Abstract
Recently, production of nanostructured scaffolds along with the therapeutic properties of aloe vera such as high ability of wound healing and anti-inflammatory properties could attract great attention. In this article, gum tragacanth, poly(ɛ-caprolactone), and aloe vera (5% wt) were electrospun for fabricating poly(ɛ-caprolactone)/aloe vera and poly(ɛ-caprolactone)/gum tragacanth/aloe vera scaffolds. Scanning electron microscope results showed that increasing aloe vera to nanofibers composition, decreased average diameter from 1118 ± 53 to 501 ± 69 nm for poly(ɛ-caprolactone), and 184 ± 34 to 123 ± 22 nm for poly(ɛ-caprolactone)/gum tragacanth mats. Fourier transform infrared spectroscopy and differential scanning calorimetry analyses revealed that aloe vera has been loaded in nanofibers structure properly. By addition aloe vera, the tensile strength and tensile strain of poly(ɛ-caprolactone)/gum tragacanth nanofibers increased from 0.21 to 0.75 and 25 to 32%, respectively. Aloe vera loaded nanofibers exhibited satisfactory degradation and wettability too. Moreover, MTT assay demonstrated more fibroblast cell proliferation for poly(ɛ-caprolactone)/aloe vera and poly(ɛ-caprolactone)/gum tragacanth/aloe vera nanofibers compared to poly(ɛ-caprolactone) and poly(ɛ-caprolactone)/gum tragacanth nanofibers. However, cells had favorable attachment and proliferation on aloe vera loaded nanofibers.
Introduction
Skin is a very intricate organ and hence producing a bioengineered skin model, which replicates the essential physiological characteristics for replacing the injured or damaged parts has been a challenging aim [1]. The most important characteristics of wound dressing is acceleration of wound healing by providing a moist environment and elimination of trauma and infection [2]. Recently, several new wound dressing products were presented to accelerate the wound healing process with simulating of biological, topographical, and physical properties. Among them, nanotechnology-based polymeric biomaterials have gained prominence interest owing to the enhanced porous structure, large surface to volume ratio and reduced side effects on injured parts. Electrospinning is one of the most efficient, versatile, cost-effective, and straightforward methods for fabricating micro- to nano-sized biomimetic nanofibers with high specific surface area, high porosity, high interconnectivity of pores, remarkable mechanical and biological properties, which give them extremely huge capability for applications such as reduction environmental pollutions [3,4], tissue engineering, drug delivery, and wound healing [5]. Produced nanofibers from synthetic polymers have exhibited favorable processability, whereas their low potential in cell attachment, their hydrophobic nature and absence of surface cell recognition sites limits their application in biomedical fields. On the other hand, natural polymers presented adequate biocompatibility, efficient interactions with cells, mimicking the morphological structure of natural ECM as well as high hydrophilicity. But their low mechanical strength and fast degradation rate restricts their utilization in tissue engineering [6]. However to overcome these shortcomings, blending synthetic and natural polymers seems to be a favorable way for exploiting great advantages of both polymers [7]. The same scaffolds with maintaining their original structures in determined time point, helped to release drug and extracts controllable and quicker. Recently, the inclusion of therapeutic medicinal plants in alternative medicine has been increasing in different societies as a way to improve the quality of people’s life with the advantage of being 100% natural [8]. Aloe vera (AV), as the oldest therapeutic herb belonging to the Liliacea family [9], has the ability to promote healing of a variety of skin disorders including infections, burns, dermatologic conditions because of its antimicrobial effect [10], reducing pain [11], enhanced cell proliferation [12], and epithelizing action on damaged skin tissue since ancient times. AV has been composed of amino acids, enzymes, vitamins, polysaccharides (pectins, cellulose, hemicellulose, glucomannan, acemannan, and mannose derivatives), and other low molecular weight substances [13–15]. Use of nanofibrous scaffolds loaded with AV for biomedical applications especially for treatment of wounds after skin damages such as burn and skin ulcers has evoked huge interest. AV incorporated chitosan membrane displayed satisfactory degradation, wettability, higher antibacterial property, mechanical strength, and favorable cell compatibility with primary human dermal fibroblast [16]. Suganya et al. [17] fabricated AV loaded poly(ɛ-caprolactone) (PCL) nanofibers for the treatment of different skin disorders, which they were low cost scaffolds compared with other ones such as PCL/collagen nanofibers and also showed desirable biological and healing properties. Nevertheless, loading AV in the blend nanofiber containing natural and synthetic polymers has not been studied yet.
