Abstract
Natural polymers such as chitosan and Aloe vera are widely used in novel wound dressings due to their biocompatibility and biodegradability. A problem associated with these polymers is their poor mechanical behavior. Efforts have been made to improve the mechanical properties by mixing synthetic polymers such as PVA, but the role of chitosan and Aloe vera in the final dressing is dimmed. The techniques are also time-consuming and costly and there is still a need for an acceptable and affordable wound dressing which can be made through easily accessible techniques. A new but very simple method is introduced in this work for incorporating PVA nanofibers with Aloe vera-containing chitosan films. Using this method the levels of Aloe vera and chitosan in the system can be optimized at higher scales while benefiting from PVA best mechanical properties as a composite layer. Higher amounts of Aloe vera and chitosan in the system lead to lower product costs and more biocompability. The biological properties of films were examined through cell cytotoxicity and antibacterial tests and compared with Atomic force microscopy results. Physical and mechanical properties of films containing PVA nanofibers were characterized by water vapor permeability, swelling ratio, and tensile tests. The morphology of fibers before and after applying on the films was also observed by scanning electron microscopy. According to the results, this combination of natural and synthetic polymers has led to an affordable, biocompatible, and flexible film for wound dressing applications.
Introduction
Due to the biocompatibility and biodegradability, natural polymers are widely used in novel wound dressings. Chitosan is an antibacterial, biocompatible polysaccharide obtained by removing the acetyl group of Chitin found in the external skeleton of crustaceans. It has the similar structure of glycosaminoglycan (Scheme 1(a)), which causes tissue integrity and has a tremendous effect on the process of wound healing [1]. The simple processability of this biopolymer to produce a variety of shapes has made it a favorite material to produce a variety of wound dressings. Chitosan is hydrolyzed by body enzymes, especially lysozyme and its decomposition results in the release of amine polysaccharides, which will either burn through body metabolism or repelled [2]. Oligomers derived from chitosan decomposition have been identified in the acceleration of wound healing [3].
Although previous studies confirm the good antimicrobial properties of chitosan, there is no evidence of the significant effect of this biopolymer on fibroblast’s growth and proliferation. Most studies have reported chitosan as non-toxic, but neutral in cell growth stimulation [4,5]. In addition, the production process of chitosan is costly and fully deacetylated chitosan is difficult to produce on a large scale [6]. Therefore, combining it with other natural polymers that enhance cell proliferation can improve its imperfections for wound dressing applications.
Aloe vera is an herbal extract used for ages in inflammation cures. The 99% of its gel is water and the rest is rich in vitamins, enzymes, and nutrients. Most of them (about one-quarter of the remaining 1%) is made up of mono/polysaccharides. The main polysaccharide is Glucomannan (Scheme 1(b)) which is a long-chain molecule, including glucose and mannose [7]. The Salicylic acid and antiprostaglandin present in the gel prevent the activity of bradykinin in the wound, resulting in pain-killing effects [8]. Antifungal properties of Aloe vera were used to treat the diabetic foot ulcers with drug resistance [9].
In comparison with chitosan, Aloe vera does not have significant antimicrobial properties, but instead, it increases cell proliferation [4]. Moreover, it is very brittle in dry form and does not have as good flexibility as chitosan in film formation.
The main challenge in using natural polymers for wound dressing applications is their poor mechanical properties not only in the point of low strength but mainly in the point of elongation and adaption to the patient movements. One approach is to incorporate natural polymers with synthetic polymers. The point is to select a biocompatible and biodegradable synthetic polymer.
Polyvinyl alcohol (PVA) with the formula of [CH2CH(OH)]n is synthesized through partial or full hydrolysis of polyvinyl acetate [10]. It has good elongation and it is a suitable material for electrospinning because of its viscosity, electrical conductivity, and surface tension. What makes this polymer suitable for medical applications is its hydrophilicity and polar nature which makes it biocompatible and biodegradable.
