Abstract
Flame retardant textiles are increasingly in demand. There have been a number of approaches by which textile material is made flame retardant. The plant extracts imparting such properties to lingo-cellulosic material has been studied in this work. The paper reports the application of green coconut (Cocosnucifera Linn) shell extract which is a natural waste source onto jute fabric. The acidic coconut shell extract was applied in neutral and alkaline conditions on jute fabric in different concentrations. The emerging fabric showed good flame retardant properties which were measured by different standard flammability tests. The limiting oxygen index value found to increase by 48% after application of alkaline coconut shell extract as such and on concentrating the coconut shell extract, it was found to increase 81%. The thermo gravimetric behaviour and degradation mechanism were studied by using thermo gravimetric analysis in nitrogen atmosphere. The presence of different elements, chemical groups and the structural topography of the untreated and coconut shell extract-treated lingo-cellulosic fabric were analysed by attenuated total reflection-Fourier transform infrared, Scanning electron microscopy, energy dispersive X-ray analysis and phytochemical analysis tests. In addition to the flame retardant property, the treated fabric showed natural colour (measured by colour strength value) and antibacterial property against both gram positive and gram negative bacteria.
Keywords
Introduction
The chemical composition of jute (lignocellulose) differs in a major way from that of cotton (cellulose), in terms of the presence of lignin and hemicelluloses. Therefore, jute fibres/yarns/fabrics are used to develop a number of value-added and diversified products by means of chemical processing and modification. Jute is usually used to make sacks and coarse cloth. The fibres are also used to make different home furnishing items like curtains, chair coverings, carpets, area rugs, hessian cloth, jute brattice cloth in mines, furnishing at public halls and theatre. Fibre is well known for its elegant sound and heat insulation, low thermal conduction and anti-static properties. Jute fibres are also naturally decomposable and can be used in high-performance technical material, geotextile arena, etc. Value addition of jute fabric is done by carrying out bleaching and associated modifications for obtaining improved whiteness and photo-stability. Other properties such as fire retardancy, rot resistance and water repellency are also very popular as far as the value addition of jute textile is concerned [1–4]. In the last 10 decades repeated attempts have been made to make jute a fire retardant. Jute having limiting oxygen index (LOI) of 21% easily catches fire due to cellulosic nature. Various synthetic chemicals have been used for imparting flame retardancy to the jute fabric. Phosphorous-based flame retardants along with nitrogenous and sulphur compound are the most effective formulations so far reported [5]. Consequently, during the last five decade, different flame retardant synthetic chemicals based on the composition of sulphur, nitrogen and phosphorous had come into the market for imparting flame retardancy to jute. Among all these flame retardants, Proban process and Pyrovatex process are widely dominating the market [6]. However, such formulations need to be applied in an adverse acidic condition and more add-on% is required for flame retardancy. Therefore, the jute fabric loses its tensile strength tremendously and also becomes stiffer. Besides, such a treatment is expensive, non-ecofriendly due to the involvement of severe chemicals, high-temperature curing and toxic formaldehyde emission during treatment [7]. Recently researchers have used eco-friendly sodium metasilicate nanohydrate for making fire retardant jute material [8]. Composition of the polycarboxylic acid and nano zinc oxide [9] also has been developed by the researchers to make the fire resistance process eco-friendly. However, such treatment cannot give satisfactory handle, strength and durable fire resistant to the fabric. Recently, researchers have also reported the use of plasma treatment with various polymerisation gases to impart fire retardancy to the cellulosic fabric. The plasma process is water free and eco-friendly but this process is very much costlier and the imparted flame retardant property is not wash durable [10]. Hence efforts have been made to prepare more environment-friendly, cost-effective fire retardant products, that when applied to cellulosic or lingo-cellulosic fabrics, will maintain its quality and flame retardant durability to a great extent. To this end, recently researchers have used nitrogen, sulphur-based ammonium sulphamate for making fire retardant for cellulosic and ligno-cellulosic fabric [5, 11–12]. These chemicals too are not completely eco-friendly and the treated fabric lost fire retardancy after washing.
Till date very few researches have reported on the use of natural extracts for imparting fire retardancy to ligno-cellulosic textile materials. In this regard, whey proteins, chicken feather-based protein, starch, chitosan, clay, DNA bio-molecule, natural green waste such as spinach leaves and banana pseudostem sap have been used to impart fire retardancy to cellulosic and ligno-cellulosic fabric [13–16]. To the best of our knowledge, no work has been carried out using coconut shell extract (CSE) obtained from a natural bio-resource waste, for making fire retardant to ligno-cellulosic material. Besides, fire retardancy we have also reported the use of CSE as antibacterial and ultraviolet protection finishing property along with inherent colouration of CSE on ligno-cellulosic material.
Materials and methods
Material and CSE application
A plain woven raw ex-loom Hessian 100% jute fabric, with the natural golden colour of 281.6 g/m2 areal density with 6 warp/cm and 5 weft/cm and having 256 tex warp and 256 tex weft yarn counts were used in this study. Green coconut waste was collected from the local market at Mumbai, India. The mesocarp of green coconut was cut into pieces, and then sap was extracted out using a grinder. Original fresh CSE solution after extraction looked yellowish brown in colour and showed the pH value of 4.5. It was made neutral (pH 7) and alkaline (pH 10) by the addition of anhydrous sodium carbonate (Na2CO3). Fresh scoured jute fabrics were then impregnated with different concentrations of CSE (original as it is extracted CSE (CA), double concentrated (CB) by evaporating the extract to half of its volume) at 90℃ with material to liquor ratio of 1:15 for 60 min. Treated jute fabric was then dried in air at room temperature. Thereafter samples were conditioned for 48 h at 65% relative humidity (RH) at 27℃, before testing.
Phytochemical analysis
Chemical tests for the identification and screening of bioactive chemical constituents in the CSE were carried out with the extracts using the standard procedure [17]. For each test of saponin, phenols, tannins, terpenoid, flavonoids and glycoside, 2 mL of CSE was used for analysis. Acetic acid glacial extra pure, chloroform LR, hydrochloric acid, sulphuric acid (99.8%) and ferric chloride hexahydrate LR were received from S. D. Fine Chemicals Limited (SDFCL) Mumbai, India. Zinc dust (A.W. 65.37) was received from RFCL Limited, Gujarat, India.
Gas chromatography-mass spectrometry (GC-MS) analysis
GC-MS analysis was done with the instrument GC-MS-QP 2010 Ultra, Shimadzu.
