Abstract
Human hairs are considered as major waste from barbershops. It contains an important protein called keratin. Keratin from human hair exhibits biocompatibility, wound-healing property, biodegradability, and antibacterial property. Silver and gold nanoparticles are commonly used in wound-healing applications. These nanoparticles possess excellent functional properties like antibacterial, wound healing and biocompatibility and applicable in biomedical field. In this study, silver and gold nanoparticles were used along with human hair keratin for the production of antibacterial materials. The functional properties of these two materials are synergized for wound-healing applications. Silver and gold nanoparticles were synthesized by chemical reduction method. The synthesized nanoparticles were characterized using UV–Visible, particle size, and zeta potential analyses. The prepared materials were characterized by scanning electron microscopy, Fourier transform infrared, and energy-dispersive X-ray spectroscopic analyses and they were measured for the physical properties such as air permeability, water contact angle, moisture content, and water absorbency. Furthermore, the antibacterial activities were evaluated against burn wound bacteria. Superior antibacterial activities and adequate physical properties found in keratin/nanoparticles immobilized cotton samples.
Introduction
Nanobiotechnology refers to the intersection of nanotechnology and biology. Recently, this technology is being used in disease diagnosis, target drug delivery system, and clinical areas [1]. Nanobiotechnology has high potential applications in medical sciences for improving health care among human beings around the world. Novel nanoparticles and nanodevices are introduced to have a positive impact on human health. Wong [2] studied the characteristics and applications of silver nanoparticles. Research community have found that silver nanoparticles have effective antibacterial properties against gram-positive and gram-negative bacteria. Guzman et al. [3] synthesized the silver nanoparticles by chemical reduction method. Kim et al. [4] stated that the silver nanoparticles have superior antibacterial properties. The silver nanoparticles showed improved wound-healing nature and anti-inflammatory effects on wound sites when compared to control samples [5]. Kwan et al. [6] found that the wounds treated with silver nanoparticles have better healing effect and good mechanical strength. Lima et al. [7] observed that gold nanoparticles have strong antibacterial activity against
The present research work aims at evaluating the human hair keratin-coated cotton gauze fabric for their bandage characteristics and antimicrobial activities against burn wound bacteria such as
Samples were named as follows: sample 1 for control fabric, sample 2 for silver nanoparticles immobilized keratin-coated fabric, and sample 3 for gold nanoparticles immobilized keratin-coated fabric.
Experimental methods
Materials
The materials needed for the research were collected from the vicinities in and around Tiruchengode, Tamil Nadu and certain chemicals were bought from Mumbai. The human hair sample required for the research was obtained from native barbershops in Tiruchengode, sterilized cotton gauze fabric was sourced from G. K. Surgical Private Limited, Rajapalayam and chemicals such as ethanol, methanol, hydrochloric acid, chloroform, urea, sodium carbonate, ethylene di-ammine tetra acetic acid, sodium hydroxide, silver nitrate were purchased from Chemico Chemicals, Erode. Sodium metabisulphate was purchased from Loba Chemie [P] Ltd, Mumbai while sodium dodecyl sulphate, tri sodium citrate, chloroauric acid, and other auxiliaries were procured from Merck Specialties [P] Ltd, Mumbai. These chemicals were used without further purification. Double-distilled deionized water was used in the research work.
Synthesis of silver nanoparticles by chemical reduction method
Silver nanoparticles were prepared by chemical reduction method [21]. About 50 mL silver nitrate solution (0.001 M) was taken in a conical flask. It was then heated to boiling temperature for 10 min. Trisodium citrate (5 mL of 1% concentration) solution was added drop by drop to the above-prepared solution with constant stirring. The solution was then heated to boil with continuous stirring until the color changed to greenish yellow. The change in color indicated the formation of silver nanoparticles. The formation of nanoparticles in the solution was analyzed by UV–Visible spectroscopy (Varian model 5000, Dandong Analog Radiative Instrument Co. Ltd, China).