There has been a lot of researches over the last decade approving the efficacy of mixing chitosan and Aloe vera [11–13], but the resulting product cannot meet the mechanical properties essential for a wound dressing or if it meets the mechanical properties the amount of chitosan in the system is a lot higher than Aloe vera which is not economical in large scales. The addition of 15% chitosan nanoemulsion to a PVA–Aloe vera mixture containing only 5% Aloe vera powder has led to an increase in tensile and modulus [14]. Also, it is believed that the use of Aloe vera powder is less effective than its fresh gel [4].
There were also some approaches in mixing PVA with Aloe vera or chitosan during electrospinning [15,16], but it affects the electrospinning of PVA and makes the process very time consuming and complicated, while PVA alone is known for its comfortable and easy electrospinning process. In a recent study Aloe vera concentration ranging from 2% to 4% is considered being the optimum range that could produce the electrospun PVA–Aloe vera nanofibers with the tensile properties in the range of skin tissue [17]. Electrospinning of these three materials together needs more PVA rather than chitosan and Aloe vera practically and the role of chitosan and Aloe vera in the system is dimmed. Lower amounts of Aloe vera in these systems lead to more final product costs.
In this work, Aloe vera-containing chitosan films were prepared to induce cell proliferation and reduce the final product costs. As it was expected, the low mechanical properties of Aloe vera in the dry form would reduce the flexibility and softness of chitosan films. An optimal ratio is obtained to maintain the film forming properties of chitosan. Finally, PVA nanofibers were used to meet the requirements of a soft and flexible wound dressing. A new but very simple method is used in this work to introduce PVA nanofibers to the system. Using this method, electrospun PVA nanofibers are prepared separately under its normal electrospinning conditions and loaded on the films as a nanofiber layer which maintains its structure and can introduce the mechanical properties of a composite to the system.
Materials and methods
Materials
Farm cultivated Aloe barbadensis miller leaves were supplied from Morvarid Khalij Fars Corporation (Iran). Fresh juicy leaves with a length of 40 to 50 cm were selected. PVA (Mw: 85,000–124,000 g/mol, 99+% hydrolyzed, powder (10–30 mesh)) and medium molecular weight (190–310 kDa) chitosan powder (40–80 mesh) with 75–85% DD were purchased from Sigma Aldrich Co. (USA). Lactic acid (pharmaceutical grade, liquid form, 90% purity), citric acid (pharmaceutical grade, powder (40–60 mesh), Mw: 192.12 g/mol), NaOH (analytical grade, Pellet form, Mw: 40 g/mol), potassium dihydrogen phosphate (analytical grade, Mw: 136.08 g/mol, powder (40–60 mesh)), magnesium chloride hexahydrate (analytical grade, Mw: 203.3 g/mol) and ethanol (pharmaceutical grade, 99% purity) were purchased from Merck Chemicals (Germany) and used as received.
Methods
Aloe vera gel preparation
Aloe vera leaves were cut at the end and left vertically for about an hour in a water container to remove any aloin inside the leaves’ vessels. The outer green membranes were then removed and the juicy pulp inside the leaf was extracted. The pulps were cut to small pieces and mixed for 5 min in a bladed stirrer to obtain a sticky mucilage. This viscose extract was filtered to remove any impurities remained. Citric acid was added (0.2 wt. %) as a stabilizer and the pH of the Aloe vera extract was adjusted to 3.6. Stabilized Aloe vera extract was kept in the refrigerator (8℃) and used within 24 h.
Aloe vera–chitosan film preparation
Chitosan–Aloe vera sample codes.

Electrospinning of PVA nanofibers
Optimum parameters obtained for PVA electrospinning.

Laying PVA nanofibers on the films
After the determination of optimum ratio for chitosan–Aloe vera film formation (more than 50% Aloe vera leads to a fragile film), a solution with this ratio was prepared again as described and poured on a glass substrate through film applicator at 30℃. After ∼2 h the solution becomes semi-dry. This is the best moment to put the nanofibers on the film. The point is not to apply them when the film is too dry or too wet in order to maintain the shape of the fibers as much as possible. The semi-dry chitosan–Aloe vera film with PVA nanofibers was left to be dried completely and peeled off the substrate afterward (Figure 1).
(a) Laying PVA nanofibers on the chitosan–Aloe vera semi-dry films. (b) The final dry film containing nanofibers.
Figure 2 represents a summarized flowchart for the preparation of chitosan–Aloe vera films containing PVA nanofibers.
