Abstract
Ferrocenylamine complexes have found increasing applications in the fields of catalysis in various organic reactions, industry, medical treatments and enzyme–activity determinations. Therefore, information related to the thermal and thermo-oxidative stability of these compounds is important for such applications; however, this information is currently limited. Twenty previously prepared palladium and platinum ferrocenylamine complexes with systematic structural variations are examined for their thermal (under nitrogen) and thermo-oxidation stability (under atmospheric air) using thermogravimetry (TG), differential thermal analysis (DTG), and differential scanning calorimetry (DSC) techniques. Degradation products are identified by comparing thermogravimetric analysis and theoretical calculations. Structure–stability studies are also discussed. The results show that all the compounds have high thermal and thermo-oxidative stabilities of up to 265 and 173 °C, respectively. Electron–donating substituents enhance the thermal and thermo-oxidative stabilities of the palladium complexes (t-Bu, selenide electrophiles and dielectrophiles), while those with destabilizing effects are aromatic substituents (Ph and tolyl). Most platinum ferrocenylamine sulfides and selenides show higher thermal and thermo-oxidative stabilities than their corresponding palladium analogs. All the prepared compounds show high thermal and thermo-oxidative stability which reinforces their catalytic and industrial applications. However, their thermal stability is higher than their thermo-oxidative stability.
Keywords

Introduction
Ferrocenylamines possess rich chemistry and a wide range of applications because of their electron-rich aromatic nature, the nucleophilicity of the amino group, their high electrochemical potential, and their unique stereostructure as sandwich-like compounds.1,2 Many ferrocenylamine derivatives have been synthesized and frequently incorporate metals, for example, palladium,3–5 platinum, 6 copper, 7 bismuth, 8 silver, 9 gold,9,10 and rhodium. 11 Their complexes are widely used as catalysts in various organic reactions for example, selective hydrogenation,12–15 cross–coupling reactions,12,16,17 and for the synthesis of ferrocene-fused heterocycles. 18 The special structure of chiral ferrocenylamines is useful in stereoselectivity, for example, asymmetric hydrogenation, 19 hydrosilylation,3,5 aldol reactions, 20 cycloaddition, 10 allylic alkylation, 7 C–H arylation, 4 and double C–H functionalization, 21 as well as for electrochemical enantiodifferentiation. 22 Industrial applications involve production of redox–active anion sensors, 23 supercapacitor electrodes, 24 corrosion inhibitors, 25 and electro- and photo-responsive materials. 26
In addition, the biological impact of ferrocenylamine derivatives has been explored and they were found to exhibit anticancer,1,27–29 antimalarial, 30 antifungal, 31 antibacterial, 29 antiplasmodial, and antiproliferative activities, 32 in addition to being carbonic anhydrase inhibitors 33 and being able to electrochemically determine β-galactosidase 34 and alkaline phosphatase activities. 35
In this study, 20 ferrocenylamine complexes with systematic structural variations have been prepared and examined for their thermal (under nitrogen) and thermo-oxidation (under atmospheric air) stability. The work sheds some light on their suitability for use under various conditions which might be required in some industries, for example, the petrochemical and pharmaceutical industries. The structural factors examined are the incorporated metal (palladium or platinum), the alkyl substituent (Me, Et, n-Pr, n-Bu, t-Bu and i-Pent) or aryl (Ph and tolyl) group and the type (sulfide or selenide) and number (mono or di) of electrophilic substituents. Degradation products were also identified.
Results and discussion
The thermal and thermo-oxidative stabilities of the palladium and platinum ferrocenylamine catalysts were examined up to 600 °C. The structures and notations of the studied compounds are presented in Figure 1. Structural variations include the metal (Pd and Pt) and the electrophilic substituents (SR and SeR) on the cyclopentadienyl rings (Cp). The electrophilic substitution can also be one (mono) or two (di), one on each Cp ring. The observed fragmentations of each compound along with the calculated and experimental percentages of each fragment molecular weight are given in Table 1 and Table S1 (see the supporting information) for thermal degradation and in Table 2 and Table S2 (see the supporting information) for thermo-oxidative degradation.

Structures of the prepared palladium and platinum ferrocenylamine sulfides and selenides.
Thermal degradation data of the palladium and platinum ferrocenylamine sulfides and selenides.

MW: molecular weight.
Palladium and platinum ferrocenylamine sulfides and selenides.
Frag denotes Fragment.
Di denotes dielectrophilic substituents.
Thermo-oxidation data of the palladium and platinum ferrocenylamine sulfides and selenides.