Synthesis of gold nanoparticles by chemical reduction method
Gold nanoparticles were synthesized by chemical reduction method [22]. In this process, 50 mL (0.01 wt%) chlorauric acid solution was taken in a 100 mL conical flask. The solution was then heated to boiling temperature with continuous stirring. The trisodium citrate (1 wt%) solution was then added to auric solution with constant stirring until the color of the solution changed from yellow to black and then to red. The change in color indicated the formation of gold nanoparticles as confirmed by UV–visible spectroscopy (Varian model 5000, Dandong Analog Radiative Instrument Co. Ltd, China).
Isolation of keratin protein from human hair (Sulphidosis method)
Five grams of prepared and chemically conditioned hair samples were cut into small pieces. Human hair sample was dipped in 100 mL solution containing urea, sodium dodecyl sulphate, and metabisulphite at 65℃ for 2 h. The pH of the solution was adjusted to 6.5 by adding acetic acid solution. The solution was then dialyzed against distilled water using cellulose membrane for 3 days at 30℃. Finally, the purified keratin was stored at 2℃ in a refrigerator to maintain their stability.
The powder form of keratin was obtained by freeze drying of purified keratin solution at −46℃ for 3 days. The powdered form of keratin was used for characterization process [23].
Coating of keratin and silver nanoparticles
The keratin and silver nanoparticles were taken in the ratio of 3:2 and coated on gauze fabric. In this process, 360 mL of keratin solution (10 mg/mL concentration) was taken in a tray. Sterilized gauze fabric was padded twice in keratin solution using two bowl padding mangle at 30℃. The padded fabric was then dried at 80℃ for 5 min. Keratin-coated cotton fabric was then padded twice in 240 mL silver colloidal solution using the same equipment. Furthermore, the padded fabric was dried at 80℃ for 5 min and wrapped in aluminum foil and sealed with the help of a zip lock cover.
Coating of keratin and gold nanoparticles
The keratin and gold nanoparticles were taken in the ratio of 3:2 and coated on gauze fabric. In this process, a measured volume of 360 mL keratin solution (10 mg/mL concentration) was taken in a tray. Sterilized gauze fabric was padded twice with keratin solution using two bowl padding mangle at 30℃. The padded fabric was then dried at 80℃ for 5 min. Furthermore, the keratin-coated fabric was then padded twice with 240 mL gold colloidal solution using the same equipment. The padded fabric was dried at 80℃ for 5 min and wrapped by aluminum foil and sealed with the help of a zip lock cover.
Characterization of nanomaterials
Silver and gold nanoparticles were characterized using different techniques such as UV–Visible spectroscopy, particle size analysis, and zeta potential.
Characterization of silver and gold nanoparticles
UV–Visible spectroscopy
Silver and gold nanoparticles were characterized by UV–Visible spectroscopy from 200 to 700 nm. An ultraviolet-visible (UV–Vis) spectrophotometer (Varian model 5000, Dandong Analog Radiative Instrument Co. Ltd, China) was used to determine the surface plasmon resonance (SPR) of gold and silver nanomaterials. The absorbance spectrum of nanoparticles was recorded from 200 to700 nm.
Particle size and zeta potential analysis
The particle size of silver and gold particles was analyzed using Nanophox particle size analyzer, Sympatec, Germany. Zeta potential instrument was used to determine the surface charge of nanoparticles either in solid or solution (colloids) forms. The surface charge of nanoparticles was measured using Malvern Zeta Potential Instrument (Zetasize Nano Range), UK.
Characterization of nanoparticles (silver and gold) immobilized keratin-coated fabric
The samples were characterized using different techniques such as Fourier transform infrared spectroscopy (FTIR), scanning electron microscope (SEM), and energy-dispersive X-ray spectroscopy (EDX).
FTIR analysis
Sample 1, sample 2, and sample 3 were subject to infrared spectroscopy and the spectra were recorded using a NICOLET-6700 FTIR spectrophotometer.
SEM–EDX analysis
SEM HITACHI S-8840 was used to observe the surface morphology and uniform distribution of keratin and nanoparticles on the surface of samples. The elemental analysis was performed using the EDX.
Performance testing of bandage fabric
Sample 1 (control fabric), sample 2 (silver nanoparticles immobilized keratin-coated fabric), and sample 3 (gold nanoparticles immobilized keratin-coated fabric) were subject to air permeability (ASTM D737-96), water contact angle (ASTM D2495-07(2012)), moisture content (ASTM D2495-07(2012), and water absorbency (AATCC Test Method 79) tests. These tests were performed as per standard test methods.