Flowchart of steps for preparation of chitosan–Aloe vera films containing PVA nanofibers.
Statistical analysis
The data presented are expressed as mean ± standard deviation and analyzed for significance with Minitab 17 statistical software using one-way analysis of variance (ANOVA). Pairwise comparisons were conducted using the Tukey method. The p-values of less than 0.05 were considered to be statistically significant.
Characterization
Thickness
The film thickness was measured using a digital micrometer (Mitutoyo, Japan), at five different points of each sample to the nearest 0.001 mm, and the average was recorded.
Water vapor permeability
The water vapor permeability (WVP) of films was determined using a modified ASTM E96 procedure. A cup containing 25 ml of distilled water with an external diameter of 25 mm and the internal diameter of 20 mm (exposed area: 1.2 × 10−3 mm2) was used. Prepared films were cut to a diameter of 25 mm, placed like a cap on the bottle and sealed. The whole system was placed inside a desiccator containing saturated magnesium chloride (MgCl2) solution in order to provide a 35% relative humidity at 35℃ for 24 h. The tests were conducted in triplicates. The reduced weight of the system (to the nearest 0.0001 mg) was recorded as the WVP index which was calculated using equation (1).

Swelling ratio
The swelling ratio of films was determined as the maximum percentage of absorption in films by placing them inside PBS solution for specific periods of time (2 min) in 37℃. The wet sample weight measurements were continued until no changes were observed in three consecutive measurements. The tests were conducted in triplicates. The swelling ratio was calculated using equation (2).

Mechanical properties
All mechanical properties were determined using a universal mechanical testing machine (SANTAM-STM-20, Iran) according to ASTM D-2370. The film dimensions were 1 × 7 cm with the specified thickness measured at three points along the strip. The measurements were performed using a crosshead speed of 10 mm/min. The values are the mean of five repetitions.
Scanning electron microscopy
The cross-section of samples was observed by a Tescan-Vega© scanning electron microscopy (SEM) (Czech Republic) in three different magnifications (1000, 5000, 10,000). The films were freeze-fractured in liquid nitrogen and coated with gold-palladium using a sputter coater (EMITECH- K450X, England) before SEM analysis.
Atomic force microscopy (AFM)
A non-contact Dualscope/Rasterscope C26 (DME, Denmark) atomic force microscope was used to investigate the surface topography of samples. Samples measuring 3 × 3 µm2 with specified front sides were scanned. All image analysis and surface parameters were conducted using DME-SPM software (Version 2.1.1.2). The mean surface roughness (Sa) of the films was calculated using equation (3), where 
Cell viability
In vitro cytotoxicity of films against L929 mouse fibroblast cells was evaluated using MTT assay according to ISO 10993-5. Samples were placed in the center of 24-well plates and the cell suspension was seeded on them at the density of 50,000 cells/well and incubated in 5% CO2 for 24 h at 37℃. After that, Tetrazolium salt (MTT) solution (1 mg/ml) was poured on cells, and the culture medium was dark incubated for 3–5 h. The number of live cells per well was measured using ELISA Reader to determine the absorbance of each well at λ540. Cell viability was expressed as the percentage of absorbance relative to control. The control was comprised of cells not exposed to the films. Experiments were performed in quadruplicates.
Antibacterial test
The inhibitory property of samples against 
Results and discussion
Influence of Aloe vera on biological properties of chitosan
Cell cytotoxicity
All samples were not only non-toxic to fibroblasts but also caused a little cell proliferation in most cases (Figure 3). Although chitosan seems to be non-toxic, it cannot significantly increase cellular activity, and it seems that the presence of Aloe vera can improve this activity to some extent.
MTT test results for samples with different compositions of chitosan and Aloe vera. Statistical significance of 
Silva et al. [4] also showed that fibroblasts have a spherical morphology in contact with pure chitosan films, and they accumulate on the surface as it was seen in our results too (Figure 4(a)). However, for the chitosan–Aloe vera hybrid films, this morphology changes into a spindle shape, and the cells exhibit a good proliferation (Figure 4(b) and (c)).