UV–visible spectrophotometer analysis
UV–visible spectrophotometer analysis of CSE for diluted solution sample was done by scanning from 300 to 700 nm wavelength with the instrument UV-1800, Shimadzu.
Determination of add-on %
Before any physical and chemical characterization, both the treated and the untreated jute fabrics was conditioned at 65% RH and 27℃ temperature for 48 h. Add-on % of the CSE treated fabrics was carried out by gravimetric principle, using the following formula. After add-on testing, the treated fabric was evaluated for flammability tests.
Flammability assessment
Flammability properties of the untreated and treated samples were evaluated by standard methods. For LOI analysis, ASTM D2863 test method was used. In vertical burning behaviour and properties were measured as per the IS 1871 method A.
Thermogravimetric analysis (TGA)
Thermogravimetric (TG) curves of both the untreated and the treated fabrics were obtained by using a differential thermo gravimetric analyser, DTG-60 H with simultaneous DTA-TG apparatus of Shimadzu having a heating rate of 10℃/min in a nitrogen atmosphere.
Attenuated total reflection (ATR) Fourier transform infrared (FTIR) analysis
ATR–FTIR spectroscopy of the untreated and the treated jute were recorded using a Shimadzu FTIR-8400 S spectrometer at a resolution of 1 cm−1 in the wavelengths of 800–4000 cm−1.
Scanning electron microscope (SEM) and energy dispersive X-ray (EDX) analysis
The surface morphology of the untreated and CSE treated jute fabric was analysed using SEM and EDX analysis. The presence of elements and their respective mass percentage were analysed using Analytical Scanning Electron Microscope, Model: JEOL-6380LA. The SEM image of char analysis was done on FEG-SEM of TESCAN.
Antibacterial activity
Antibacterial susceptibility testing was done using the well-diffusion assay, according to the standard of the National Committee for Clinical Laboratory Standards. Antibacterial activity was qualitatively evaluated by AATCC 147 test method, measuring the size of the zone of inhibition of bacterial growth around the well. The antibacterial activity was quantitatively evaluated against Staphylococcus aureus (AATCC 25923) and Escherichia coli (AATCC 25922) according to the AATCC 100-2004 test method. Colonies of bacteria recovered were counted on the agar plate and the % reduction of bacteria (R) was calculated by the following equation
Ultraviolet protection factor (UPF) analysis
The UPF values of the untreated jute fabric and dyed fabric with the extract were measured using a Shimadzu UV-2600 series in the range of 280–400 nm, Model UV-2600(A11665101436). The UPF value of each fabric was determined from the total spectral transmittance based on AS/NZS 4399:1996 method.
Colouration on jute fabric
Colour depth of the samples was evaluated measuring the reflectance values, using computer colour matching, Spectra Scan 5100+ system at λmax. The reflectance values were determined using the following equation
Assessment of fastness properties
Washing fastness of CSE-treated dyed sample at different pH was carried out according to ISO 105 C-10:2006 method. Similarly, light fastness and rubbing fastness of the same samples were also assessed according to ISO 105-B02:2013 and ISO 105-X 12:2002 methods, respectively.
Evaluation of flame-retardant activity
The flame-retardant activity of the finished samples was evaluated as per ISO 2 method, the wherein sample was washed with 5 g/L of soap in a solution with liquor ratio 50:1, at 50℃, for 45 min, then rinsed properly with water and dried. Thereafter, the samples were conditioned in a desiccator for 24 h, before conducting flammability tests.
Determination of tensile strength
Effect of treatment on breaking load of the jute fabric was evaluated by measuring breaking load of the samples in H5KS Single Column Universal Tester (Tinius Olsen) as per the ASTM D 5035-1995 method.
X-ray diffraction (XRD) analysis
XRD analysis of the untreated and treated samples were recorded in Shimadzu 6100, equipped with CuKα radiation (λ = 1.54 Å) in the 2θ.
Results and discussion
Vertical and LOI flame retardancy analysis
Being lingo-cellulosic in nature, untreated jute fabric showed LOI value of 21%, reflecting easy flammability of the substrate. Any material showing LOI value of equal or more than 26% can be considered as fire retardant [18]. The LOI and flammability values of the untreated and the CSE treated jute fabrics at different pH (for colour refer Figure 1) with two concentrations are given in Table 1. After application of the CSE, the LOI value was found to increase significantly. When the fabrics were treated with CA, the LOI value was found to increase to 25%, 28% and 31% for acidic, neutral and alkaline pH, respectively, which is almost 19%, 33% and 48% higher than that obtained with the untreated sample. This is attributed to the increase in swelling of jute as pH varied from acidic to alkaline pH, which consequently increased add-on % of CSE and the LOI value. When the concentration of the CSE was doubled by evaporation (CB), the LOI value increased for all the cases at different pH. This is due to the fact that as the add-on percentage increased it caused a further increase in the LOI to 27%, 34% and 38% for acidic, neutral and alkaline pH, respectively. This increase was of the order of 28%, 62% and 81% of the untreated fabric. In the vertical flammability measurement, it was observed that the untreated sample ignited readily, and burnt entirely with flame within the 90 s. However, the CSE treated sample (CA) showed the presence of flame only for 8 s (partial burning) followed by combustion with afterglow for 281 s, and thus, recording a total burning time of 289 s for alkaline pH (10) whereas in case of CB, it does not show any flame and burnt completely in the 30 s as depicted in Figure 2(a) and (b).
Extraction process and colour of coconut shell extract (CSE) at different pH. Vertical flammability test of the untreated and CSE-treated jute fabrics. CA: CSE at its original concentration; CB: double concentrated CSE; CSE: coconut shell extract; Flame contact time = 10 s; sample size = 25 × 4 cm. Vertical flammability of the coconut shell extract (CSE)-treated jute fabric in (a) CSE at its original concentration (CA) and (b) double concentrated CSE (CB).

Untreated as well as CSE-treated jute samples (acidic and neutral pH) showed no char length as both the CA samples were burnt completely. On the contrary, CB-treated jute fabric samples showed char length at different pH. Residual char mass that remained after burning has been represented in Figure 3. Minimum char length was observed having a length of 50 mm and 12 mm for CA and CB, at pH 10, respectively. All the CSE-treated jute fabric samples showed hard char mass compared to the char mass of untreated jute fabric which might be due to the more deposition of inorganic material on the treated lignocellulosic polymeric material.
Residual char mass remained after completion of the vertical flammability test for CSE at its original concentration (CA) and double concentrated CSE (CB) of flame contact time 10 s.
Vertical flammability test of the CSE-treated jute fabrics for CB.

CSE: coconut shell extract; sample size = 25 × 4 cm.