MW: molecular weight.
Palladium and platinum ferrocenylamine sulfides and selenides.
Frag denotes fragment.
Di denotes dielectrophilic substituents.
Thermal stability
On thermal treatment under nitrogen, all the compounds showed two fragmentation steps, except for PdSC6H4Me (three steps) and PtSeMe (four steps). The TG, DTG and DSC thermograms of PtSeMe are presented in Figure 2. The TG and DTG thermograms show the four degradation steps at 197, 234, 276 and 366 °C. Calculations revealed that the fragments of each step are NMe2, Cp, Me and 2Cl respectively; the DSC thermogram showed that the four thermodegradation steps are endothermic. As in most compounds, the substituted cyclopentadienyl ring degraded before the unsubstituted ring, with the amino group (NMe2) more frequently evolving first. However, evolution of chlorine atoms is observed only in a few of the prepared compounds with temperatures ⩾300 °C (PdSMe).

The TG, DTG and DSC thermograms of PtSeMe.
Figure 3 shows the thermal stability of both palladium and platinum compounds. All the compounds showed high thermal stability ⩾162.5 °C (PdStolyl) where the first fragment peak range was 162.5–265 °C, while the second peak range was 234–405 °C. For the palladium sulfides, the stability increases on increasing the size of the alkyl group up to a t-Bu group, where PdSt-Bu showed the highest thermal stability with degradation peaks at 265 and 405 °C; then, the stability decreases with alkyl size or aromatic substitution. The high stability of PdSt-Bu could be attributed to the high electron–donating ability of the t-Bu group. Mono (PdSeMe) and di (PdDiSeMe) selenides showed higher stabilities than their respective sulfides (PdSMe and PdDiSMe), with thermal stabilities of 174, 198, 166 and 180.5 °C, respectively. The platinum series showed different trends where the highest stability in the series was observed for PtSMe (222 °C); however, the diselenide PtDiSeMe exhibited better stability (214 °C) than its disulfide (PtDiSMe, 189 °C) and monoselenide (PtSeMe, 197 °C) analogs. Platinum methyl selenides (PtSeMe, PtDiSeMe) and sulfides (PtSMe, PtDiSMe) also showed greater stability (197, 214, 222, and 189 °C) than their palladium counterparts (174, 198, 166, and 180.5 °C).
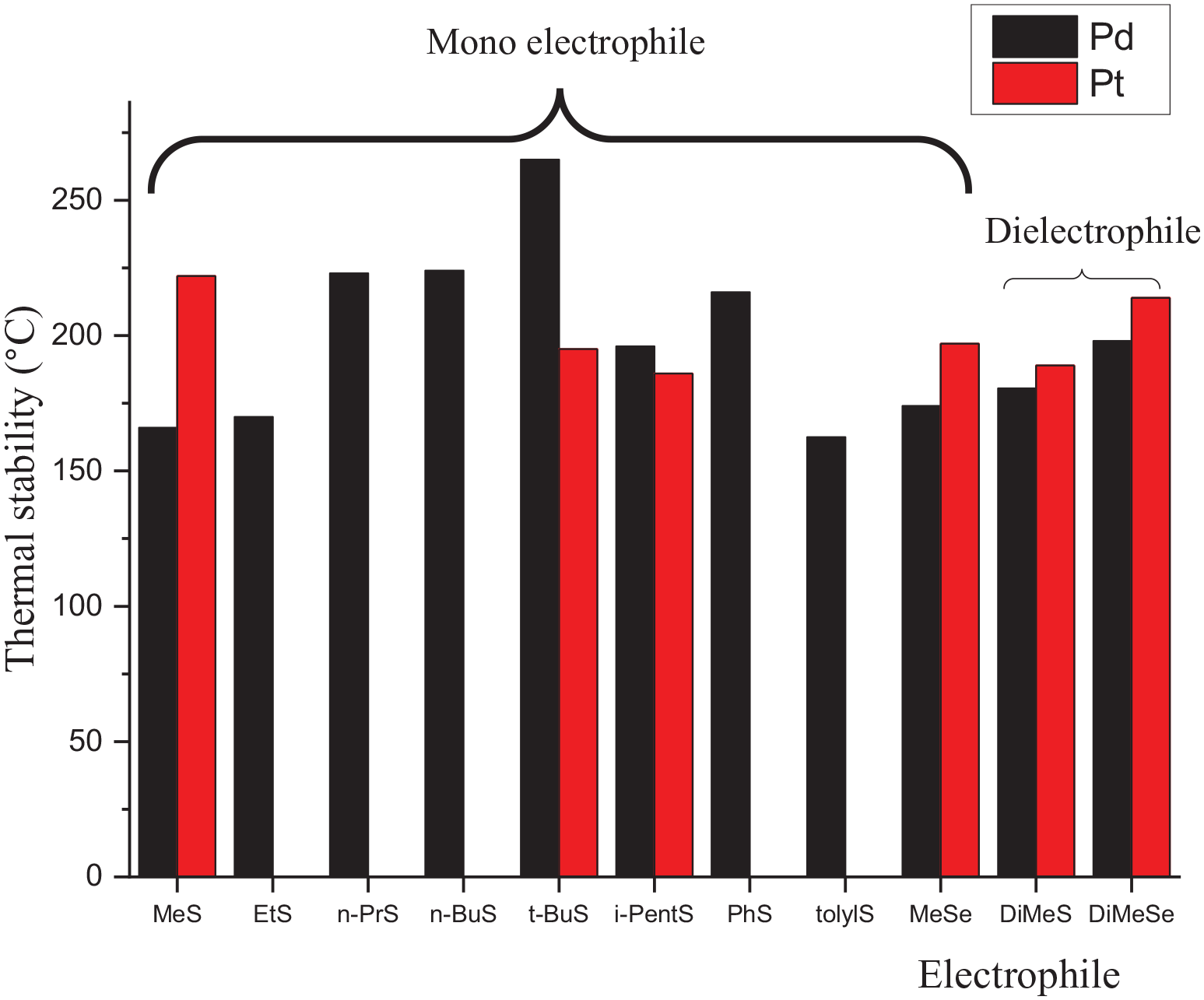
Thermal stabilities of the palladium and platinum ferrocenylamine sulfides and selenides.
Thermo-oxidative stability
The thermo-oxidative stability of the ferrocenylamine catalysts was examined under air conditions and the results are presented in Table 2 and Figure 4. The results indicate that both the palladium and platinum series express less thermo-oxidative stability compared with their thermal stability, where the first and second peaks appeared in the ranges 63 (PtDiStolyl)–173 (PtSMe) and 95 (PtDiStolyl)–419 (PdDiSMe) °C, respectively. In addition, many compounds, especially those with dielectrophilic substitution, degraded in 3–6 steps and several compounds evolved all the organic moieties, for example, PdSt-Bu, PdSi-Pent, PtSt-Bu, PtSi-Pent, PtSeMe, and PdDiSeMe. These examples indicate the more visible of the thermal oxidation than thermal degradation of the organic moiety; however, the inorganic residues remain un-oxidized since their weights remain unchanged.