Cell culture images and AFM micrographs of the surface of (a,d) plain CS6% film, (b,e) CS6%–Aloe vera 25% film, (c,f) CS6%–Aloe vera 50% film.
Many studies have suggested the non-toxicity of chitosan to fibroblasts [20]. They have also reported neutrality in confirming the non-toxicity of this substance [5]. This can be due to the unipolar nature of chitosan, which cannot interact well with the bipolar extracellular matrix of the proteins in the medium [21]. Adding other polysaccharides or proteins to chitosan can improve its cellular activity [22,23].
Previous researches have shown that even nanometric changes in surface roughness can lead to cell growth and proliferation [24,25]. They had also shown that the adhesion and orientation of cells on the surface depends on the depth of valleys and peaks and not on their distance from each other [26]. Comparing cell culture images for CS6% films with AFM results (Figure 4(d) to (f)) can describe the reason for better cell proliferation after Aloe vera addition. The surface roughness parameter (Sa) has increased from ∼0.45 nm to ∼3 nm by adding Aloe vera up to 50%. Sharp and short valleys and peaks on the surface of plain chitosan films changes to smooth and deep ones by adding Aloe vera (Scheme 2), which provides a better condition for cell adhesion and proliferation.
Chemical structure of (a) chitosan and (b) Glucomannan (the main polysaccharide in Aloe vera extract). Effect of Aloe vera on the surface roughness of chitosan films.

The highest cellular activity was seen in CS6% samples containing 50% Aloe vera. Lower levels of Aloe vera could not affect cellular activity significantly. The chitosan concentration in the system is also important and it should not be too low (as in CA7) which leads to a very diluted system remaining a flat surface without peaks and valleys in dry form. In fact, an optimum combination between chitosan concentration and the amount of Aloe vera in the system should be obtained to have a suitable surface for cell adhesion.