Thermogravimetric analysis
The TG curve of untreated and CSE-treated jute samples at different pH (refer Figure 4 and Table 3) evidenced three stages of progression. The thermogram of the untreated (A) jute sample depicts three stages of mass loss. In the initial stage at a temperature below 200℃, mass loss occurred mainly due to the removal of bound and unbound absorbed moisture from the cellulose polymer. However, the main weight loss (pyrolysis) occurred in the temperature range 260–500℃. In this range, mass loss peak has been observed at 330℃ due to pyrolysis of cellulose [19]. Above 400℃, dehydration and char formation occurred [20]. It could be seen that the untreated jute sample (A) lost approximately 86% of its mass below 400℃ whereas in the case of alkaline pH, CSE-treated jute fabric (D) showed 67% mass loss. TG curves showed that by increasing the pH of the application, the thermal stability of the treated fabric has been increased as the amount of char mass left at a higher temperature has been increased and the degradation point has been shifted gradually towards the lower temperature. TG curve also showed that increasing the concentration of CSE also increased the thermal stability of the CSE-treated fabric with more char mass generation (refer Figure 4(b) and Table 3).
(a) Thermogravimetric analysis plots of (A) untreated jute, (B) coconut shell extract (CSE)-treated jute fabric at pH 4.5, (C) pH 7 and (D) pH 10 for CSE at its original concentration (CA). (b) Thermogravimetric analysis plots of (A) untreated jute, (B) CSE-treated jute fabric at pH 4.5, (C) pH 7 and (D) pH 10 for double concentrated CSE (CB). Different TGA parameters for untreated and CSE-treated jute fabric. CA: CSE at its original concentration; CB: double concentrated CSE; CSE: coconut shell extract.

It can also be observed that the treated fabric D started forming char and the quantity of char residue remained higher than the untreated fabric at any higher temperature up to 500℃. It increased from 15% to 33%. It shows that the thermal stability of natural ligno-cellulosic fabric increased when it was treated with CSE with alkaline pH, which has a positive effect on prior dehydration and char formation. Therefore, it can be concluded from the TG curves that CSE improved the thermal behaviour of jute fabric if it is applied at alkaline pH. It might be due to the more pick up of CSE by the jute fabric which reflects the presence of more inorganic elements such as sodium, silicon, magnesium, phosphorus and nitrogen as confirmed by EDX analysis (refer Figure 9).
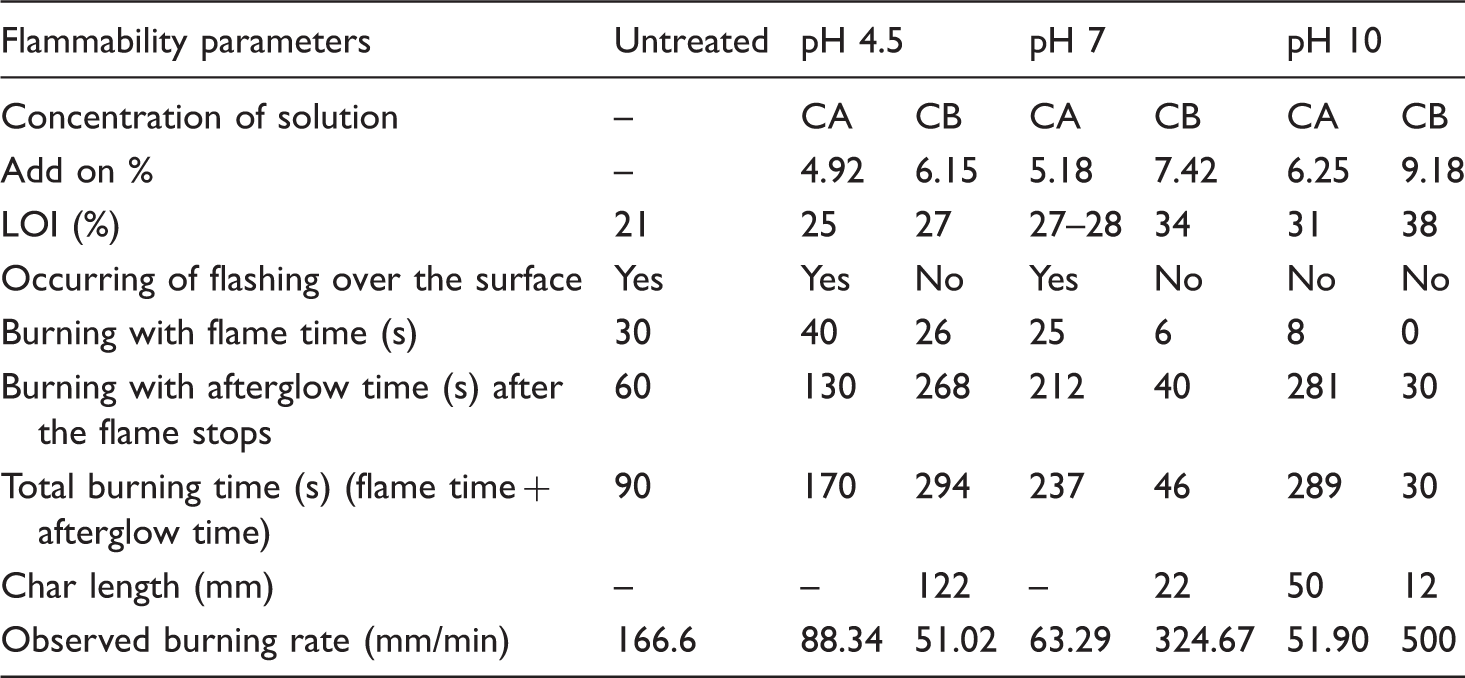
ATR-FTIR analysis
The peaks observed in the range of 800 and 1300 cm−1 wavelength were mainly due to the presence of inorganic salts in dried CSE as shown in Figure 5(a) [21]. FTIR spectrum of CSE sap has shown the peak at 3271 cm−1 which is due to stretching vibration of phenolic hydroxyls (O–H bond) and broad area indicates the intramolecular hydrogen bonding among the polyhydroxy aromatic compounds (Figure 5(a)). The peak observed at 2940 cm−1 is due to ‘C–H’ stretching frequencies. Around 2860–2800 cm−1, methoxy bands are observed, supported by a band near 1270 cm−1 [22]. The peak at 1608 cm−1 and neighbouring bands are due to the presence of aromatic rings and 1045 cm−1 due to –O–C aliphatic stretching vibration in the CSE, respectively. The IR absorption bands observed at 1000-1100 cm−1 might be assigned to the phosphate group antisymmetric stretching modes [25]. The small peaks observed at 1176 and 873 cm−1 might be due to the presence of sulphates [21]. These types of elements in the inorganic salts have also been detected in the EDX analysis of the CSE and the treated jute fabric. The CSE-treated fabrics have similar peak pattern as untreated jute as shown in Figure 5(b) since the add-on of the CSE on fabric is insufficient to give ATR-FTIR sensitive peaks.
