 device used for testing of low stress mechanical properties.
Mechanical properties
The TIRA test 2300 Labor Tech machine was used to measure the tensile properties of casein treated fabrics (15 × 5 cm sample size) as per ISO 1924-2 standard. The tearing strength of fabrics was measured using Elmendorf tester under impact loading as per ASTM D 1424-96 standard.
Results and discussion
Effect of pH on viscosity of casein suspension
Figure 1(a) shows the shear rate vs. viscosity for the alkaline and acidic casein suspensions prepared at different casein concentrations. As the shear rates increased, all samples showed an initial decrease on the viscosity and then formed a horizontal plateau. The viscosity curves showed the shear thinning behavior from 0 to 50 s−1 shear rates for almost all samples, which suggested the disintegration of casein into smaller particles [23,24]. The only exception was 150 g/L acidic casein suspension which showed extended shear thinning behavior till 150 s−1 shear rate. From Figure 1(b), the viscosity was found to increase with increase in casein concentration under both pH conditions. However, the acidic casein suspensions exhibited significant increase in viscosity than the alkaline casein suspensions across all range of casein concentrations. This behavior can be attributed to the different structural organization of casein macromolecules under acidic and alkaline pH conditions [25,26].
(a) Viscosity of casein suspension in alkaline and acidic conditions. (b) Images of casein suspension in alkaline and acidic conditions.
Surface structure of casein treated cotton fabrics
The effect of different concentration and pH of casein suspension on casein add-on of cotton fabrics is shown in Figure 2(a). The add-on was found to increase in linear pattern with increase in casein concentration due to maximum wetting and binding of individual fibers. Further, the suspension of casein prepared under acidic pH delivered higher add-on of casein than that of alkaline pH, which can be explained from observed higher viscosity of casein suspension under acidic pH (see Figure 1). Another reason for the higher add-on of casein under acidic pH is static attraction, compared to the repulsion between cotton fiber and casein under alkaline condition (see section “Mechanism of casein behavior under different pH” and Figure 9).
Surface structure of casein treated cotton fabrics. (a) Casein add-on, (b) FTIR analysis.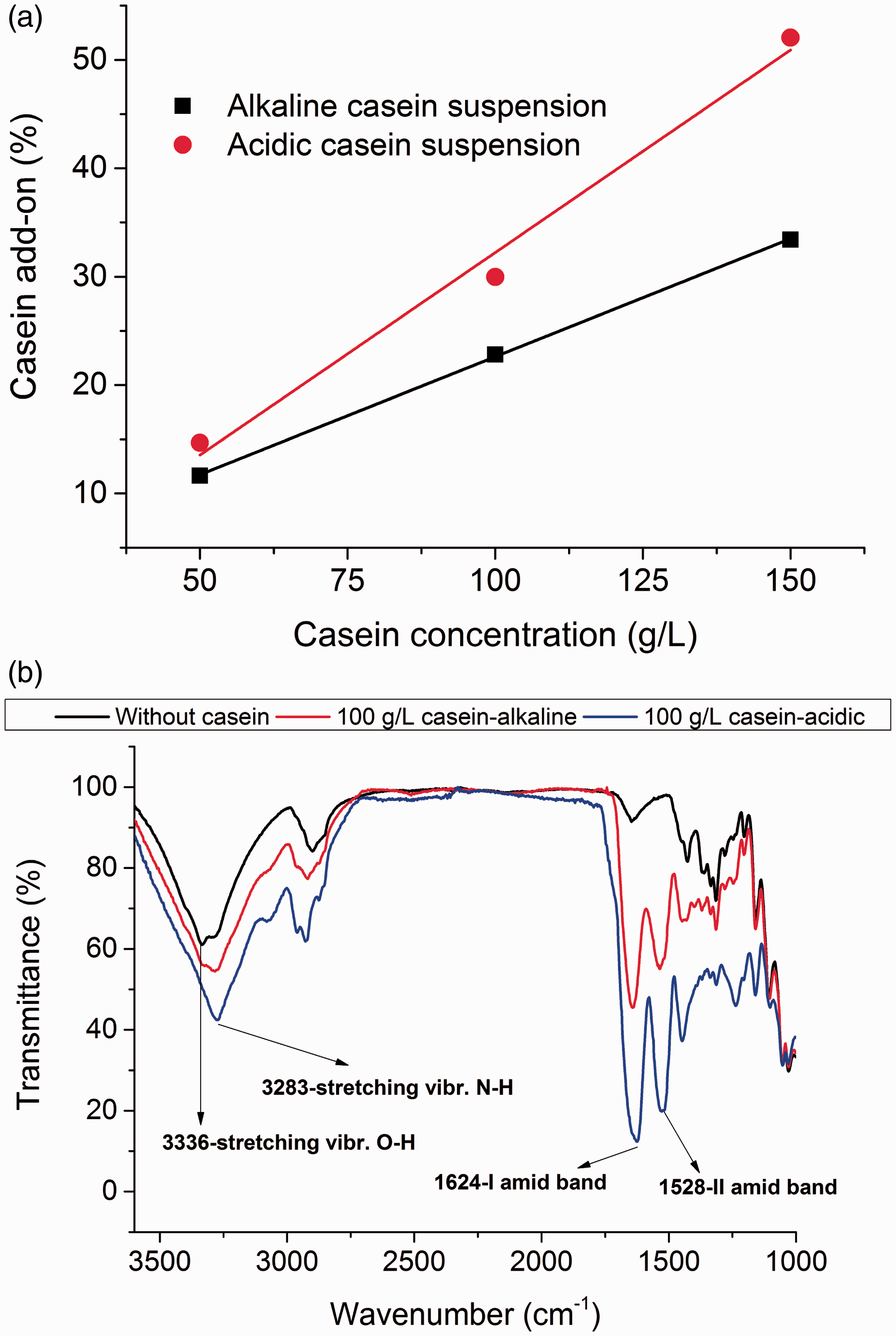