This article concentrates on the production of nanofibers using PCL/gum tragacanth (GT)/AV and studies their capability for application in wound healing process. PCL is an aliphatic biodegradable and biocompatible polyester with good tensile properties. However, its poor hydrophilicity and slow degradation kinetics limit its application in biomedical field [18]. GT is a natural low cost polysaccharide polymer, which is extensively utilized in Chinese and Iranian folk medicine. GT has showed favorable capability for wound healing in the form of blended nanofibers with PCL or PVA [19–21]. It also exhibited high potency for regeneration of periodontal defect and peripheral nerve damage [22,23]. Curcumin loaded PCL/GT nanofibers exhibited good potency for healing of diabetic wounds in in-vivo experiments [24].
In this study, for the first time, natural biopolymer GT with perfect biological properties is used as a hydrophilic natural carrier for AV. The cooperative association of GT and native AV gel was explored as an approach to create blended membranes with excellent biological properties, which could be useful as active wound dressings. Blending of GT/AV with PCL provides a proper product with improved mechanical and degradation characteristics. This study contains the characterization and analysis of cell growth and proliferation to show the efficiency of these scaffolds for application in skin tissue regeneration.
Experimental procedure
Materials
PCL (Mw 80 kDa) was bought from Sigma-Aldrich. Ribbon type GT with high quality, gathered from the stems of fluccosus species of Astragalus bushes. After drying and purification of this raw gum, fine powder was prepared. AV was purchased from Sigma Aldrich. Human dermal fibroblasts (HDFs) were obtained from American Type Culture Collection. Dulbecco modified Eagle’s medium (DMEM), fetal bovine serum (FBS), penicillin streptomycin solution, and trypsin-ethylene diamine tetra acetic acid were purchased from Gibco, Invitrogen Corp., USA.
Electrospinning method
Electrospinning was performed to prepare blend nanofibers of PCL/GT/AV. PCL and GT was dissolved in acetic acid (90% AA) to form 20 and 7% of both polymeric solution, respectively. PCL/GT (2:1) solution was prepared by addition proper amounts of both prepared polymer solutions. For production of AV containing nanofibers, 5% (w/w) AV (according to the total weight of applied polymer in solution) was added to PCL/GT and PCL solution and stirred for 1 h. Prepared solution was transferred into individual 20-mL syringe attached to a stainless steel spinneret (19 G) and a high voltage of 15 kV was applied to the tip of the spinneret. The flow rate and distance from the needle tip were set at 1.0 mL/h and 14 cm, respectively. Random nanofibers were formed on aluminum wrapped collector, dried overnight under vacuum and used for more analysis. Figure 1 shows the set-up of electrospinning process.