(a) Fourier transform infrared (FTIR) analysis of coconut shell extract. (b) FTIR analysis of (A) untreated jute (B) treated jute at pH 4.5 (C) pH 7 and (D) pH 10 for double concentrated CSE (CB).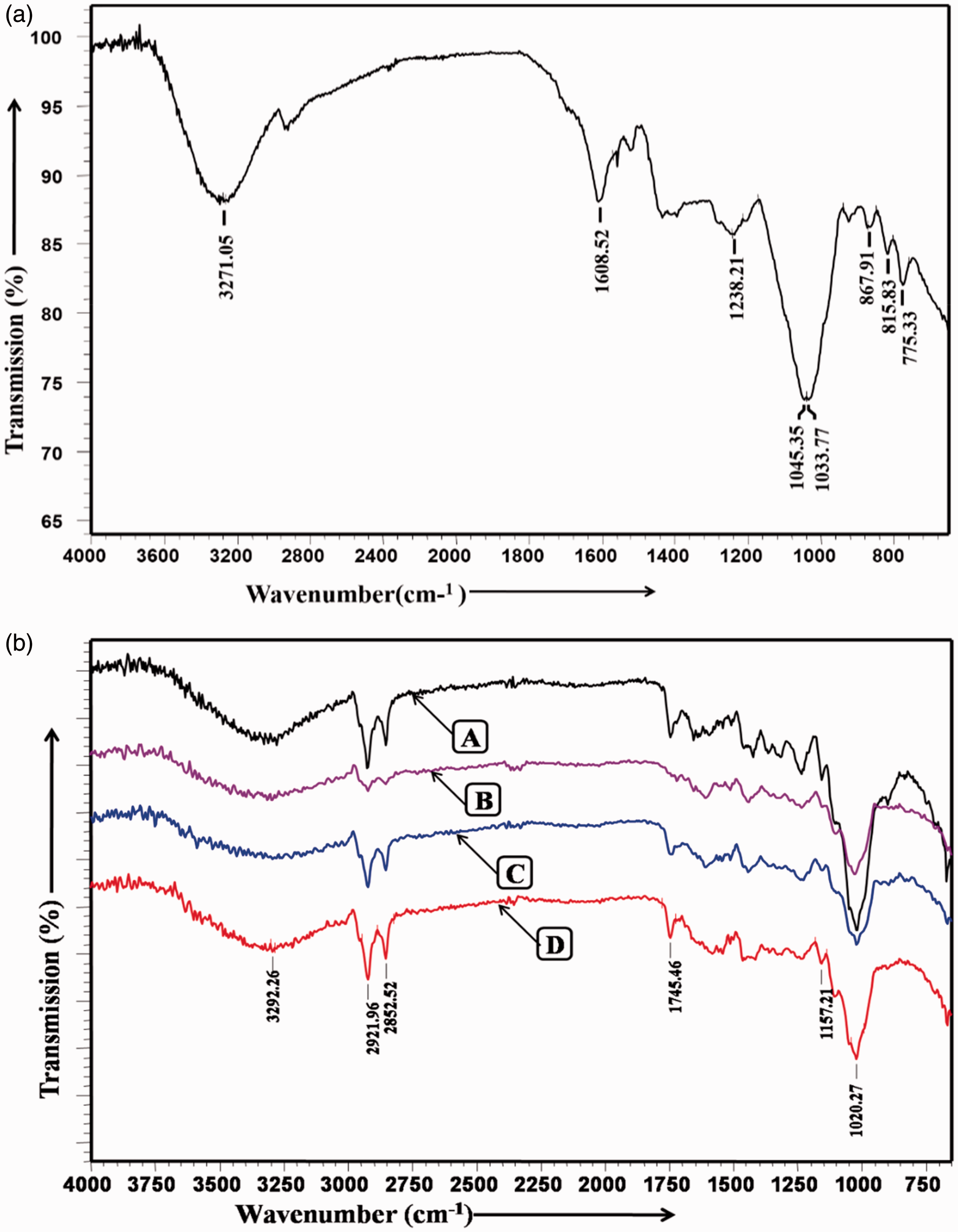
SEM analysis
The SEM images of the dried CSE untreated and treated jute fabric at alkaline pH are shown in Figure 6. It can be seen from Figure 6(A) that untreated jute sample showed clean surface, without any deposition whereas in Figure 6(B, C, and D) after the CSE treatment (CB), the coating of CSE could easily be visible which was distributed over the entire surface of the fabric. The coating was more at pH 10 as it was visible clearly on Figure 6(D) which was also responsible for more LOI value and thus gives more flame retardant property.
Scanning electron microscopy (SEM) images of (A) untreated jute (B) coconut shell extract (CSE)-treated jute at pH 4.5, (C) pH 7 and (D) pH 10 for double concentrated CSE (CB).
Char analysis of untreated and treated jute fabric with SEM and ATR-FTIR
Char residual analyses of the untreated and the CSE-treated jute samples were carried out and the results are presented in Figure 7. It showed that while the untreated jute fabric has the light-weight, ash-like white char mass (A), the treated jute fabric showed a hard, solid and a blackish char mass at different pH for concentrated solution, i.e. CB. However, at pH 10 CSE-treated jute showed harder, blackish carbonaceous char. To understand this phenomenon more vividly, SEM analysis of the char mass samples was carried out at different pH (B, C, D). Accordingly, char models for untreated and treated jute have been represented in Figure 7. Untreated jute char showed a net-like open capillary structure due to which volatile flammable gases can pass through easily and help to burn the jute fabric continuously (A1). However, treated jute char showed dense bubble like char structures formed of closed cells containing pockets of gases (B1, C1, D1). This closed cell char structure helped to restrict the flow of flammable gases and prevent them from coming out in contact with the flame source.
Char analysis and morphology of untreated (A, A1), coconut shell extract (CSE)-treated jute fabric at pH 4.5 (B, B1) 7 (C, C1) and 10 (D, D1) for double concentrated CSE (CB).