Thermo-oxidative stabilities of the palladium and platinum ferrocenylamine sulfides and selenides.
On the other hand, many aspects of the thermo-oxidative behaviors are similar to those of the thermal properties. First, the substituted cyclopentadienyl ring usually degraded during the first step, followed by the unsubstitute Cp ring, while chlorine atoms were expelled in only a few compounds during the second or third steps at temperatures ⩾ 340 °C (for PdSMe, as under the nitrogen conditions). Second, as in the thermal degradation, the highest thermo-oxidative stability of the palladium monosubstituted sulfides was observed for PdSt-Bu (146 °C); while the highest for both the palladium and platinum series was PtSMe (173 °C). Third, the diselenide PdDiSeMe exhibited higher stability (165 °C) than its disulfide (PdDiSMe, 143 °C) and monoselenide (PdSeMe, 141 °C) analogs. Fourth, the least stable members of both the palladium (PdSPh, PdStolyl) and platinum (PtDiStolyl) series were those with aromatic substituents (75.0, 130, 63, and 151 °C, respectively). Finally, platinum mono- and disubstituted ferrocenylamine sulfides and selenides showed better thermo-oxidative stabilities than those of the palladium analogs, with the most stable compound being PtSMe.
Mechanistic insights
Little is known about thermal and thermo-oxidation degradation mechanisms as well as the degradation products of ferrocene derivatives. Ferrocene itself is highly stable up to 400 °C 36 and has a mild oxidation potential on losing an electron from the central ferrous ion to form a ferrocenium cation.36,37 The introduction of an electron–windrowing group on the cyclopentadienyl ring (Cp) reduces the thermal stability of ferrocene through destabilization of the π-bond between the iron atom and the Cp rings. 38 This complements our findings that the effective electron–donating groups, t-Bu and selenide (compared to sulfide analogous), provide better thermal stability. In addition, it was found that in primary amines, the thermal stability increases in the order: diamine > alkanolamine > amino acid salt, while steric hindrance around the amine group protects the amines against thermal degradation. 39 Also consistent with the present results, thermal degradation of ferrocene-containing polyamides was found to be a multistep process with the first oxidation step taking place on the carbon vicinal to the nitrogen atom, 40 which supports scission of the dimethylamine group in most of the compounds during the first degradation step.