Antibacterial activity test (Kirby–Bauer Test)
Antibacterial efficacy of sample 1, sample 2, and sample 3 were evaluated against
Results and discussion
UV–Visible spectroscopic analysis – Silver nanoparticles
UV–Visible spectroscopy was used to characterize the silver nanoparticles. The absorption spectrum of silver nanoparticles is shown in Figure 1. The SPR at 420 nm indicated the formation of silver nanoparticles and the average particle size was confirmed by particle size analyzer. Maribel et al. [25] revealed the formation of silver nanoparticles by the surface plasmon absorption spectra at 412 nm by UV–Visible spectroscopy. The researchers stated that position and absorption spectra are dependent on the particle size, shape, and dielectric constant of the surrounding medium [26]. Ashritha [27] observed the UV–Visible absorbance peak at 430 nm for silver nanoparticles.
UV–Visible spectra of silver nanoparticles.
UV–Visible spectroscopy analysis – Gold nanoparticles
UV–Visible spectrum of gold nanoparticles is shown in Figure 2. Gold nanoparticles exhibited a single absorption peak in the visible range between 450 and 500 nm, because of the SPR peak at 479 nm. UV–Visible absorption spectrum revealed the formation of gold nanoparticles by showing the surface plasmon absorption peak at 479 nm. Harihar et al. [22] observed the UV–Visible spectra between 510 and 550 nm for gold nanoparticles. Amir et al. [28] observed the spectra of gold at 530 nm by UV–Visible spectroscopy.
UV–Visible spectra of gold nanoparticles.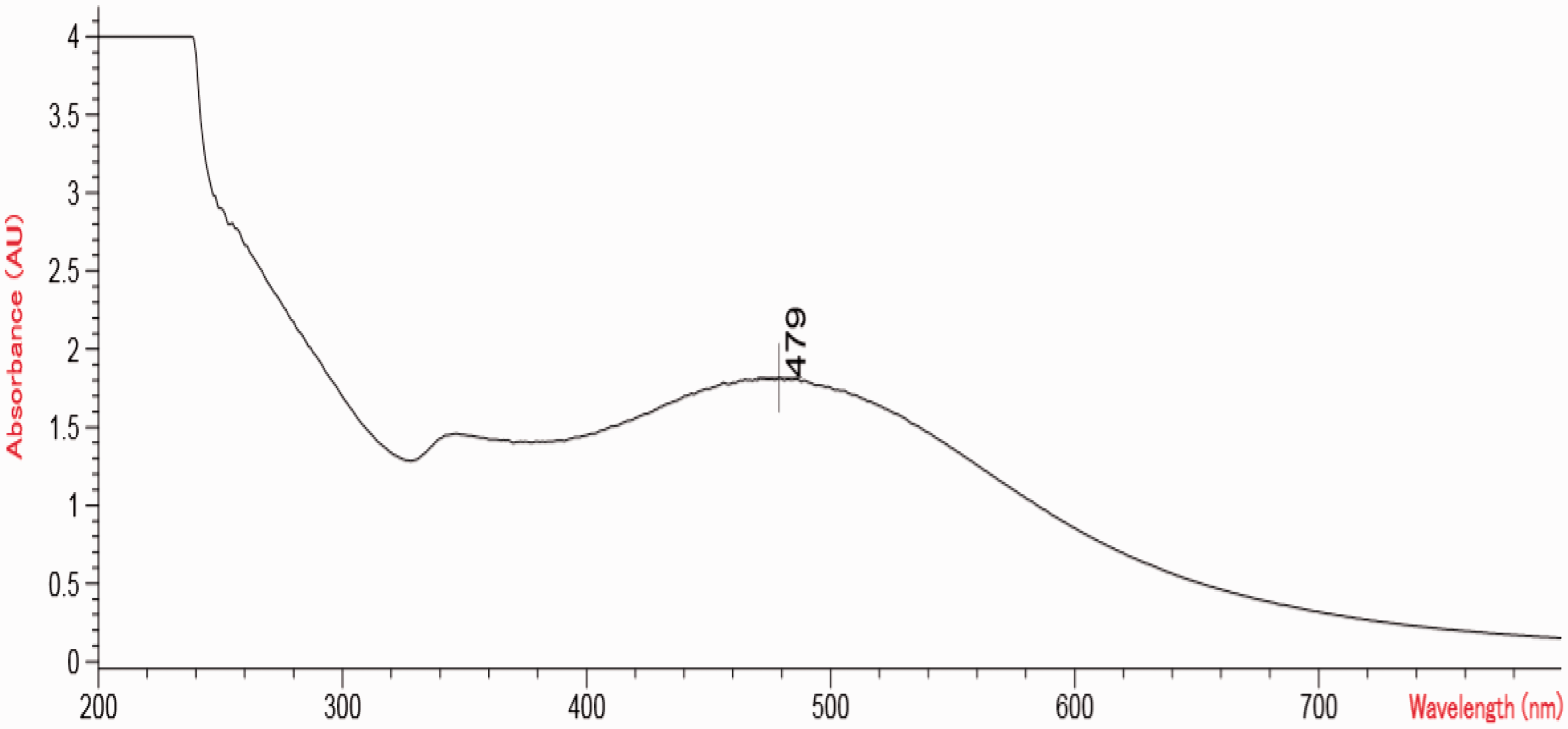
Particle size and zeta potential analysis
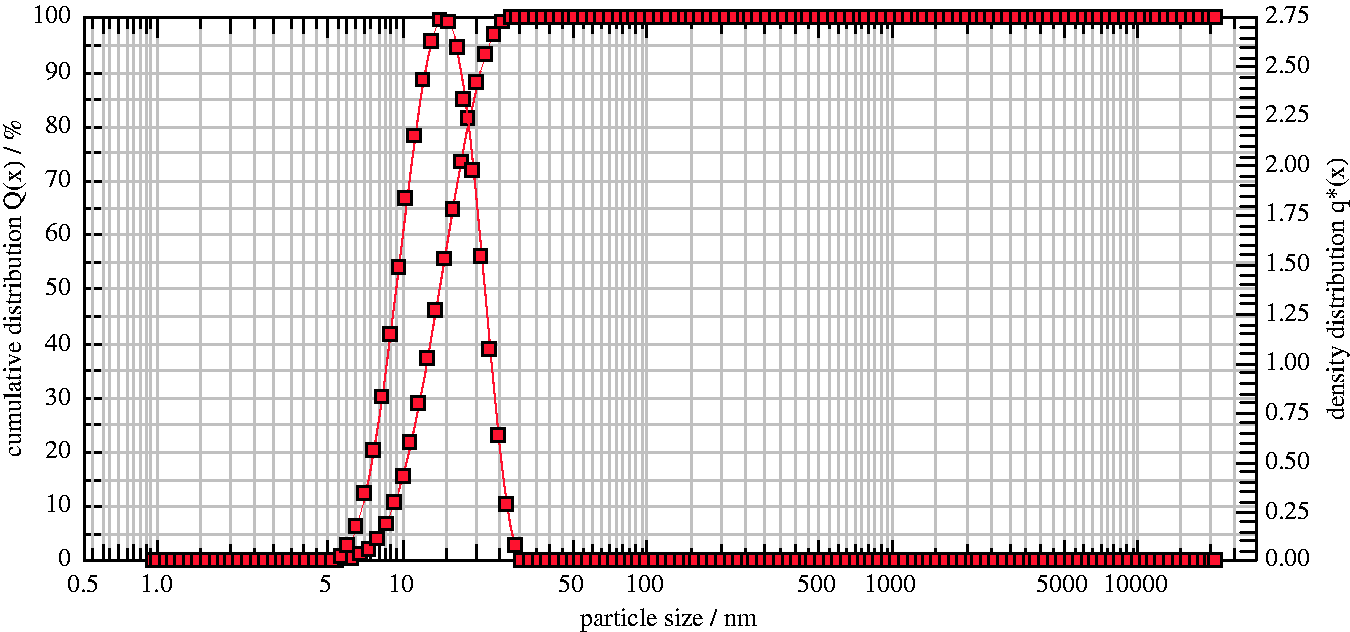
The nanoparticle size of silver and gold nanoparticles is shown in Figures 3 and 4, respectively. The size of silver nanoparticles is in the range of 50–110 nm with an average size of 71.8 nm and gold nanoparticles is in the range of 8–30 nm with mean size of 14.0 nm.