On comparison of the ATR-FTIR spectra of burnt fabric untreated and treated samples as shown in Figure 8, it is seen that the peak at 1440 cm−1 (attributed to aromatic –C = C– skeletal vibration) in untreated jute char changes to include a 1480 cm−1 component at acidic pH char, and includes peak appearance at 1560 cm−1 with increasing pH from neutral to alkaline (increasing aromatic carbon content with treated sample) and peak disappearance at 1040 cm−1 and 1080 cm−1 corresponding to C6 skeletal vibration and C–O–C pyranose ring skeletal vibration which is present in untreated char and decreases very much in intensity in the treated jute samples. This change in peak region 1040–1080 cm−1 corresponds to the decreased amount of formation of levoglucosan, its formation being inhibited by the retardant which is most pronounced at acidic pH [23].
Attenuated total reflection–Fourier transform infrared (ATR–FTIR) analysis of the char of untreated jute (A) and coconut shell extract (CSE)-treated jute at pH 4.5 (B), pH 7 (C) and at pH 10 (D) for double concentrated CSE (CB).
EDX analysis
Elemental analysis was carried out by the EDX spectroscopic analysis of the dried CSE (A), untreated and CSE treated jute fabric (B & C) which are represented in Figure 9. It was found that the untreated jute contains the presence of carbon (55.21%), oxygen (43.85%), silicon (0.73%) and traces of sulphur (0.21%) atoms as the technique used cannot detect hydrogen atom. Pure dried CSE contains carbon (26.15%), oxygen (13.15%), nitrogen (28.1%), potassium (12.27%), silicon (3.31%), chlorine (2.56%), manganese (2.96%), nickel (5.27%), iodine (1.73%), magnesium (1.65%), aluminium (0.85%), phosphorous (1.58%) and sodium (0.42%). CSE-treated jute sample at pH 10 for CB also contains carbon (39.86%), oxygen (30.14%), potassium (7.13%), sodium (5.42%), silicon (5.98%), sulphur (1.89%), aluminium (2.28%) and nickel (6.76%). These elements may be present in oxide or salt form as chloride, silicate, phosphate, phosphite, etc. on the CSE-treated jute fabric which is synergistically responsible for fire retardant property.
Energy dispersive X-ray analysis (EDX) images showing the peak for (A) dried coconut shell extract (CSE), (B) untreated jute and (C) treated jute at pH 10 for double concentrated CSE (CB).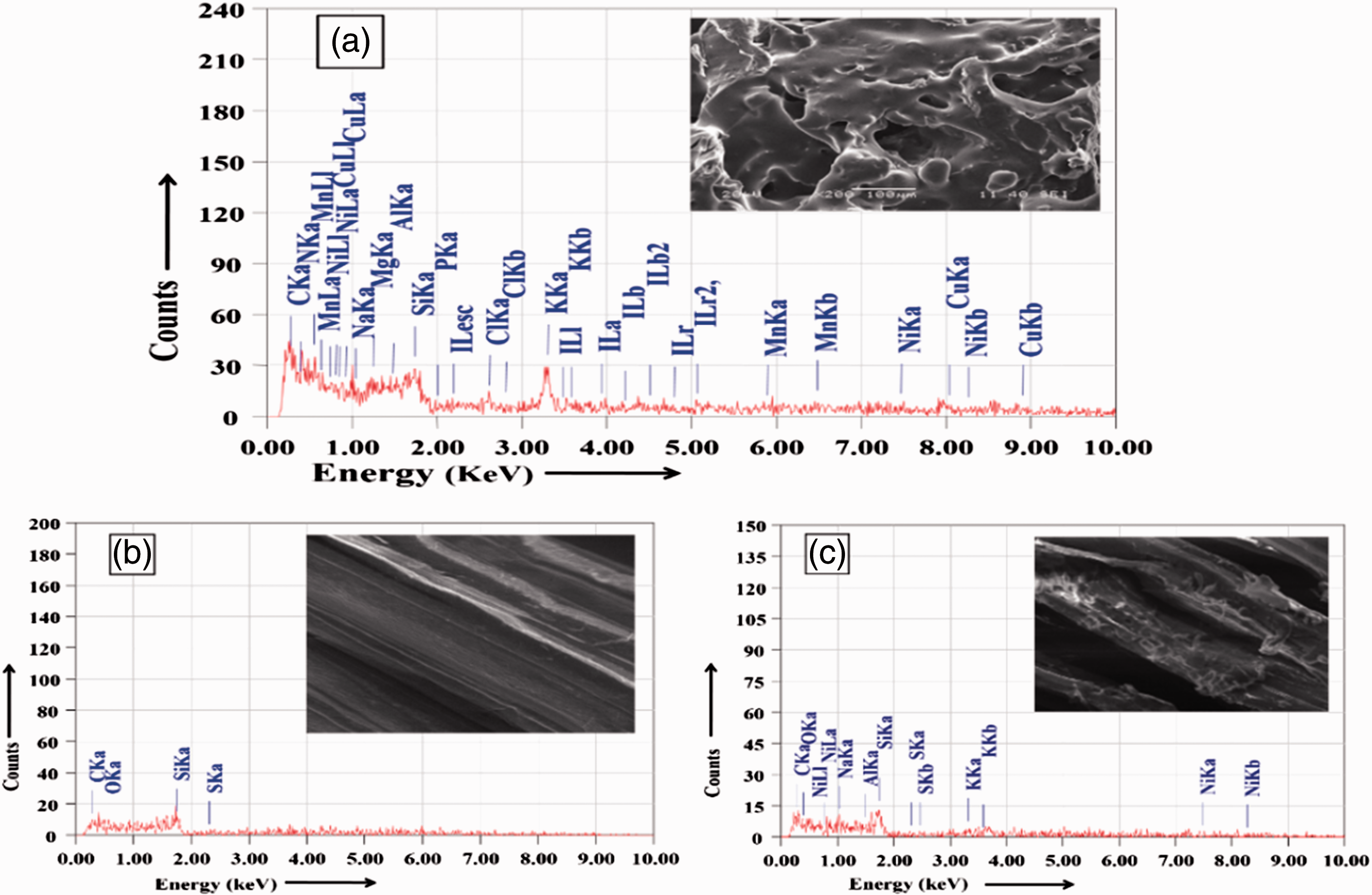
Phytochemical analysis
CSE was placed in a test tube and shaken vigorously for a few time. The formation of stable foam was taken as an indication of the presence of saponin. Two millilitres of 2% solution of ferric chloride (FeCl3) was mixed with CSE. A blue-green or black colouration indicated the presence of phenols and tannins. CSE was mixed with 2 mL of chloroform followed with 2 mL of concentrated sulphuric acid carefully added and then shaken gently. A reddish brown colouration of the interphase was formed which showed a positive result for the presence of terpenoid (Salkowski’s test). The extract was mixed with zinc dust and concentrated hydrochloric acid was added drop wise. It gives red colour after few minutes indicating the presence of flavonoids (zinc-hydrochloride reduction test). CSE was mixed with 2 mL of glacial acetic acid containing two drops of 2% FeCl3. The mixture was added into another tube containing 2 mL of concentrated sulphuric acid. The presence of brown ring at the interphase indicates the presence of glycosides. The summarised phytochemical screening of chemical constituents of CSE has been used in this study on a qualitative basis. The results revealed the presence of active compounds in the CSEs. All the tests showed positive component for phenols, tannin, saponin, terpenoid, glycoside and flavonoid as shown in Figure 10.