Later, FTIR analysis was carried out to confirm the presence of casein protein on the cotton fabrics. Figure 2(b) shows the FTIR spectra of cotton treated with 100 g/L casein suspension under both alkaline and acidic pH. For untreated cotton fabric, typical vibration modes of cellulose were detected e.g. (–OH) at ca. 3300, (–CH2) at 2900, d(OH) at 1640, d(CH2) at 1425, d(CH) at 1370, d(OH) at 1310, etc. However, additional two peaks centered at 1624 and 1528 cm−1 for respective vibrations of amide I and amide II groups were observed for casein treated cotton fabrics. Further, the greater amount of casein deposition for the acidic suspension of casein can be confirmed based on their higher peak in FTIR spectra.
SEM analysis
Figure 3(a) to (f) shows typical SEM micrographs of casein treated cotton fabrics under both pH conditions. The absence of cracks indicated better compatibility between casein and cotton fibers for both suspensions. The coating of low concentration of alkaline casein suspension exhibited distinct individual cotton fibers than the coating of acidic casein suspension (Figure 3(a) and (b)). However, the grouping of individual cotton fibers increased with increase in casein concentration (Figure 3(c) to (f)). Therefore, possible stiffening of fabric structure can be expected due to blocking of pores with increase in casein concentration. Moreover, the acidic casein suspension was found to produce continuous, homogeneous, and coherent film of casein at lower concentration due to their increased viscosity than alkaline casein suspension. This indicated higher chances of possible deterioration in wearing comfort of cotton fabrics after treatment with acidic casein suspension than alkaline suspension.
Scanning electron microscope images of casein treated cotton fabric. (a) 50 g/L alkaline casein suspension, (b) 50 g/L acidic casein suspension, (c) 100 g/L alkaline casein suspension, (d) 100 g/L acidic casein suspension, (e) 150 g/L alkaline casein suspension, (f) 150 g/L acidic casein suspension.
Thermo-oxidative stability of casein treated cotton fabrics
Thermo-oxidative characteristics of casein treated cotton fabrics.


Thermo-oxidative stability of casein treated fabrics. (a) Thermo-gravimetric analysis for alkaline casein suspension, (b) derivative thermo-gravimetric for alkaline casein suspension, (c) thermo-gravimetric analysis for acidic casein suspension, (d) derivative thermo-gravimetric for acidic casein suspension.
Flame retardancy of casein treated cotton fabric
Burn characteristics of casein treated cotton fabrics.