Particle size of silver nanoparticles. Particle size of gold nanoparticles.
Zeta potential value of silver and gold nanoparticles is shown in Figures 5 and 6, respectively. Figures show that the zeta potential value of silver and gold nanoparticles was −18.9 mV and −3.2 mV, respectively. Silver and gold nanoparticles possess negative charge due to the generation of electrostatic repulsion in the particles. The charge remains in the particles thereby preventing the aggregation of the particles. Therefore, the nanoparticles remain stable in solution without using any stabilizing agent. Saeb et al. [29] stated that the nanoparticles have either positive or negative zeta potential. They tend to repel each other and do not bind together. Melendrez et al. [30] studied the zeta potential value of metal nanoparticles. The authors found that the nanoparticles with zeta potential greater than 20 mV or less than −20 mV have sufficient electrostatic repulsion in particles thereby remaining stable in solution. Verma et al. [22] stated that negative charge of nanoparticles was due to citrate ions.
Zeta potential value of silver nanoparticles. Zeta potential value of gold nanoparticles.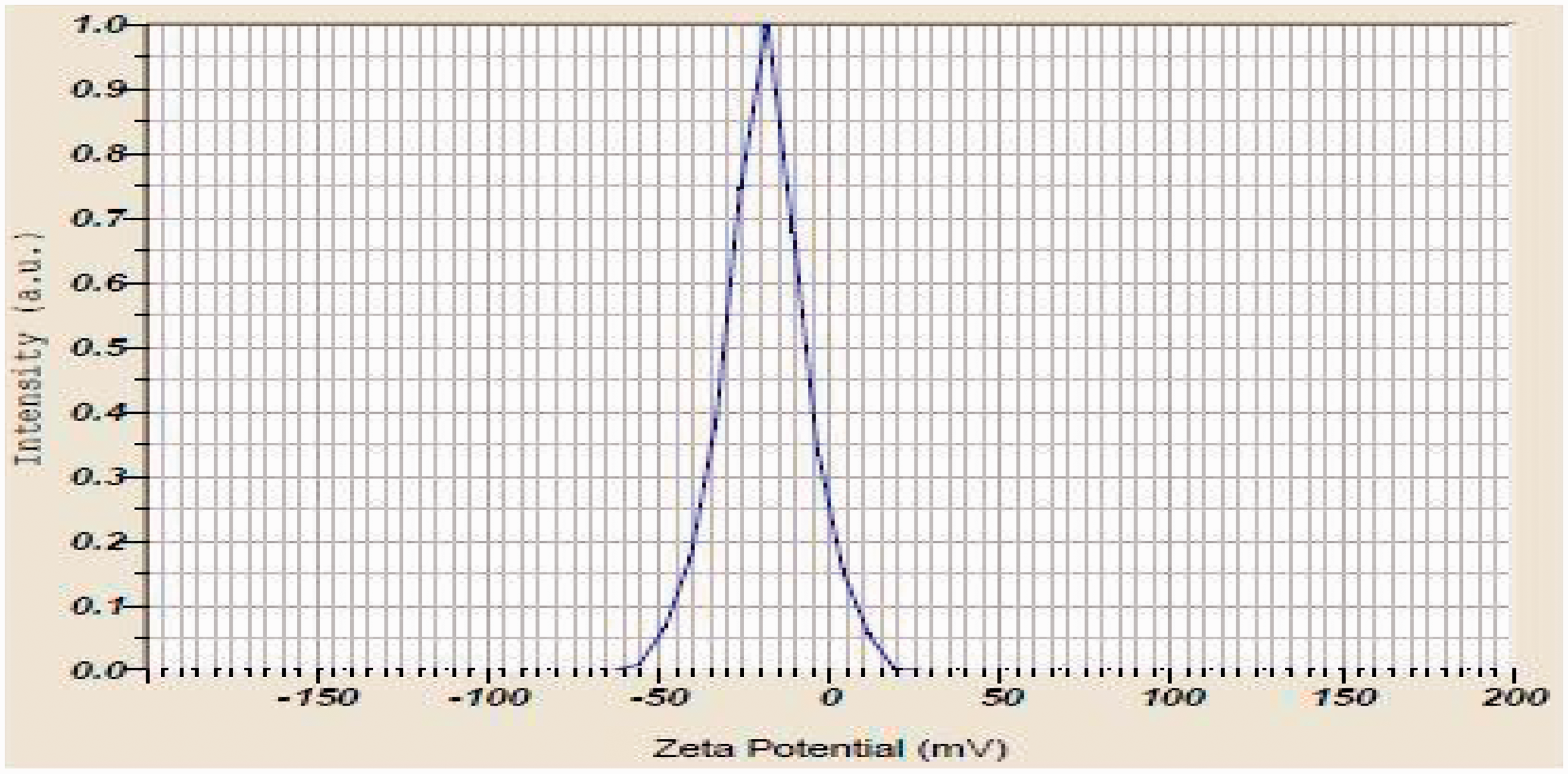

FTIR analysis
Sample 2 and sample 3 were subject to FTIR analysis. Infrared spectra were recorded using NICOLET-6700 FT-IR spectrophotometer. FTIR spectrums of control fabric (sample 1), keratin/silver nanoparticles coated fabric (sample 2), and keratin/gold nanoparticles coated fabrics (sample 3) are shown in Figure 7(a) to (c), respectively. Figure 7(b) and (c) shows the characteristics band for α-helix conformation in the amide I region at 1654 cm−1 and amide II region at 1545 cm−1, respectively. Furthermore, sample 2 and sample 3 show an amide I and amide II bands, which are the characteristics bands for α-helix conformation found in human hair keratin. Kong and Yu (2007) [42] observed the bands