Phytochemical analysis of coconut shell extract.
GC-MS analysis
The GC-MS chromatogram, along with the mass spectral identification of the two main peaks visible at retention times of 5.4 and 7.06 min as 3,5-dihydroxy-6-methyl-2,3-dihydro-4 H-pyran-4-one, molecular mass m/z 144, 29.3% by peak area as shown in Figure 11(a) and 5-hydroxymethylfurfural (HMF), molecular mass m/z 126, 70.8% by peak area as shown in Figure 11(b), respectively, explain the composition of the ethyl acetate soluble and thermally volatile fraction of the green CSE. The components were identified by matching the peaks with Computer Wiley MS libraries and confirmed by comparing mass spectra of the peaks with those from literature. This is merely a partial identification of the composition of the green CSE. These two compounds have been previously identified to occur in fruit extracts and green plant material [27]. 3,5-Dihydroxy-6-methyl-2,3-dihydro-4 H-pyran-4-one is a relatively stable degradation compound (Maillard reaction intermediate) of hexose [28]. Polyphenolics and 5-HMF are present together in wines, similar to the occurrence in CSE [29].
Chromatogram with Retention time, the mass spectrum of the peak, spectral identification for (a) compound 1 and (b) compound 2.
Polyphenolics like the anthocyanins (occurrence supported by the UV spectra of green CSE at different pH) are not found in this GC-MS spectrum due to the column and GC-MS conditions. They are likely to be the key flame retardant chemical functionalities and also responsible for the UV protective and antibacterial effects.
UV–visible spectrophotometric analysis of CSE
UV–visible spectrophotometric analysis of CSE was shown in Figure 12. Anthocyanins absorb strongly in the visible and UV spectral range, with maximum absorbance falling in the region of 465–560 nm and 280–320 nm. Their UV absorbing capacity varies depending on their specific aglycones, sugar conjugation and acylation configurations. Anthocyanins are present mostly as colourless hemiketals under mildly acidic conditions, as in the case of CSE. At pH 4.5 and 7, a band with absorbance maxima at 324 nm, undergoes a bathochromic shift to 366 nm at pH 10. At pH 4.5 and 7, the two other band maxima at 416–420 nm and at 505 nm collapse to a slight band maxima at 505 nm on increase to pH 10. As the pH is raised, kinetic and thermodynamic competition occurs between the hydration reaction of the flavylium cation and the proton transfer reactions related to the acidic hydroxyl groups of the aglycone. While the first reaction gives a colourless carbinol pseudo-base, which can undergo ring opening to a chalcone pseudo-base, the latter reactions give rise to quinonoidal bases [30].
UV–visible spectra of coconut shell extract at pH (A) 4.5, (B) 7 and (C) 10.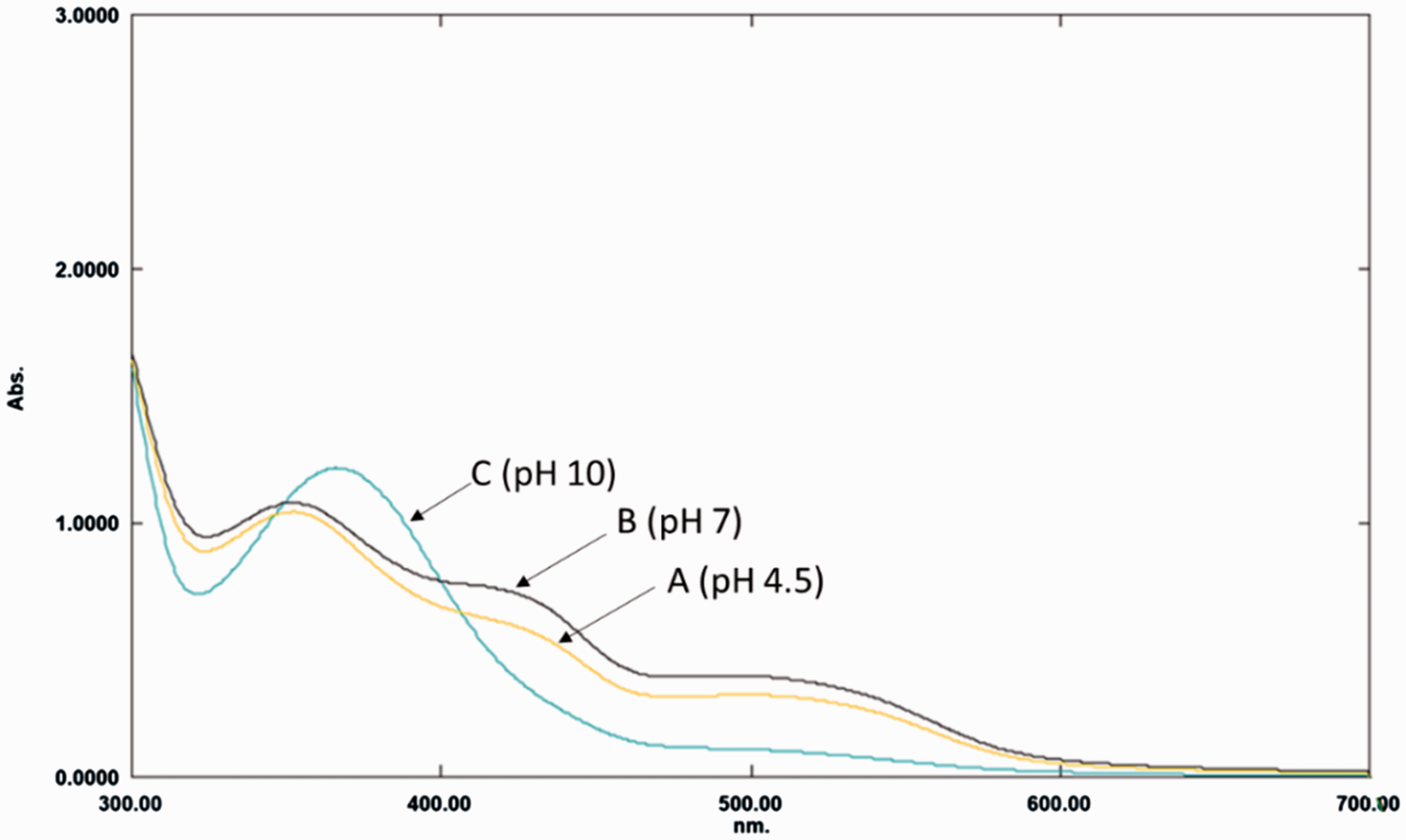
Antibacterial activity of CSE-treated jute fabric
Reduction percentage of E. coli and S. aureus on treated jute fabric.