Electrospinning process for nanofiber production.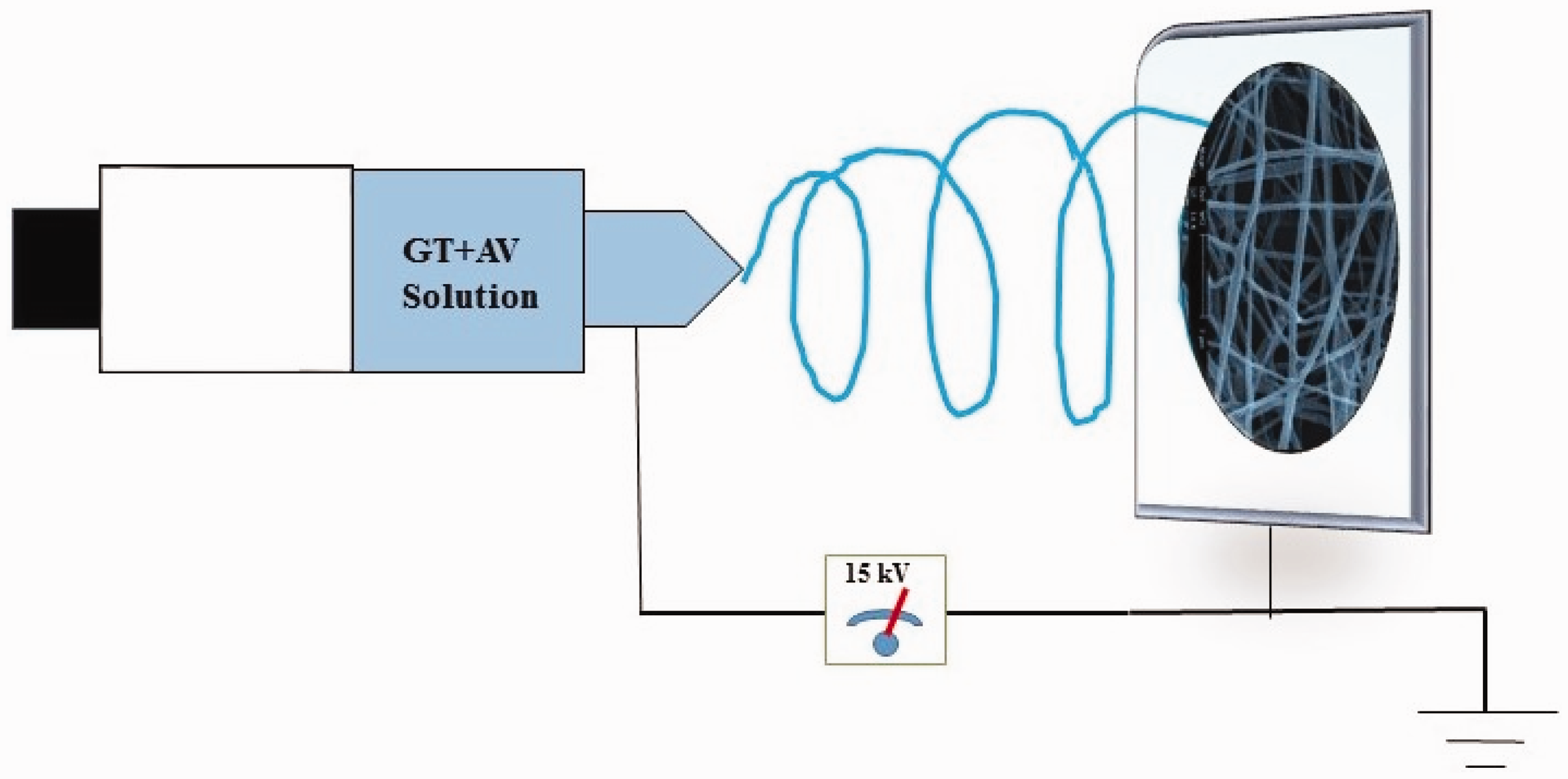
Characterization of nanofibers
The viscosity of the different polymer solutions (PCL, PCL/AV, PCL/GT, and PCL/GT/AV) was recorded using a Brookfield viscometer (Model DV-II + Pro). The morphology and structure of fabricated nanofibers were assessed under FESEM (XL30-SFEG, 12 FEI/Phillips, and Japan) at an accelerating voltage of 20 kV. Image analysis software (Image J, National Institutes of Health, USA) was utilized to analyze the diameters of the nanofibers. Fourier transform infrared spectroscopy (FTIR) analysis (Nicolet Magna-IR 560) was used to study the structural changes using KBr method for AV, PCL/GT, and PCL/GT/AV nanofibers. Hydrophilicity characteristic of fabricated nanofibers was determined via contact angle measurement instrument, and a sessile drop method-based video contact angle system (VCA Optima, AST Products, Billerica, MA) was applied for this aim. The distilled water droplet size was about 1.0 µL.
Differential scanning calorimetry (DSC; Mettler Toledo, Switzerland) was used for examination of thermal properties of samples and nanofibers were heated from −110℃ to 290℃ at a heating rate of 10℃/min under nitrogen.