Antibacterial properties
All samples, except CA7 (which is the most diluted system in terms of chitosan content), have been effective against Effects of chitosan and Aloe vera concentration changes on antimicrobial activity against two bacteria: (a) 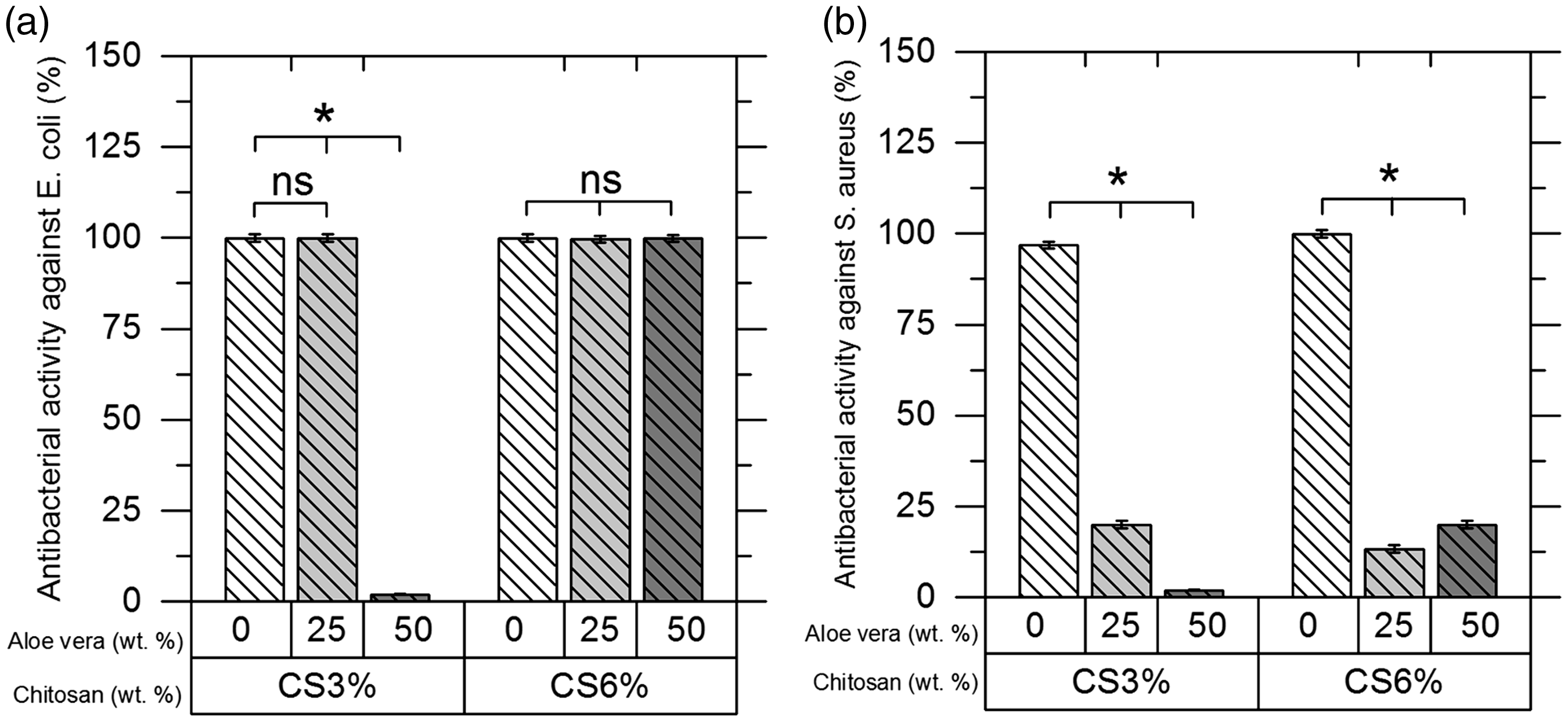
In CS3% samples, the addition of Aloe vera up to 25% had no effect on the antimicrobial activity against
Previous researches on the antibacterial properties of chitosan have shown that it is more effective against gram-negative bacteria than gram-positives [27]. Chitosan can split the cell walls of bacteria that have a negative charge through its functional NH3+ groups. The number of negative charges on gram-negative bacteria cell walls is higher than the gram-positive ones, and they absorb more chitosan content. This is the reason for more efficiency of chitosan in eliminating gram-negative bacteria [28].
The OH− groups in Aloe vera are adjacent to NH3+ chitosan chains and involves some of them. Thus, chitosan chains from the NH3+ groups approach and surround Aloe vera chains (Scheme 3). The gram-negative bacteria with higher charge density have greater power in absorbing chitosan chains. The cell wall of the bacteria is hydrophobic [29] and Aloe vera is less attracted to it. Therefore, when the sample is in contact with Schematic representation of chitosan and Aloe vera chains placement relative to the bacterial cell wall.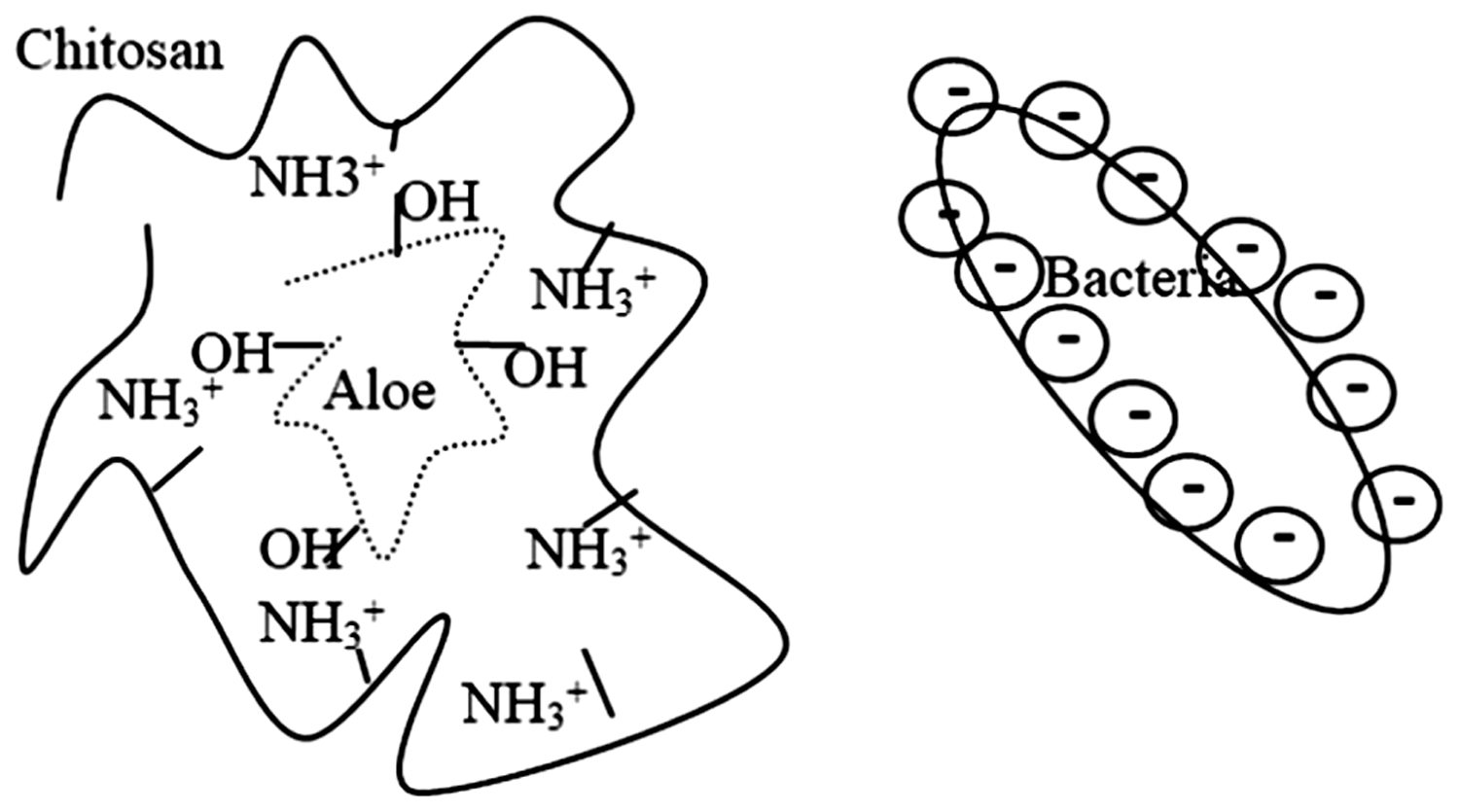
For
Morphology of the films and fibers (SEM)