CSE: coconut shell extract.

Antibacterial activity of coconut shell extract (CSE) of S. aureus and E. coli on the untreated and treated jute.
Quantitative test (AATCC 100) results as shown in Table 4 clearly indicate that the jute fabric treated with CSE showed good antibacterial properties against S. aureus and E. coli. Bacterial reduction % in the treated fabric for all pH is more than 96%. As far as the literature is concerned, green coconut shell (mesocarp) is rich in different types of plant secondary metabolites, especially antioxidants. These protect the coconut from a biotic attack that is essential for microbial infection resistance [32].
Evidence for the presence of phenolic compounds and tannin in the CSE was obtained positive with ferric chloride test. Also, the presence of saponin, terpenoid, glycoside and flavonoids was found to show positive results. Tannins are polyphenolic compounds, shown to have antibacterial activity [33]. Jute fibre has a strong ability to form chelating complexes with metal ion due to the presence of –OH and –COOH groups. Hydroxyl (–OH) group of jute fibre will form hydrogen bonding and minor Van der Waal interaction when it is treated with tannin. Moreover, metal ions, tannin and the hydroxyl group of the jute fibre form strong coordinate bonding between them on treatment with metallic salts. Besides, the large insoluble metal–tannin complex molecules are responsible for imparting antibacterial effect when it is inside the jute fibre [31].
Colour parameters of the treated jute fabric at different pH and concentration
K/S values and colour co-ordinates of dyed jute fabric with CSE.
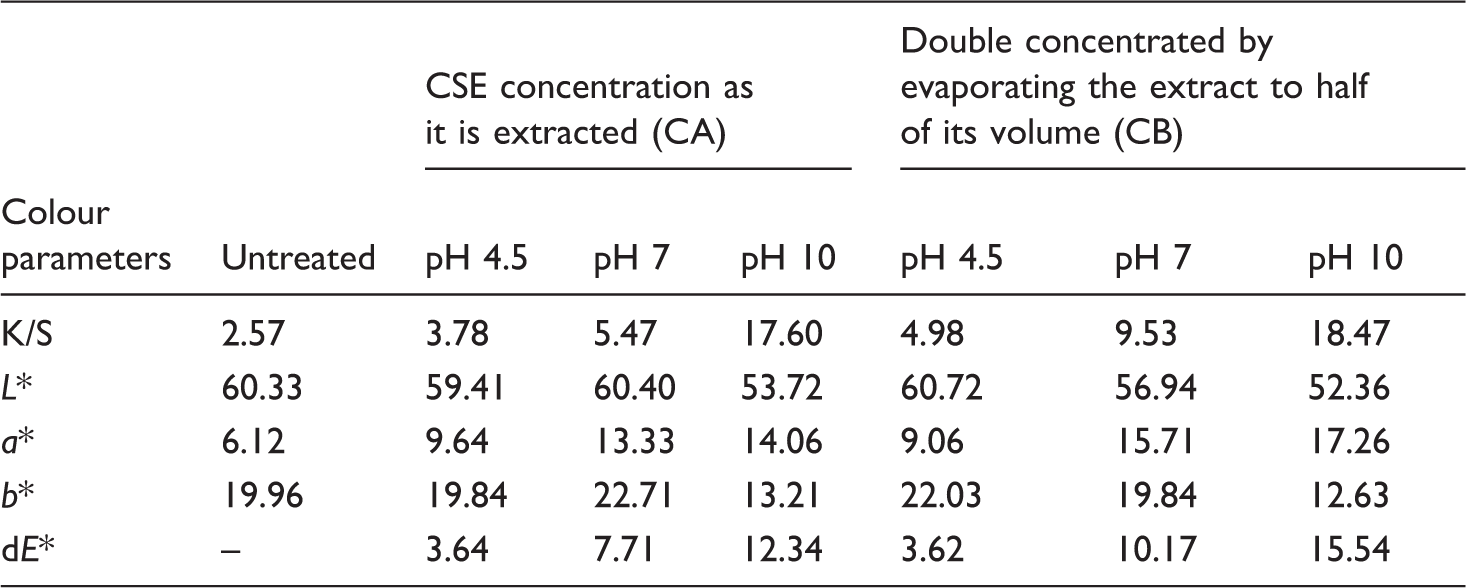
CSE: coconut shell extract; L*: lightness (0 = black, 100 = white), a*: red–green coordinates (positive values = red, negative values = green), b*: yellow–blue coordinates (positive values = yellow, negative values = blue), dE* = total colour difference.
The highest K/S value was obtained in the case of pH 10 both for CA and CB but there was an increase of 36% of K/S value in case of CB as compared to CA at pH 10. From a* and b* values, it can be seen that CSE-treated jute fabrics at pH 10 produced good improvement in colour and their values were positive which showed shifts in their tones resulting in beautiful colours. The K/S values increased with an increase in pH and concentration. The colouring substances of CSE are the natural tannins and polyphenols present in it. It can be concluded that jute fabric can be successfully dyed with CSE due to its richness in tannins.
Assessment of fastness properties of the dyed jute fabric
Fastness properties of jute fabric at different pH and concentration.

CSE: coconut shell extract.
Evaluation of flame-retardant activity
After one ISO 2 washing, LOI value of the CSE treated (pH 10 CB) fabric decreased from 38 for unwashed fabric to 31 in the washed fabric sample which is still significantly 47% higher than that noted for the untreated sample. The decrease in the flame-retardant properties in the CSE-treated sample after washing may be attributed to the fact that the CSE treatment only remains on the surface of the treated fabric and are removed partially after washing.
Ultraviolet (UV) protection property
UPF values of untreated and treated jute fabric with CSE.