The spread of flame with respect to time is important property for enhancement in margin of safety under the fire. This can give the wearer more time to take off the clothing or extinguish the fire for reducing the degree and extent of burn injuries. This work employed the image analysis tools for estimation of flame propagation over small intervals of time on casein treated samples. The effect of coating of different concentration and pH of casein suspension on burn length and burn area can be seen from Figure 5. The fabrics samples treated with acidic casein suspensions (except 50 g/L casein) revealed lesser burn length and burn area as compared to alkaline casein suspensions. The acidic suspension of 100 g/L casein presented least burning behavior among all samples, with 15% more burn length and 21% more burn area over same concentration of alkaline casein suspension after 40 s. The slower burning of samples can be explained due to release of more phosphoric acid and ammonia from protonated casein in acidic pH conditions (see section “Mechanism of casein behavior under different pH”). This can be further justified from Figure 6 which showed the photographs of carbonized areas for all samples after 10, 20, 30, and 40 s of burning time. Surprisingly, more intumescence with release of oily substances was observed while burning the cotton fabrics treated with acidic casein suspension. These observations have never been reported in previous studies; however, mentioned first time in this work.
Estimation of flame propagation by image analysis. (a) Burn length for alkaline casein, (b) burn length for acidic casein, (c) burn area for alkaline casein, (d) burn area for acidic casein. Images of burnt sample at different time intervals. (a) Alkaline casein suspension, (b) acidic casein suspension.

The morphology of burnt residue of 50, 100, and 150 g/L casein treated fabrics for both pH conditions can be observed from SEM images in Figure 7. The residue for alkaline casein suspension showed the formation of globular micrometric structures at localized spaces (Figure 7(a), (c) and (e)). On the other hand, the residue for acidic casein suspension showed the formation of more expanded globular micrometric structures (i.e. global intumescence) at enlarged spaces (Figure 7(b), (d) and (f)). The globular micrometric structures are phosphorus-rich bubbles that blow up during the combustion [29]. The presence of more bubbles in case of acidic casein suspension can be ascribed to the release of more ammonia due to protonation of casein under acidic pH conditions (see section “Mechanism of casein behavior under different pH”).
SEM images of burnt casein treated cotton fabrics. (a) 50 g/L alkaline casein suspension, (b) 50 g/L acidic casein suspension, (c) 100 g/L alkaline casein suspension, (d) 100 g/L acidic casein suspension, (e) 150 g/L alkaline casein suspension.
Durability of casein treated cotton fabrics
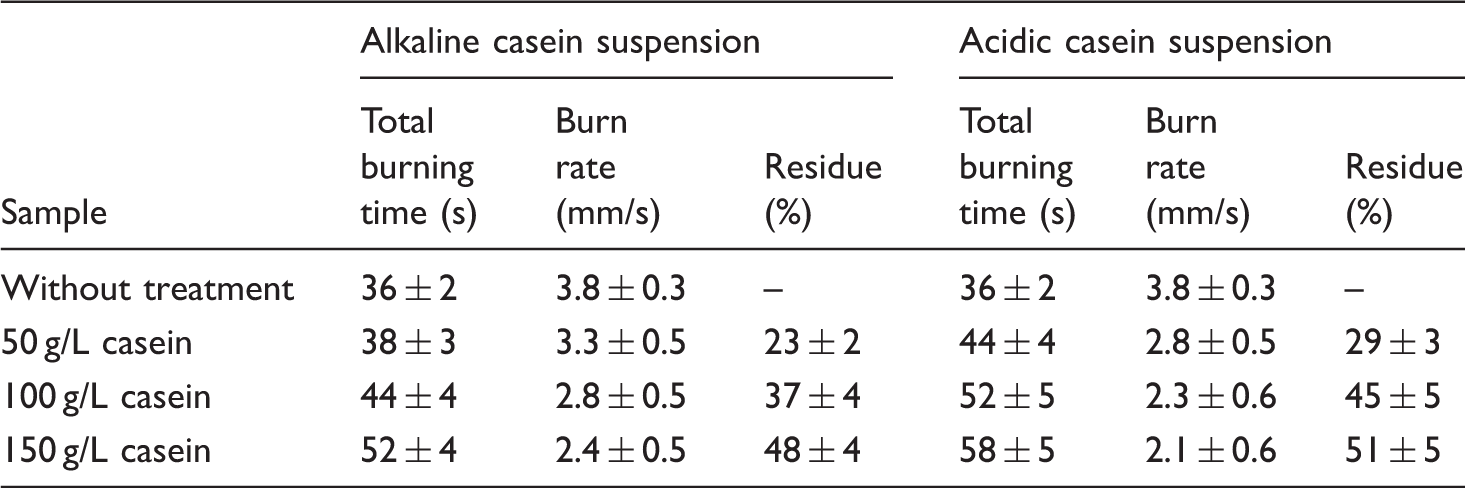
Due to water soluble nature of casein and their tendency to undergo physical-chemical modifications after interaction with light and oxygen, the casein treated textiles cannot be expected for long term durability against washing and UV ageing. However, the effect of casein suspension pH has never been studied before from durability aspects. This work presented the comparative performance of alkaline and acidic 150 g/L casein suspension treated textiles against washing and UV ageing durability.