between 1600 and 1700 cm−1 (amide I region) and between 1500 and 1560 cm−1 (amide II region) for α-helix conformation. Lyman et al. [31] observed similar peaks for keratin. Figure 7(b) shows peaks at 3333, 2930, 1150, 1080, and 1029 cm−1 correspond to silver nanoparticles. Furthermore, the broad band appearing at 3333 cm−1 assigned for O–H stretching vibration indicated the presence of hydroxyl group in reducing agent. The peak at 1029 and 1080 cm−1 corresponds to C–O stretching vibrations. The spectrum from Figure 7(c) revealed the characteristic band for gold nanoparticles. The remaining peak at 3400, 2900, and 1057 cm−1 represented the cotton fabric.
(a) FTIR of cotton fabric; (b) FTIR of human hair keratin/silver nanoparticles immobilized cotton fabric; (c) FTIR of human hair keratin/gold nanoparticles immobilized cotton fabric.
SEM and EDX analysis
Sample 1, sample 2, and sample 3 were subject to SEM and EDX analyses. SEM analysis revealed the uniform distribution and high-density coating of keratin/silver nanoparticles (Figure 8(b)) and keratin/gold nanoparticles (Figure 8(c)) on the surface of cotton fabric. Moreover, the coated fabric show smooth and swelling surface and it was due to absorbency of keratin/silver and keratin/gold nanoparticles. SEM photograph of uncoated cotton fabric is shown in Figure 8(a).
SEM image: (a) uncoated cotton fabric; (b) human hair keratin/silver nanoparticles immobilized cotton fabric; (c) human hair keratin/gold nanoparticles immobilized cotton fabric.
Elemental analysis of silver and gold nanoparticles was performed using EDX and the results are displayed in Figure 9(a) (silver) and Figure 9(b) (gold), respectively. The peaks around 0.5 keV, 1.5 keV, 2 keV, and 5 keV correspond to the binding energy of C, O, Na, and Ag, respectively. Moreover, the peaks around 0.5 ke V, 1.5 keV, 2 keV, and 6 keV correspond to the binding energy of C, O, Na, and Au, respectively.