According to the surface and cross-sectional images of the films (Figure 6(a) and (b)), Aloe vera addition to CS6% films results in the formation of uniform hybrid films with a dense structure with no porosity at the weight ratio of 1:1 (CA11). This ratio was also the best in biological results and film forming properties. Therefore, it was selected for the next step of laying nanofibers on the surface and called CAP11.
SEM images of (a) Surface of chitosan–Aloe vera (1:1) films (b) Cross-section of chitosan–Aloe vera (1:1) films (c) PVA nanofibers right after electrospinning (d) PVA nanofibers after laying on the surface of chitosan–Aloe vera (1:1) films.
As it is seen in (Figure 6(c) and (d)), PVA nanofibers have slightly swollen after laying on the semi-dry film as expected, but the general structure of the fibers is maintained. Comparing fiber diameters before and after laying on the chitosan–Aloe vera films, shows a change from 40–80 nm to 60–120 nm which are still in nanoscale. Having minimally swollen fibers depends on when we put them on the surface of the semi-dry film.
Physical properties
Water vapor permeability
Regardless of the Aloe vera content, increasing the chitosan concentration, decreased the amount of water diffusion through the film. Since chitosan is less hydrophilic than Aloe vera, such behavior was expected. The effect of adding Aloe vera as a hydrophilic agent on the permeability of films follows an increasing behavior (Figure 7(a)), but it affects the film integrity in wet conditions. (Figure 7(b) and (c)) shows chitosan–Aloe vera films with/without PVA nanofibers after 24 h of contact with water vapor. Addition of PVA nanofibers can compensate for the reduction of integrity caused by adding Aloe vera as well as increasing the WVP furthermore due to the hydrophilic nature of PVA.
(a) WVP changes in different concentrations of chitosan–Aloe vera films (b) films without PVA fibers in wet conditions (c) films containing PVA fibers in wet conditions.