Washing durability
Durability of 150 g/L casein treated cotton fabrics.


Microstructure of residue from burnt 150 g/L casein treated sample. (a) 5 cycles of water washing on alkaline casein, (b) 5 cycles of water washing on acidic casein, (c) 5 cycles of UV ageing on alkaline casein, (d) 5 cycles of UV ageing on acidic casein.

Mechanism of casein behavior under different pH.
Ageing durability
The process of ageing is defined as the action of atmospheric elements in altering the color, texture, composition or form of exposed objects, ultimately leading to disintegration or failure to perform some functions [33,34]. The well-known elements of weather are radiation, moisture, thermal conditions, and gases. Table 3 shows the results of total burning time (s) and residue (%) for alkaline and acidic 150 g/L casein suspension treated samples after exposure to humidity and ultraviolet rays. As compared to acidic casein suspension, the alkaline casein suspension treated cotton textiles showed significant loss of flame retardant properties (i.e. burning time and residue) with increase in time of ageing. This behavior can be attributed to formation of disordered structure due to cross-linking, hydrolysis, oxidation of the side chains, and migration of casein when exposed to ageing conditions. Furthermore, the phenomenon of local intumescences was not observed for alkaline casein suspension samples after prolonged exposure to UV rays (see Figure 8(c) and (d)). This was attributed to reduced ability of casein macromolecules to release phosphoric acid and ammonia in alkaline suspension. Despite better performance of acidic casein suspension, further research is required to improve their UV ageing properties. The improvement of ageing durability for hydrophilic flame retardants has been studied recently [35]. The application of TiO2 nanoparticles can effectively improve the anti-ageing property of flame retardants due to their improved photo stability, decreased diffusion of oxygen and moisture, and reduced probability of attack at the molecular chain. Therefore, the combined application of casein and TiO2 nanoparticles for improvement of ageing properties could be interesting for the future research.
Mechanism of casein behavior under different pH
The casein macromolecules can be considered as poly amino acids bearing several phosphate groups in their micellae structure [36]. Each amino acid has characteristic pH at which it carries both positive and negative charges and exists as zwitter ion. Therefore, the casein behaves as an amphoteric and exists in solution as shown in Figure 9. The durability of casein treatments can be explained from presence of their surface charge. In alkaline conditions, the casein macromolecules have net negative charge which results in less durable attachment of casein with cotton fiber surface by weaker physical bonds. On the other hand, the casein macromolecules carry net positive charge when suspended in acidic conditions, which results in relatively strong attachment of casein with cotton fiber surface and thus more durability. The occurrence of more intumescence in acidic casein suspension can be explained from easier release of ammonia due to their protonation behavior. The carboxyl groups are dissociated while the amino groups are easily protonated in casein structure under acidic conditions.
Physiological comfort and mechanical properties of casein treated cotton fabrics
The air permeability, thermal conductivity, water vapor permeability, and stiffness testing was carried out to know the physiological comfort properties of casein treated cotton fabrics.
Air permeability
Air permeability is the measure of air flow passed through a given area of a fabric and decides the physiological comfort to the great extent. Figure 10(a) shows the effect of coating of different concentration and pH of casein suspension on air permeability of cotton fabrics approximated by second order polynomial regression analysis. It can be seen that there is significant drop in air permeability for acidic suspension of casein than the alkaline suspension. The alkaline casein suspension delivered acceptable values of air permeability till 100 g/L of casein concentration; however, the air permeability of acidic casein suspension was acceptable till 70 g/L of casein concentration. The drop in air permeability of approx. 52% and 99% was found over untreated fabrics for 100 g/L concentration of casein suspension in alkaline and acidic conditions, respectively. This behavior can be explained from higher add-on of acidic casein suspension, which produced thicker casein film on the surface of cotton fabric as compared to alkaline casein suspension. The reduced air flow through the fabrics was attributed to closing or decreasing the size of pores by formation of such thick film.