EDX image: (a) human keratin/silver nanoparticles immobilized fabric; (b) human keratin/gold nanoparticles immobilized fabric.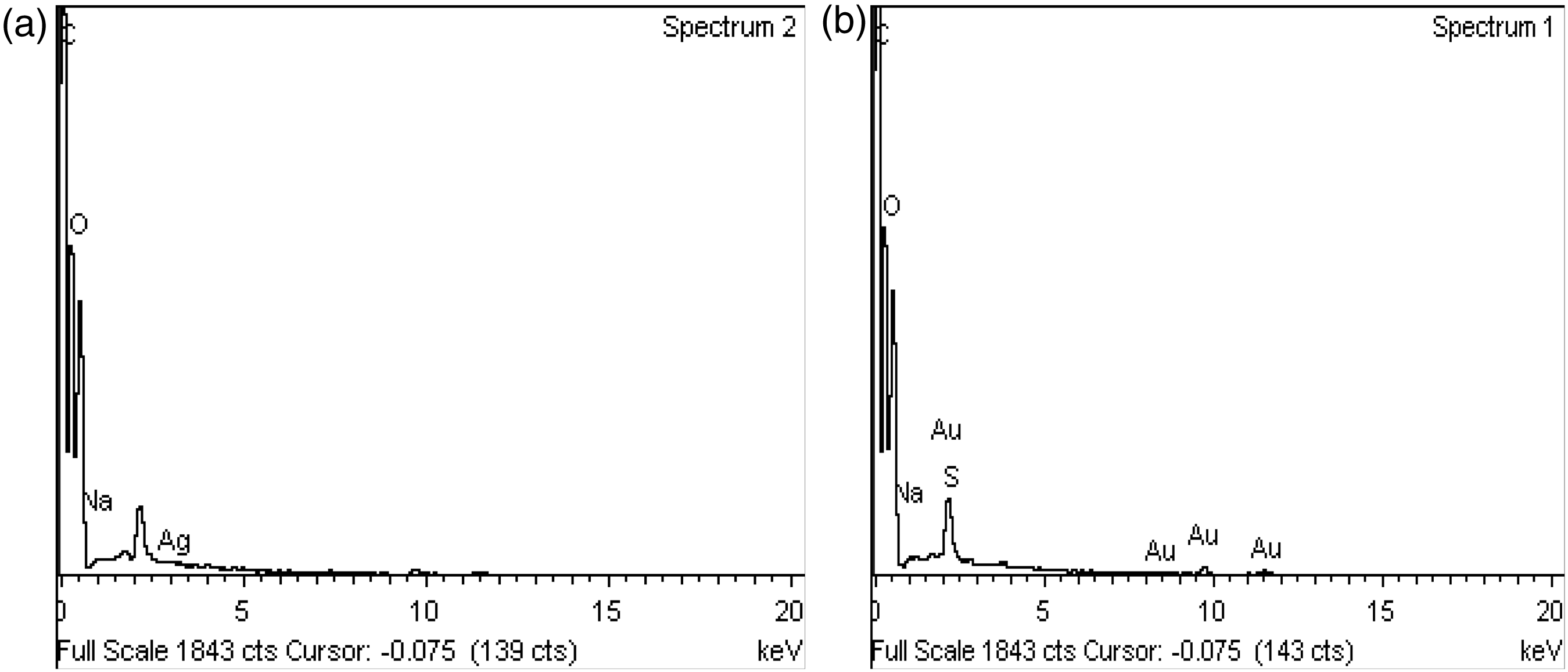
Air-permeability test (ASTM D737)
Air permeability of samples.

Contact angle test and moisture content test (ASTM D2495-07(2012)
Water contact angle of samples.