Swelling ratio
The rate of swelling and the affinity to water absorption have been increased significantly (Figure 8) as expected because Aloe vera is more hydrophilic than chitosan. Comparing optimum samples with/without Aloe vera and with/without PVA nanofibers in Table 3, the swelling ratio has been increased by 40% after adding Aloe vera to chitosan films. It increased even more (about 10%) after applying the nanofibers (CAP11). PVA nanofibers not only improve the integrity and handling of Aloe vera containing chitosan films but also have increased the water absorption capacity of films by its hydrophilicity.
Swelling ratio changes by time for different chitosan–Aloe vera films. Water absorption of samples with/without Aloe vera and with/without PVA nanofibers.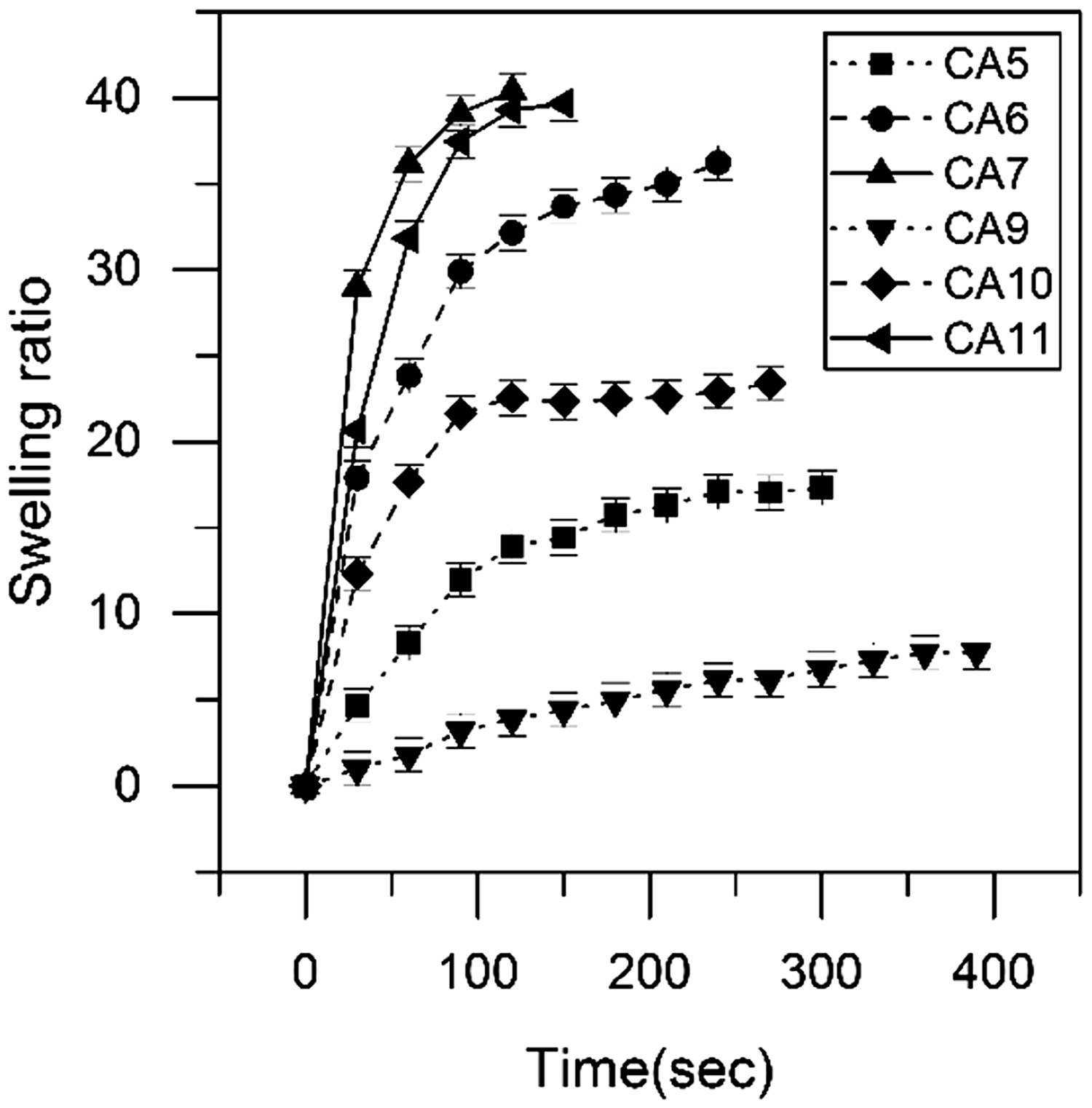

Mechanical properties
Mechanical properties of plain chitosan and chitosan–Aloe vera hybrid films in different proportions.