Physiological comfort and mechanical properties of casein treated cotton fabrics. (a) Air permeability, (b) water vapor permeability, (c) thermal conductivity, (d) stiffness, (e) tensile strength, (f) tear strength.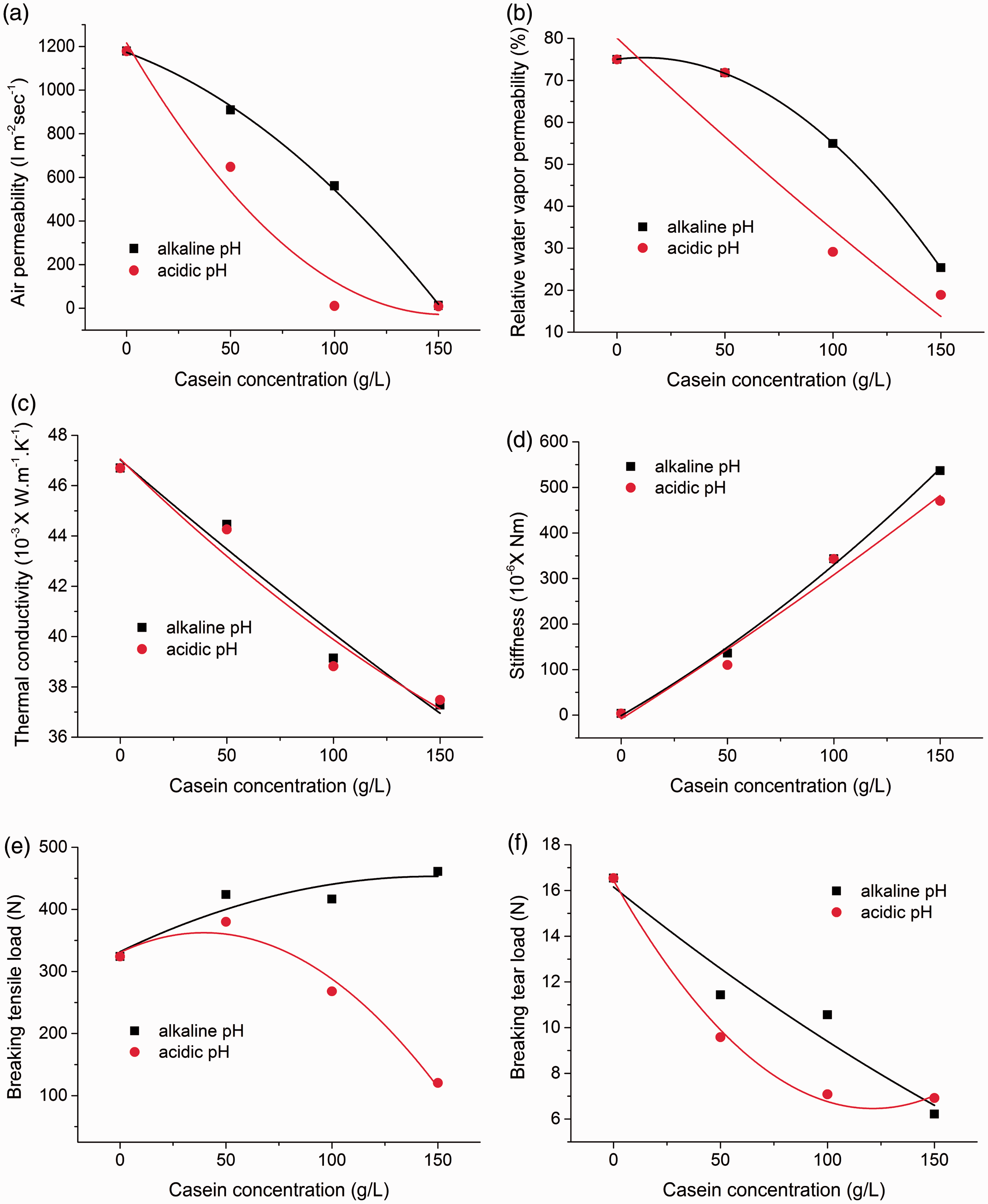
Water vapor permeability
The water vapor permeability indicates the amount of resistance against the transport of water through the given area of fabric. Figure 10(b) shows the effect of coating of different concentration and pH of casein suspension on water vapor permeability of the cotton fabric approximated by second order polynomial regression analysis. Likewise air permeability, the moisture vapor transport properties of cotton fabrics were least affected when coated with alkaline casein suspension than the acidic casein suspension. The drop in moisture vapor transport of approx. 27% and 75% was found over untreated fabric for 100 g/L concentration of casein suspension in alkaline and acidic conditions, respectively.