Tensile stress, strain, and elastic modulus of the PCL, PCL/GT, and PCL/GT/AV mats were reported by a uniaxial tensile machine (Instron5943, Canton, MA) with a load cell capacity of 10 N and cross head speed of 5 mm min−1. All nanofiber tape samples were cut in the form of rectangular shape with dimensions of 30 × 10 mm2. This test was done in room temperature and about five samples were used for detecting exact results.
Nanofibers degradation studies
To perform the biodegradability test, the collected nanofibers on aluminum foil were put in PBS (pH of 7.4) and incubated for 20 days at 37℃. After finishing 20 days, the scaffolds were washed and then dried in vacuum oven for 48 h. FESEM images were used for reporting morphology changes.
MTT assay
Cell viability and cell proliferation on PCL/AV and PCL/GT/AV nanofibrous scaffolds were done by using the colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test (MTT). Fresh culture medium was used as control. In this assay, the metabolically active cells were reacted with tetrazolium salt in MTT reagent to form a soluble formazan dye and absorbance was recorded at 570 nm using spectrophotometer. NIH 3T3 fibroblast cells were plated in 90 µL of DMEM, 10% FBS, at a density of 10,000 cells/cm2. After 2 days of cell seeding in 24-well dish, the medium was removed and 1 mL fresh medium and 100 µL MTT solution were injected to each well. Cells were incubated at 37℃ (5% CO2) for 4 h, the medium was removed, and then the formazan reaction products were dissolved in dimethyl sulfoxide. After 20 min of shaking, the solution was used for spectrophotometric analysis. The optical density of the formazan solution was reported from BioTek ELISA plate reader at 570 nm.
Cell culture
After sterilizing of scaffolds by UV, they were soaked in a culture medium for 12 h. NIH 3T3 fibroblast cells were cultured in DMEM and 10% FBS, 1% antibiotic solution at 37℃ under a humidified atmosphere of 5% CO2, 99% relative humidity (RH). About 10,000 cells/cm2 fibroblast cells were seeded on PCL/AV and PCL/GT/AV nanofibers on 24 well plates and incubated at 37℃ for 2 days. Cell adhesion and cell morphology was checked after 1 day by using light microscope. After 48 h, washing and fixing of cells was performed. Cell morphology was observed by FESEM.
Statistical analysis
Statistical analysis was performed using one-way analysis of variance (ANOVA), and significance was considered at p ≤ 0.05.
Results and discussion
Morphology of nanofibers
The morphology of fabricated scaffolds was analyzed by FESEM. Figure 2(a) to (d) exhibits the micrographs of these nanofibers containing PCL, PCL/AV5%, PCL/GT, and PCL/GT/AV 5%. Fibers with smooth surface and bead free morphology without any aggregation were produced evidently, indicating that the AV was incorporated well within the nanofibers. The average fiber diameters of PCL, PCL/AV 5%, PCL/GT, and PCL/GT 5% nanofibers were in the range of 1118 ± 53, 501 ± 69, 184 ± 34, and 123 ± 22 nm, respectively. Such as other reports, it is clear that, the loading of AV significantly reduced the fiber diameter due to increased solution conductivity and reduction in solution viscosity [15]. Viscosity measurements results showed that when GT and AV are blended with PCL, there is a reduction in viscosity of blends and fiber diameter (Table 1). The scanning electron microscope (SEM) morphology of PCL/GT/AV exhibited finer nanofibers when compared to other scaffolds and hence will be more suitable for fibroblast adhesion and proliferation.
SEM micrographs of (a) PCL, (b) PCL/AV, (c) PCL/GT, and (d) PCL/GT/AV, electrospun under applied voltage 15 kV, extrusion rate 1 mL/h, and nozzle to collector distance 14 cm. Viscosity (mPa/s) of PCL, PCL/AV, PCL/GT, and PCL/GT/A.