CSE: coconut shell extract; UPF: ultraviolet protection factor.
Improvement of UV protection is mainly attributed to the effect of CSE which behaves as a natural dye and it bears more deep red colour at pH 10. Deep colour dyeing due to the presence of tannin also helps to increase the UPF property. Moreover, good UPF property after treatment of CSE might be due to the formation of coated layer on the dyed jute fabric as shown in SEM image. EDX analysis also shows the presence of oxides of metals of aluminum, manganese and nickel on dyed jute fabric with CSE without any use of mordant which also supports the UPF property of jute fabric. Polyphenols (phytochemical analysis) and anthocyanins (UV–visible spectroscopy analysis) have been reported as potential effective agents to prevent ultraviolet radiation [35].
XRD analysis
The XRD analysis of the untreated and treated jute was studied before and after treatment at different pH as shown in Figure 14 for CB. As the results are shown in Table 8, it was observed that on comparing the intensity of peaks due to crystalline and amorphous regions, the crystallinity of the jute increased from the untreated jute fabric compared to the treated jute fabric and it also increased as the pH increased. The treatment with the CSE causes the rearrangement of the cellulose chains in a more compact manner increasing the crystalline order, though, at acidic pH, there is increased amorphous nature due to the destruction of molecular arrangement [36].
X-ray diffraction (XRD) analysis plots of (A) untreated jute, (B) coconut shell extract (CSE)-treated jute fabric at pH 4.5, (C) pH 7 and (D) pH 10. Intensity of peaks due to crystalline and amorphous regions. Software calculated Lorentzian correction normalized intensity.

Peaks indicative of cellulose I and cellulose II crystalline arrangement.

2θ: diffraction angle; dÅ: interplanar distance; RI: relative intensity; (
Tensile strength of jute fabric
It is seen that the tensile strength (refer Figure 15) has increased by treating the jute fabric with CSE, by approximately 7% to 11% in the warp direction and 3% to 12% in the weft direction. It also showed an increase in tensile strength as pH of the treatment bath increased. This is supported by the XRD data showing increased apparent crystallinity with increased pH as well as the negative Poisson’s ratio in the y–z plane of the cellulose II crystalline form, contributing to the auxetic nature [38]. The CSE extract was shown to be of a binding character on the jute fabric surface which contributes to the increase in tensile strength by being favourably well dispersed on the surface, allowing even distribution of the load.
Tensile strength of untreated and treated fabric at different pH condition.
Mechanism of imparted multifunction properties on jute fabric
CSE-treated jute fabric showed flame retardancy which may be attributed to the presence of phenols, tannin (observed from phytochemical analysis of CSE), various positively charged metal ions (potassium, magnesium, aluminium, phosphorus, silicon, chlorine, manganese, nickel, iodine), metallic salts and oxides specially in the form of chloride, phosphate, silicate, magnesium chloride and sodium chloride. The presence of these inorganic salt molecules was roughly observed in the ATR-FTIR analysis of CSE and by the peaks of the elements in the EDX analysis of CSE. Anthocyanins (a component of CSE) belong to the group of flavonoid natural dyes and contain three to six hydroxyl groups that can be methylated. There might be electrostatic interaction of the anionic lignocellulose and cationic anthocyanins in CSE. As a result, a stable complex might be formed due to the interaction [39]. It has been found that the amount of phosphorus and silicon is more in the alkaline CSE-treated fabric, and it is showing more thermal stability rather than the acidic CSE-treated fabric. As these phosphates and chlorinated products can act as fire retardants for the lingo-cellulosic substrate, the flame retardancy effect of the CSE treated jute fabric is attributed to the combined synergistic effect of the presence of different metallic salts, silicate, phosphate, nitrate and bound and unbound water molecules and different phytochemicals. Their combined presence has helped in the formation of more char and non-flammable gases like CO2, H2O, etc. as observed from the TGA. Moreover, increase in add-on % also helps to increase LOI values for the treated fabric particularly for the alkaline pH of concentrated extract solution (CB). As far as the physical observation (heavy intense black colour char mass formation after burning, earlier dehydration from the TG curve, voluminous foamy nature of CSE observed from SEM analysis) is concerned, it can be said that CSE worked on the condensed phase intumescent mechanism. On the contrary, different positive metal ions (potassium, aluminium, nickel, magnesium, silicon) present in the CSE (as observed from EDX analysis), presence of tannin, terpenoid, saponin bio-molecules jointly may rupture the negatively charged bacterial cell wall, finally killing the bacteria and provide antibacterial property [31]. Moreover, good UPF property after treatment of CSE might be due to coated layer which contains different metal oxides such as aluminum, nickel, manganese on the dyed jute fabric as shown in SEM and EDX image. Polyphenols (phytochemical analysis) and anthocyanins (UV–visible spectroscopy analysis) have been reported as potential effective agents to prevent ultra violet radiation [35]. The presence of tannin and other phytochemical groups also are responsible for the formation of colouration on jute fabric, particularly when it was applied from alkaline pH (10) at higher concentration (CB).
Conclusion
The present study highlighted with the multifunctional effect of CSE on the ligno-cellulosic material. CSE application at different pH improved the thermal stability of the treated jute fabric. CSE treatment at alkaline concentrated condition increases the thermal stability of the treated jute fabric as the treated fabric showed specific char length. Flame retardancy in the CSE-treated jute fabric might be attributed to the presence of phosphate compounds, inorganic metal salts especially sodium and potassium chloride, which helped in the production of more char and non-flammable gases. On jute fabric, CSE application was carried out in acidic, neutral and alkaline pH. Moreover, the inherent property of the cellulosic textile such as comfort, transparency and strength was preserved. Treated ligno-cellulosic fabric also showed very good antibacterial and good UPF property. Moreover, treated fabric showed natural colour and it is was found to be better in the case of alkaline pH at higher concentration. Therefore, it can be concluded that CSE acts as multifunctional reagent for ligno-cellulosic jute fabric which can be used for various applications such as making low-cost home furnishing products like public halls, theatre, sofa cover, table lamp, curtains, protective material for making tents, carpet backing, jute brattice cloth in mines and packaging materials. As green coconut shell, the raw material is widely available and mainly a waste product, this kind of research approach might lead to a good value addition from the recycling of the waste material for ligno-cellulosic and cellulosic material.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: One of the authors Mr. Pintu Pandit is indebted to University Grants Commission-Basic Scientific Research (UGC-BSR) having award letter number F.25-1/2014-15 (BSR)/No. F.5-65/2007(BSR), for the scholarship support from the Govt. of India during the period of this study.