Conclusion
It can be concluded that ferrocenylamine complexes show high thermal and thermo-oxidative stabilities up to 265 (PdSt-Bu) and 173 °C (PtSMe), respectively, which should allow these compounds to be used for industrial applications. Degradation is a multistep process that takes place in 2–4 steps usually starting with degradation of the substituted Cp ring followed by the unsubstituted Cp ring and then the chlorine atoms. Generally, both the thermal and thermo-oxidative stabilities are enhanced by the electron-donating groups, t-Bu and the selenide electrophile, as well as the presence of dielectrophiles, while they are destabilized by aromatic substituents (Ph and tolyl). Platinum ferrocenylamine sulfides and selenides showed higher thermal and thermo-oxidative stabilities than their palladium analogs.
Experimental
All reagents and starting materials were reagent grade. Solvents were freshly dried before use. All complexes were previously prepared and characterized.12,41 All reactions were performed under anhydrous argon as outlined below. Elemental analysis (CHN) was performed using a Perkin Elmer CHN 2400 analyzer.
Synthesis of the ferrocenylamine sulfide and selenide complexes
The amine (S)-C5H5FeC5H4CHMeNMe2 (1.3 g, 5.1 mmol) was dissolved in 50 mL of dried ether. The solution was cooled to −78 °C and then 4.0 mL (6.4 mmol) of n-BuLi was added dropwise via a syringe with stirring. Stirring was continued for 12 h at room temperature. The appropriate alkyl disulfide or selenide (5.9 mmol) was added dropwise via a syringe at −78 °C. After 24 h of stirring, 20 mL of saturated aqueous NaHCO3 was added and the organic layer was extracted twice with ether. The combined organic layer was dried over anhydrous sodium sulfate and evaporated. The crude product was chromatographed on a short silica gel column eluting with a hexane/CH2Cl2 solvent system.
Dielectrophile ligands were prepared as above but with a second addition of the same amounts of n-BuLi and the alkyl disulfide or selenide in the presence of TMEDA (5.5 mmol) was necessary under similar conditions.
Pd and Pt complexes were prepared by dissolving (PhCN)2PdCl2 or (PhCN)2PtCl2 in warm dry benzene (10 mL) and adding the ligand in a 1:1.1 molar ratio. The reaction mixture was stirred at room temperature for 12 h in the case of Pd complexes and for five days in the case of Pt complexes. The resulting precipitates were filtered, washed with benzene and recrystallized from hexane/CH2Cl2.
Thermal analyses
Thermal analyses, thermogravimetry (TG), differential thermal analysis (DTG), and differential scanning calorimetry (DSC) were performed using a TA 3000 Mettler system. Both dynamic (purged nitrogen gas) and static (atmospheric air) environments were applied. The operating conditions are: start temperature 50 °C, end temperature 600 °C, and heating rate 20 °C/min. For TG measurements, open crucibles were used, while for DSC analysis, sealed pans with lids containing a hole pierced with a thick pin were used. The peak temperature was obtained from DTG analysis. Fragments were assigned based on the fragment percentage of the molecular weight (MW) in the TG analysis; all experimental fragment percentages differ from the calculated values by <3.0% of the MW.
Supplemental Material
sj-docx-1-chl-10.1177_17475198221141979 – Supplemental material for Thermal and thermo-oxidative stability of a series of palladium and platinum ferrocenylamine sulfides and selenides
Supplemental material, sj-docx-1-chl-10.1177_17475198221141979 for Thermal and thermo-oxidative stability of a series of palladium and platinum ferrocenylamine sulfides and selenides by Hussein M Ali and Isra H Ali in Journal of Chemical Research
Supplemental Material
sj-docx-2-chl-10.1177_17475198221141979 – Supplemental material for Thermal and thermo-oxidative stability of a series of palladium and platinum ferrocenylamine sulfides and selenides
Supplemental material, sj-docx-2-chl-10.1177_17475198221141979 for Thermal and thermo-oxidative stability of a series of palladium and platinum ferrocenylamine sulfides and selenides by Hussein M Ali and Isra H Ali in Journal of Chemical Research
Footnotes
Author contributions
Both authors have contributed to all research steps including the design, experimental work, interpreting results, discussion, and writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