Hydrophilicity of nanofibers
The surface properties of the nanofibers were also investigated for reporting hydrophilic nature of the fabricated membranes. The effect of AV loading on the contact angles of both PCL and PCL/GT nanofibers is shown in Figure 2. It can be observed that the water contact angle of PCL and PCL/AV was 124° (Figure 2(a)) and 103° (Figure 2(b)), respectively. The contact angle of the PCL/GT and PCL/GT/AV nanofibers was measured to be 84° and 55°, respectively (Figure 3(c) and (d)). This indicates that incorporation of GT to PCL nanofibers increased hydrophilic properties of PCL/GT nanofibers. This indicates that incorporation of AV in PCL and PCL/GT nanofibers changed the surface characteristics of electrospun fibers because of the hydrophilic and soluble nature of AV. AV with number of protein, polysaccharides, and other compounds in its structure could expose hydrophilic groups on the membranes [25]. However, existence of GT in PCL/GT formulation increased hydrophilic properties of nanofibers too.
Water contact angle values of (a) PCL, (b) PCL/AV, (c) PCL/GT, and (d) PCL/GT/AV nanofibers.
DSC results
Figure 3 presents the results of DSC analysis of electrospun matrices of PCL, PCL/GT, PCL/GT/AV nanofibers, and GT powder. The samples were dried in oven at 70℃ for 10 min prior to DSC analysis. For PCL mat, the endothermic peak assigned to the melting of crystalline phase, which it is observed at 59.5℃. However, its glass transition temperature is about −57℃. The DSC graph of GT exhibited the peak temperature at 120℃ and 259℃, which it is related to loss of water and thermal decomposition temperature of GT polymer, respectively. PCL/GT scaffold showed very slight increase in melting and degradation temperature compared with GT and the observed temperatures were 63℃ and 267℃, respectively. The increase in the degradation temperature showed that the stability of nanofibers has raised. For PCL/GT/AV nanofibers, melting and degradation temperature was observable at 62℃ and 256℃, respectively. As it can be seen in the graph, the AV decreased the mat stability (Figure 4).
DSC results of GT powder and PCL, PCL/GT, and PCL/GT/AV nanofibrous scaffolds.
FTIR results
The main peaks of AV (Figure 5) can be observed at 3411 (N–H stretching), 2933 and 2861 (aliphatic group in AV structure), 1729 (O-acetyl esters), 1608 (asymmetrical COO stretching), 1255 (glucan units), and 1090–1037 cm−1 (glycosidic bond). In PCL/GT structure, the observed broad band at 3430 cm−1 could correspond to stretch vibrations of O–H groups in the GT structure. The bands at 2930 and 2856 cm−1 related to stretching vibrations of methylene groups. The sharp peak at 1726 cm−1 could be assigned to carbonyl stretching vibrations in aldehydes, ketones, and carboxylic acids. In PCL/GT/AV scaffold, the O–H stretching has shifted to the lower frequency side and a sharp peak at 3433 cm−1 was observed. A slight bending at 1727 cm−1 is due to the presence of carbonyl group in PCL and another peak at 1166 cm−1 is because of C–O–C group. The spectrum of PCL/GT/AV confirms the successful incorporation of AV to PCL/GT backbone by detection of PCL, GT, and AV characteristic functional groups.
FTIR analysis of AV powder and PCL/GT and PCL/GT/AV nanofibers.
Mechanical properties of nanofibers
The fabricated scaffolds should have appropriate mechanical properties to tolerate forces when they are applied in the injured parts [25]. Figure 6 presents the stress–strain figures of PCL, PCL/AV 5%, PCL/GT, and PCL/GT/AV 5% scaffolds. Tensile strength for PCL, PCL/AV 5%, PCL/GT, and PCL/GT/AV 5% nanofibers were 0.99, 0.32, 0.21, and 0.75 MPa and can withstand a strain of 102, 137, 25, and 32%, respectively. In PCL and PCL/GT nanofibers, the addition of AV increased the strain of mat. For PCL/AV structures, the strength decreased after addition of AV, but for PCL/GT/AV nanofibers, this amount increased compared to PCL/GT nanofibers. AV gel is rich in polysaccharides, so enhanced mechanical strength in PCL/GT/AV compared with PCL/GT nanofibers could be related to similarity in GT and AV structure and their favorable compatibility. By using these results, we conclude that blending PCL/GT with AV 5% gave suitable mechanical characteristic to these scaffolds and improved their strength and strain properties.