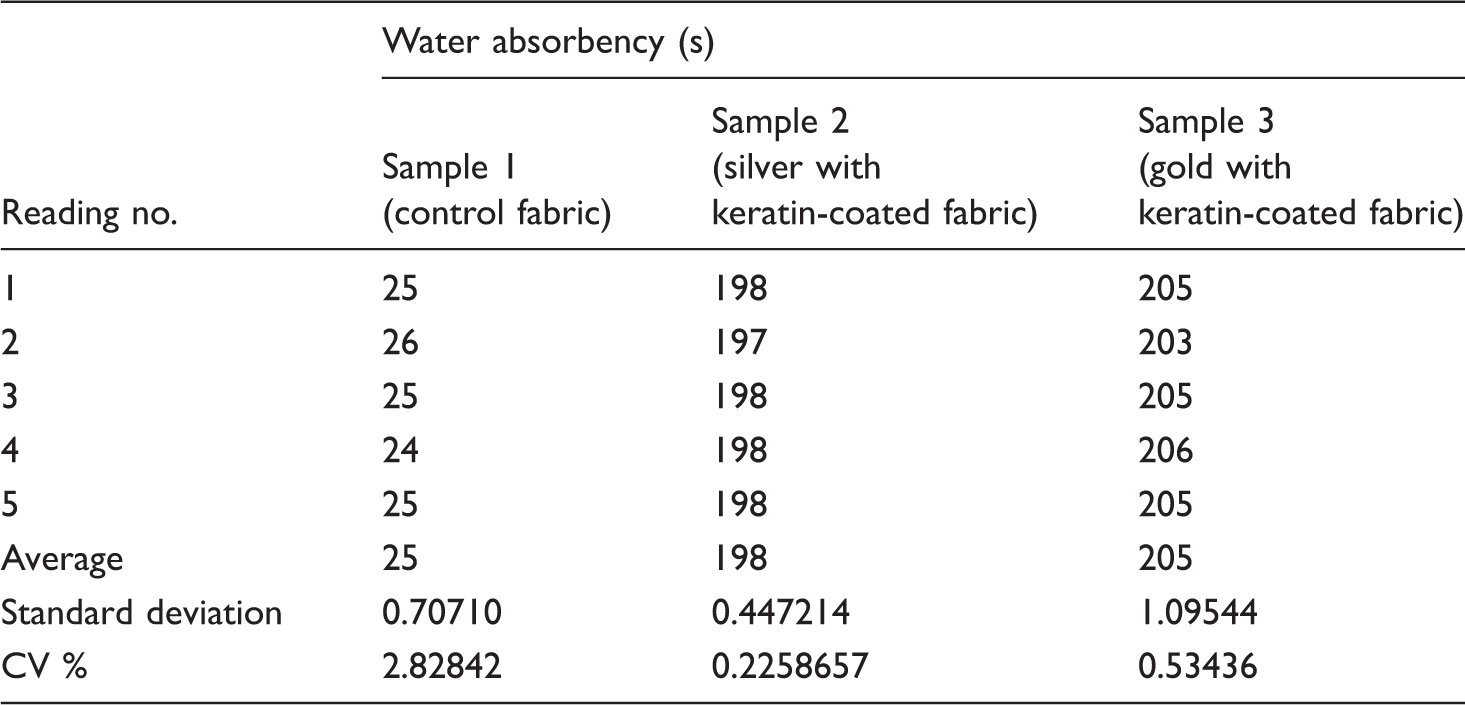
Water absorbency test (AATCC Test Method 79)
Water absorbency of samples.
Antibacterial activity test (Kirby–Bauer test)
Antibacterial test by zone of inhibition was carried out to determine the level of inhibition by silver and gold nanoparticles along with keratin biopolymer. The zone of inhibition (cm) around sample 1, sample 2, and sample 3 against the test organisms are shown in Figure 10. Sample 2 and sample 3 show greater antibacterial properties (i.e. > 3.2 cm zone of inhibition) against the test organisms on the first day. Zone of inhibition (cm) was increased in sample 2 and sample 3 from first day to third day of the test. Antibacterial test result revealed that the strongest antibacterial activities were found in sample 2 and sample 3 against Evaluation of antibacterial activities of the samples.
Conclusion
The coated cotton fabrics were developed using a combination of nanomaterials and biopolymer for wound-healing applications. Silver and gold nanoparticles were synthesized by chemical reduction method. The nanoparticles were characterized using UV–Visible spectroscopy, particle size analyzer, and zeta potential measurement. The UV–Visible spectra at 420 nm and 479 nm indicated the formation of silver and gold nanoparticles with an average size of 71.8 nm and 14.59 nm, which was confirmed by particle size analyzer. The silver and gold nanoparticles possess negative zeta potential value of −18.9 mV and −3.2 mV, respectively. Furthermore, FTIR analysis revealed the presence of keratin, silver nanoparticles, and gold nanoparticles in coated samples. SEM photograph showed the uniformly distributed and high-density coated keratin and nanomaterials on the surface of sample 2 and sample 3, respectively. Moreover, sample 2 and sample 3 possess not only high percent of oxygen and carbon, which was confirmed by EDX analysis but also adequate physical properties and superior antibacterial activities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