Thermal conductivity
It is a measure of the rate at which heat is transferred through unit area of the fabric across unit thickness under a specified temperature gradient. Figure 10(c) shows the effect of coating of different concentration and pH of casein suspension on thermal conductivity of the cotton fabric approximated by second order polynomial regression analysis. The similar linear trend of reduction in thermal conductivity with increase in casein concentration can be observed for both alkaline and acidic suspensions of casein. This behavior can be attributed to the reduction in porosity of cotton fabric surface with increase in casein concentration.
Stiffness
Stiffness is a tendency of the fabric to keep standing without support. It is a special property of the fabric for desirable draping and also influences the sensorial comfort of clothing. It can be evaluated from bending properties of the fabric using bending length and flexural rigidity. Figure 10(d) showed the effect of coating of different concentration and pH of casein suspension on the bending properties of cotton fabrics. For both alkaline and acidic casein suspensions, the stiffness was found to increase in linear trend with increase in concentration of casein. The increased inter-fiber/yarn friction and abrasion at fiber/yarn cross-over points can be the cause of such behavior due to deposition of casein over cotton structure [37]. No loss in draping behavior and satisfactory sensorial comfort behavior of cotton fabrics can be expected below 100 g/L concentration of casein suspension.
Mechanical properties
Figure 10(e) and (f) shows the effect of coating of different concentration and pH of casein suspension on tensile strength and tear strength for casein treated cotton fabrics, respectively. With increase in casein concentration, the tensile strength was increased for alkaline casein suspension. However, it was reduced for acidic casein suspension. The increased tensile strength for alkaline casein suspension can be explained from contribution of load bearing capacity of attached thin casein layer. Whereas, the reduced tensile strength for acidic casein suspension can be attributed to possible hydrolysis of cotton fibers. Furthermore, the reduction in tear strength was found for both alkaline and acidic casein suspensions with increase in casein concentration. The increase in rigidity, grouping of cotton fibers, and possible hydrolysis of cotton structure could be the cause for reduced tear strength values [38]. For 100 g/L casein, the reduction in mechanical properties were not significant (p < 0.05) for alkaline casein suspension but significant for acidic casein suspension.
Conclusions
The study presented the application of casein protein for environment friendly flame retardant finish of cotton fabrics. The effect of coating of different concentration and pH of aqueous casein suspension on flame retardancy, physiological comfort, and mechanical properties were investigated. When the casein suspension was prepared under acidic pH conditions, its viscosity was found higher than that of alkaline casein suspension. This resulted in higher casein add-on after coating of acidic casein suspension. From TGA, the shift of Tonset10% values to lower temperature indicated stronger sensitization of the cellulose decomposition by treatment of casein macromolecules under acidic pH. The samples coated with acidic casein suspension produced higher amount of residue than alkaline casein suspension and it increased with increase in casein concentration. From image analysis results, the acidic suspension of 100 g/L casein presented least burning behavior among all samples, with 15% more burn length and 21% more burn area of same concentration of alkaline casein suspension after 40 s. From SEM microstructures, the residue of alkaline casein suspension showed the formation of globular micrometric structures at localized spaces only. On the other hand, the uniform formation of more expanded globular micrometric structures (i.e. global intumescence) with release of oily substances was observed for residue of acidic casein suspension. The presence of more bubbles in case of acidic casein suspension can be ascribed to the release of more ammonia due to protonation of casein under acidic pH conditions. Furthermore, the treatment of casein under acidic pH conditions suggested their relatively better durability against water washing and UV ageing as compared to alkaline pH. However, due to blockage of fabric porosity by higher viscosity of casein in acidic pH, their physiological comfort properties deteriorated significantly over alkaline casein suspension. Therefore, the consideration to both flame retardancy and physiological comfort can be given by selecting moderate concentration of casein suspension in both alkaline and acidic conditions. In particular, 70–100 g/L concentration of alkaline casein suspension and lower concentration of acidic casein suspension between 50 and 70 g/L can provide adequate flame retardant performance with acceptable physiological comfort and mechanical properties. In this way, the present work of casein treatments on cotton textiles can be employed to prevent second-degree burn injuries from low intensity heat flux accidents (such as cigarette fires or car/motor bike racing). The coated textiles could be suitable for short term applications in clothing, home textiles, technical textiles, etc.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research project of Student grant competition of Technical University of Liberec no. 21202 granted by Ministry of Education, Youth and Sports of Czech Republic.