Mechanical properties of electrospun nanofibers.
Degradation behavior of nanofibers
Electrospun polymeric nanofibers are increasingly being applied to produce scaffolds for tissue engineering applications as they present high surface area-to-volume ratio and high porosity. The nanofibers exhibited random orientation with a porous structure and interconnected pores. An ideal wound dressing product should exhibit gradual degradation and reabsorption of it by the host [16]. After passing 20 days, PCL/AV nanofibers showed no cracks and morphology change (Figure 7(a)). For PCL/GT/AV nanofibers, especially in the case of finer nanofibers, minor cracks were observable, but they maintained their original structure (Figure 7(b)).
Biodegradation behavior of (a) PCL/AV and (b) PCL/GT/AV nanofibers.
MTT test for fabricated nanofibers
Extracted materials from plants are volatile in nature and have extensively used in nanofibers structure because of their favorable properties [26]. Figure 8 presents the results of cell proliferation studies carried out on electrospun scaffolds using fibroblast cells. Higher cell proliferation was observable on electrospun AV loaded PCL and PCL/GT nanofibers compared to pure PCL and PCL/GT scaffolds. It was found that PCL/AV and PCL/GT/AV nanofiberous matrix increased cell proliferation 30%, 20% on day 2 and 21.5%, 18.5% on day 4 compared to PCL and PCL/GT nanofibers, respectively. AV has significant growth factors, which results in higher cell proliferation [27]. However, because of the excellent biological properties of GT, PCL/GT scaffolds showed higher cell proliferation compared to PCL nanofibers. Proliferation value for PCL/GT/AV was more than other scaffolds, which it can be related to synergistic association of GT and AV in its structure.
Proliferation of fibroblast cells on electrospun nanofibers.
Cell morphology on the nanofibers
The fibroblast cell morphology of in vitro-cultured fibroblasts on AV loaded electrospun PCL and PCL/GT scaffolds was characterized for 2 days of culture by preparation them for SEM studies (Figure 9). PCL-AV 5% scaffolds showed good adherence and spindle-like morphology of fibroblast cells. For GT/PCL/AV scaffolds, higher cell growth, adherence, and well spread morphology of cells was observable on day 2 when compared to PCL/AV scaffold. The higher hydrophilic nature of PCL/GT/AV scaffolds and excellent biological properties of AV and GT in scaffold composition were reasons for better adhesion, migration, and proliferation of fibroblasts cells.
Cell morphology of nanofibers (a) PCL/AV, (b) PCL/GT/AV; A1 and B1 with 200× magnification; A2 and B2 with 100× magnification.
Conclusion
In this work, novel herbal biodegradable PCL/GT/AV nanofibers were characterized for skin tissue engineering. Composite scaffolds from PCL/GT/AV nanofibers with random topography were produced, and the mechanical, chemical, and cyto-chemical properties of the nanofibers have been investigated. Our results showed that AV decreased PCL and PCL/GT nanofibers diameter and improved hydrophilic properties of these scaffolds. Mechanical strength measurements revealed that AV raised the tensile strength and strain of PCL/GT nanofibers. PCL/GT/AV nanofibers with suitable mechanical strength, hydrophilic nature, and maintaining original structure after biodegradation test was used for cell culture studies. In vitro cell culture studies exhibited higher proliferation and proper cellular phenotype on electrospun PCL/GT/AV nanofibers. The PCL/GT/AV biocompatible scaffold will serve as a favorable scaffold for moderate or high exudate wounds because of the relatively low cost and natural origin in addition to hydrogel properties of GT and moisture absorption of scaffolds, which are typically needed for light to moderate exudate wounds. All of these results confirmed the potential of utilizing GT/PCL/AV scaffolds as a regenerative approach for healing different skin disorders and application in skin tissue engineering.
Footnotes
Acknowledgement
The authors would like to express their sincere thanks to the deputy of research of University of Bonab for the financial and technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Bonab (Grant No. 94/I/ER/2825).