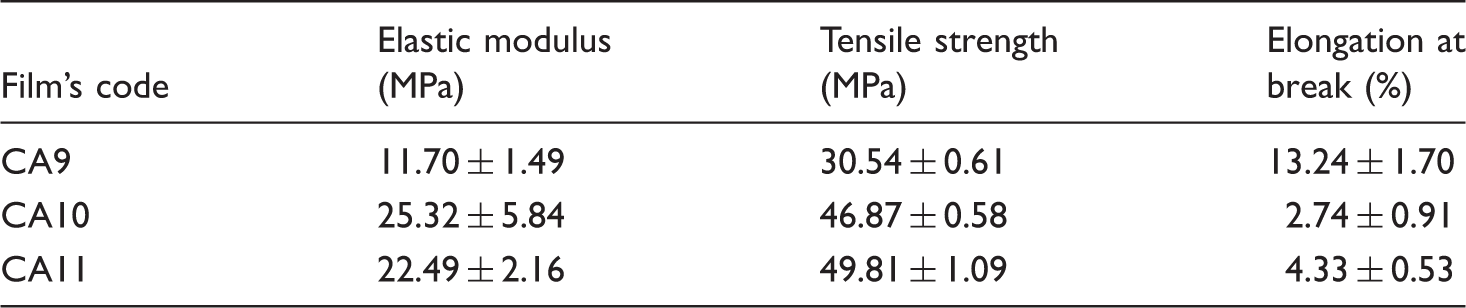
Table 5 and Scheme 4 show what happens to the mechanical properties of films before and after applying PVA nanofibers. A detailed comparison between CA11 and CAP11 in Table 5, indicates a significant reduction in modulus (∼8 times) which was expected considering the lower modulus of PVA than chitosan and Aloe vera [30,31]. This significant reduction is also seen in tensile strength (∼5 times) and stress at yield (∼3 times) which means that the stress–strain curve has settled down (Scheme 4).
Changes in the stress–strain curves of chitosan–Aloe vera films before and after applying PVA nanofibers. Mechanical properties of optimum chitosan–Aloe vera films before and after applying PVA nanofibers.

While the strain at yield has increased almost 99%, a slight increase of 34% has occurred in elongation at break. This means that the length of the plastic zone in which the film cannot return to its original dimensions has decreased, and the length of the elastic zone has increased instead. An ideal wound dressing has a longer elastic zone and can return to its original state after elongation.
The increase of the strain at yield and the decrease of stress at yield at the same time indicate that the sample stretches more under less force until the irreversible changes of dimensions. In other words, the length of the elastic zone has increased while its slope is decreased.
Conclusion
A problem associated with chitosan wound dressings is the final price and limited spreading of cells over their surface. Aloe vera-containing chitosan films were prepared to induce cell proliferation and reduce production costs. The cytotoxicity and antibacterial tests showed an improvement in cell viability and inhibitory properties in hybrid films. The optimal ratio in which the mechanical properties of films were acceptable to use as wound dressings while retaining their cellular and antibacterial properties was 1:1. Adding more than 50% Aloe vera to the formulation leads to a fragile film. PVA nanofibers were used in favor of the mechanical properties expected for a wound dressing in order to reduce the modulus and softening the system. Addition of PVA nanofibers can compensate for the reduction of integrity caused by adding Aloe vera as well as increasing the water absorption capacity furthermore due to the hydrophilic nature of PVA. According to the results, chitosan–Aloe vera–PVA hybrid films can be a promising cost-effective material for wound healing applications.
Footnotes
Acknowledgements
We wish to express our gratitude to Iran Polymer and Petrochemical Institute (IPPI) for their help in the completion of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
